Abstract
Background:
The recent development of new antileukemic therapies (anti-CD20 monoclonal antibodies, Bruton tyrosine kinase inhbitors, phosphoinositide 3-kinase inhibitors, and B-cell lymyphoma-2 antagonists) improved the progression-free survival (PFS) compared with selected standard regimens in clinical trials for patients with relapsed/refractory chronic lymphocytic leukemia (CLL). Unfortunately, the relative efficacy of all possible therapeutic options remains unknown because there is no direct evidence for all possible comparisons.
Objectives:
We aimed to compare the efficacy and safety of novel agents, chemotherapy, and immunotherapy using a Bayesian network meta-analysis (NMA).
Design:
Systematic literature review with Bayesian NMA.
Methods:
An extensive systematic literature review of randomized clinical trials for relapsed/refractory CLL was performed. We searched for articles indexed in medical databases (MEDLINE, Embase, The Cochrane Library) and gray literature that could be further implemented into the Bayesian NMA.
Results:
The systematic search identified 15 randomized trials that formed networks comparing PFS, overall survival (OS), overall response rates, and serious adverse events. Our study showed that all regimens containing novel agents significantly prolonged PFS compared with standard chemoimmunotherapy and immunotherapy. Among targeted drugs, venetoclax (VEN) + rituximab (RTX) had comparable efficacy in terms of PFS to zanubrutinib (ZAN) [hazard ratio (95% credible interval), 1.10 (0.59–2.08)], acalabrutinib (ACA) [0.78 (0.47–1.30)], ibrutinib (IBR) monotherapy [0.72 (0.41–1.27)], and other IBR-based regimens. ZAN was superior to IBR monotherapy [0.65 (0.49–0.86)] but not to ACA [0.71 (0.49–1.02)]. There were no significant differences in OS in any of the above comparisons.
Conclusion:
All novel therapies have better efficacy than chemoimmunotherapy and immunotherapy regimens. Among novel agents, the relative efficacy of VEN + RTX was similar to all BTKi, while ZAN was superior to IBR and comparable to ACA.
Trial registration:
PROSPERO CRD42022304330.
Keywords
Introduction
Chronic lymphocytic leukemia (CLL) is the most prevalent type of leukemia in Western countries. 1 It primarily affects elderly patients, with a median age at diagnosis of 70 years. 2 The main sign of the disease is persistent monoclonal B-cell lymphocytosis in the peripheral blood and bone marrow, often coexisting with lymphadenopathy and enlargement of the liver and spleen. CLL is an indolent disease with frequent relapses, and patients usually require multiple lines of treatment over the years. 3 The most common causes of CLL-related death are infections, disease progression, and secondary cancer. 4
Despite the growing incidence of CLL over the last 30 years, the mortality rates significantly decreased in North America (from 1.22 to 0.98 per 100,000) and Western Europe (from 1.06 to 0.93 per 100,000) between 1990 and 2019. 1 According to the National Cancer Institute statistics, 5-year relative survival rates increased from 80.1% in 2000 to 88.5% in 2018. 5 One of the main reasons for improving patient survival was the development of new antileukemic therapies: initially anti-CD20 antibodies and then new targeted therapies such as Bruton tyrosine kinase inhibitors (BTKi) (ibrutinib, IBR), 6 phosphoinositide 3-kinase inhibitors (PI3Ki) (idelalisib, IDE), 7 and B-cell lymphoma-2 (BCL2) antagonists (venetoclax, VEN). 8 In the last decade, the US Food and Drug Agency and the European Medicines Agency approved six new targeted drugs indicated for relapsed/refractory CLL, including second-generation B-cell receptor (BCR) inhibitors [e.g. acalabrutinib (ACA), zanubrutinib (ZAN) and duvelisib (DUV)]. Clinical trials for third-generation agents [e.g. pirtobrutinib (PIR), ublituximab (UBL)] are also ongoing.9,10
Pivotal phase III clinical trials examining the efficacy and safety of IBR (RESONATE 6 ), IDE (Study116 7 ), and VEN (MURANO 8 ) confirmed their superiority over standard immunotherapy or chemoimmunotherapy in relapsed/refractory CLL. More recent trials were conducted to assess the efficacy of BCR inhibitors with chemoimmunotherapy (TUGELA, 11 HELIOS 12 ) and to compare different BTKis (ALPINE, 13 ELEVATE-RR 14 ). Unfortunately, only a few randomized trials comparing novel drugs from different pharmacotherapeutic classes were conducted, which limits the possibility of drawing conclusions about the relative efficacy of all targeted therapies. Direct evidence from real-world practice suggests differences between these novel agents. For example, Mato et al. 15 showed that IDE might be inferior to IBR and VEN in patients with relapsed/refractory CLL. However, data from retrospective studies are limited by the heterogeneous characteristics of patients who receive different therapeutic regimens according to their preferences, the type of previous treatment lines, and the availability of reimbursement for novel therapies.16–19
As data on the relative effectiveness of novel targeted therapies are essential for clinicians and healthcare policymakers, we have proposed to use a Bayesian network meta-analysis (NMA) 20 to compare treatments for relapsed/refractory CLL simultaneously. Thus, the primary objective of this study was to compare the safety and efficacy of various regimens for CLL, including novel agents, monoclonal antibodies, and chemotherapy.
Methods
A systematic review was conducted to identify randomized clinical trials that could be further used for the NMA. The review was performed according to 2020 PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guidelines 21 and their extension for NMAs 22 (Supplemental File 1). The protocol for the systematic review was previously registered in the PROSPERO database (CRD42022304330). Here, we presented critical information about the systematic review methodology. Additional information was included in the supplement.
Data sources and searches
A systematic search of electronic medical databases (MEDLINE, EMBASE, and CENTRAL) was performed on 27 January 2022, and updated on 10 October 2023. To retrieve unpublished data for studies, we also hand-searched clinical trial registries (ClinicalTrials.gov, EU Clinical Trial Registry, World Health International Trials Registry Platform Search Portal), conference materials (American Society of Hematology, European Hematology Association, American Society of Clinical Oncology, European Society of Medical Oncology), and websites of medicines regulatory authorities (Food and Drug Agency, European Medicines Agency) and health technology assessment agencies (National Institute for Health and Care Excellence, German Federal Joint Committee). The references of the included full-text articles were checked to identify any additional relevant publications. We also attempted to contact the corresponding authors of the included studies if any critical data were missing. We did not restrict our search to any date. Detailed sources and search strategies for each database were presented in Supplemental File 2 (Supplemental Table 1).
Study selection
We searched for clinical trials conducted in adult patients with relapsed/refractory CLL who previously received at least one treatment line. Studies involving mixed populations (previously treated and untreated) were included if the results for a subgroup of patients with relapsed/refractory CLL were available. Evaluated interventions (as monotherapy or combination therapy) were as follows:
anti-CD20 monoclonal antibodies: obinutuzumab, ofatumumab (OFA), rituximab (RTX), UBL;
BTKi: ACA, IBR, PIR, ZAN;
PI3Ki: IDE, DUV, umbralisib;
BCL2 antagonists: VEN;
other therapies: dinaciclib, lenalidomide.
Eligible comparators included an alternative monotherapy or drug combinations, any other active treatment, best standard or supportive care, placebo, or physician’s choice (PC). To ensure the best quality of the NMA, we included only randomized clinical trials published in English. The publication of a full-text article, a report, or a study protocol was required for inclusion in the systematic review. Studies published as conference abstracts only were excluded. The outcomes of interest were progression-free survival (PFS), overall survival (OS), overall response rates (ORRs), and safety. We also searched for other efficacy endpoints, including minimal residual disease, quality of life, and subsequent treatment lines, but it was impossible to perform the NMA for these outcomes. As for safety, here we presented the results of a comparative analysis of serious adverse events (SAEs). A detailed comparison of safety will be published separately in the future.
Selection and extraction processes
Two reviewers (MM and MR) independently selected abstracts and full texts based on predefined inclusion and exclusion criteria (Supplemental Table 2). Full-text articles were included only if both reviewers considered them eligible. Disagreements between reviewers were resolved by consensus. Data extraction was performed by one reviewer (MM) using a prespecified form and then verified by another author (MR). Any discrepancies in extraction were discussed and resolved. The following data regarding methodology were extracted from the included studies: design, interventions, outcomes, key inclusion and exclusion criteria, characteristics of patients, and previous treatment lines.
Endpoints extracted for this NMA included PFS, OS, ORRs, and SAEs. Outcomes associated with survival were presented as a median time to event, the proportion of patients event-free at specified time points (6 months, 12 months, and then annually up to 5 years), and hazard ratios (HRs) with confidence intervals (CIs). If the percentages of individual time points were not reported in the article, we read Kaplan–Maier curves using the CurveSnap software to obtain data. The ORRs and safety outcomes were shown as the number and proportion of patients with an outcome. Endpoints assessed by investigators and independent committees were drawn separately. Full-text articles were treated as the primary source for data extraction. Data from other publications were only extracted if new outcomes or data cut-offs were presented.
Data analysis and synthesis
The NMA was performed using the Bayesian approach based on the Markov chain Monte Carlo method. The GeMTC package 23 for the R software, implemented in the online interface (gemtc.drugis.org) and R console, was used for analyses. The NMA results were expressed as HRs with credible intervals (CrIs) for endpoints associated with survival and risk ratios (RRs) for dichotomous endpoints. We used contrast-based data for survival endpoints. RRs were estimated based on the results of individual arms. To assess consistency between the NMA and individual studies, we also calculated RRs for direct comparisons using the OpenMetaAnalyst software (Brown University, Providence, USA).
For all comparisons, the fixed effect model was selected as the deviance information criterion between fixed and random models was less than five, and the results of fixed models were more consistent with the original data. The model settings were as follows: initial chains, 4; burn-in, 50,000; inference, 100,000; and thinning, 5. The parameters of convergence diagnostics indicated optimal settings. We also calculated rank probabilities for every intervention, endpoint, and possible rank. Rank probabilities were then summarized using the surface under the cumulative ranking probabilities (SUCRA). To determine the benefit–risk balance of each treatment, we used the cluster analysis technique to group the therapies according to the SUCRA values for PFS, OS, and SAEs.
Primary NMAs included data for the longest available follow-up for each endpoint. If PFS was assessed by independent review committees (IRCs) and investigators for the same follow-up, we included IRC data for the primary analysis. Separate analyses for IRC and investigator assessments for PFS were conducted as sensitivity analyses. Separate NMAs were performed for ORRs assessed by investigators and IRCs. As partial remission with lymphocytosis (PRwL) is a characteristic response for patients treated with BTKi, we also assessed ORR with PRwL as a sensitivity analysis.
Quality assessment
We assessed the risk of bias in all included studies using the Cochrane revised risk-of-bias tool for randomized trials (RoB2). 24 This tool allowed us to determine the risk of bias as low or high or express some concerns about trial design, conduct, and reporting in five domains. We assessed the risk of bias separately for each study and endpoint. The overall risk score was considered the highest reported risk from any domain. If investigators and IRCs reported progression-related outcomes simultaneously, we considered only the IRC perspective for the summary to avoid duplicating the evaluation for the same endpoints. The risk of bias for the main networks was visualized using the CINeMA (Confidence in Network Meta-Analysis) tool. 25
Results
Search results
The systematic search identified 20 studies described in 206 articles and other reports, meeting our eligibility criteria. Only 15 trials were included in the qualitative analyses (ALPINE,13,26–28 ASCEND,29–34 Burger 2019, 35 DUO,36,37 ELEVATE-RR, 14 GENUINE, 10 HELIOS,12,38–40 Huang 2018, 41 MaBLE, 42 MURANO,8,43–47 OMB114242,48,49 RESONATE,6,50–53 Study116,7,54–56 Study119, 57 and TUGELA 58 ). Four excluded trials lacked the arm with connection to the network (CLLUmbrella2, 59 COMPLEMENT2,60,61 LUCID, 62 and REACH 63 ). Another pilot study (PN012 64 ) was excluded because of the small sample size and heterogeneous results in the comparator arm. We extracted 40 individual reports for the included studies to obtain results for NMAs. A detailed study flow diagram is shown in Figure 1.

Study flow diagram according to PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 2020.
Characteristics of included studies
Most included studies were phase II or III open-label multicenter trials. Only three trials were double-blind (HELIOS, Study116, and TUGELA), and one trial was single-center (Burger 2019). The interventions primarily included BTKi (IBR, ACA, and ZAN) and PI3Ki (IDE and DUV) used as monotherapy or combined with anti-CD20 (OFA and RTX) and bendamustine (BEND). The main comparators for these treatments were immunotherapy (OFA and RTX) or chemoimmunotherapy (BEND + RTX). Novel targeted therapies were used in both treatment arms only in four trials (ALPINE, ASCEND, ELEVATE-RR, and GENUINE). One trial examined the effectiveness of a BCL2 antagonist combined with anti-CD20 (VEN + RTX). Two studies compared only chemoimmunotherapy (MaBLE and OMB114242), while the reference arm received PC in two trials. The OMB114242 study defined PC as non-OFA regimens, including chemotherapy, immunotherapy, and steroids. In the ASCEND trial, investigators chose between IDE + RTX and BEND + RTX. The availability of results for the above subgroups was critical for performing NMAs for all identified studies.
Nearly all studies were designed as superiority trials with PFS as the primary endpoint. As for their homogeneity, critical eligibility criteria were similar between studies and typical for patients with relapsed/refractory CLL. Only three trials had more specified criteria regarding cytogenic abnormalities in eligible populations. The ELEVATE-RR and GENUINE trials included only patients with high-risk cytogenetic profiles (del17p, del11q), while subjects with confirmed del17p were excluded from the HELIOS study. The patient’s age, sex, clinical staging, and performance status were relatively similar in the included studies. The potential source of heterogeneity could be the outlying frequencies of del17p and del11q in the ELEVATE-RR, GENUINE, and HELIOS studies. In the remaining studies, the frequency of cytogenetic abnormalities was similar (~15–30%). The number of previous treatment lines ranged from 1 to 16, with a median of 1 or 2 for individual studies. The median number of previous therapies was higher in OMB114242, Study116, and Study119 (three to four lines). Detailed characteristics of the individual studies are provided in the supplement (Supplemental Tables 3–8).
Risk-of-bias assessment
Most included studies had a low or unclear overall risk of bias for PFS, ORR, and OS. The primary concerns for the risk-of-bias assessment were the lack of details of the randomization process and possible deviations from the intended intervention. Two trials (Huang 2018 and MaBLE) had an overall high risk of bias for efficacy endpoints because we could not rule out that the open-label design did not affect the assessment of progression-related endpoints. For the same reason, the risk of bias for SAEs was high for all open-label studies. Detailed data on the risk of bias are shown in the supplement (Supplemental Figures 1–9).
Progression-free survival
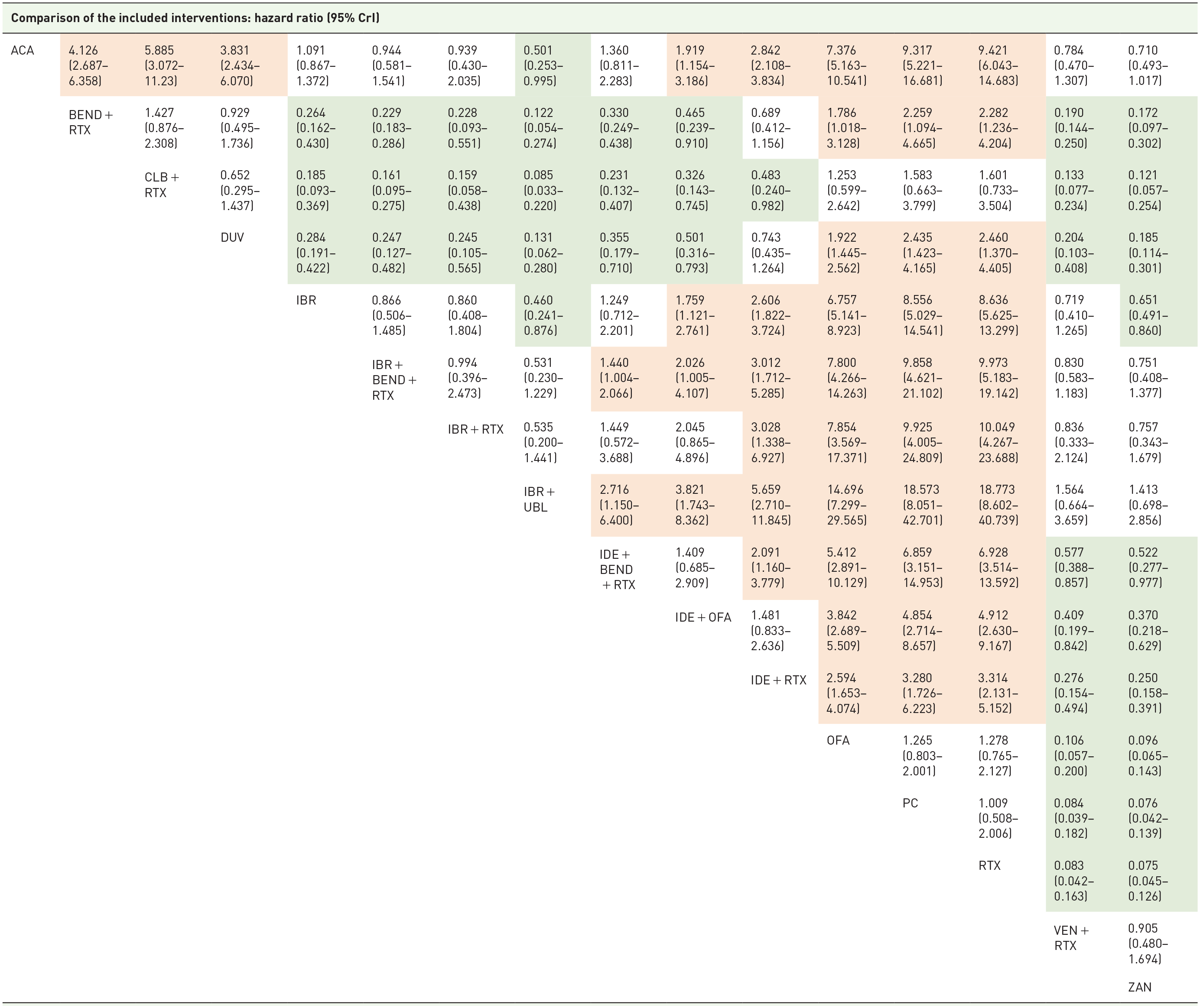
The NMA for the longest available follow-up showed significant differences between treatments. All monotherapies with BTKi were significantly superior to chemotherapy and immunotherapy regimens, DUV, and combinations of IDE with anti-CD20 (Table 1). ZAN was also superior to IBR [HR (95% CrI) = 0.65 (0.49–0.86)] and IDE + BEND + RTX triple combination therapy [HR (95% CrI) = 0.52 (0.28–0.98)]. No statistically significant differences between ZAN and ACA were observed [HR (95% CrI) = 0.71 (0.49–1.02)]. The triple combination therapy IBR + BEND + RTX had similar efficacy to IBR [HR (95% CrI) = 0.87 (0.51–1.49)] but was superior to IDE + BEND + RTX [HR (95% CrI) = 0.69 (0.48–0.996)].
Relative efficacy matrix for PFS – primary analysis.

Each cell gives the effect of the column-defining intervention relative to the row-defining intervention. Colors represent direction of statistical significance (light green – column-defining intervention significantly better than row-defining intervention, light orange – column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; BEND, bendamustine; CLB, chlorambucil; CrI, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; PFS, progression-free survival; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
Similarly to BTKi, PFS was significantly improved by VEN + RTX compared with all chemoimmunotherapy regimens, anti-CD20 monotherapy, and PI3K-based therapies. No differences between VEN + RTX and therapies based on BTKi were identified, including a combination therapy of IBR with a novel anti-CD20 antibody – UBL [HR (95% CrI) = 1.56 (0.66–3.66)]. The combination therapy IBR + UBL was significantly better than the other therapies except for ZAN, VEN + RTX, IBR + RTX, and IBR + BEND + RTX. As for chemoimmunotherapy comparisons, we found that RTX, CLB + RTX, and PC had similar efficacy in improving PFS. On the other hand, BEND + RTX was associated with a significantly longer PFS compared with RTX [HR (95% CrI) = 0.44 (0.24–0.81)], OFA [HR (95% CrI) = 0.56 (0.32–0.98)], and PC [HR (95% CrI) = 0.44 (0.21–0.91)]. The SUCRA value indicating the greatest probability of outperforming the other regimens were the highest for IBR + UBL, ZAN, and VEN + RTX (0.96, 0.89, and 0.85).
Sensitivity analyses showed that most primary analysis results were consistent with IRC and investigator assessments (Supplemental Tables 13 and 14). The main differences for PFS assessed by IRCs were noted for OFA, IDE + OFA, and IDE + RTX results. In contrast to the primary analysis, the comparisons of IDE + OFA with ACA, IBR, IBR + BEND + RTX, VEN + RTX, and BEND + RTX suggested similar efficacy of treatments. Nonsignificant differences were observed for the following comparisons: IDE + RTX versus OFA, IDE + RTX versus PC, OFA versus BEND + RTX, and OFA versus PC. The highest SUCRA value for PFS assessed by IRCs was reported for IBR + UBL and VEN + RTX (0.92, 0.89). In the NMA for PFS assessed by investigators, differences for IDE + RTX versus RTX and CLB + RTX were nonsignificant, while anti-CD20 antibodies (OFA, RTX) were significantly better than PC.
Overall survival
As expected, most OS comparisons showed similar efficacy between treatments (Table 2). All monotherapies with BTKi and VEN + RTX significantly improved OS compared with RTX, OFA, and PC. ZAN and ACA were superior to DUV, while VEN + RTX had a longer OS than BEND + RTX [HR (95% CrI) = 0.40 (0.26–0.62)], CLB + RTX [HR (95% CrI) = 0.27 (0.11–0.66)], and DUV. Monotherapy with RTX showed a worse outcome than IDE + RTX [HR (95% CrI) = 2.22 (1.20–4.13)], IBR + BEND + RTX [HR (95% CrI) = 4.30 (1.04–18.02)], and IBR + UBL [HR (95% CrI) = 4.38 (1.64–11.76)]. BEND + RTX was also inferior to both triple therapies consisting of BCR inhibitors. As for PFS, the highest SUCRA values for OS were reported for VEN + RTX (0.93).
Relative efficacy matrix for OS – primary analysis.

Each cell gives the effect of the column-defining intervention relative to the row-defining intervention. Colors represent direction of statistical significance (light green – column-defining intervention significantly better than row-defining intervention, light orange – column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; BEND, bendamustine; CrI, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; PFS, progression-free survival; RTX, rituximab; UBL, ublituximab; VEN, venetoclax; ZAN, zanubrutinib.
Because the network for primary OS contained data from the ASCEND trial from a relatively short follow-up (16.1 months), we performed sensitivity analyses, including the results from the mixed treatment arm (IDE/BEND + RTX) for the 36-month follow-up, which resulted in 2 separate networks for IBR and BEND + RTX. The division of the network did not impact the results of the comparisons based on IBR network (Supplemental Tables 15 and 16), except for the difference in the comparison of DUV and ACA, which lost significance [HR (95% CrI) = 1.89 (0.99–3.58)]. The separate NMA via BEND + RTX also showed that CLB + RTX is associated with shorter patient survival than IDE + BEND + RTX [HR (95% CrI) = 2.38 (1.003–5.56)]. The absence of VEN + RTX in the sensitivity network resulted in higher SUCRA values for ACA and IBR + UBL (0.92).
Overall response rate
Networks for ORR were conducted separately for IRC and investigator assessments. Because the ORR in the ASCEND trial was reported only for the combined IDE/BEND + RTX, it was impossible to include studies in which BEND + RTX was the comparator arm.
NMA results for IRC assessments showed that patients treated with BTKi responded to treatment significantly more often than those receiving treatments based on PI3Ki or immunotherapy. Among BTKi, the ORR was significantly lower in patients treated with IBR compared to ZAN [RR (95% CrI) = 0.88 (0.81–0.95)] or IBR + UBL [RR (95% CrI) = 0.79 (0.61–0.96)]. No significant differences between ACA and IBR + UBL and ZAN were observed (Supplemental Table 17). In the sensitivity analysis for ORR with PRwL, the relative efficacy of all therapies remained the same except for IBR versus IBR + UBL [RR (95% CrI) = 0.84 (0.69–1.01)] and ACA versus IDE/BEND + RTX [RR (95% CrI) = 1.14 (1.03–1.27)] comparisons (Supplemental Table 18).
We found that ZAN and ACA were superior to IBR and standard immunotherapy based on investigator assessment. No differences were observed between ZAN and ACA [RR (95% CrI) = 1.04 (0.93–1.16)] or between these drugs and the IDE/BEND + RTX combined arm (Supplemental Table 19). Interestingly, differences between IBR and ACA diminished after PRwL was included in the ORR comparison [RR (95% CrI) = 0.97 (0.91–1.02)], while ZAN performed better than IBR + RTX combined therapy [RR (95% CrI) = 1.13 (1.002–1.269)] (Supplemental Table 20).
Serious adverse events
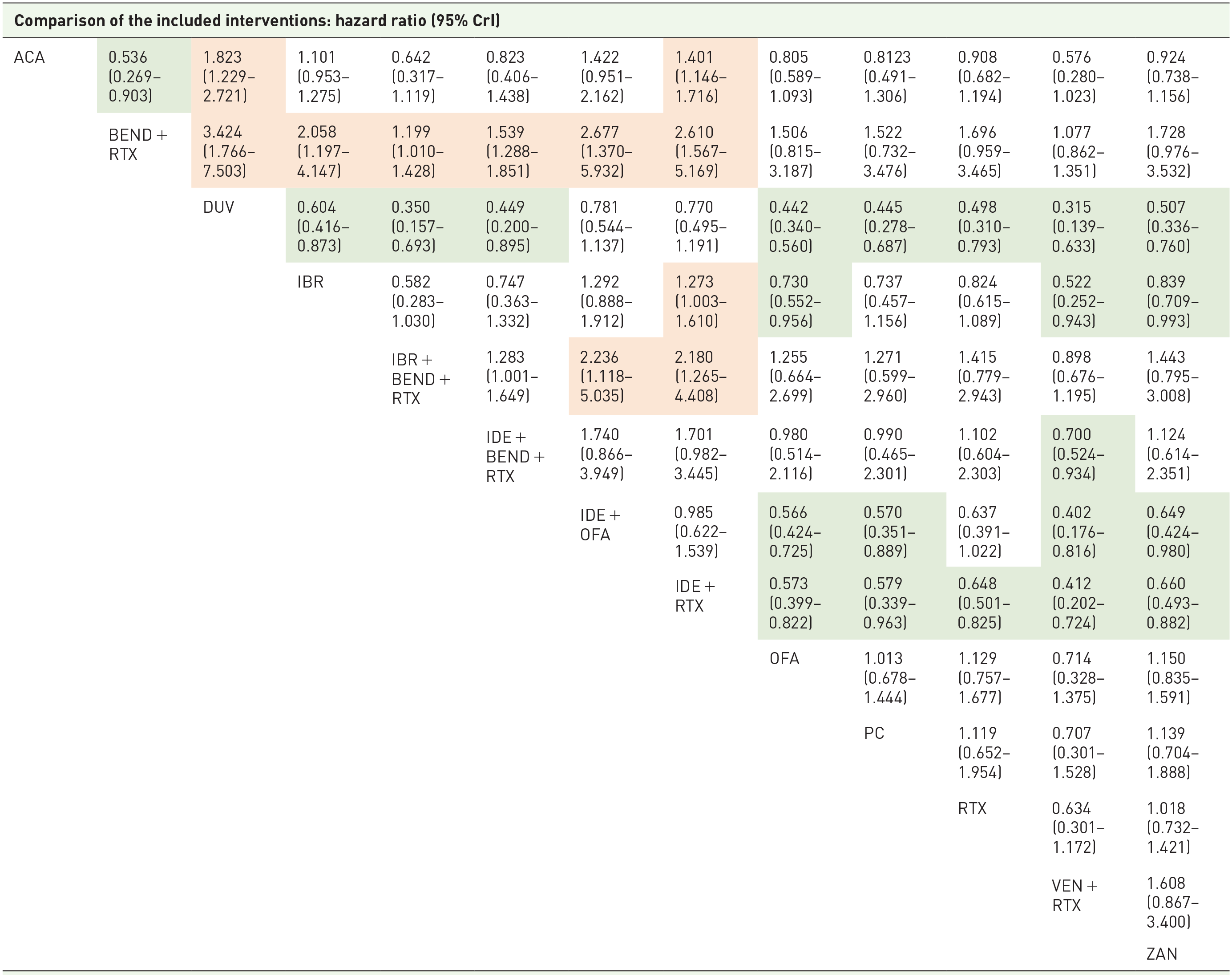
The BEND + RTX regimen showed the highest SUCRA values for SAEs (0.951), meaning it had the lowest probability of toxicity among all the treatments. The SAE rates for BEND + RTX were significantly lower than the other targeted therapies except for ZAN [RR (95% CrI) = 0.58 (0.28–1.02)] and VEN + RTX [RR (95% CrI) = 0.93 (0.74–1.16)] (Table 3). We found that VEN + RTX is a highly acceptable regimen, no different from anti-CD20 monotherapy, and is less toxic than all PI3Ki-based therapies and IBR.
Relative efficacy matrix for SAEs.
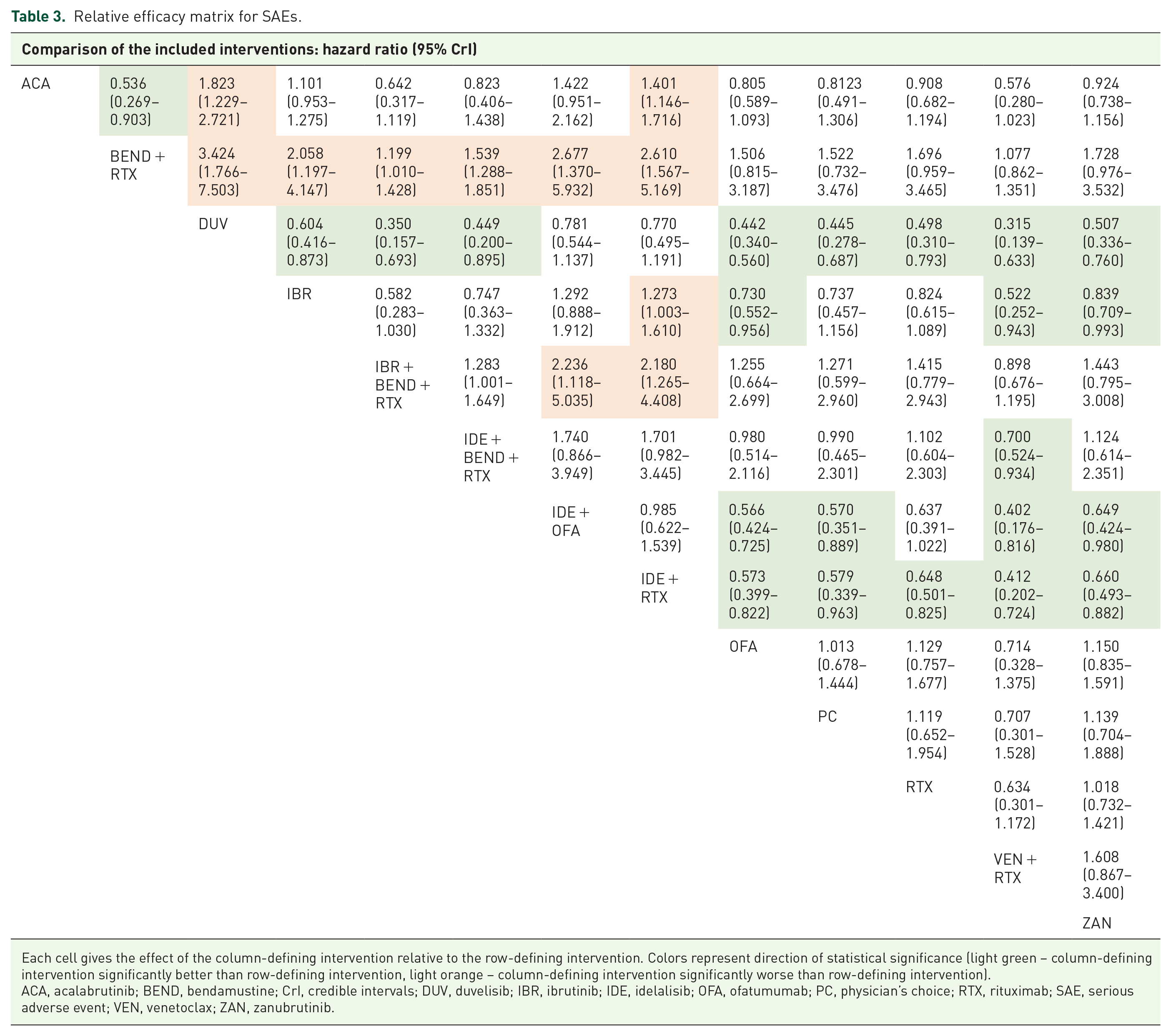
Each cell gives the effect of the column-defining intervention relative to the row-defining intervention. Colors represent direction of statistical significance (light green – column-defining intervention significantly better than row-defining intervention, light orange – column-defining intervention significantly worse than row-defining intervention).
ACA, acalabrutinib; BEND, bendamustine; CrI, credible intervals; DUV, duvelisib; IBR, ibrutinib; IDE, idelalisib; OFA, ofatumumab; PC, physician’s choice; RTX, rituximab; SAE, serious adverse event; VEN, venetoclax; ZAN, zanubrutinib.
DUV had the lowest SUCRA value for SAEs (0.02) and was inferior to chemoimmunotherapy, VEN + RTX, and BTKi therapies. However, its relative safety was comparable to IDE with anti-CD20 regimens. IBR and ACA showed similar SAE rates [RR (95% CrI) = 1.01 (0.95–1.28)], while IBR was inferior to ZAN [RR (95% CrI) = 1.19 (1.01–1.41)] and OFA [RR (95% CrI) = 1.37 (1.05–1.81)].
Overall benefit score
To examine the relative effectiveness of therapies, we conducted an exploratory cluster analysis based on SUCRA values for the following pairs: PFS and OS, OS and SAEs, and PFS and SAEs (Figure 2). The analysis showed that UBL + RTX, ZAN, and VEN + RTX had the highest probability of being the best treatment option for both PFS and OS. Moreover, VEN + RTX showed the best benefit-risk balance for efficacy and safety.

Clustered ranking plots of SUCRA values for OS, PFS, and SAE.
Discussion
Our study provided extensive data on the efficacy and safety of new targeted therapy, immunotherapy, and chemoimmunotherapy. Using an indirect comparison method, we showed that the therapies containing novel agents prolong PFS significantly compared with all immunotherapy and chemoimmunotherapy regimens included in this study. While this may be obvious for clinicians with extensive experience in managing patients with CLL, to our knowledge, this is the first study to provide firm evidence by examining a wide range of CLL therapies using Bayesian statistics. Additionally, our study showed that the VEN + RTX regimen has similar efficacy to therapies containing BTKi. Nevertheless, the probabilities based on SUCRA values suggest that VEN + RTX, ZAN, and IBR + UBL may be preferred regimens. These findings align with the most recent National Comprehensive Cancer Network guidelines, 65 which favor ZAN, VEN, and ACA for treating patients with relapsed/refractory CLL. The guidelines did not mention IBR + UBL as a recommended option, probably because UBL had not been approved for use yet.
In our research, therapies based on PI3Ki generally performed worse than those containing BTKi and VEN. This result may be due to the lower efficacy of PI3Ki but also partially because IDE- and DUV-treated patients (DUO, Study116, Study119) were a few years older and more pretreated than patients in other studies. Interestingly, we also noted that the statistical inference for some comparisons (e.g. based on IDE or OFA) changed depending on whether progression and response evaluations were performed by IRCs or by investigators. However, these differences may be partially assigned to the changes in network geometry due to data availability; it should be noted that significant discrepancies in the assessment between investigators and IRCs also occurred. For example, the median PFS for ACA in the ELEVATE-RR 36 study was about 8.5 months lower for IRC compared with the investigator’s assessment for the same follow-up. Differences were also observed in ORRs; for example, according to investigators, an additional 25% of patients treated with IBR showed a response in the RESONATE trial. 6
This study does not clarify which results (IRCs or investigators) should be considered more reliable. As most included studies were open-label, IRC assessments seem more objective and associated with a lower risk of bias. On the other hand, investigators usually provide data for longer follow-ups, which may be necessary for comparing treatments that do not produce rapid remissions but stabilize the clinical course of the disease. Another important aspect that influenced comparisons was the inclusion of PRwL in the ORR assessment. Our study revealed that potential significant differences between BTKi therapies observed by investigators diminished after PRwL was considered in the assessment.
During our systematic search, we identified a few attempts of NMA for relapsed/refractory CLL, but all were based on a limited number of trials. Molica et al. 66 indirectly compared VEN + RTX and BCR inhibitors added to BEND + RTX based on data from MURANO, TUGELA, and HELIOS trials. The authors reported that PFS for VEN + RTX and IBR + BEND + RTX were superior to IDE + BEND + RTX, which aligns with our results. Moreover, there were no significant differences in OS for all comparisons, as in our study. Recently, Chanan-Khan et al. 67 published a conference abstract describing an indirect comparison of ZAN, other BTKi, and VEN + RTX. As in our study, PFS assessed by investigators for ZAN therapy was longer than that for ACA and IBR but not VEN + RTX, while OS was similar for all therapies. Consistent results of the networks characterized by various geometries, models, and input data (different durations of follow-up) suggest that VEN + RTX and ZAN may be considered the preferred therapeutic options among the already approved regimens based on data from clinical trials.
Considering the limitations of our study, such as an indirect comparison and clinical trials as data sources, our results should be interpreted with caution. While we did not observe significant heterogeneity in the network and consistency between direct and indirect comparisons was satisfactory (Supplemental Tables 9–12), most of the obtained results were based only on indirect comparisons between single studies. Data availability was the primary concern in this research. The entire network was connected by the combined comparator arm of the ASCEND trial, and the availability of results for the IDE + RTX and BEND + RTX subgroups determined the network geometry and possibility of including all identified studies for comparisons. According to our protocol, we planned to examine the consistency of effects in different subgroups, for example, by lines of therapies, reasons for treatment initiation, and specific cytogenetic abnormalities. However, these ancillary analyses were not feasible due to the lack of reported data in many of the included trials. For example, only 7 of the 15 studies reported some outcomes, with the available results referring to different types of assessment (investigator-assessed or IRC) and other categories analyzed (e.g., 1 line versus >1 line of treatment, 1–3 versus >3 lines of treatment). To overcome this limitation, we calculated interaction tests for subgroup comparisons in individual studies, which showed no significant interactions in almost all cases (Supplemental Table 33). This suggests that relative effects between analyzed therapies remained comparable within treatment lines in primary trials. Nonetheless, we cannot exclude the potential impact of the treatment line and other factors on NMA results.
Another potential limitation is the risk of bias identified in the included studies. Although the methodological quality of the trials was usually adequate, the lack of blinding may have impacted the outcome assessment in some cases. The generalizability of clinical trial results to routine practice also raises concerns about the applicability of our research. 68 Although randomized clinical trials are considered the best available evidence, we cannot overlook that all studies have an inherent selection bias as they require subjects to provide informed consent to participate. Therefore, we look forward to the publication of observational studies reporting on the effectiveness of all novel targeted agents in real-world practice. By comparing real-world data from observational studies and NMAs, we will gain more insight into the relative efficacy and safety profiles of novel and standard therapies in relapsed/refractory CLL.
Despite the above limitations, our study has several strengths, such as the extensive literature search covering not only published articles but also the gray literature. Moreover, compared with previous NMAs, we used more mature data and included more trials. Our results may aid clinicians in the management of individual patients as well as healthcare authorities in making informed decisions regarding public health.
Conclusion
To conclude, our study showed that all novel therapies have better efficacy than chemoimmunotherapy and immunotherapy regimens. Among novel agents, the relative efficacy of VEN + RTX was similar to all BTKi, while ZAN was superior to IBR and comparable to ACA. Our results may aid clinicians in the management of individual patients as well as healthcare authorities in making informed decisions regarding public health.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241263710 – Supplemental material for Network meta-analysis of novel targeted therapies for relapsed/refractory chronic lymphocytic leukemia
Supplemental material, sj-docx-1-tam-10.1177_17588359241263710 for Network meta-analysis of novel targeted therapies for relapsed/refractory chronic lymphocytic leukemia by Magdalena Monica, Monika Reczek and Paweł Kawalec in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241263710 – Supplemental material for Network meta-analysis of novel targeted therapies for relapsed/refractory chronic lymphocytic leukemia
Supplemental material, sj-docx-2-tam-10.1177_17588359241263710 for Network meta-analysis of novel targeted therapies for relapsed/refractory chronic lymphocytic leukemia by Magdalena Monica, Monika Reczek and Paweł Kawalec in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.