Abstract
Background:
Claudin-18 isoform 2 (CLDN18.2) is expressed in multiple cancers and is a promising target for antitumor therapy. However, there is limited knowledge regarding the prevalence and characteristics of CLDN18.2-positive colorectal cancer (CRC).
Objectives:
To determine the clinicopathological and molecular features of patients with CLDN18.2-positive CRC.
Design:
Single-center retrospective study.
Methods:
A total of 805 patients who underwent surgical resection for pathological stage I
Results:
Among these patients, 17 (2.1%) had CLDN18.2-positive CRC. Right-sided CRC was significantly more common in patients who were CLDN18.2 positive than in those who were CLDN18.2 negative (76.5% vs 28.3%, p < 0.0001), as was mucinous or poorly differentiated adenocarcinoma (17.6% vs 3.0%; 17.6% vs 2.2%, p < 0.0001), T3–4 disease (100% vs 84.3%, p = 0.075), lymphatic invasion (64.7% vs 24.2%, p < 0.0001), BRAF V600E mutation (29.4% vs 4.1%, p < 0.0001), and deficient mismatch repair (MMR) status (47.1% vs 10.0%, p < 0.0001). Multivariate analysis did not identify CLDN18.2 expression status to be an independent predictor of relapse-free survival (RFS) or overall survival (OS).
Conclusion:
Approximately 2% of all CRC cases in this study were CLDN18.2 positive and had unfavorable features (e.g., mucinous or poorly differentiated adenocarcinoma, T3–4 disease, lymphatic invasion, BRAF V600E mutation) and deficient MMR status. CLDN18.2 positivity did not have a significant impact on RFS or OS.
Introduction
Colorectal cancer (CRC) is a major leading cause of death from cancer worldwide. 1 The standard treatment for resectable CRC is radical resection followed by adjuvant therapy if indicated.2,3 Patients with unresectable CRC undergo chemotherapy with or without molecular-targeting agents based on molecular features, such as the presence of BRAF V600E mutation 4 or RAS mutation,5,6 and DNA mismatch repair (MMR) status.7,8 However, the prognosis of unresectable CRC is poor, with a 5-year survival rate of less than 20%. 9 Therefore, the development of novel therapeutic agents that can improve survival in patients with unresectable CRC is eagerly awaited.
Claudin-18 isoform 2 (CLDN18.2) is a subtype of claudin-18 that belongs to a family of tight junction proteins involved in multiple processes, including control of paracellular permeability. 10 CLDN18.2 is recognized for its high expression in gastric epithelial cells, and its expression in gastric cancer cells has also been documented. 10 Therefore, CLDN18.2 has been attracting attention as a biomarker. Zolbetuximab, the first-in-class CLDN18.2-targeted antibody, improved overall survival (OS) when added to chemotherapy in patients with CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma in the phase III SPOTLIGHT and GLOW trials.11,12 CLDN18.2 expression is also observed in other types of cancers, including those of the pancreas, biliary tract, and lungs.13 –16 Development of CLDN18.2-targeted treatment is ongoing in patients with advanced solid malignancies and includes a monoclonal antibody (ClinicalTrials.gov identifier: NCT04400383), bispecific antibodies (ClinicalTrials.gov identifier: NCT05482893 and NCT04900818), an antibody–drug conjugate (ClinicalTrials.gov identifier: NCT05009966), and CAR T cells (ClinicalTrials.gov identifier: NCT03874897).
The CLDN18.2 positivity rate in CRC has been reported to range from 0.9% to 14%.16,17 –19 Previous reports have shown an association between the expression of CLDN18.2 and the expression of MUC5AC.17,18 However, the prevalence and clinicopathological features of CLDN18.2-positive CRC have not been investigated in large-scale studies. Furthermore, little is known about the clinically relevant molecular features of CLDN18.2-positive CRC.
Therefore, in this study, we sought to determine the clinicopathological and molecular features of CLDN18.2-positive CRC and the prevalence of CLDN18.2 positivity in surgically resected CRC samples.
Materials and methods
Clinical specimens
Tissue microarrays were constructed as previously reported, 20 using surgical specimens from all patients treated for pathological stage I–III CRC at the National Cancer Center Hospital (Tokyo, Japan) between 1997 and 2019. The reporting of this study conforms to the ESMO Guidance for Reporting Oncology Real-World evidence. 21
Clinicopathological information
We retrospectively reviewed the medical records of all patients treated during the study period and collected data on baseline demographics (sex and age), pathological findings (tumor location, histological subtype, mucinous component, pathological stage, resection margin status, lymphovascular invasion), perioperative chemotherapy (neoadjuvant and adjuvant therapy), and patient outcomes (relapse-free survival (RFS) and OS). KRAS, NRAS, and BRAF V600E mutation status was determined by PCR assay as previously reported. 20 DNA MMR status was determined by immunohistochemistry of MMR proteins as previously reported. 20
The staging was based on the Union for International Cancer Control TNM classification (8th edition). Right-sided primary tumors were defined as those originating in the cecum, ascending colon, or transverse colon. Left-sided primary tumors were defined as those originating in the descending colon, sigmoid colon, rectosigmoid colon, or rectum. RFS was measured as the interval between the day of surgical resection for primary CRC and the first relapse of disease, all-cause mortality, or censored at the last date of confirmed survival without disease relapse. OS was calculated as the interval between the day of surgical resection and death from any cause or censored at the last date of confirmed survival.
Expression of CLDN18.2
We evaluated CLDN18.2 expression, using the CLDN18 antibody (Clone 43-14A; Roche Ventana, Oro Valley, AZ, USA) with the HISTO STAINER automated immunostainer (Nichirei Biosciences, Tokyo, Japan) according to the manufacturer’s protocol. Clone 43-14A was used as the primary antibody and incubated overnight at 4°C without dilution. Histofine® Simple Stain MAX-PO (MULTI) (Nichirei Biosciences, Tokyo, Japan) was used as a secondary antibody and incubated for 30 min at room temperature. A sample of normal gastric mucosa was used as a positive control. CLDN18.2 positivity was defined as the presence of moderate (complete membrane staining that is either non-uniform or weak but with obvious circumferential distribution) to strong (complete membrane staining) expression in at least 1% of the tumor cells.
Statistical analysis
Patients were divided into a CLDN18.2-positive group (⩾1%) and a CLDN18.2-negative group (<1%) based on the CLDN18.2 expression level. The associations of CLDN18.2 expression with patient characteristics, pathological findings, and perioperative chemotherapy were examined using the Chi-squared and Mann–Whitney U tests. OS and RFS were estimated using the Kaplan–Meier method, and the difference in survival after complete R0 resection of stage II or III CRC was compared according to CLDN18.2 status using the log-rank test. Multivariate analyses were performed using a Cox proportional hazard model to explore the prognostic significance of CLDN18.2 expression in terms of survival outcomes.
All statistical analyses were performed using SPSS version 22.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA). All tests were two-tailed, and the significance level was set at a p-value of 0.05.
Results
Expression of CLDN18.2
CLDN18.2 expression levels in the 805 patients are shown in Figure 1. A total of 17 (2.1%) patients showed expression of CLDN18.2 (⩾1%) and 788 (97.9%) did not (<1%). CLDN18.2 expression was ⩾75% in two patients (0.25%). Representative micrographs of staining patterns for CLDN18.2 expression in CRC are shown in Figure 2.

Expression of CLDN18.2 in patients with CRC.

Staining patterns for CLDN18.2 expression. Representative micrographs of staining patterns for (a) 0%, (b) 20%, (c) 60%, and (d) 90% CLDN18.2 expression in CRC (original magnification 40× and 100×).
Patient characteristics
Patient characteristics according to CLDN18.2 expression status are shown in Table 1. Right-sided colon cancer was significantly more common in patients with CLDN18.2-positive CRC than in those with CLDN18.2-negative CRC (76.5% vs 28.3%, p < 0.0001), as were the proportions with mucinous and poorly differentiated adenocarcinoma (17.6% vs 3.0% and 17.6% vs 2.2%, respectively, p < 0.0001) and the proportion with a high mucinous component (⩾10%) (47.1% vs 12.9%, p < 0.0001). T3–4 disease tended to be more common in patients with CLDN18.2-positive CRC than in those with CLDN18.2-negative CRC (100% vs 84.3%, p = 0.075). There were no cases of stage I disease among patients with CLDN18.2-positive CRC. Lymphatic invasion was significantly more common in patients with CLDN18.2-positive CRC than in those with CLDN18.2-negative CRC (64.7% vs 24.2%, p < 0.0001).
Patient characteristics.

CLDN18.2, claudin-18 isoform 2; CRC, colorectal cancer.
KRAS, NRAS, and BRAF V600E mutation and DNA MMR status
Table 2 shows the genetic (KRAS, NRAS, BRAF V600E) mutation status and DNA MMR status according to whether patients had CLDN18.2-positive CRC or CLDN18.2-negative CRC. The proportion of patients with KRAS or NRAS mutation was not significantly different between those with CLDN18.2-positive CRC and those with CLDN18.2-negative CRC (KRAS, 41.2% vs 41.9%, p = 0.95; NRAS, 5.9% vs 3.3%, p = 0.56). BRAF V600E mutation was significantly more common in patients with CLDN18.2-positive CRC than in those with CLDN18.2-negative CRC (29.4% vs 4.1%, p < 0.0001), as was deficient MMR status (47.1% vs 10.0%, p < 0.0001).
KRAS, NRAS, and BRAF V600E mutation status and DNA MMR status.

CLDN18.2, claudin-18 isoform 2; CRC, colorectal cancer.
Supplemental Table 1 shows the characteristics of the 17 patients with CLDN18.2-positive CRC. Lynch syndrome was confirmed in one patient. In these 17 patients with CLDN18.2-positive CRC, patients with deficient MMR status have a numerically higher proportion of harboring BRAF V600E mutation compared with those with proficient MMR status (50% vs 11.1%, p = 0.079) (Table 3).
Association of BRAF V600E mutation status with DNA MMR status in patients with CLDN18.2-positive CRC.
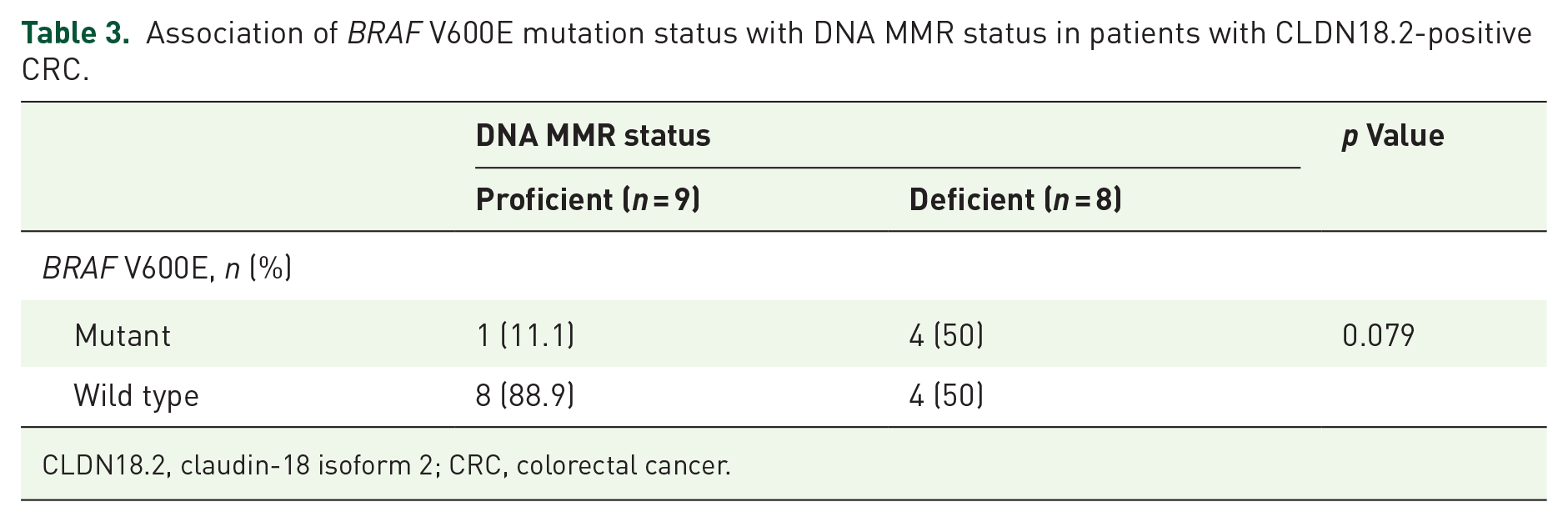
CLDN18.2, claudin-18 isoform 2; CRC, colorectal cancer.
RFS and OS
We compared RFS and OS in 706 patients in whom R0 resection was completed for stage II or III CRC according to whether CLDN18.2 status was positive or negative (Supplemental Table 2). There was no statistically significant difference in RFS and OS between patients with CLDN18.2-positive CRC and those with CLDN18.2-negative CRC (RFS, hazard ratio (HR) 1.02, 95% confidence interval (CI) 0.25–4.16, p = 0.98; OS, HR 2.22, 95% CI 0.41–11.9, p = 0.16) (Figure 3(a) and (b)).

Comparison of RFS and OS in patients with stage II or III CRC according to CLDN18.2 expression status. (a) There was no statistically significant difference in RFS according to whether CRC was CLDN18.2
We also compared RFS and OS in the stage II or III CRC population according to BRAF V600E mutation status. Among the patients with BRAF V600E mutation, there was no statistically significant difference in RFS and OS between patients with CLDN18.2-positive CRC and those with CLDN18.2-negative CRC (RFS, HR 1.68, 95% CI 0.13–22.6, p = 0.63; OS, HR 1.78, 95% CI 0.13–25.2, p = 0.59) (Supplemental Figure 1(A) and (B)). In the population without BRAF V600E mutation, there was also no statistically significant difference in RFS and OS between patients with CLDN18.2-positive CRC and those with CLDN18.2-negative CRC (RFS, HR 1.56, 95% CI 0.27–8.80, p = 0.53; OS, HR 2.48, 95% CI 0.28–21.8, p = 0.19) (Supplemental Figure 2(A) and (B)).
Prognostic significance of CLDN18.2
In multivariate analyses, male sex (p = 0.023), left-sided primary CRC (p = 0.043), stage III disease (p = 0.001), lymphatic invasion (p = 0.036), venous invasion (p = 0.009), and RAS mutation (p < 0.0001) were significantly associated with shorter RFS (Table 4), while male sex (p = 0.031), older age (p < 0.0001), stage III disease (p = 0.01), no adjuvant therapy (p = 0.007), and proficient MMR (p = 0.018) were significantly associated with shorter OS (Table 5). CLDN18.2 expression was not identified to be an independent predictor of RFS or OS in patients with CRC.
Multivariable analyses of RFS.

CI, confidence interval; CLDN18.2, claudin-18 isoform 2; HR, hazard ratio; MMR, mismatch repair; RFS, relapse-free survival.
Multivariable analyses of OS.

CI, confidence interval; CLDN18.2, claudin-18 isoform 2; HR, hazard ratio; MMR, mismatch repair; OS, overall survival.
Discussion
CLDN18.2 is emerging as a promising target in oncology. In this study, we investigated the prevalence of expression and clinicopathological features of CLDN18.2 in more than 800 patients with stage I–III CRC. Immunohistochemical analysis showed that the prevalence of CLDN18.2 was 2.1% for ⩾1% expression and 0.25% for ⩾75% expression, indicating that expression of this isoform is rare in CRC. CLDN18.2 expression was associated with unfavorable clinicopathological features, including mucinous or poorly differentiated adenocarcinoma, T3–4 disease, lymphatic invasion, right-sided origin, and BRAF V600E mutation.
Expression of CLDN18.2 may affect tight junction permeability, possibly increasing the diffusion of nutrients and extracellular growth factors that promote cancer cell growth, especially in the gastric mucosa.16,17 Our finding that CLDN18.2-positive CRC is rare is consistent with previous research.16,17 –19 However, the prevalence of CLDN18.2 expression in patients with CRC has not been investigated until now in large-scale studies, and the cutoff value for positivity was variable in previous reports.16,17 –19 The present study demonstrates the prevalence of CLDN18.2 in more than 800 patients with CRC, which is the largest population investigated thus far.
In our study, the CLDN18.2-positive population was associated with unfavorable clinicopathological features, including mucinous or poorly differentiated adenocarcinoma, T3–4 disease, and lymphatic invasion. Furthermore, our findings indicate an association between CLDN18.2 expression and BRAF V600E mutation. Moreover, a small number of patients with CLDN18.2-positive CRC had deficient MMR status without BRAF V600E mutation. CLDN18.2 expression was not identified as an independent prognostic factor for RFS and OS, which is in line with previous reports of CLDN18.2 not being a prognostic predictor in patients with pancreatic ductal adenocarcinoma22,23 and gastric cancer.24,25
It is hypothesized that ectopic CLDN18.2 expression in CRC is associated with a gastric phenotype. This hypothesis is supported by previous studies of CRC that found a positive correlation between CLDN18 and MUC5AC (a gastric marker mucin) expression and a negative correlation between CLDN18 and CDX2, which is a known factor in the transformation of intestinal stem cells into gastric stem cells.17,26,27 BRAF V600E mutation is associated with CDX2 inactivation. 28 Moreover, microsatellite instability-high CRC cases are reported to have a high frequency of CDX2 loss and expression of gastric-type markers such as MUC5AC and CLDN18. 29 These observations support our findings that CLDN18.2 expression is enriched in patients with BRAF V600E mutated or deficient MMR status.
Development of CLDN18.2-targeted treatment in patients with advanced solid tumors is ongoing. Drug development targeted for patients with CLDN18.2-positive CRC is also desirable, given the association with CLDN18.2 expression and unfavorable clinicopathological features. Although patients with CLDN18.2-positive CRC constitute a rare population, there may be a significant number of these patients, given that CRC is one of the most common cancers worldwide.
This study had some limitations. First, it had a single-center retrospective design and only included patients with pathological stage I–III CRC. Second, despite being a large study that included more than 800 patients, the number of CLDN18.2-positive patients was relatively small. Third, analysis based on tissue microarray might affect the evaluation of CLDN18.2 expression. Further research is required to determine the optimal threshold of CLDN18.2 expression for developing treatments targeting CLDN18.2 in CRC.
Conclusion
In this study, only about 2% of all CRC cases were CLDN18.2-positive. These CRC cases were associated with unfavorable features (e.g., mucinous or poorly differentiated adenocarcinoma, T3–4 disease, lymphatic invasion, BRAF V600E mutation) and deficient MMR status. Although patients with CLDN18.2-positive CRC are a rare population, they may be significant in number considering the high incidence of CRC. Therefore, our findings support the need for clinical development of a targeting strategy for CLDN18.2-positive CRC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-jpg-2-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-jpg-3-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-jpg-4-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-5-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-jpg-5-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-6-tam-10.1177_17588359241286774 – Supplemental material for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study
Supplemental material, sj-pdf-6-tam-10.1177_17588359241286774 for Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study by Shigemasa Takamizawa, Hidekazu Hirano, Atsuo Takashima, Hirokazu Shoji, Toshiharu Hirose, Natsuko Okita, Kouya Shiraishi, Shigeki Sekine, Yasuyuki Takamizawa, Yukihide Kanemitsu and Ken Kato in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
