Abstract
Background:
Stereotactic body radiation therapy (SBRT) in treating non-small-cell lung cancer (NSCLC) exhibits a remarkable therapeutic efficacy. However, its effectiveness in overcoming resistance to epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) in patients with advanced EGFR mutations (EGFRm) NSCLC remains uncertain.
Objective:
We aimed to analyze the effect of SBRT on patients with first-line EGFR-TKIs.
Design and methods:
Eligible patients with advanced NSCLC initially diagnosed with EGFRm were enrolled. Patients in the EGFR-TKIs group received only the first-generation EGFR-TKIs until disease progression or death, while the others in the EGFR-TKIs + SBRT group received EGFR-TKIs and early SBRT (dose of 40–60 Gy/5–8 F) targeting the primary lung tumor at 1 month after EGFR-TKIs. The primary endpoint was progression-free survival (PFS), and the secondary endpoints were treatment-related adverse effects, overall survival (OS), and sites of initial failure.
Results:
A total of 184 advanced NSCLC patients with EGFRm were enrolled, including 39 patients in the EGFR-TKIs + SBRT group and 145 patients in the EGFR-TKIs group. The median PFS was 15.50 months in the EGFR-TKIs + SBRT group compared to 9.33 months in the EGFR-TKIs group (p = 0.0020). However, the median OS was 29.10 months in the EGFR-TKIs + SBRT group and 26.33 months in the EGFR-TKIs group, with no significant difference observed (p = 0.22). SBRT is an independent positive prognostic factor for PFS in advanced EGFRm NSCLC. EGFR exon 19 deletion mutation (16.33 vs 11.55 months, p = 0.0087) and fewer metastases (0–5) (31.94 vs 9.59 months, p = 0.0059) were associated with improved PFS in EGFR-TKIs + SBRT versus EGFR-TKIs. Combination therapy increased radiation pneumonitis mainly in Grades 1–2 (89.74% vs 0.0%). The EGFR-TKIs + SBRT group mainly had new site failure (57.10% vs 32.10%) rather than the original site failure.
Conclusion:
Early SBRT for primary lung tumors may overcome targeted resistance in advanced EGFRm NSCLC patients combined with EGFR-TKIs without serious toxicities, especially for EGFR exon 19-del.
Trial registration:
ChiCTR-OIN-17013920.
Keywords
Introduction
Lung cancer is a prevalent and highly fatal malignant disease globally, with a significant incidence and mortality rate. 1 Non-small-cell lung cancer (NSCLC) constitutes around 80% of all lung cancer cases, and epidermal growth factor receptor (EGFR) mutations are identified in approximately 45%–55% of NSCLC cases within the Chinese population. 2 Tyrosine kinase inhibitors (TKIs) are considered the standard therapy for advanced lung cancer patients with EGFR mutations (EGFRm), which have been shown to greatly enhance patient survival of TKIs. First-generation targeted drugs, such as icotinib, erlotinib, and gefitinib, have been shown to achieve 9–13 months of progression-free survival (PFS) in advanced NSCLC patients with EGFRm. 3 The mechanisms of resistance to the first-generation EGFR-TKIs are diverse and not yet fully understood, including EGFR-dependent signaling pathway mechanisms, EGFR-independent signaling pathway mechanisms, and small-cell transformation mechanisms, among which the EGFR p.T790M mutation is the most common resistance mechanism.4,5 The initial advancement in locations where the disease originated (primary/metastatic) was categorized as the original site failure (OF), accounting for 50% of patients. The increased diameter of the primary tumor in the lung was more strongly associated with the incidence of OF. 6 The lung was the most common site of initial progression.7,8 Reducing the occurrence rate of OF and prolonging PFS of the first-generation EGFR-TKI treatment in NSCLC is a matter worthy of attention.
Stereotactic body radiation therapy (SBRT) plays a crucial role in the treatment of NSCLC, owing to its attributes of delivering high doses, minimal segmentation, and exceptional precision. These distinctive features contribute significantly to the efficacy and impact of SBRT as a treatment modality for NSCLC. Several studies have demonstrated that in elderly patients with early-stage lung cancer who decline surgery, the 5-year survival rate associated with SBRT treatment is comparable to that of surgical interventions. These findings suggest that SBRT can serve as a viable alternative for patients who are unsuitable for or refuse surgery, providing comparable long-term survival outcomes.9,10 Local treatment improves survival in patients with oligoprogressive and oligometastatic lung cancer, prolonging PFS and OS compared to maintenance treatment. 11 In advanced EGFRm NSCLC, local radiotherapy intervention can extend the PFS of oligoprogressive patients by approximately 6 months. 12 In vitro and in vivo studies have shown that radiotherapy can sensitize EGFR-TKI treatment. Li et al. 13 conducted a study that revealed a statistically significant decrease in the half maximal inhibitory concentration (IC50) of TKIs therapy in NSCLC cell lines harboring EGFR p.T790M mutation when combined with radiation, compared to the cell lines without radiation. These findings suggest that local radiotherapy intervention has the potential to reduce TKI resistance in these cell lines.
Therefore, this study was developed and aimed to explore the clinical value of early SBRT combined with EGFR-TKIs on primary lung tumors in advanced EGFRm NSCLC patients.
Materials and methods
Study design and patient selection
This pilot observational study included a total of 39 patients enrolled in the registered clinical trial phase II trial (registration number: ChiCTR-OIN-17013920 and the date of registration date: December 14, 2017). In addition, clinical information from 145 advanced NSCLC patients harboring EGFRm, collected from our hospital between August 2016 and December 2019, was included in the analysis. The key eligibility criteria for the study participants were as follows: (1) age ⩾18 and ⩽85 years; (2) histological diagnosis of stage IIIB–IV NSCLC; (3) Eastern Cooperative Oncology Group performance status of 0 or 1; (4) harboring specific EGFRm (exon 21 p.L858R or exon 19 deletion); (5) patients with brain metastases were eligible if they were asymptomatic or completed treatment for brain metastases, such as whole-brain radiation or stereotactic radiosurgery, at least 14 days prior to starting study treatment; and (6) individuals who achieved partial response (PR) or stable disease (SD) after 1-month EGFR-TKI treatment according to the Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1). Major exclusion criteria included a history of previous thoracic radiotherapy or prior TKI therapy, apart from the study treatment. The first participant was enrolled on August 24, 2016. The reporting of this study followed the principles of the ESMO Guidance for Reporting Oncology Real-World Evidence (GROW). The research protocol received approval from the Taizhou Hospital Ethics Committee. Individual consent for this retrospective analysis was waived.
Treatment
The patients in the study were divided into two groups: the EGFR-TKIs treatment group and the EGFR-TKIs combined with the SBRT (EGFR-TKIs + SBRT) group. Patients in the EGFR-TKIs group were administered a first-generation EGFR-TKI (icotinib 125 mg orally three times a day or gefitinib 250 mg orally once daily) until disease progression or death. This group was exclusively treated with EGFR-TKIs.
In the EGFR-TKIs + SBRT group, SBRT was initiated approximately 1 month after starting the oral EGFR-TKI treatment until disease progression or death. The total radiation dose administered during SBRT ranged from 40 to 60 Gy, delivered in 5–8 fractions.
Assessment and endpoints
Tumor imaging was conducted at the beginning of the study (baseline), 1 month after treatment initiation, and then every 3 months afterward. The assessment of tumor response was based on RECIST v1.1 criteria.
According to RECIST v1.1 criteria, a PR was defined as a reduction of at least 30% in the sum of the longest diameter of the target lesions, as measured by computed tomography, compared to the baseline measurement. Progressive disease (PD) was defined as an increase of at least 20% in the sum of the maximum diameters of the target lesions or the appearance of one or more new lesions following treatment initiation. SD was defined as a status that falls between PR and disease progression.
Pattern failure models, as described in Al-Halabi et al., 6 were classified as OF, including progression in initial primary or metastatic lesions, or new site failure (NF). Simultaneous occurrence of OF/NF was labeled as original and new site failure (ONF).
Adverse effects (AEs) were monitored during treatment and for a specified duration afterward. AEs included nausea, vomiting, diarrhea, rash, paronychia, increased liver enzymes, increased creatinine, and radiation pneumonia. The severity of AEs was graded using the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. Survival was assessed every 4 weeks during follow-up.
The primary endpoint of the study was PFS, which measured the time from EGFR-TKI treatment to either disease progression or death. Secondary endpoints were pattern failure models, AEs, and OS, which measured the time from the initiation of EGFR-TKIs treatment to death.
Statistical analysis
All statistical analyses were conducted using the Statistical Package for the Social Sciences version 20.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics, including median (minimum and maximum), were used to summarize the median age, follow-up time, and interval time from EGFR-TKI treatment to SBRT intervention. Kaplan–Meier curves were employed to analyze the PFS and OS rates. Differences between groups were evaluated using the log-rank test. Hazard ratios (HRs) and corresponding 95% confidence intervals (CIs) were calculated. The univariate and multivariate characteristics of PFS and OS were assessed using a Cox proportional hazard model. A significance level of p < 0.05 was used to determine statistical significance, and all tests were two-sided.
Results
Patient characteristics
A total of 184 patients with advanced NSCLC harboring EGFRm were included in this study, including 145 patients in the EGFR-TKIs group and 39 patients in the EGFR-TKIs + SBRT group. There were no significant differences in clinical characteristics between the two groups (Table 1, Figure 1).
Baseline characteristics of patients.

ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor; PR, partial response; SBRT, stereotactic body radiation therapy; SD, stable disease.

Flow chart of patient enrollment.
Efficacy
The median follow-up times for EGFR-TKIs + SBRT and EGFR-TKIs groups were 27.20 (6.17–61.23) and 25.37 months (4.90–69.57), respectively (p = 0.21). The median time on EGFR-TKI induction (prior to SBRT) was 1.27 months (0.73–2.47). Before the radiotherapy, the median lung primary tumor size is 2.28 cm (0.76–5.32). The median PFS was 15.50 months in the EGFR-TKIs + SBRT group, significantly longer than the 9.33 months in the EGFR-TKIs group (HR = 0.57, 95% CI: 0.41–0.78, p = 0.002). However, the median OS did not show a significant difference between the two groups (29.10 vs 26.33 months, HR = 0.76, 95% CI: 0.51–1.14, p = 0.22) (Figure 2).

Overall PFS (a) and OS (b) were compared by the Kaplan–Meier methods between patients who underwent SBRT or not.
In EGFR exon 21 p.L858R subgroup, the median PFS was 12.53 months in the EGFR-TKIs + SBRT group and 8.03 months in the EGFR-TKIs group (HR = 0.66, 95% CI: 0.41–1.05, p = 0.11) (Figure 3(a)), and the median OS was 22.54 versus 24.10 months (HR = 1.14, 95% CI: 0.63–2.07, p = 0.65) (Figure 3(b)). In EGFR exon 19 deletion subgroup, the median PFS of the EGFR-TKIs + SBRT group versus EGFR-TKIs group was 16.33 versus 11.55 months (HR = 0.51, 95% CI: 0.33–0.80, p = 0.0087) (Figure 3(c)), and the median OS was 49.70 versus 29.67 months (HR = 0.55, 95% CI: 0.31–0.97, p = 0.073) (Figure 3(d)).

Subtype analysis of PFS and OS was compared by the Kaplan–Meier methods between patients who underwent SBRT or not. SBRT. (a) PFS of patients with L858R mutation in EGFR-TKIs + SBRT group and EGFR-TKIs group. (b) OS of patients with L858R mutation in EGFR-TKIs + SBRT group and EGFR-TKIs group. (c) PFS of patients with a 19-del mutation in EGFR-TKIs + SBRT group and EGFR-TKIs group. (d) OS of patients with a 19-del mutation in EGFR-TKIs + SBRT group and EGFR-TKIs group. (e) PFS of patients with fewer metastasis (0–5) in EGFR-TKIs + SBRT group and EGFR-TKIs group. (f) OS of patients with fewer metastasis (0–5) in EGFR-TKIs + SBRT group and EGFR-TKIs group. (g) PFS of patients with multi-metastasis (>5) in EGFR-TKIs + SBRT group and EGFR-TKIs group. (h) OS of patients with multi-metastasis (>5) in EGFR-TKIs + SBRT group and EGFR-TKIs group.
For patients with fewer number of metastases (number of metastases: 0–5) in EGFR-TKIs + SBRT, the median PFS was 31.94 versus 9.59 months in the EGFR-TKIs group (HR = 0.26, 95% CI: 0.12–0.56, p = 0.0059), while the median OS was undefined versus 30.57 months (HR = 0.27, 95% CI: 0.10–0.73, p = 0.057) in the two groups (Figure 3(e) and (f)). There was no significant difference in median PFS or OS between the EGFR-TKIs group and the EGFR-TKIs + SBRT group in terms of multiple metastases (number of metastases: >5) (Figure 3(g) and (h)).
Furthermore, the Cox regression multivariable analysis (Tables A1 and A2) showed that SBRT was a significantly independent positive predictor of better PFS, with an HR of 0.64 (95% CI: 0.43–0.94, p = 0.021), instead of OS (HR of 0.87, 95% CI: 0.56–1.36, p = 0.55). Oligometastatic lesions were found to independently predict PFS in advanced lung cancer patients. In addition, EGFR exon 19 deletion was identified as an independent predictive factor for both PFS and OS in patients with advanced lung cancer.
Safety
AEs are summarized in Table 2. The addition of thoracic SBRT to TKIs for advanced NSCLC patients with EGFRm was well tolerated without severe toxicities. There were no ⩾grade 3 toxicities in either cohort. The incidence of radiation pneumonia was significantly increased in the EGFR-TKIs + SBRT group compared to the EGFR-TKIs group (89.74% vs 0%), but the incidents were mainly of Grade 1–2. The most common AE was rash, and there was no difference between the two groups in terms of rash, nausea and vomiting, diarrhea, paronychia, elevated aminotransferase, or elevated creatinine (all p > 0.05).
Adverse events.

EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor; N/A, not applicable; SBRT, stereotactic body radiation therapy.
Sites of initial failure and second genetic testing
As shown in Table 3, a total of 137 patients in the EGFR-TKIs group had disease progression, mainly in OF, and the initial progression patterns of OF, NF, and ONF were 46.0%, 32.1%, and 21.9%, respectively. By contrast, a total of 35 patients in the EGFR-TKIs + SBRT group had disease progression, mainly in NF, and the initial progression patterns of OF, NF, and ONF were 14.3%, 57.1%, and 28.6%, respectively (p = 0.002).
Pattern of initial failure.
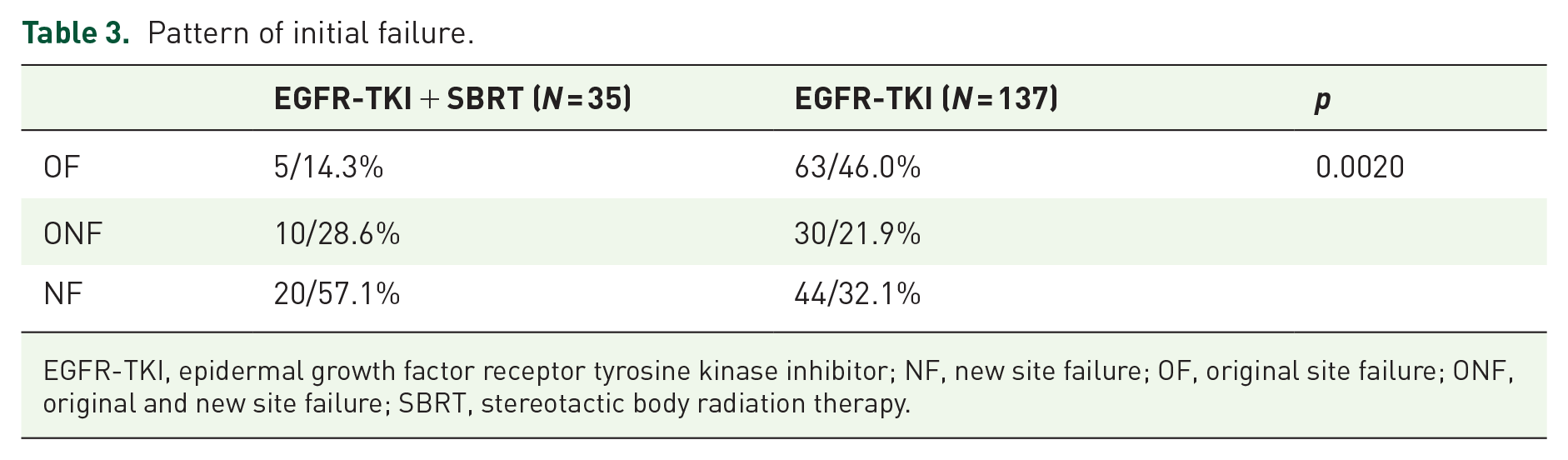
EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor; NF, new site failure; OF, original site failure; ONF, original and new site failure; SBRT, stereotactic body radiation therapy.
As shown in Figure A1, 53.10% (77/145) of patients in the EGFR-TKIs group and 65.71% (23/35) of patients in the EGFR-TKIs + SBRT group underwent secondary gene testing after the initial progression (p = 0.34). There were 66.23% (51/77) versus 86.96% (20/23) patients with EGFR p.T790 M positivity in the two groups (p = 0.068).
Discussion
This was a meaningful study to elucidate the value of early SBRT in advanced NSCLC patients harboring EGFRm with a relatively large sample size. SBRT combined with EGFR-TKIs was proven to prolong the median PFS with tolerated side effects, especially for those with EGFR exon 19 deletion. Although this combination therapy led to a significant increase in the incidence of radiation pneumonitis in advanced EGFRm NSCLC patients, as compared to EGFR-TKIs alone, it is important to note that the majority of AEs observed were of Grade 1–2 severity. OF was mainly observed in EGFR-TKIs-treated NSCLC while NF mainly occurred in the EGFR-TKIs + SBRT group, suggesting the failure model transfer.
The EGFR-TKIs + SBRT group demonstrated a significantly longer median PFS compared to the EGFR-TKIs group (15.50 vs 9.33 months, HR = 0.57, p = 0.0020). However, the inclusion of early SBRT did not lead to improved median OS in advanced NSCLC with EGFRm (29.10 vs 26.33 months, p = 0.22). In line with our findings, Wang et al. 14 proved that EGFR-TKIs plus thoracic SBRT significantly extended PFS compared with EGFR-TKIs monotherapy (19.4 s vs 13.7 months, p = 0.034), while the impact on OS has not been established yet (p = 0.557). Another recent real-world study demonstrated that administering SBRT to the primary tumor at the time of maximal response of EGFR-TKIs in certain patients with EGFRm non-oligometastatic NSCLC resulted in a significantly improved median PFS1 compared to the delayed radiotherapy (RT) group, where RT was administered at the occurrence of oligoprogression (22.3 vs 12.9 months, p = 0.0031). 15 However, there was no significant difference in OS between these two groups (46.6 vs 51.3 months, p = 0.54).
Recent studies have indicated that the addition of local consolidation therapy along with SBRT may enhance both PFS and OS in patients with oligometastatic and oligoprogressive EGFRm NSCLC.16,17 Consistent with our results, subgroup analysis revealed that in patients with fewer metastases (0–5), superior PFS (31.94 vs 9.59 months, p = 0.0059) and extended OS (immature vs 30.57 months, p = 0.057) were exhibited in the EGFR-TKIs + SBRT group compared with the EGFR-TKIs group. However, in the multi-metastases group, SBRT intervention did not have a significant impact on PFS or OS (13.97 vs 9.33 months, p = 0.10, and 27.90 vs 25.37 months, p = 0.97, respectively). Regarding genetic analysis, in the subgroup with EGFR exon 19 deletion, the EGFR-TKIs + SBRT group showed superior PFS compared to the EGFR-TKIs group (16.33 vs 11.55 months, HR = 0.51, p = 0.0087), and there was a noticeable trend toward improved OS in the EGFR-TKIs + SBRT group, although the difference was not statistically significant (p = 0.073). However, in the EGFR exon p.L858R mutation group, SBRT administration did not improve either PFS or OS compared to monotherapy treatment with TKIs. Consistent with the results of previous studies, this may be related to various factors, such as the structural characteristics of EGFR exon p.L858R, biological characteristics, and incorporation of other mutations. Perhaps EGFR-TKIs combined with chemotherapy (NEJ009 18 ) and EGFR-TKIs combined with antiangiogenic agents (NEJ026 19 and CTONG1509 20 ) may be better for those with EGFR exon p.L858R. Based on this scenario, the selected population who are more suitable for upfront SBRT combined with EGFR-TKI treatment still needs to be further explored and validated in larger and prospective clinical trials.
Previous studies have generated conflicting results regarding the optimal site for local intervention with SBRT in advanced EGFRm NSCLC patients. While many studies have focused on analyzing the effects of radiotherapy on all oligometastases or all lesions in the lung, the benefits of local intervention in the primary lesions are undeniable. For instance, Peng et al. 7 reported the results of radiotherapy combined with targeted therapy in the EGFRm oligometastatic patients treated with at least 3 months of EGFR-TKIs. The PFS of the combined therapy group and EGFR-TKIs group were 17.4 and 8.9 months, respectively (p = 0.042), indicating a delay in targeted resistance with the combined therapy. Moreover, they found that the PFS was associated with the radiation field, with the primary irradiation group exhibiting a PFS of 21.8 months, the metastatic irradiation group showing a PFS of 10.6 months, and the group receiving irradiation for both the primary and metastatic lesions having a PFS of 18.3 months. This suggested that primary tumor irradiation significantly delayed targeted resistance. Similarly, Xu et al. 8 reported that in the oligometastatic EGFRm NSCLC, superior PFS and OS of all lesions treated with consolidation therapy were observed compared with the partial consolidation therapy or no consolidation therapy group (p < 0.001), and consolidation therapy for all lesions has the greatest survival benefit on combination therapy. They also observed that the OS in the primary tumor intervention group and the primary tumor without intervention group were 40.5 and 31.5 months (p < 0.001), respectively, indicating that the consolidation of the primary tumor had survival benefits for patients. Encouragingly, our study investigated the efficacy of SBRT specifically on intrapulmonary primary tumors in patients with advanced EGFRm NSCLC, and the results were positive, suggesting a decrease in normal tissue damage along with therapeutic benefits.
Meanwhile, this study also analyzed the pattern of initial disease progression in patients with advanced EGFRm NSCLC treated with SBRT combined with EGFR-TKIs, and the results showed that EGFR-TKIs combined with SBRT significantly reduced the occurrence of primary metastases (OF: 14.30% vs 46.0%) and had more incidence of NF (57.1% vs 32.1%), demonstrating that SBRT has a good ability of local control. In addition, the second genetic testing after initial progression showed that there was no significant difference in EGFR p.T790M mutation between EGFR-TKIs + SBRT treatment and EGFR-TKIs monotherapy (86.96% vs 66.23%, p = 0.068). SBRT in this study may prolong the PFS of patients with advanced EGFRm NSCLC by reducing the resistance of primary lung tumors in the lung. However, Wang et al. 14 reported that the frequency of the EGFR p.T790M mutation seemed to increase in patients treated with TKIs and additional thoracic SBRT. The underlying mechanism of SBRT combined with EGFR-TKIs still needs to be explored, and multiple groups of irradiation fields in prospective studies should be established.
On the other hand, the optimal timing of radiotherapy intervention during EGFR-TKI treatment remains unclear. For brain oligometastasis, oncologist experts agreed that the earlier the SRS or surgical local treatment, the better the prognosis of symptomatic NSCLC patients. For intrapulmonary lesions, many studies believe that the optimal intervention time is at the time of maximum tumor response, but the best tumor response time is often difficult to determine. Therefore, more researchers believe and propose that local intervention at the beginning of TKI treatment or before the emergence of drug resistance can not only eliminate tumor heterogeneous clonal cells and reduce tumor dissemination but also reduce radiation damage to normal lung tissue by reducing tumor burden. A retrospective analysis of 105 samples showed that the tumor response appeared approximately 2 months after EGFR-TKI treatment. 21 Wu et al. 22 proposed that the average time of tumor response was also approximately 7.4 weeks. Similarly, there were 24 cases of PR and 15 cases of SD in the EGFR-TKIs + SBRT group after one-month administration of EGFR-TKIs, and the median time between SBRT intervention and targeted therapy was 1.27 months (0.73–2.74).
According to the theory of tumor clonal evolution, changes in the internal and external tumor environment cause tumor cells sensitive to TKI treatment to gradually die, and the residual clonal tumor cells gradually increase in the main clonal tumor cells, which results in TKI resistance. Therefore, researchers propose that earlier local intervention in advanced EGFRm patients before the emergence of drug resistance in tumors may delay the time of EGFR-TKIs resistance and maximize the survival benefit of patients.23,24 SBRT combined with EGFR-TKIs may not only effectively exert local tumor control and prolong PFS in patients with advanced NSCLC but also reduce tumor burden and decrease normal tissue damage in patients. 25 However, a lack of prospective clinical evidence on the optimal time of early SBRT local intervention in advanced EGFRm NSCLC patients is an urgent problem. Zheng et al. found that radiotherapy of all lung lesions (all metastases ⩽10) within 2 weeks after oral EGFR-TKIs has a good control effect on stage IV NSCLC patients with EGFRm. PFS was approximately 13 months, and the toxicity and side effects were well tolerated. 21
However, there were some shortcomings in this study. First, this study was a single-center, unrandomized study with a small number of patients, particularly in the EGFR-TKIs + SBRT groups. The results of this study will need to be verified by a multicenter phase III prospective project with a larger sample size. Second, this study reported the initial progression pattern of therapies but failed to analyze the related risk factors for the failure pattern in depth. Finally, the effective intervention sites and optimal intervention time of SBRT for delaying EGFR-TKI resistance in advanced EGFRm NSCLC patients need to be established as a multigroup control for further exploration and verification.
Conclusion
In summary, the combination of early SBRT with EGFR-TKI therapy has demonstrated feasibility and safety in delaying EGFR-TKI resistance in patients with advanced EGFRm NSCLC. In addition, this combined treatment approach has shown the ability to change the failure pattern from OF failure to NF failure, indicating a favorable local control effect of SBRT. Future research focusing on the role of SBRT in the context of osimertinib treatment for advanced EGFRm NSCLC is warranted. It is worth mentioning that a phase III clinical trial (NCT03727867) has been developed by our investigators to further investigate this area.
Footnotes
Appendix A
Univariate and multivariate analyses of factors associated with OS.
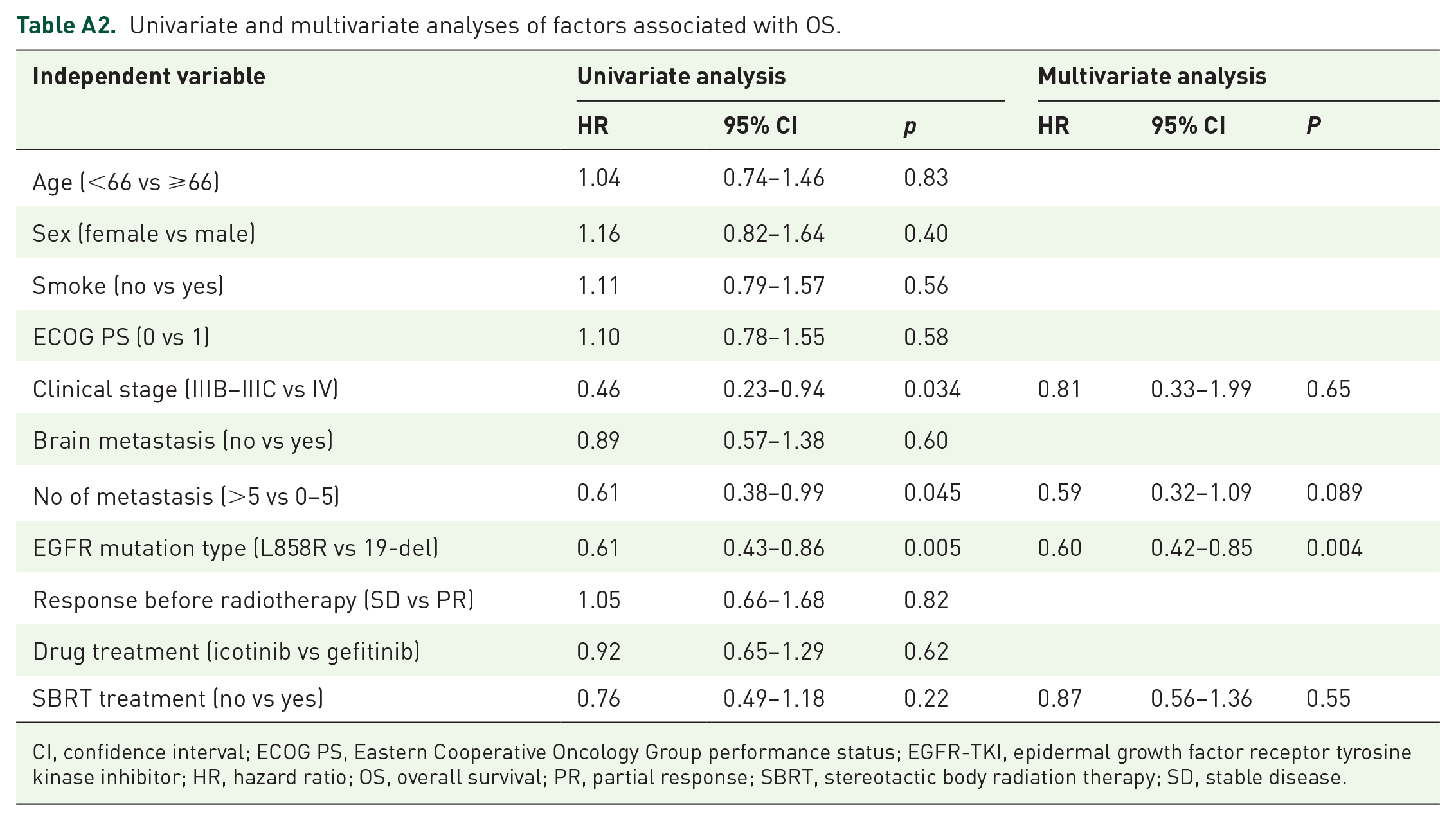
| Independent variable | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | P | |
| Age (<66 vs ⩾66) | 1.04 | 0.74–1.46 | 0.83 | |||
| Sex (female vs male) | 1.16 | 0.82–1.64 | 0.40 | |||
| Smoke (no vs yes) | 1.11 | 0.79–1.57 | 0.56 | |||
| ECOG PS (0 vs 1) | 1.10 | 0.78–1.55 | 0.58 | |||
| Clinical stage (IIIB–IIIC vs IV) | 0.46 | 0.23–0.94 | 0.034 | 0.81 | 0.33–1.99 | 0.65 |
| Brain metastasis (no vs yes) | 0.89 | 0.57–1.38 | 0.60 | |||
| No of metastasis (>5 vs 0–5) | 0.61 | 0.38–0.99 | 0.045 | 0.59 | 0.32–1.09 | 0.089 |
| EGFR mutation type (L858R vs 19-del) | 0.61 | 0.43–0.86 | 0.005 | 0.60 | 0.42–0.85 | 0.004 |
| Response before radiotherapy (SD vs PR) | 1.05 | 0.66–1.68 | 0.82 | |||
| Drug treatment (icotinib vs gefitinib) | 0.92 | 0.65–1.29 | 0.62 | |||
| SBRT treatment (no vs yes) | 0.76 | 0.49–1.18 | 0.22 | 0.87 | 0.56–1.36 | 0.55 |
CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor; HR, hazard ratio; OS, overall survival; PR, partial response; SBRT, stereotactic body radiation therapy; SD, stable disease.
Acknowledgements
The authors sincerely thank the patients and their families, investigators, all employees, and the institution involved in this study.
