Abstract
Background:
Sunitinib, a multitarget tyrosine kinase inhibitor, showed encouraging antitumor activity and manageable toxicity in patients with advanced midgut neuroendocrine tumors (NETs) in earlier results from phase I and II trials.
Patients and methods:
In this phase II trial, patients with a nonresectable grade 1 or 2 midgut progressive NET and Eastern Cooperative Oncology Group performance status 0–1 were randomly assigned 1:1 to receive 37.5 mg sunitinib or a placebo, combined with 120 mg lanreotide autogel every 28 days. The planned sample size was 104 patients. The primary outcome was investigator-assessed progression-free survival (PFS).
Results:
The study was stopped early because of insufficient patient recruitment. Between January 2013 and December 2016, 44 patients were enrolled and received sunitinib (n = 22) or placebo (n = 22). The median age was 63.7 years (Q1–Q3 range, 56.6–68.1) and 26 patients (59.1%) were male. The main localization was ileum (N = 37, 84.1%) and the majority were grade 2 (n = 25, 56.8%). The median follow-up was 36.7 months (95% confidence interval (CI) 34.6–48.2). The median PFS was 9.84 months (95% CI 6.8–23.3) with sunitinib and 11.47 months (95% CI 5.4–15.3) with placebo (hazard ratio (HR) = 0.80, 95% CI 0.41–1.56, p = 0.51). There was no difference in overall survival between treatment arms (HR = 0.81, (95% CI 0.32–2.01), p = 0.64). The objective response rate was 9.1% with sunitinib and 0.0% with placebo, and 19 patients (86.4%) had stable disease. Thirty-nine patients (88.6%) completed the baseline QLQ-C30 questionnaire. Baseline health-related quality of life level was similar between treatment arms, except for physical and emotional functioning which were higher (p = 0.089) and lower (p = 0.023) in the sunitinib arm, respectively. Trends toward longer time until a definitive deterioration in favor of the sunitinib arm were observed for 10 out of 15 dimensions (HRs < 1), with a significant result for financial difficulties (HR = 0.31, (90% CI 0.10–0.94)). Twenty-seven patients (61.4%) had at least one adverse event grade ⩾3 (sunitinib: 72.7%, placebo: 50.0%), with only one patient grade 4 for hypertension and vomiting. Eleven deaths non-related to treatment occurred (sunitinib arm: n = 5, placebo arm: n = 6).
Conclusion:
Our study does not provide enough evidence to conclude the role of sunitinib in advanced midgut NETs, primarily due to a lower-than-expected number of enrolled patients. While we cannot entirely rule out the efficacy of sunitinib, lanreotide alone may play a significant role.
Trial registration:
EudraCT: 2012-001098-94.
Plain language summary
Neuroendocrine tumours of the small intestine are a rare condition with a very different prognosis and treatment from the more “classic” tumours (known as intestinal adenocarcinomas). A wide variety of treatments can be proposed, depending on the extent of the disease, the specific characteristics of the tumour (degree of proliferation) and how far it has progressed. Treatments may include surgical removal of tumours, administration of hormones (somatostatin analogues), “conventional” chemotherapy agents (cytotoxics), targeted systemic treatments, embolisation/chemotherapy applied directly to liver metastases, radioactive agents (PPRT), etc. Sunitinib is a drug that prevents the synthesis of vessels by certain tumours, thereby preventing them from feeding and proliferating. We compared this drug with a placebo, in combination with a somatostatin analog named lanreotide (phase II study). The main objectives studied were the rate and duration of tumor control (named progression free survival or PFS, corresponding to the length of time during the treatment of a tumour that a patient lives with the disease but it does not get worse. In a clinical trials, this is one classical way to see how well a new treatment works) and the quality of life parameters. The full recruitment of 104 patients planned for this study performed in 11 French center could not be reached, no doubt because of the rarity of this disease and the existence, at the same time, of therapeutic trials with other treatments. Despite some favourable trends for sunitinib on quality of life, the study was not conclusive, partly due to the use of octreotide in all patients, which may have interfered with the experimental treatment.
Introduction
Neuroendocrine tumors (NETs) are relatively rare malignancies, although their incidence has steadily risen over the last decades, to reach approximately 7 cases per 100,000 in the United States. 1 Midgut well-differentiated NETs are one of the most frequent localizations, especially those arising from the small intestine. Because of the indolent nature of this disease, the majority of patients with midgut NETs are diagnosed with distant metastases. 1 Midgut NETs are characterized by their intrinsic ability to produce vasoactive peptides and hormones such as serotonin, which can be responsible for carcinoid syndrome. Patients with midgut NETs have a 5-year overall survival (OS) rate of approximately 70%, even in the presence of metastases.1,2
Currently, there are limited therapies with proven benefits for patients with unresectable, advanced midgut NETs. Treatment options primarily include somatostatin analogs, which may relieve symptoms related to hormonal hypersecretion and participate in controlling the tumor.3,4 Upon progression on first-line somatostatin analogs, everolimus 5 and peptide-radionuclide receptor therapy with 177Lu-DOTATATE 6 have demonstrated a significant prolongation of progression-free survival (PFS) in patients with NETs. Moreover, transarterial liver embolization has been associated with symptomatic and antitumor response, but its use relies on a low level of evidence. 7 Cytotoxic chemotherapy has limited efficacy in this setting. 8 Hence, establishing treatment strategies in patients with advanced midgut NETs remains challenging, as underlined by the national and international treatment guidelines.9–11
One characteristic of digestive NETs is their rich vascularization, making them a relevant therapeutic target. Sunitinib is an orally administered tyrosine kinase inhibitor targeting the receptors of vascular endothelial growth factors (VEGFR-1, 2, 3), platelet-derived growth factors (PDGFRα/β), and the KIT, FLT3, and RET signalization pathways, 12 providing a robust rationale for its evaluation in patients with NETs. In 2011, sunitinib was proven efficient in patients with pancreatic NETs in a phase III trial, 13 and was therefore approved by the Food and Drug Administration in this setting. Earlier results from phase I and II trials showed encouraged antitumor activity and manageable toxicity of sunitinib in patients with advanced midgut NETs.14,15
Based on these observations, we conducted a phase II, randomized, double-blinded trial SUNLAND to assess the safety and efficacy of sunitinib in patients with advanced midgut NETs.
Patients and methods
Study design and participants
SUNLAND was a multicenter, randomized, double-blinded phase II study (ClinicalTrials.gov identifier: NCT01731925) performed in 11 French centers and 2 Belgian centers. The study was approved by the institutional review board or ethics committee of each participating center and conducted in accordance with the Good Clinical Practice standards and Declaration of Helsinki, and relevant local regulations. SUNLAND was promoted and coordinated by the Multidisciplinary Groupe in Oncology (GERCOR) and supported by the European Neuroendocrine Tumor Society and the French Neuroendocrine Tumor National Network (RENATEN).
Eligible patients were aged ⩾18 years of age, had nonresectable, locally advanced or metastatic pathologically confirmed grade 1 (G1) or G2 midgut NET, an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1, life expectancy of ⩾3 months, and adequate organ function. Main biological limits were as follows: serum aspartate aminotransferases ⩽2.5 × upper limit of normal (ULN) and ⩽5 × ULN when liver function abnormalities were due to underlying malignancy, total serum bilirubin ⩽1.5 × ULN, serum albumin ⩾3.0 g/dL, absolute neutrophil count ⩾1500/μL, platelets ⩾100,000/μL, hemoglobin ⩾9.0 g/dL, and serum creatinine ⩽1.5 × ULN. Tumors were graded according to the World Health Organization 2010 classification (G1: Ki-67 ⩽2%; G2: Ki-67 >3% and ⩽20%). 16 Other inclusion criteria included documented progression and measurable disease according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 based on local assessment, and as assessed on contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI) scans within 12 months prior to baseline and at baseline. Main exclusion criteria included G3 NETs, pancreatic or bronchial NETs, uncontrolled cardiac complications due to carcinoid syndrome, obstructive intestinal tumor, treatment with lanreotide dose ⩾120 mg monthly, prior anti-VEGF therapy, unstable systemic disease, recent (⩽6 weeks) treatment with another anti-tumoral agent or loco-regional therapy, or concomitant treatment with CYP3A4 inhibitors/inducers (7/12 days prior to study drug administration, respectively).
Randomization
Eligible patients were randomly assigned in a 1:1 ratio to receive either sunitinib or placebo, both in combination with lanreotide autogel. The random allocation sequence was generated through a computer random number generator. Randomization using a minimization technique was stratified according to center, the Ki-67 index as determined by a local pathologist (⩽5% vs >5%), the degree of liver involvement (<50% vs ⩾50%) as determined by the investigator, and the time interval documenting tumor progression (⩽6 vs >6 months).
Procedures
Patients received once-daily oral sunitinib at the dose of 37.5 mg (sunitinib arm) or placebo (placebo arm), combined with subcutaneous injection of 120 mg lanreotide autogel every 28 days, until disease progression, unacceptable toxicity, and/or withdrawal of patient consent. Tumor evaluation was performed every 2 months by investigators using contrast-enhanced CT or MRI scans. The imaging modality of subsequent assessments was consistent with the baseline modality for each patient. Patients were followed for survival status after progression every 3 months until the final OS data cutoff date, designated as the date on which 70% of enrolled patients died or after 3 years of follow-up, whichever came first.
The following assessments were performed at screening, every 2 months until progression, at the end of treatment, and 28 days after the last treatment dose: vital signs, ECOG PS, physical examination, 12-lead electrocardiogram, and laboratory tests (blood hematology, biochemistry, thyroid function, chromogranin A, and urine 5-hydroxyindoleacetic acid). Adverse events (AEs) and laboratory abnormalities were collected throughout treatment until 28 days after the final dose. AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0. Health-related quality of life (HRQoL) was assessed using the European Organisation for Research and Treatment of Cancer QoL Questionnaire C30 (EORTC QLQ-C30) cancer-specific questionnaire at baseline, every 2 months during the treatment period, and at the end of the study. The QLQ-C30 includes 30 items and assesses a global health status, 5 functional scales (physical, role, emotional, cognitive, and social functioning), and 9 symptomatic scales (fatigue, nausea and vomiting, pain, dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties). Scores vary from 0 (worst) to 100 (best) for global health status and functional scales, and from 0 (best) to 100 (worst) for the symptomatic scales.
Outcomes
The primary outcome was investigator-assessed PFS defined as the time between randomization and the first disease progression by RECIST 1.1, or death from any reason in the absence of documented progression, whichever occurred first. Secondary outcomes were OS (the time between randomization and death of any cause), duration of response, objective response rate (ORR; the proportion of patients with complete or partial response per RECIST 1.1), safety, and time until definitive HRQoL score deterioration (TUDD); the time between randomization and a first HRQoL deterioration of at least 5 points as compared to the baseline score, without further improvement of more than 5 points as compared to the baseline score 17 ).
Statistical analysis
Sample size determination was based on the 6-month PFS rate, using Simon’s minimax two-step design, with a unilateral alpha type I error of 5% and a power of 90% and under the null hypothesis and alternative hypothesis of 55% and 75% PFS rate at 6 months in the experimental arm, respectively. Given the assignment ratio of 1:1 and an anticipated drop-out rate of 5%, the enrolment of 104 patients was required. The study had to be stopped if ⩽20 (60.6%) or ⩽32 alive patients (65.3%) were progression-free after the inclusion of 33 or 49 patients, respectively, with 6 months of follow-up in both arms.
Categorical variables were described as frequencies (percentages) and quantitative variables were described as median (interquartile range 25%–75% Q1–Q3). Efficacy was assessed in the intent-to-treat population, composed of all randomized patients. Kaplan–Meier estimation method was used to obtain the estimates of median event-free time associated with each treatment, with corresponding 95% confidence intervals (CIs) estimated using the standard errors derived by Greenwood’s formula. Safety was assessed in all patients who received at least one dose of the study treatment. HRQoL scores at baseline were compared using the t-test or Mann–Whitney nonparametric test. Median TUDD for each score was computed with its 90% CI. Univariate Cox proportional hazard analyses were used to estimate hazard ratios (HRs) and 90% CI of the treatment group effect.
Role of the funding source
This work was supported by Pfizer and Ipsen, which provided study treatments. The study was designed by academic investigators. Data collection and statistical analyses were done by the sponsor (GERCOR). The initial draft of the manuscript was prepared by the first author in collaboration with the sponsor. All authors were involved in the data collection, analysis and interpretation, and critical revision of the manuscript. The corresponding author had full access to the data and had final responsibility for the decision to submit it for publication.
Study reporting
The reporting of this study conforms to guidelines for reporting randomized studies from the CONSORT 2010 checklist (Supplemental Table S1).
Results
Patient characteristics
The study was stopped early because of insufficient patient recruitment. Between January 7, 2013 and December 7, 2016, a total of 44 patients were randomized to receive sunitinib (n = 22) or placebo (n = 22). The median age of patients with NETs was 63.7 years (Q1–Q3 range, 56.6–68.1) and there were more males than females (59.1% vs 40.9%). Patient and tumor characteristics were similar at baseline (Table 1). The primary midgut NETs were mainly located in the ileum (84.1%) and were classified as G2 (56.8%) with a median Ki-67 index of 3% (Q1–Q3 range, 1.5–8.9). There were 47.7% and 52.3% patients with ECOG PS of 0 and 1, respectively, and a similar proportion of patients in both groups had diarrhea (59.1% and 52.3%), flushing (50% and 54.5%), or abdominal pain (36.4% and 45.5%), respectively, related to carcinoid syndrome. Thirty-six patients (81.8%) previously underwent primary tumor surgery, and 26 patients (59.4%) had metastases surgery and/or percutaneous destruction (Table 1). All patients, but 2 (9.1%) in the sunitinib arm and 5 (22.7%) in the placebo arm, received prior systemic therapy. Overall, 37 (84.1%) and 5 (11.4%) patients were treated with long-acting somatostatin analogs and/or chemotherapy, respectively.
Patient baseline characteristics according to treatment arm.
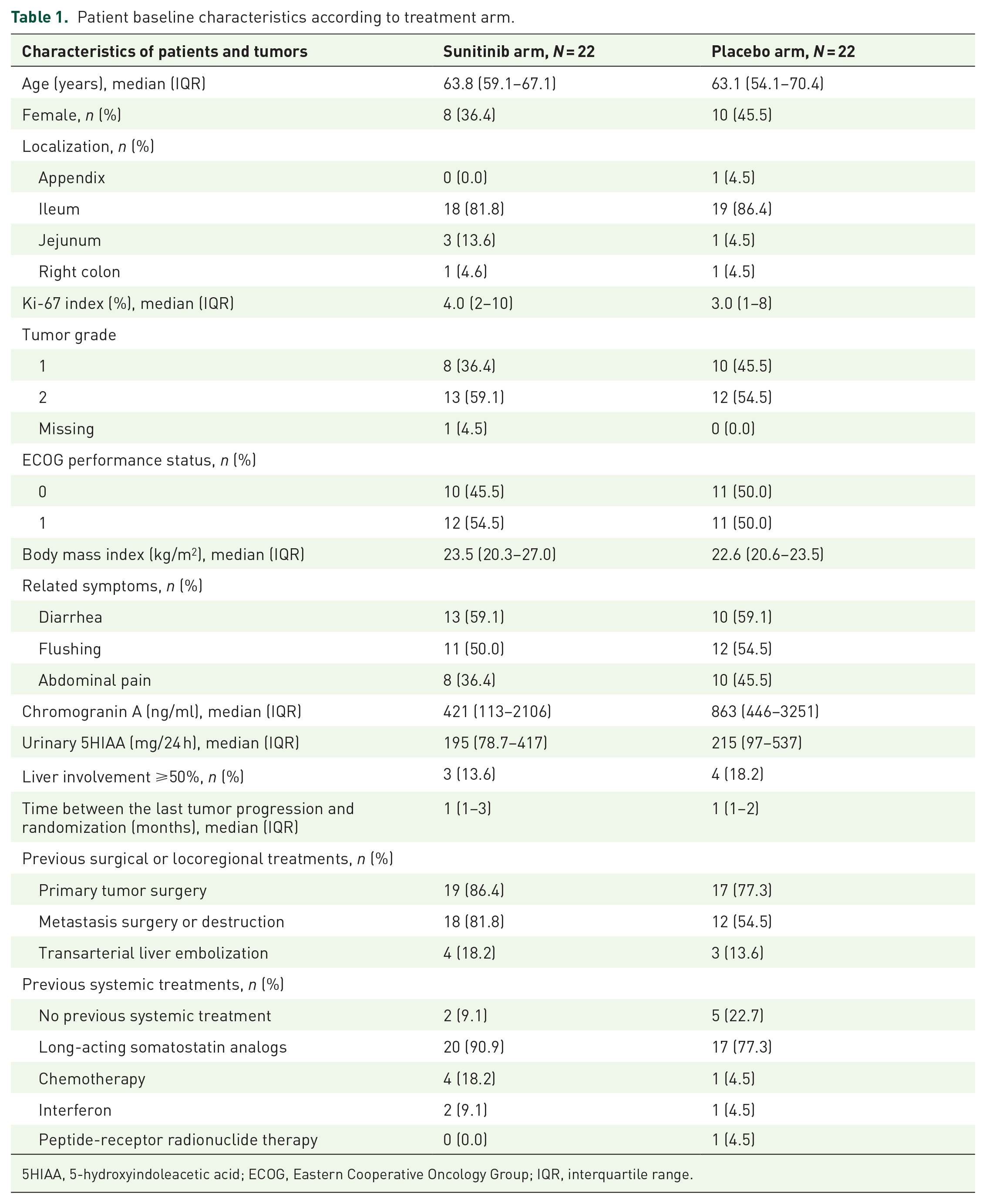
5HIAA, 5-hydroxyindoleacetic acid; ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range.
Treatment received and safety
The median number of cycles completed was 9 (Q1–Q3 range, 6–16) in the sunitinib arm and 10 (Q1–Q3 range, 4–15) in the placebo arm and the median treatment duration was 7.5 months (Q1–Q3 range, 4.6–15.0) and 9.2 months (Q1–Q3 range, 2.8–13.8) (p = 0.9535), respectively.
A total of 27 patients (61.4%) experienced at least 1 grade ⩾ 3 AEs; 16 (72.7%) in the sunitinib arm and 11 (50.0%) in the placebo arm (Table 2). Grade 4 hypertension (n = 1) and vomiting (n = 1) were observed in 2.3% of all patients. Fatigue, oral mucositis, asthenia, dysgeusia, diarrhea, nausea, and hand-foot syndrome were the most observed non-hematologic toxicities (Table 2). Overall, 21 serious AEs were reported in 14 patients and 12 follow-up visits. Among them, 18 were treatment-unrelated and 3 (anemia, acute coronary syndrome, hearing impairment) were classified as expected serious adverse reactions to treatment. Eleven deaths occurred (sunitinib arm: n = 5, placebo arm: n = 6), and all were related to NET progression or intercurrent disease. No treatment-related deaths were recorded.
Most frequent adverse events according to the treatment arm.

Efficacy and tumor response
Median follow-up was 36.7 months (95% CI (34.6–48.2)). At the last contact date, 9 alive patients without progression (20.4%) were censured, 6 patients without documented progression (13.6%) died, and 29 patients (65.9%) had tumor progression. Of the latter group of patients, 16 (55.2%) were alive, 12 (41.4%) died, and 1 (3.4%) was lost to follow-up.
Median PFS was 9.84 months (95% CI (6.8–23.3)) in the sunitinib arm and 11.47 months (95% CI (5.4–15.3)) in the placebo arm (Figure 1). The corresponding 6-month PFS rates were 77.3% (95% CI (53.7–89.8)) and 68.2% (95% CI (44.6–83.4)), respectively (Figure 2). There was no difference in PFS between the two arms (HR = 0.80, 95% CI (0.41–1.56), p = 0.51).

Progression-free survival according to treatment arm.
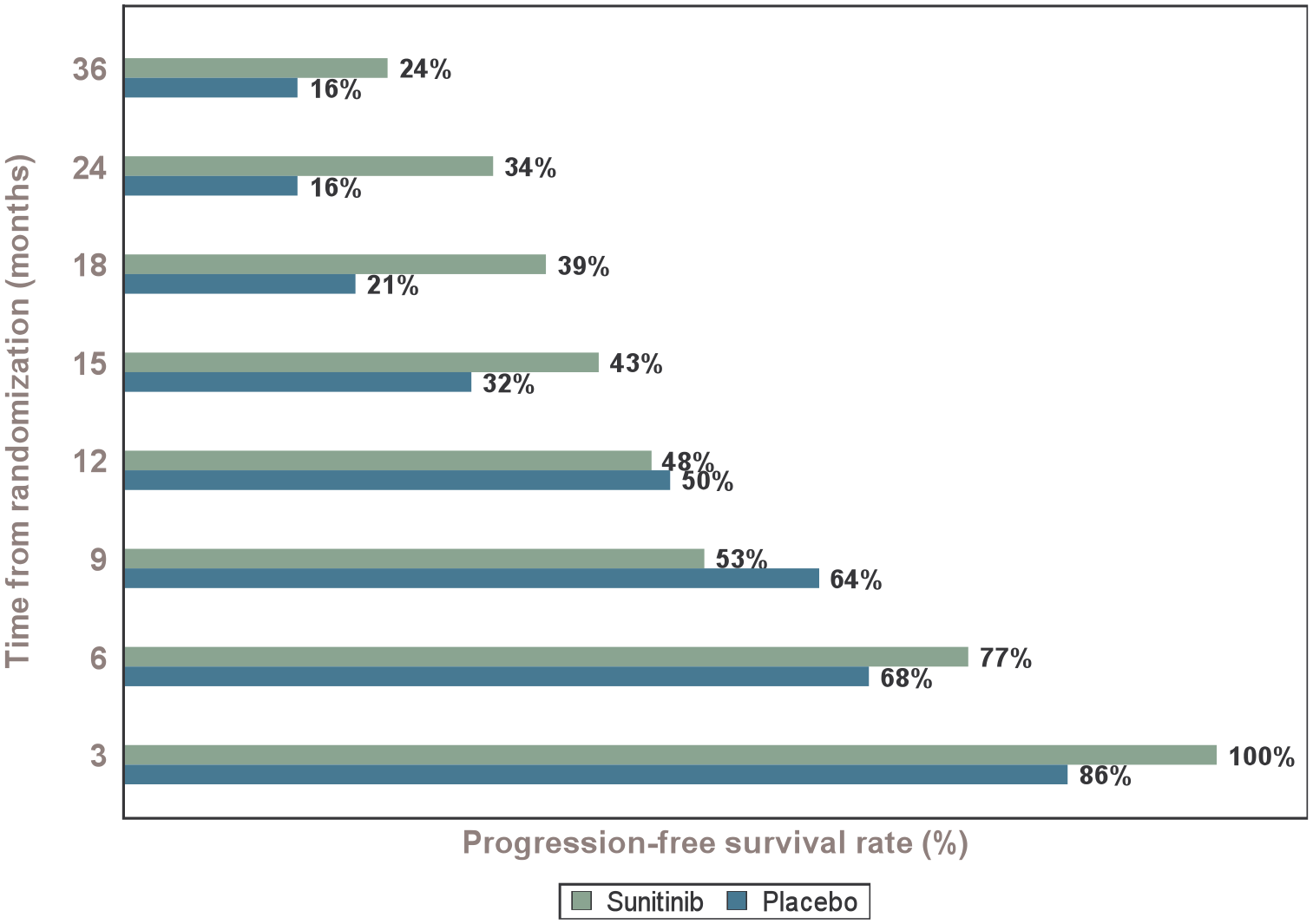
Progression-free survival rates of patients at times of interest.
Median OS was not reached in either arm at the end of the follow-up. The 6, 12, and 24-month OS were 100.0%, 100.0%, and 75.6% (95% CI (47.7–90.2)) in the sunitinib arm and 95.6% (95% CI (71.9–99.3)), 86.4% (95% CI (63.4–95.4)), and 72.4% (95% CI (48.6–86.6)) in the placebo arm, respectively (Figure 3). There was no difference in OS between the two arms (HR = 0.81, 95% CI (0.32–2.01), p = 0.64).

Overall survival according to treatment arm.
The ORR was 9.1% (2/22) in the sunitinib arm (one complete response and one partial response) and 0.0% in the placebo arm. Moreover, 19 patients (86.4%) had stable disease as the best response in each treatment arm.
Health-related quality of life
A total of 39 patients (88.6%) completed the QLQ-C30 questionnaire; 19 patients (86.4%) in the placebo arm and 20 patients (90.9%) in the sunitinib arm. Baseline HRQoL scores were similar between treatment arms, except for physical and emotional functioning which were higher (p = 0.089) and lower (p = 0.023) in the sunitinib arm (Supplemental Table S2).
The median deterioration in months for all scores is presented in Supplemental Table S3. Overall, trends toward longer TUDD in favor of the sunitinib arm were observed for 10 out of 15 dimensions with HRs (sunitinib vs placebo) <1. Among them, the TUDD result was significantly lower for patients treated with sunitinib only for the financial difficulties dimension (HR = 0.31, (90% CI 0.10–0.94)). The TUDD curves of the five targeted HRQoL scores (global health status, physical functioning, emotional functioning, fatigue, and diarrhea) are shown in Figure 4(a)–(e).

Time until definitive quality of life deterioration for (a) global health status. (b) Physical functioning. (c) Emotional functioning. (d) Fatigue. (e) Diarrhea scores according to treatment arm.
Discussion
The SUNLAND study was designed to prospectively assess the potential role of sunitinib in patients with advanced midgut NETs. The interim analysis did not allow us to draw a definitive conclusion about the role of sunitinib in this setting, primarily due to the lack of statistical power related to insufficient patient enrolment. Nevertheless, we observed that sunitinib tended to temporarily slow the disease progression and identified a small subset of patients who achieved tumor response in the sunitinib arm, but not in the placebo arm. Moreover, analyses of EORTC QLQ-C30 data showed a trend that 10 out of 15 HRQoL dimensions had longer TUDD in favor of sunitinib. However, we cannot exclude that some of these effects were linked to the somatostatin analogs rather than the addition of sunitinib into the experimental arm.
Compared with pancreatic NETs, a limited number of efficient treatment options are available in patients with advanced midgut NETs. Long-acting somatostatin analogs can increase PFS, as shown by the two phase III trials PROMID and CLARINET.3,4 Given the results of these studies, they are recommended as the first line in patients with metastatic midgut NETs, even if their role has been better demonstrated in slow-growing tumors (Ki-67 <10%) with metastatic liver involvement <50%.9–11 Otherwise, the efficacy of everolimus (the RADIANT-4 and 177Lu-DOTATATE (NETTER-1 studies5,6)) has been shown in progressive metastatic midgut NETs. Transarterial liver embolization can be relevant in selected NET patients who have a prominent live involvement and provide an ORR of 30% and a 20-month PFS.7,18 In addition, the role of systemic chemotherapy is very limited in this setting, except in the case of progressive, highly proliferative, and/or bulky disease, notably after the failure of other treatment options. 8 Alkylating agents have poor efficacy 19 ; the 5-fluorouracil–dacarbazine combination has provided only modest results (ORR: 14%, median PFS: 9 months).8,20,21 Interestingly, the best results were those obtained using capecitabine combined with the antiangiogenic bevacizumab. In the BETTER phase II study, 22 among 49 patients with pretreated advanced gastrointestinal NETs, ORR and disease control rate were 18% and 88%, respectively, and the median PFS was 23.4 months, underlying the relevance of targeting angiogenesis in these patients.
In 2011, Raymond et al. 13 reported that a single-agent sunitinib had an 11.4-month PFS (vs 5.5 months in the placebo arm, HR = 0.42, 95% CI (0.26–0.66), p < 0.001) in patients with progressive, advanced pancreatic NETs and was subsequently approved as a standard treatment in this setting. In addition, a post hoc analysis of that study showed a prolonged OS versus placebo (HR = 0.73, 95% CI (0.50–1.06), p = 0.094; 38.6 vs 29.1 months). 23 The lack of statistically significant difference was likely due to a high rate of crossover to sunitinib in 69% of the patients in the placebo arm. 23 In a randomized phase II study reported by Kulke et al., 14 among 41 patients with “carcinoid tumors” (19 with midgut NET), 43.9% had tumor shrinkage, but only 1 (2.4%) had RECIST-based ORR. The disease control rate was 85.4% and median PFS was 10.2 months (95% CI (9.2–17.5)).
In the SUNLAND study, the ORR in the patients who received sunitinib was low (9.1%). This highlights the challenge of assessing the efficacy of cytostatic treatments, particularly in slowly growing tumors such as midgut NETs. 24 Alternative imaging such as “Choi-like” criteria, measuring changes in tumor density could be more relevant to evaluate the antitumor efficacy of sunitinib.25,26 In our study, the 6-month PFS rate was 77.3% in the sunitinib arm, which was above the prespecified threshold defining efficacy. Despite the lack of differences in PFS between the two arms, higher PFS rates with sunitinib, at least after 1 year from randomization, suggest that some patients may benefit from this drug. However, an additive role of lanreotide in the combination arm on the antitumor effect remains possible.
Given the lanreotide antitumor effect, 3 evaluation of co-administered targeted therapy is very challenging. In the RADIANT-2 study, the everolimus and long-acting octreotide combination was not superior to octreotide plus placebo in patients with advanced NETs and carcinoid syndrome. 27 Interestingly, in the later RADIANT-4 trial, everolimus showed superiority over placebo alone in patients with non-functioning NETs of lung or gastrointestinal origin. 5
In our study, 72.7% of the patients in the sunitinib arm had at least one grade ⩾3 AE (vs 50.0% in the placebo arm). However, these AEs were manageable and did not impact TUDD, which was longer in the sunitinib arm for 10 HRQoL dimensions of the QLQ-C30. The toxicity of treatments has to be cautiously taken into account to avoid the discouragement of patients who receive long-term administration for a slowly growing disease.
Since the beginning of the SUNLAND study, multiple drugs have been tested in advanced midgut NETs, particularly multisite tyrosine kinase inhibitors (e.g., cabozantinib, lenvatinib, axitinib, pazopanib), checkpoint inhibitors (e.g., pembrolizumab, avelumab, spartalizumab), and peptide-receptor radionuclide therapy (e.g., 177Lu-oxodotreotide, 177Lu-edotreotide). 28 The high number of prospective studies in this rare condition likely explains the difficulties of completing some trials, as was the case for SUNLAND. Recently, surufatinib showed promising efficacy. This novel multikinase inhibitor targets not only angiogenesis (VEGFR-1, 2, 3) but also fibroblast growth factor receptor-1 that contributes to resistance to VEGF blockade therapy and colony-stimulating factor-1 receptor, which participates in immune evasion. In the SANETep phase III study, surufatinib significantly prolonged PFS over placebo in patients with advanced extra-pancreatic NETs (gastrointestinal tract, 47%), suggesting that targeting angiogenesis alone might not be sufficient in this setting. 29 Combination of this drug with the programmed death 1 inhibitor toripalimab showed antitumor activity in pretreated NETs. 30
Conclusion
In conclusion, the data from our study do not allow us to draw definitive conclusions on the role of sunitinib in advanced midgut NETs, primarily due to lower-than-anticipated numbers of enrolled patients mainly due to competing studies in this rare condition. Although the efficacy of sunitinib cannot be entirely ruled out in this patient population, the possibility that lanreotide alone plays a more significant role than the addition of sunitinib remains plausible.
Supplemental Material
sj-doc-2-tam-10.1177_17588359241290140 – Supplemental material for SUNLAND: a randomized, double-blinded phase II GERCOR trial of sunitinib versus placebo and lanreotide in patients with advanced progressive midgut neuroendocrine tumors
Supplemental material, sj-doc-2-tam-10.1177_17588359241290140 for SUNLAND: a randomized, double-blinded phase II GERCOR trial of sunitinib versus placebo and lanreotide in patients with advanced progressive midgut neuroendocrine tumors by Pascal Hammel, Denis Smith, Pauline Afchain, Sophie Dominguez-Tinajero, Jean-François Seitz, Astrid Lievre, Eric Van Cutsem, Eric Assenat, Frédéric Di Fiore, Marc Peeters, Iradj Sobhani, Eric Raymond, Emilie Charton, Dewi Vernerey, Louis De Mestier and Catherine Lombard-Bohas in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-1-tam-10.1177_17588359241290140 – Supplemental material for SUNLAND: a randomized, double-blinded phase II GERCOR trial of sunitinib versus placebo and lanreotide in patients with advanced progressive midgut neuroendocrine tumors
Supplemental material, sj-docx-1-tam-10.1177_17588359241290140 for SUNLAND: a randomized, double-blinded phase II GERCOR trial of sunitinib versus placebo and lanreotide in patients with advanced progressive midgut neuroendocrine tumors by Pascal Hammel, Denis Smith, Pauline Afchain, Sophie Dominguez-Tinajero, Jean-François Seitz, Astrid Lievre, Eric Van Cutsem, Eric Assenat, Frédéric Di Fiore, Marc Peeters, Iradj Sobhani, Eric Raymond, Emilie Charton, Dewi Vernerey, Louis De Mestier and Catherine Lombard-Bohas in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Medical writing support for the development and submission of this manuscript, under the direction of the authors, was provided by Magdalena Benetkiewicz (Sc.D.) and was funded by GERCOR.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
