Abstract
Background:
The emergence of new antileukemic drugs, including Bruton tyrosine kinase inhibitors (BTKis), phosphoinositide 3-kinase inhibitors (PI3Kis), and B-cell lymphoma 2 antagonists (BCL-2a), has significantly improved the outcomes for patients with relapsed/refractory chronic lymphocytic leukemia (CLL). Despite advances in treatment efficacy, the comprehensive safety profile of these novel agents versus traditional chemotherapy and immunotherapy has not been adequately explored, and there have been few direct comparisons.
Objectives:
This study aimed to compare the safety profiles of novel therapeutic agents, chemotherapy, and immunotherapy in patients with relapsed/refractory CLL using a Bayesian network meta-analysis (NMA).
Methods:
A systematic literature review was conducted to identify randomized clinical trials on relapsed/refractory CLL. The search encompassed major medical databases (MEDLINE, Embase, and CENTRAL) and gray literature, with the aim to integrate the findings into a Bayesian NMA framework for safety outcome assessment.
Design:
Systematic literature review with Bayesian NMA.
Results:
The systematic search identified 14 randomized trials that formed networks for the comparison of safety outcomes. No differences were shown between therapies in terms of overall adverse events (AEs). However, bendamustine + rituximab had a more favorable safety profile for grade ⩾3 AEs when compared with ibrutinib (risk ratio 0.62 (95% credible interval 0.40–0.86)), acalabrutinib (0.69 (0.45–0.94)), zanubrutinib (0.64 (0.42–0.91)), and venetoclax + rituximab (0.87 (0.79–0.96)). The frequency of grade ⩾3 AEs, serious AEs, and treatment discontinuations and deaths due to AEs was comparable between acalabrutinib, zanubrutinib, and venetoclax + rituximab. There were no significant differences in the safety profiles regarding hematological events, events affecting the quality of life, and infections for most comparisons of venetoclax + rituximab with BTKis. Among BTKi-specific events, zanubrutinib was associated with a higher risk of hypertension (2.96 (1.74–5.16)) and bleeding (1.38 (1.06–1.81)) than acalabrutinib. No differences in the risk of atrial fibrillation were found between acalabrutinib and zanubrutinib (1.56 (0.74–3.34)).
Conclusion:
Our findings showed that venetoclax + rituximab, acalabrutinib, and zanubrutinib have acceptable safety profiles, which indicates that they may be the preferred therapeutic options in the setting of relapsed/refractory CLL.
Trial registration:
PROSPERO CRD42022304330.
Keywords
Introduction
Chronic lymphocytic leukemia (CLL) is the most common leukemia in Western countries. 1 It affects mainly elderly patients with a median age at diagnosis of 70 years. 2 The disease is characterized by persistent monoclonal B-cell lymphocytosis, which often coexists with lymphadenopathy, hepatomegaly, and splenomegaly. Although CLL has an indolent nature with multiple relapses during the clinical course, its major complications, such as frequent infections, may lead to death. 3
A recent development and approval of antileukemic targeted therapies such as Bruton tyrosine kinase inhibitors (BTKis; ibrutinib, acalabrutinib, and zanubrutinib) and BCL2 antagonists (venetoclax) have been pivotal in improving survival in patients with relapsed/refractory CLL. Phase III clinical trials examining the benefit-risk ratio of ibrutinib (RESONATE 4 ), acalabrutinib (ASCEND 5 ), and venetoclax (MURANO 6 ) confirmed their superiority over standard immunotherapy or chemoimmunotherapy in relapsed/refractory CLL. While these advancements have significantly improved patient outcomes, they have also brought new challenges in terms of understanding the safety profiles of these therapies, especially in often heavily pretreated patients. Direct-comparison studies have reported different safety outcomes of these therapies, underlying the need for a comprehensive analysis to guide clinical decision-making.
For example, ibrutinib has been associated with a higher incidence of cardiovascular adverse events (AEs), including atrial fibrillation and hypertension, which may be important in certain populations. Idelalisib, a PI3K inhibitor (PI3Ki), has shown a tendency for immune-mediated toxicities such as pneumonitis and colitis. Venetoclax, a BCL2 antagonist, has been linked with tumor lysis syndrome, especially in high-risk patients. These differences in safety profiles, as highlighted by direct-comparison studies, complicate the choice of treatment in patients with relapsed/refractory CLL. Therefore, when deciding on the most appropriate therapeutic option for CLL, it is important to consider not only the efficacy of each therapy but also its safety profile.
Most studies directly comparing the safety profile of therapies have been limited in scope and have not included the full range of available therapeutic options. Therefore, a network meta-analysis (NMA) is necessary. Using an NMA, multiple treatments can be compared simultaneously (even in the absence of direct head-to-head trials) by creating a network of comparisons across a range of studies. This approach is particularly valuable in the setting of CLL, where multiple agents with different mechanisms of action have emerged.
In this study, we used a Bayesian NMA 7 to synthesize the available evidence from both direct and indirect comparisons with the aim to provide a more comprehensive understanding of the relative safety of various regimens for CLL, including novel agents, monoclonal antibodies, and chemotherapy. Our findings may have implications not only for clinicians navigating the complex treatment landscape, but also for patients seeking an effective and safe treatment, as well as for health policy makers looking to ensure the optimal allocation of healthcare resources. This study attempts to fill the existing knowledge gap and to clarify the relative safety of therapies used in patients with relapsed/refractory CLL.
Methods
A systematic review was conducted according to the 2020 Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines 8 (Supplemental File 1) and their extension for NMAs 9 to identify randomized clinical trials reporting safety outcomes to be included in the subsequent NMA. The protocol of the study was previously registered in the PROSPERO database (CRD42022304330). The data concerning the efficacy of therapies have already been published previously elsewhere. 10 This paper presents an extensive analysis focused solely on safety, covering a broader range of data that extends beyond the capacity suitable for inclusion in the previous publication.
Data sources and searches
A systematic search of electronic medical databases, including MEDLINE, Embase, and CENTRAL, was conducted initially on January 27, 2022, with the last update on October 10, 2023. Additionally, various sources of unpublished data were explored, including clinical trial registries (ClinicalTrials.gov, EU Clinical Trial Registry, and International Trials Registry Platform Search Portal), conference proceedings (American Society of Hematology, European Hematology Association, American Society of Clinical Oncology, and European Society of Medical Oncology), and the websites of regulatory (Food and Drug Agency and European Medicines Agency) and health technology assessment agencies (National Institute for Health and Care Excellence and German Federal Joint Committee). The references of the included full-text articles were reviewed to identify any additional relevant publications. Our search strategy was not limited to a specific date. Detailed search queries for each database were presented in Supplemental File 2 (Supplemental Table 1).
Study selection
We searched for clinical trials conducted in adult patients with relapsed/refractory CLL who had received at least one treatment line. Studies involving mixed populations (both previously treated and untreated) were included only if at least 80% of participants had relapsed/refractory CLL. The evaluated interventions, either as monotherapy or in combination with other drugs, included:
anti-CD20 monoclonal antibodies: obinutuzumab, ofatumumab, rituximab, ublituximab;
BTKis: acalabrutinib, ibrutinib, pirtobrutinib, zanubrutinib;
PI3Kis: idelalisib, duvelisib, umbralisib;
BCL2 antagonists: venetoclax;
other therapies: dinaciclib, lenalidomide.
Eligible comparators for our study included alternative monotherapies or drug combinations, any other active treatment, the best standard or supportive care, a placebo, or the therapy of the physician’s choice. To ensure the best quality of the NMA, we restricted our inclusion criteria to randomized clinical trials that were published in English. A study was considered for inclusion if it was available as a full-text article, report, or study protocol. Studies published only as conference abstracts were excluded to ensure that data analysis is comprehensive and reliable. The outcomes of interest focused on the general safety profile, including the rate of overall AEs, grade ⩾3 AEs, serious AEs (SAEs), AEs leading to treatment discontinuation, and AEs resulting in death. Additionally, we selected specific AEs based on a preliminary feasibility study conducted to assess the feasibility of the NMA. The specific AEs included overall and grade ⩾3 anemia, thrombocytopenia, neutropenia, diarrhea, nausea, fatigue, atrial fibrillation, hypertension, bleeding, infections, and pneumonia. The risk of major bleeding and secondary malignancies was also assessed.
Study selection and data extraction
Abstracts and full-text articles were independently screened by two reviewers (M.M. and M.R.), according to predefined inclusion and exclusion criteria (Table 1). Full-text articles were included only if both reviewers considered them eligible. Any disagreements between reviewers were resolved by consensus. Data were initially extracted by one reviewer (M.M.) using a prespecified form. Subsequently, they were verified for accuracy by another reviewer (M.R.). Discrepancies identified during this process were discussed and resolved by consensus. The following data were extracted from the included studies: study design, interventions, key inclusion criteria, and safety outcomes presented as the number and percentage of patients with a specific AE. Full-text articles and regulatory reports were treated as the primary source for data extraction. Data from other publications were extracted if new outcomes or data cut-offs were presented.
Detailed inclusion and exclusion criteria.

AE, adverse events; BCL2, B-cell lymphoma 2; CD20, cluster of differentiation 20; CLL, chronic lymphocytic leukemia; PI3K, phosphoinositide 3-kinase.
Data analysis and synthesis
NMAs were conducted using the Bayesian framework based on the Markov chain Monte Carlo method. The GeMTC package 11 for the R software was used. The results of the NMA were presented as risk ratios (RRs) with credible intervals (CrIs). Additionally, we calculated RRs for direct comparisons using the OpenMetaAnalyst (Brown University, Providence, RI, USA) software to check consistency between the NMA and individual studies. Node-splitting analyses were also conducted to assess inconsistencies within the networks.
We performed NMAs both as fixed and random models. For both models, the model parameters were set as follows: initial chains, 4; burn-in, 50,000; inference, 200,000; and thinning, 10. These settings were confirmed by the Brooks–Gelman–Rubin convergence diagnostics as optimal, with a potential scale reduction factor of ~1. When the difference in the deviance information criterion values between the fixed and random models exceeded 3, we opted for a model with lower deviance information criterion values because it provided a better fit with lower complexity. Conversely, if the difference was less than 3, we chose the fixed effect model, as it more accurately reflected the outcomes of primary studies without adding significant complexity. The results for alternative models were presented in the Supplemental Appendix. Rank probabilities for each intervention and outcome, along with their possible ranks, were calculated and summarized using the surface under the cumulative ranking probabilities (SUCRA).
Our primary NMAs included data for the longest available follow-up and treatment duration for each outcome. To examine the effect of treatment duration on the NMA results, we performed additional NMAs restricted to data for up to 2 years of therapy as sensitivity analyses, using the same model (fixed/random) as in the primary analysis. This allowed a more focused comparison between therapies used until the occurrence of unacceptable toxicity or disease progression and those with predefined treatment duration. Additionally, in cases where AEs occurred in all patients within a treatment arm or where there were no events reported, a correction of one event was applied.
Quality assessment
The risk of bias in all included studies was evaluated using RoB 2—a revised Cochrane risk of bias tool for randomized trials. 12 The tool allowed us to categorize the risk of bias as either low or high and to express certain concerns regarding trial design, execution, and reporting across five distinct domains. The overall risk score was based on the highest level of risk identified in any of these domains.
Results
Search results
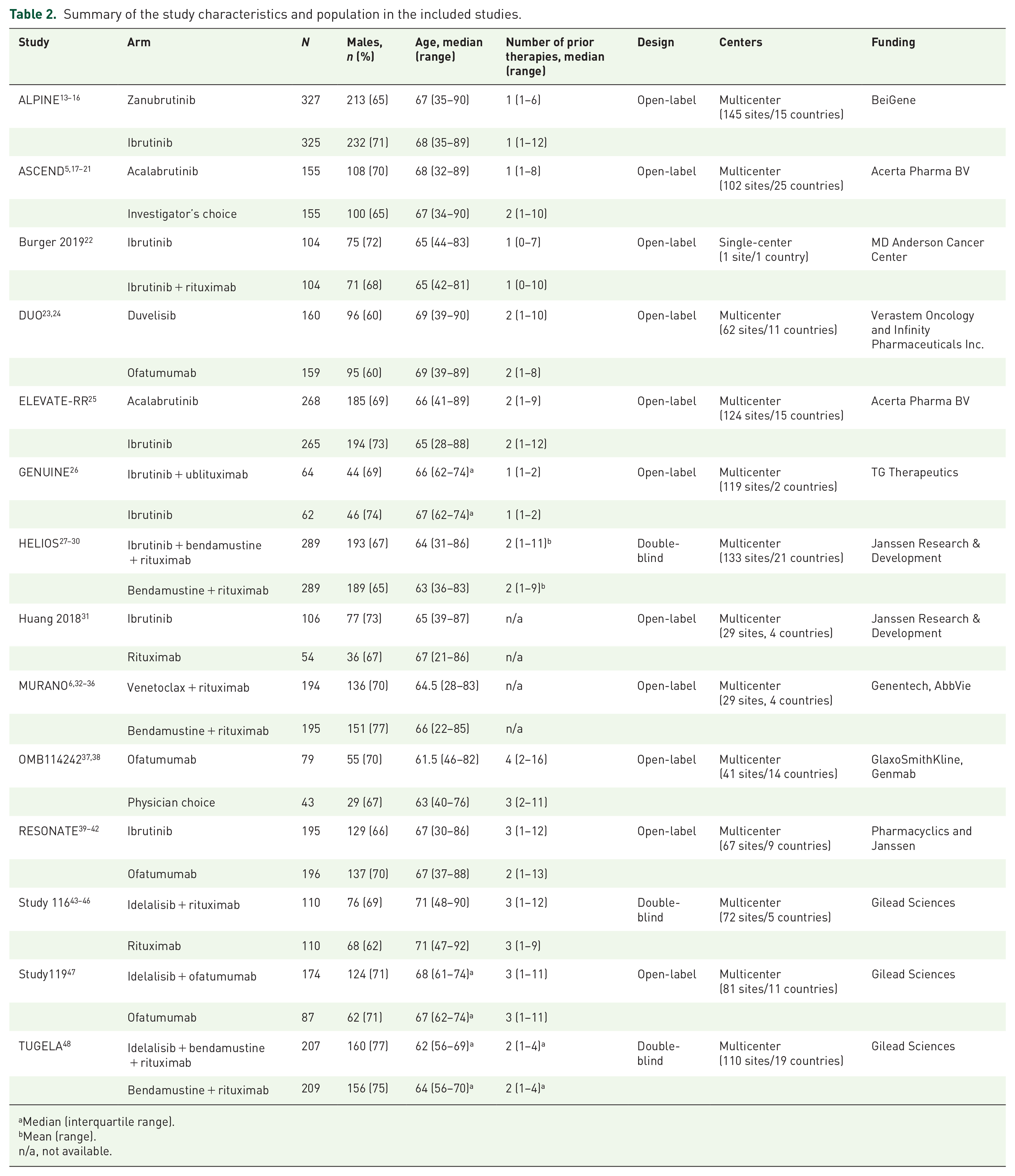
The systematic search identified 20 studies described in 206 articles and other reports that met our eligibility criteria for the systematic review. However, only 14 trials were included in qualitative analyses: ALPINE,13–16 ASCEND,5,17–21 Burger 2019, 22 DUO,23,24 ELEVATE-RR, 25 GENUINE, 26 HELIOS,27–30 Huang 2018, 31 MURANO,6,32–36 OMB114242,37,38 RESONATE,4,39–42 Study116,43–46 Study119, 47 and TUGELA 48 (Table 2). Four trials (CLLUmbrella2, 49 COMPLEMENT2,50,51 LUCID, 52 and REACH 53 ) were excluded because they lacked an arm connected to the network. Two additional studies were excluded because of the population: PN012 54 was a pilot study with a small sample size, while MaBLE 55 predominately involved treatment-naïve patients and did not report safety outcomes for the relapsed/refractory CLL population. In total, 29 individual reports from the included studies were extracted for the NMAs. The process of study selection and exclusion is shown in Figure 1.
Summary of the study characteristics and population in the included studies.
Median (interquartile range).
Mean (range).
n/a, not available.

PRISMA 2020 flow diagram.
Characteristics of included studies
Most studies included in our analysis were phase II or III open-label multicenter trials. There were three double-blind studies (HELIOS, Study116, TUGELA) and one single-center study (Burger 2019). Treatments mainly involved BTKis (ibrutinib, acalabrutinib, and zanubrutinib) and PI3Kis (idelalisib and duvelisib) used as monotherapy or in combination with anti-CD20 antibodies (ofatumumab, rituximab, and ublituximab) with or without bendamustine. The main comparators included immunotherapy, bendamustine + rituximab, or ibrutinib. One trial focused on the BCL2 antagonist venetoclax combined with anti-CD20 (venetoclax + rituximab) (Figure 2).

Network plot of the included studies.
Safety evaluations, consistent across most studies, were conducted on patients who received at least one dose of the study drugs. AEs were classified according to the National Cancer Institute Common Terminology Criteria for Adverse Events ver. 4.0x with information collected for up to 28–30 days after treatment discontinuation. The exception was the OMB114242 trial, which extended the AE-reporting period to 60 days and did not specify the AE classification used.
The median treatment duration varied across studies, ranging from 2.1 months for the physician’s choice (chemotherapy, immunotherapy, and/or steroids) in the OMB114242 trial to 65.6 for ibrutinib in the RESONATE trial. The median duration was largely determined by the planned duration of therapy. Immunotherapies and chemotherapies, such as ofatumumab, rituximab, or bendamustine + rituximab, had a shorter median duration (around 5–6 months), reflecting their fixed-cycle treatment regimens. In contrast, BTKis, like ibrutinib, were administered continuously until disease progression or unacceptable toxicity. The dosing of analyzed therapies was consistent for each regimen across different studies.
None of the included studies demonstrated a low risk of systematic error for safety outcomes. The evaluation identified either some concerns or a high risk of bias. The primary areas impacting the risk of bias assessment were the open-label design of trials, which could influence patients’ and researchers’ expectations regarding the reporting of AEs, and an insufficient description of reporting potential deviations from intended interventions, including the use of additional cointerventions or medications to counteract specific AEs. In the included studies, an as-treated analysis was used to assess safety-related outcomes. However, this approach did not significantly impact the risk of bias assessment, as in most cases, the as-treated analysis excluded only individual patients who did not receive a single dose of the medication. Moreover, patients in the as-treated groups received their therapies in accordance with their initial randomization. Detailed characteristics of the studies and risk of bias assessment are shown in the Supplemental Appendix (Supplemental Tables 2–8, Supplemental Figures 1 and 2).
Overall safety profile
Bendamustine + rituximab was identified as having the highest likelihood of being the least toxic regimen across various safety outcomes, as demonstrated by high SUCRA values for overall AEs (0.83), grade ⩾3 AEs (0.96), and SAEs (0.95). Of note, the probability of AE-related treatment discontinuation and death was the lowest for the physician’s choice (SUCRA: 0.94) and venetoclax + rituximab (SUCRA: 0.86), respectively. Conversely, duvelisib emerged as potentially the most toxic drug across most safety outcomes, except for AE-related treatment discontinuation, where the highest toxicity was shown for the combination of idelalisib + rituximab (SUCRA: 0.08) (Figure 3).

SUCRA values for each analyzed safety outcome.
A comparative analysis revealed that while there were no significant differences in overall AE rates between therapies (Table 3), bendamustine + rituximab showed significantly lower rates of grade ⩾3 AEs and SAEs than most treatments (Table 4), indicating its relative safety. The only exceptions were noted for the comparison with ibrutinib + bendamustine + rituximab (0.95 (0.88–1.03)), ofatumumab (0.74 (0.47–1.10)), rituximab (0.78 (0.50–1.11)), and the physician’s choice (0.83 (0.48–1.40)). BTKi-based regimens, including acalabrutinib, ibrutinib, zanubrutinib, ibrutinib + bendamustine + rituximab, and venetoclax + rituximab were associated with the lower rates of grade ⩾3 AEs, as compared with duvelisib and idelalisib in combination with rituximab or ofatumumab.
Relative matrix table for AEs.
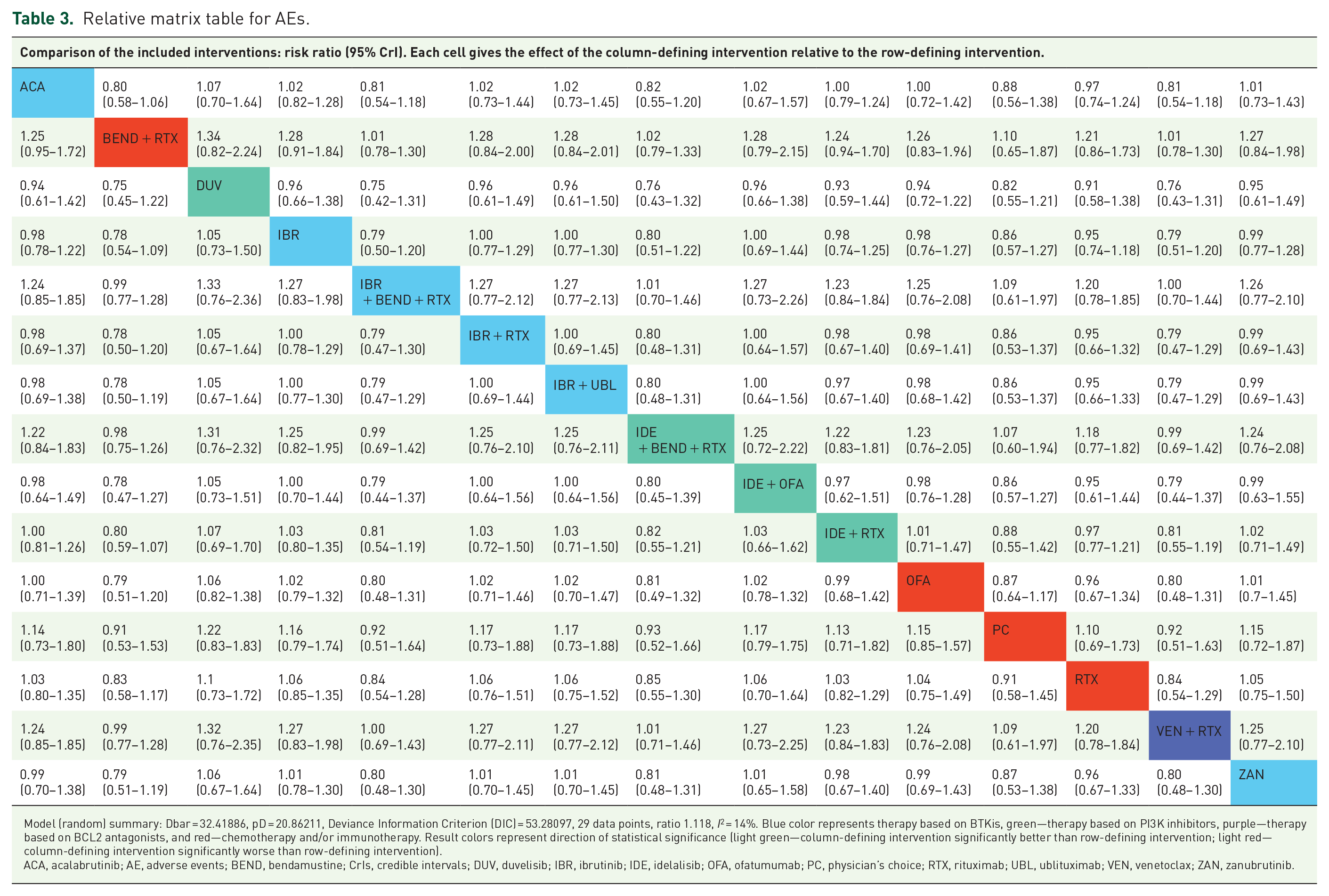
Model (random) summary: Dbar = 32.41886, pD = 20.86211, Deviance Information Criterion (DIC) = 53.28097, 29 data points, ratio 1.118, I2 = 14%. Blue color represents therapy based on BTKis, green—therapy based on PI3K inhibitors, purple—therapy based on BCL2 antagonists, and red—chemotherapy and/or immunotherapy. Result colors represent direction of statistical significance (light green—column-defining intervention significantly better than row-defining intervention; light red—column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; AE, adverse events; BEND, bendamustine; CrIs, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
Relative matrix table for grade ⩾3 AEs.

Model (fixed) summary: Dbar = 29.80088, pD = 27.38373, Deviance Information Criterion (DIC) = 57.18460, 29 data points, ratio 1.028, I2 = 6%. Blue color represents therapy based on BTKis, green—therapy based on PI3K inhibitors, purple—therapy based on BCL2 antagonists, and red—chemotherapy and/or immunotherapy. Result colors represent direction of statistical significance (light green—column-defining intervention significantly better than row-defining intervention; light red—column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; AE, adverse events; BEND, bendamustine; CrIs, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
Among the novel treatments, grade ⩾3 AEs were significantly higher in patients treated with ibrutinib compared with acalabrutinib (1.11 (1.01–1.24)), ibrutinib + bendamustine + rituximab (1.55 (1.10–2.39)), and venetoclax + rituximab (1.42 (1.004–2.19)), but not when compared with zanubrutinib (1.05 (0.94–1.16)). No significant differences in grade ⩾3 AEs were observed between venetoclax + rituximab and second-generation BTKis.
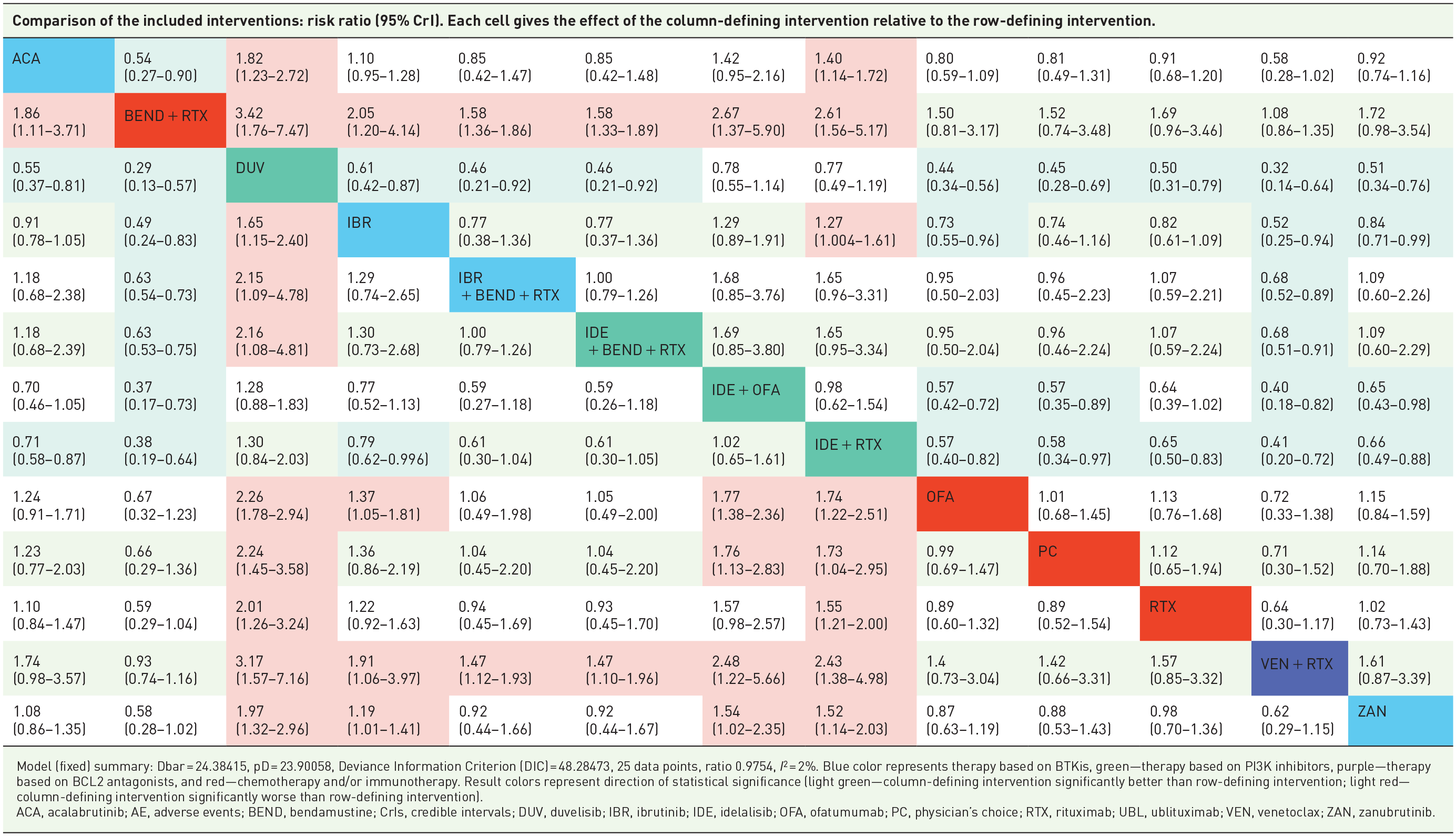
Similar patterns were noted when comparing the rates of SAEs among therapies (Table 5). The SAE rates for bendamustine + rituximab were significantly lower than those for the targeted therapies, except for zanubrutinib (0.58 (0.28–1.02)) and venetoclax + rituximab (0.93 (0.74–1.16)). Zanubrutinib and venetoclax + rituximab demonstrated lower SAE rates compared with ibrutinib, idelalisib + rituximab, and duvelisib.
Relative matrix table for SAEs.

Model (fixed) summary: Dbar = 24.38415, pD = 23.90058, Deviance Information Criterion (DIC) = 48.28473, 25 data points, ratio 0.9754, I2 = 2%. Blue color represents therapy based on BTKis, green—therapy based on PI3K inhibitors, purple—therapy based on BCL2 antagonists, and red—chemotherapy and/or immunotherapy. Result colors represent direction of statistical significance (light green—column-defining intervention significantly better than row-defining intervention; light red—column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; AE, adverse events; BEND, bendamustine; CrIs, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
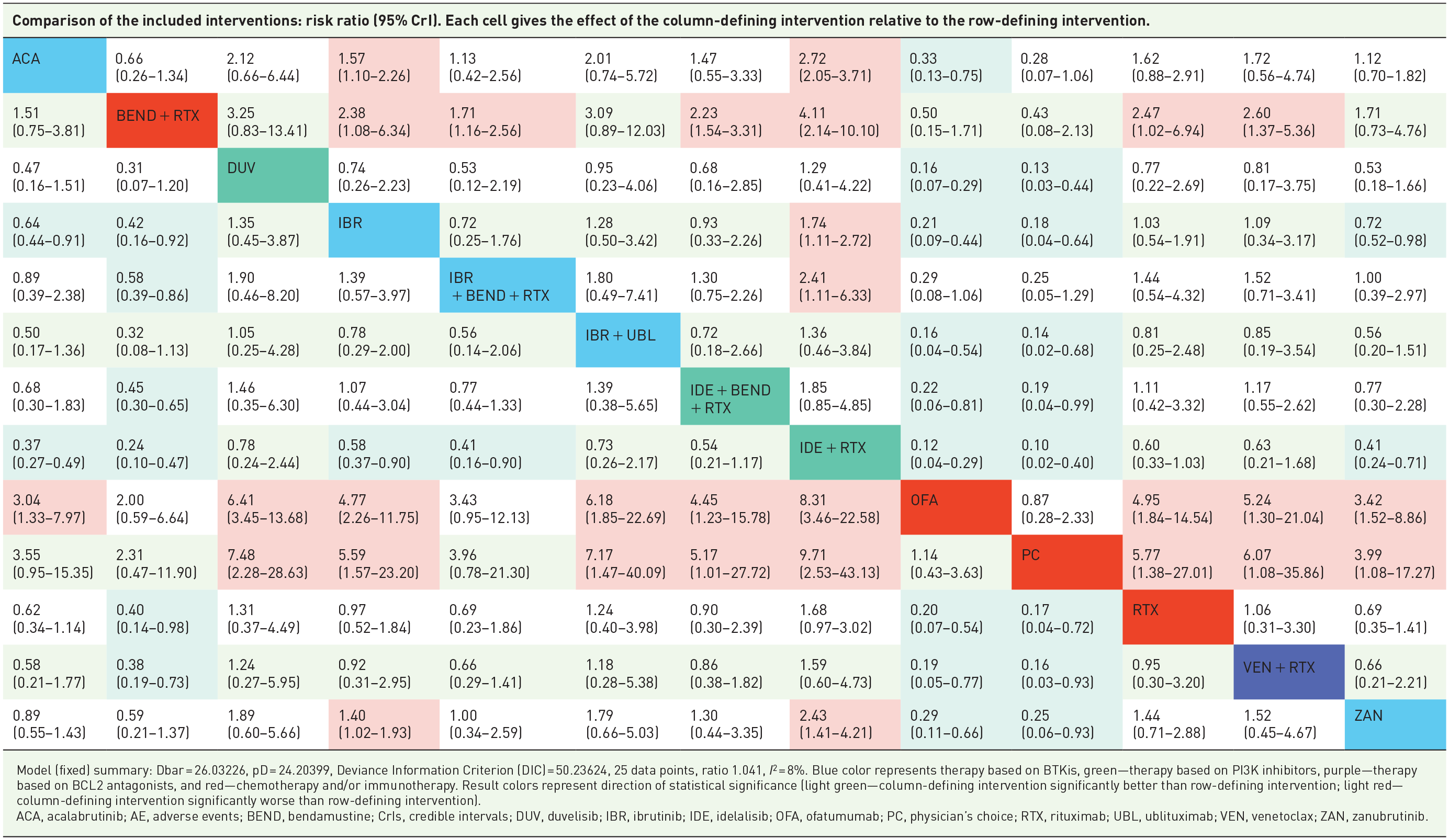
In patients with relapsed/refractory CLL, treatment discontinuation due to AEs was more common in those receiving idelalisib + rituximab compared with other treatments, such as acalabrutinib (2.72 (2.05–3.71)), ibrutinib (1.74 (1.11–2.71)), zanubrutinib (2.43 (1.41–4.21)), ibrutinib + bendamustine + rituximab (2.41 (1.11–6.33)), and bendamustine + rituximab (4.11 (2.14–10.10)) (Table 6). Venetoclax + rituximab showed no significant differences in the rates of treatment discontinuation due to AEs (1.59 (0.60–4.73)) compared with all BTKi-based regimens. Second-generation BTKis (i.e., acalabrutinib and zanubrutinib) showed significantly lower rates of treatment discontinuation due to AEs than the first-in-class ibrutinib. Additionally, discontinuations were more frequent in triple therapies that included a B-cell receptor inhibitor combined with bendamustine + rituximab versus bendamustine + rituximab alone.
Relative matrix table for AE-related treatment discontinuation.

Model (fixed) summary: Dbar = 26.03226, pD = 24.20399, Deviance Information Criterion (DIC) = 50.23624, 25 data points, ratio 1.041, I2 = 8%. Blue color represents therapy based on BTKis, green—therapy based on PI3K inhibitors, purple—therapy based on BCL2 antagonists, and red—chemotherapy and/or immunotherapy. Result colors represent direction of statistical significance (light green—column-defining intervention significantly better than row-defining intervention; light red—column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; AE, adverse events; BEND, bendamustine; CrIs, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
Duvelisib was associated with significantly higher rates of AE-related deaths compared with the remaining therapies, except ibrutinib + rituximab (2.41 (0.23–21.59)) and idelalisib + rituximab (1.44 (0.40–4.95)). The frequency of AE-related deaths was similar across all BTKis and venetoclax + rituximab (Table 7).
Relative matrix table for AE-related death.
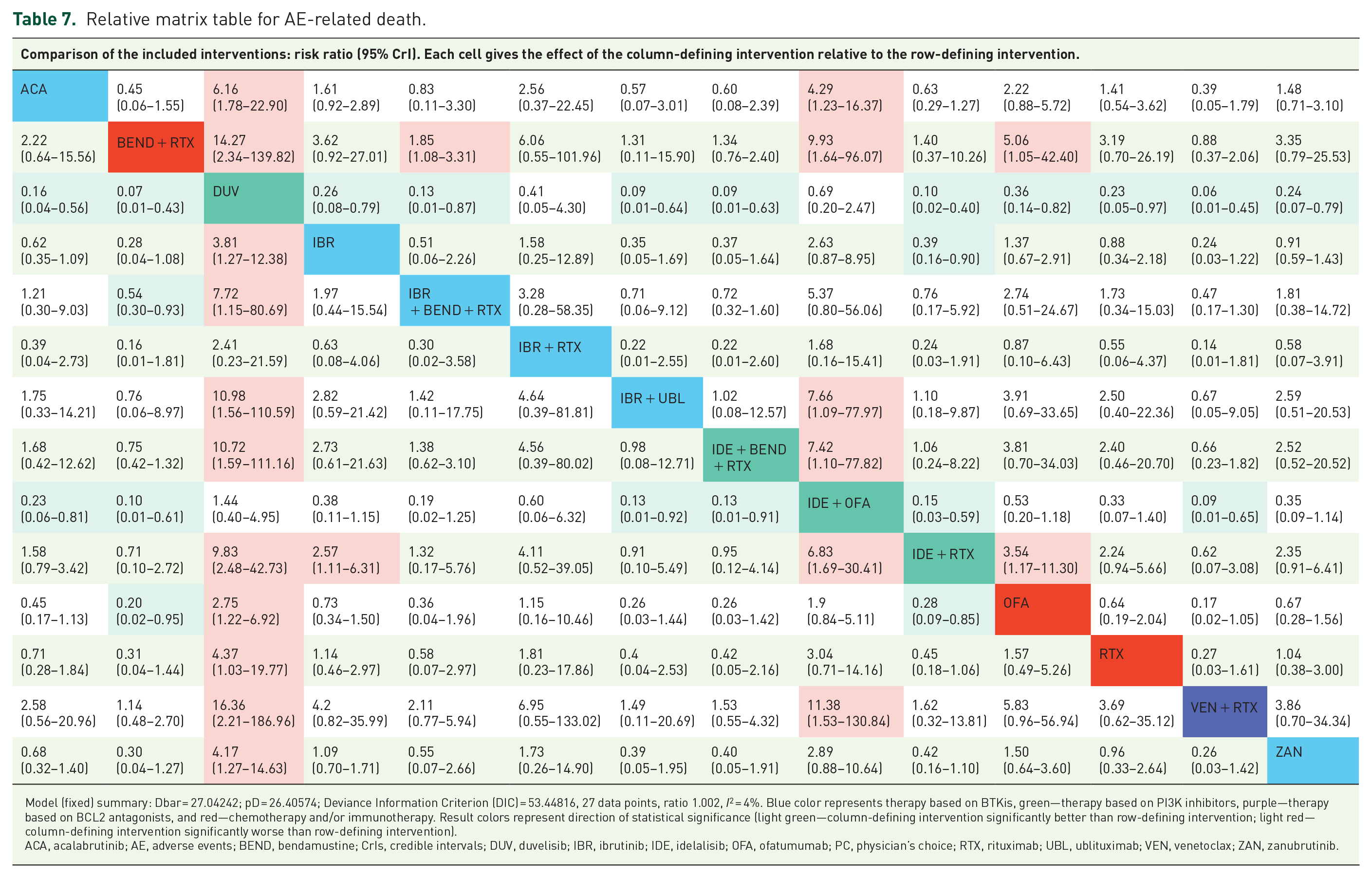
Model (fixed) summary: Dbar = 27.04242; pD = 26.40574; Deviance Information Criterion (DIC) = 53.44816, 27 data points, ratio 1.002, I2 = 4%. Blue color represents therapy based on BTKis, green—therapy based on PI3K inhibitors, purple—therapy based on BCL2 antagonists, and red—chemotherapy and/or immunotherapy. Result colors represent direction of statistical significance (light green—column-defining intervention significantly better than row-defining intervention; light red—column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; AE, adverse events; BEND, bendamustine; CrIs, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
A sensitivity analysis focusing on trials that assessed AEs for up to 2 years revealed that network geometry and treatment duration significantly influenced the outcomes of individual therapy comparisons. For shorter durations of treatment, the initially observed differences between acalabrutinib and bendamustine + rituximab became nonsignificant. Additionally, some differences that previously indicated a safety benefit in terms of SAEs for zanubrutinib, acalabrutinib, and venetoclax + rituximab compared with ibrutinib and idelalisib + ofatumumab were no longer significant in the sensitivity analysis. Detailed comparisons are shown in the Supplemental Appendix (Supplemental Tables 83–87).
Hematological AEs
Venetoclax + rituximab showed the lowest probability of causing anemia (SUCRA: 0.90), indicating that it was the most favorable option in terms of anemia risk. Conversely, duvelisib had the highest probability of causing anemia (SUCRA: 0.19), making it the least favorable option. Significant differences in the probability of anemia between individual therapies were not common, and there were no significant differences between venetoclax + rituximab, zanubrutinib, and ibrutinib, suggesting that these treatments have similar risk profiles.
However, significant differences were identified in the comparison between acalabrutinib and idelalisib + rituximab (1.75 (1.03–3.10)), and between acalabrutinib and ofatumumab (1.67 (1.003–2.81)) (Supplemental Table 59). Moreover, ofatumumab showed a lower risk of anemia compared with duvelisib (0.45 (0.25–0.76)) and idelalisib + ofatumumab (0.45 (0.21–0.85)). As for grade ⩾3 anemia, the combination of ibrutinib + bendamustine + rituximab had the highest SUCRA value (0.96), indicating its superiority over acalabrutinib (0.25 (0.05–0.89)), ibrutinib (0.22 (0.04–0.85)), duvelisib (0.10 (0.01–0.54)), and other therapies (Supplemental Table 60) in terms of minimizing the risk of severe anemia. Additionally, duvelisib performed worse than idelalisib + rituximab (5.15 (1.35–21.16)), ofatumumab (2.51 (1.18–5.98)), and venetoclax + rituximab (5.70 (1.08–38.28)) regarding the risk of severe anemia.
Venetoclax + rituximab was identified as having the lowest probability of causing overall thrombocytopenia and grade ⩾3 thrombocytopenia, with SUCRA values of 0.78 and 0.83, respectively. No significant differences were observed in the overall analysis of thrombocytopenia risk across treatments, suggesting that they have a relatively uniform risk profile for this AE (Supplemental Table 61). However, when focusing on grade ⩾3 thrombocytopenia, significant differences emerged in comparisons involving ofatumumab. Ofatumumab was found to have a significantly lower risk of severe thrombocytopenia compared with acalabrutinib (0.35 (0.12–0.90)), duvelisib (0.23 (0.05–0.75)), ibrutinib (0.42 (0.17–0.91)), and idelalisib + rituximab (0.26 (0.07–0.90)) (Supplemental Table 62). No other comparisons showed significant differences.
In the analysis of neutropenia risk across various therapies for CLL, ofatumumab had the lowest probability of inducing neutropenia (SUCRA: 0.95). Significantly lower rates of neutropenia were noted for ofatumumab compared with the remaining therapies, except acalabrutinib (0.72 (0.42–1.19)), bendamustine + rituximab (0.55 (0.27–1.16)), ibrutinib + bendamustine + rituximab (0.52 (0.25–1.11)), and the physician’s choice (0.93 (0.51–1.80)) (Supplemental Table 63). Acalabrutinib demonstrated better performance in reducing the risk of neutropenia compared with ibrutinib + ublituximab (0.46 (0.22–0.89)), idelalisib + rituximab (0.56 (0.41–0.76)), rituximab (0.67 (0.46–0.96)), and zanubrutinib (0.63 (0.41–0.96)). No significant differences in overall neutropenia rates were observed between venetoclax + rituximab and BTKi monotherapies, indicating that they have a comparable risk profile for this AE. However, venetoclax + rituximab exhibited the highest probability of grade ⩾3 neutropenia (SUCRA: 0.11), with significantly higher rates compared with acalabrutinib (2.37 (1.20–4.25)), bendamustine + rituximab (1.47 (1.20–1.82)), ibrutinib + bendamustine + rituximab (1.38 (1.07–1.80)), ofatumumab (3.74 (1.61–8.20)), and the physician’s choice (3.32 (1.18–9.29)). Of note, no differences in the risk of grade ⩾3 neutropenia were observed between acalabrutinib, ibrutinib, and zanubrutinib, suggesting that these BTKis have a similar safety profile for severe neutropenia (Supplemental Table 64).
Sensitivity analyses tailored to treatment duration revealed that differences in the rates of anemia between acalabrutinib and idelalisib + rituximab, as well as ofatumumab, were no longer significant when considering shorter durations. Similarly, some differences noted for duvelisib and ibrutinib + bendamustine + rituximab regarding grade ⩾3 anemia were less significant in the sensitivity analysis. In the sensitivity analysis of grade ⩾3 thrombocytopenia, the differences between therapies were much more common, especially for comparisons with venetoclax + rituximab and duvelisib. For shorter follow-up periods, acalabrutinib was associated with lower rates of neutropenia compared with ibrutinib, idelalisib + bendamustine + rituximab, idelalisib + ofatumumab, and venetoclax + rituximab. Interestingly, the rates of grade ⩾3 neutropenia were also more common with acalabrutinib compared with bendamustine + rituximab and ibrutinib + bendamustine + rituximab. The results are described in detail in the Supplemental Appendix (Supplemental Tables 88–93).
AEs impacting the quality of life
In the comparative analysis of treatments for CLL, the rates of nausea (including grade ⩾3) and diarrhea were generally similar across therapies (Supplemental Table 65, Supplemental Tables 67–68). However, when focusing specifically on grade ⩾3 diarrhea, acalabrutinib showed significantly lower rates compared with several other treatments including duvelisib (0.08 (0.01–0.75)), ibrutinib (0.22 (0.06–0.66)), ibrutinib + ublituximab (0.11 (0.01–0.61)), idelalisib + ofatumumab (0.04 (<0.01–0.61)), idelalisib + rituximab (0.06 (0.02–0.18)), and zanubrutinib (0.04 (<0.01–0.45)) (Supplemental Table 66). Zanubrutinib, on the other hand, was associated with higher rates of grade ⩾3 diarrhea compared with ibrutinib + rituximab, ofatumumab, and rituximab. However, in sensitivity analyses for the treatment duration of up to 2 years, differences between acalabrutinib and zanubrutinib, as well as between duvelisib and ibrutinib, became nonsignificant (Supplemental Tables 95–98). The highest SUCRA values for overall diarrhea and nausea were observed for bendamustine + rituximab (0.77) and rituximab alone (0.76). Conversely, the probability of grade ⩾3 nausea and diarrhea was the lowest with idelalisib + ofatumumab (0.81) and ofatumumab alone (0.81).
Fatigue, a common AE impacting the quality of life, exhibited similar rates among most of the treatments analyzed. However, higher rates of fatigue were reported for bendamustine + rituximab with and without the addition of ibrutinib compared with ibrutinib + rituximab, idelalisib + rituximab, rituximab, and zanubrutinib as well as for acalabrutinib when compared with ibrutinib + rituximab (2.10 (1.04–4.44)) (Supplemental Table 69). Despite these findings, no significant differences in the rates of fatigue were observed between acalabrutinib, ibrutinib, zanubrutinib, and venetoclax plus rituximab (venetoclax + rituximab). Concerning grade ⩾3 fatigue, acalabrutinib exhibited higher rates of fatigue in comparison with duvelisib, ibrutinib, ibrutinib + rituximab, idelalisib + ofatumumab, ofatumumab, and ibrutinib + ublituximab (Supplemental Table 70). On the contrary, lower rates of severe fatigue were shown for ibrutinib + rituximab versus either bendamustine + rituximab or idelalisib + bendamustine + rituximab. When considering shorter follow-up periods, the sensitivity analysis revealed no significant differences between therapies either for overall or grade ⩾3 fatigue (Supplemental Tables 99–100). SUCRA values indicated that ibrutinib + rituximab had the lowest impact on the fatigue-related quality of life (0.89).
Infections
The analysis of the risk of infection associated with various treatments for CLL showed that bendamustine + rituximab had the highest SUCRA value for overall infections (0.87), indicating it as the treatment with the lowest probability of infections. Venetoclax + rituximab was identified as the therapy with the lowest probability of grade ⩾3 infections (SUCRA: 0.95), pneumonia (SUCRA: 0.93), and grade ⩾3 pneumonia (SUCRA: 0.90), suggesting a favorable risk profile for infections, particularly for more severe cases.
A comparative analysis showed that bendamustine + rituximab had lower rates of infection than acalabrutinib (0.68 (0.45–0.94)), duvelisib (0.52 (0.32–0.80)), ibrutinib (0.65 (0.43–0.90)), idelalisib + bendamustine + rituximab (0.83 (0.72–0.95)), idelalisib + ofatumumab (0.65 (0.40–0.98)), idelalisib + rituximab (0.67 (0.44–0.92)), and zanubrutinib (0.67 (0.43–0.94)) (Supplemental Table 71). However, sensitivity analyses adjusted for treatment duration indicated that the differences in infection rates between bendamustine + rituximab and both acalabrutinib and zanubrutinib became less significant, suggesting that treatment duration influences the comparative risk of infection (Supplemental Table 101). The difference in infection rates remained significant only when comparing bendamustine + rituximab with ibrutinib. No significant differences in the rate of overall infections were observed between acalabrutinib, ibrutinib, and zanubrutinib, either in the longest or 2-year follow-up, highlighting that these treatments have a similar infection risk profile.
The rates of grade ⩾3 infections were significantly lower for venetoclax + rituximab compared with acalabrutinib (0.29 (0.08–0.74)), duvelisib (0.19 (0.05–0.61)), ibrutinib (0.30 (0.08–0.79)), idelalisib + bendamustine + rituximab (0.51 (0.32–0.82)), idelalisib + rituximab (0.25 (0.07–0.64)), and zanubrutinib (0.32 (0.09–0.87)) (Supplemental Table 72). After adjustment for shorter treatment duration, the differences in infection rates between venetoclax + rituximab and all BTKis became less significant, indicating that the duration of treatment may impact the relative risk of infection (Supplemental Table 102). However, the differences remained significant when comparing venetoclax + rituximab to PI3Ki-based regimens, suggesting a consistently lower risk of infections with venetoclax + rituximab in these comparisons.
For pneumonia, including grade ⩾3 events, the rates were comparable between venetoclax + rituximab and zanubrutinib, indicating a similar risk profile (Supplemental Tables 73–74). However, a lower risk of pneumonia was observed with venetoclax + rituximab compared with acalabrutinib and ibrutinib, highlighting the favorable profile of venetoclax + rituximab for pneumonia. After adjustment for treatment duration, differences between venetoclax + rituximab and BTKi therapies became nonsignificant, except the comparison with ibrutinib for grade ⩾3 pneumonia, where the risk remained higher for ibrutinib (Supplemental Tables 103–104).
BTKi-specific AEs
Considering BTKi-specific AEs, bendamustine + rituximab demonstrated the lowest probability of bleeding (0.88) and hypertension (0.96), according to SUCRA values. On the other hand, ofatumumab showed the lowest probability of atrial fibrillation (SUCRA: 0.96) and major bleeding (SUCRA: 0.91), highlighting its favorable safety profile in terms of these AEs. Acalabrutinib was associated with a lower risk of bleeding compared with ibrutinib (0.74 (0.61–0.89)) and zanubrutinib (0.72 (0.55–0.94)), indicating its more favorable safety profile for bleeding (Supplemental Table 75). However, no significant differences in bleeding risk were observed between zanubrutinib and ibrutinib (0.98 (0.81–1.17)), suggesting a similar risk profile of these two BTKis. Regarding severe bleeding events, the risk of grade ⩾3 bleeding and major bleeding remained similar across all BTKi therapies (Supplemental Tables 76 and 77). Significant differences were noted only for ofatumumab, which showed a markedly lower risk when compared with acalabrutinib (0.17 (0.03–0.67)), ibrutinib (0.15 (0.03–0.44)), and zanubrutinib (0.17 (0.03–0.66)).
Among BTKis, acalabrutinib demonstrated a significantly lower risk of hypertension compared with other BTKi-based treatments. Specifically, patients treated with acalabrutinib showed lower rates of hypertension compared with those on ibrutinib (0.38 (0.23–0.53)), ibrutinib + rituximab (0.42 (0.24–0.71)), ibrutinib + ublituximab (0.29 (0.10–0.83)), and zanubrutinib (0.34 (0.19–0.58)) (Supplemental Table 78). The analysis of grade ⩾3 hypertension revealed similar trends, with acalabrutinib showing a significantly lower risk than ibrutinib (0.47 (0.22–0.92)), ibrutinib + rituximab (0.45 (0.19–0.999)), and zanubrutinib (0.35 (0.15–0.77)) (Supplemental Table 79). No significant differences were observed between ibrutinib and zanubrutinib, either for overall hypertension or for grade ⩾3 events, suggesting that these two BTKis have a similar risk profile for hypertension.
Atrial fibrillation was more common in patients receiving ibrutinib compared with those on zanubrutinib (2.70 (1.55–4.95)) and acalabrutinib (1.73 (1.09–2.83)) (Supplemental Table 80). There were no significant differences in the rates of atrial fibrillation between acalabrutinib and zanubrutinib; however, both medications showed better outcomes than the combination therapy of ibrutinib and ublituximab. In the sensitivity analysis, the difference in the rates of atrial fibrillation between ibrutinib and acalabrutinib was no longer significant, whereas the difference remained significant for the comparison with zanubrutinib (Supplemental Table 94). Nevertheless, the risk of grade ⩾3 atrial fibrillation was similar across all BTKi monotherapies (Supplemental Table 81).
Secondary malignancies
Patients receiving ofatumumab had the lowest probability of developing a secondary malignancy (SUCRA: 0.87), while those receiving acalabrutinib had the highest probability (SUCRA: 0.06). This suggests that the risk of secondary malignancy was higher for acalabrutinib.
The rates of secondary malignancies were significantly higher in patients treated with acalabrutinib, ibrutinib, and zanubrutinib than in those treated with ofatumumab and idelalisib + rituximab (Supplemental Table 82). Specifically, idelalisib + rituximab was linked to a lower risk of secondary malignancy compared with ibrutinib (0.31 (0.09–0.99)), acalabrutinib (0.22 (0.06–0.76)), and zanubrutinib (0.32 (0.10–0.91)). Also bendamustine + rituximab demonstrated lower rates of secondary malignancies than acalabrutinib (0.27 (0.04–0.88)).
Discussion
Our study aimed to evaluate the safety profiles of therapies used in the treatment of relapsed/refractory CLL, with a special emphasis on newly approved targeted therapies such as ibrutinib, acalabrutinib, zanubrutinib, and venetoclax + rituximab. Our findings indicate that the safety profiles vary significantly across pharmacotherapeutic groups, and there are substantial differences between individual therapies.
In terms of the overall safety profile, there were no differences in the frequency of overall AEs, likely because most included studies reported at least one AE in all patients. This may be attributed not only to the adverse effects of the drugs but also to the presence of disease symptoms, advanced age of patients, and comorbidities, which predispose to reporting negative events. However, differences were noted in the rates of grade ⩾3 AEs, SAEs, and AE-related treatment discontinuations and deaths, especially in the comparisons with bendamustine + rituximab and PI3Ki-based regimens, including duvelisib. The results of the NMA highlighted the favorable safety profile of bendamustine + rituximab, which is reflected in clinical practice guidelines recommending this regimen, particularly for older patients and those with comorbidities.56,57 On the other hand, the relatively high toxicity of duvelisib justifies its use in later treatment lines once the options with a more favorable safety profile have been exhausted.
No significant differences were found for the newer targeted therapies within the BTKi class (acalabrutinib, zanubrutinib) and venetoclax + rituximab for any of the overall safety outcomes, suggesting that these therapies have a similar safety profile. Importantly, there were no differences in the frequency of treatment discontinuation due to AEs, even though venetoclax + rituximab is used for a maximum of 2 years, whereas acalabrutinib and zanubrutinib are used until disease progression or unacceptable toxicity occurs. These findings for the overall safety profile suggest that general safety considerations might be less important in therapy selection between acalabrutinib, zanubrutinib, and venetoclax + rituximab than the patient’s preference or economic aspects, including the reimbursement status of these drugs in different countries. Our NMA showed that the frequency of SAEs and/or grade ⩾3 AEs was higher for ibrutinib than for acalabrutinib, zanubrutinib, and venetoclax + rituximab. Therefore, from the perspective of safety, the second-generation BTKis and venetoclax + rituximab can be considered preferable to ibrutinib.
When choosing the appropriate therapy for patients, attention should be paid not only to general but also to detailed safety profiles. For hematological events, no significant differences were found between BTKi monotherapies and venetoclax + rituximab in terms of the risk of anemia and thrombocytopenia, including grade ⩾3 events. However, differences were noted between acalabrutinib and zanubrutinib for overall neutropenia, and between venetoclax + rituximab for grade ⩾3 neutropenia, suggesting that acalabrutinib might be a more optimal option for patients at increased risk of symptomatic neutropenia and neutropenic fever. Moreover, no significant differences were found in the rates of AEs potentially affecting the quality of life (fatigue, nausea, and diarrhea) between BTKi monotherapies and venetoclax + rituximab. The only differences were the lower rates of grade ⩾3 diarrhea and fatigue for the comparison of acalabrutinib with ibrutinib and of grade ⩾3 diarrhea for the comparison of acalabrutinib with zanubrutinib. However, the results should be interpreted with caution because these AEs are very rare and are seen only in individual patients. Moreover, the NMA included a limited number of studies, which resulted in wide CrIs. For venetoclax + rituximab, pneumonia (including grade ⩾3 events) was reported less frequently than for acalabrutinib and ibrutinib. However, this finding may have been influenced by a longer treatment duration for BTKi. Within the range of AEs typical for BTKis, ibrutinib was associated with slightly higher rates of atrial fibrillation than acalabrutinib and zanubrutinib, although no differences were observed for grade ⩾3 events. However, NMA results suggest that acalabrutinib may be superior to ibrutinib in terms of the risk of hypertension and bleeding, with a comparable frequency of grade ⩾3 and major bleedings. Other comparisons, some of which showed significant results, have less practical relevance because the drugs have not been approved for indications covering CLL (e.g., idelalisib + ofatumumab, ibrutinib + ublituximab) or they became less popular due to changes in the standard of care and the emergence of newer therapeutic options (e.g., idelalisib + rituximab, rituximab, and ofatumumab).
To our knowledge, this is the first comprehensive NMA discussing the safety of therapies in patients with relapsed/refractory CLL. Previous meta-analyses focused mainly on comparing the efficacy of treatments and involved a limited number of comparisons or included previously untreated patients.58–61 Only a recent meta-analysis by Liu et al. 62 partially addressed safety issues related to BTKi therapies, but it included only six studies and assessed fewer endpoints than our meta-analysis (diarrhea, neutropenia, hypertension, cardiac events, SAE, and grade ⩾3 AEs). Moreover, it included previously untreated patients, who may have a slightly different tolerance to therapy than heavily treated patients, and this may have affected the results. Liu et al. 62 conducted the NMA using a frequentist approach, and the meta-analysis model was not specified, which makes it impossible to reliably compare the results of both meta-analyses. Nevertheless, for some endpoints, significant discrepancies between our results and those of Liu et al. 62 were noted. For example, Liu et al. 62 showed that therapy with zanubrutinib and acalabrutinib was associated with an increased risk of hypertension compared with ibrutinib (3.20 (1.69–6.06) and 3.12 (1.85–5.27), respectively), whereas our meta-analysis found the hypertension risk to be significantly lower for acalabrutinib (0.38 (0.23–0.58)) and similar for zanubrutinib (1.11 (0.82–1.50)), as compared with ibrutinib. Our findings are consistent with the results of the direct comparison in ELEVATE-RR (0.38 (0.23–0.58)) and ALPINE (1.11 (0.82–1.50)) studies. The results reported by Liu et al. 62 are even more puzzling because second-generation BTKis are commonly considered to have a non-inferior safety profile for cardiac events as compared with ibrutinib. There were also some differences in indirect comparisons for neutropenia and diarrhea. Additionally, we were unable to determine if cardiovascular events in the meta-analysis by Liu et al. 62 included only atrial fibrillation or other events as well.
Our meta-analysis included a wide range of AEs and therapies used in relapsed/refractory CLL. We conducted an extensive literature review, and used not only published clinical studies but also drug registration reports in which the safety profile was often more comprehensively described and included longer follow-up periods than in published articles. In our meta-analysis, most networks did not show significant heterogeneity (I2 < 20%) or inconsistency. The only inconsistency in the main networks was noted for grade ⩾3 AEs, treatment discontinuation due to AEs, and grade ⩾3 thrombocytopenia. Therefore, these results should be interpreted with caution.
Our study has several limitations, mainly related to the limitations of primary studies. It was not possible to compare all therapies for all endpoints because of data gaps for certain AEs, especially cardiovascular ones, in some studies. Cardiovascular AEs were generally not reported in studies for PI3Kis and venetoclax + rituximab. Additionally, the assessed AEs concerned all events, regardless of their causal relationship with the therapy. This is due to the fact that treatment-related AEs were very rarely reported in primary studies; thus, it was impossible to create a network for meta-analysis based on proven or suspected treatment-related AEs. Another limitation is the duration of individual therapies. The meta-analysis included both therapies with a limited duration of use (e.g., a few cycles) and long-term therapies used until disease progression or unacceptable toxicity occurred. However, all events in the studies analyzed were assessed as treatment-emergent AEs; thus, the results of this meta-analysis reflect the actual differences in the frequency of AEs during these therapies. It was not possible to conduct other types of analyses adjusting for the timing of individual events (e.g., based on the incidence rate ratio) due to the lack of reporting of such data in primary studies. To investigate the impact of therapy duration on the meta-analysis outcome, we conducted a sensitivity analysis in which studies and results for a treatment duration longer than 24 months were excluded. However, it was not possible to standardize all studies in terms of treatment duration, and the exclusion of studies from the network changed its geometry, which may have influenced the results. Therefore, the results of the sensitivity analyses should be treated as exploratory. Additionally, for certain AEs, especially grade ⩾3, there were wide confidence intervals for comparisons, largely because these events are rare. Therefore, caution is warranted when interpreting the differences between these therapies.
Despite the above limitations, our study provides valuable insights. The results may be particularly interesting to clinicians, but they may also inform health technology assessment and reimbursement procedures. A better understanding of the differences in the safety profile between therapies can help build economic models and estimate AE-related treatment costs.
Conclusion
Our analysis showed that novel therapeutic options, such as acalabrutinib, zanubrutinib, and venetoclax + rituximab, have relatively similar safety profiles. Only a few differences were noted for selected AEs. Nevertheless, further studies with long-term follow-up are needed to comprehensively assess the safety of the newest therapies. Large registry studies and the analysis of databases collecting information on drug-related AEs would be particularly valuable.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241285988 – Supplemental material for Comparative safety of novel targeted therapies in relapsed/refractory chronic lymphocytic leukemia: a network meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241285988 for Comparative safety of novel targeted therapies in relapsed/refractory chronic lymphocytic leukemia: a network meta-analysis by Magdalena Monica, Monika Reczek and Paweł Kawalec in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241285988 – Supplemental material for Comparative safety of novel targeted therapies in relapsed/refractory chronic lymphocytic leukemia: a network meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359241285988 for Comparative safety of novel targeted therapies in relapsed/refractory chronic lymphocytic leukemia: a network meta-analysis by Magdalena Monica, Monika Reczek and Paweł Kawalec in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.