Abstract
Background:
Extranodal natural killer/T-cell lymphoma (ENKTCL) has a unique treatment principle. However, the optimal combination of drugs along with radiotherapy (RT) is unknown.
Design:
Retrospective cohort study.
Objectives:
We screened multiple drug combinations to identify the most efficacious therapeutic combinations.
Methods:
We reviewed 3105 patients who received 40 chemotherapy regimens with different combinations of 9 drug classes and/or RT. Least absolute shrinkage and selection operator and multivariable Cox regression analyses were used to screen efficacious single drugs and identify optimal combinations for overall survival (OS). Inverse probability of treatment weighting (IPTW) and multivariable analyses were used to compare survival between treatment regimens.
Results:
Screening and validation revealed RT, asparaginase (ASP), and gemcitabine (GEM) to be the most efficacious single modality/drug. RT remained an important component of first-line treatment, whereas ASP was a fundamental drug of non-anthracycline (ANT)-based regimens. Addition of RT to non-ANT-based or ASP/GEM-based regimens, or addition of an ASP-drug into ANT-based or GEM/platinum-based regimens, improved 5-year OS significantly. Use of ASP/GEM-based regimens was associated with significantly higher 5-year OS (79.9%) compared with ASP/ANT-based (69.2%, p = 0.001), ASP/methotrexate-based (63.5%, p = 0.011), or ASP/not otherwise specified-based (63.2%, p < 0.001) regimens. The survival benefit of ASP/GEM-based regimens over other ASP-based regimens was substantial across risk-stratified and advanced-stage subgroups. The survival benefits of a combination of RT, ASP, and GEM were consistent after adjustment for confounding factors by IPTW.
Conclusion:
These results suggest that combining ASP/GEM with RT for ENKTCL is an efficacious and feasible therapeutic option and provides a rationale and strategy for developing combination therapies.
Introduction
Extranodal natural killer (NK)/T-cell lymphoma (ENKTCL) is the most prevalent subtype of peripheral T-cell lymphoma in China.1–3 It is associated with Epstein–Barr virus infection, mostly arises in the upper aerodigestive tract (UADT), and frequently presents with early-stage disease.4–6 The prognosis of ENKTCL has improved significantly over the last decade, largely as a result of the introduction and widespread use of upfront radiotherapy (RT) and non-anthracycline (ANT)-based chemotherapy.7–12 Five-year overall survival (OS) is 85%–92% for low-risk early-stage disease, 55%–80% for intermediate- and high-risk early-stage disease stratified by the nomogram-revised risk index (NRI), 6 and 30%–40% for advanced-stage ENKTCL.7–14
ENKTCL is unique among aggressive lymphomas in terms of treatment principle. It is primarily resistant to ANT-based chemotherapy,15,16 but sensitive to RT. 7 Non-ANT-based chemotherapy and RT are the backbone of first-line treatment for ENKTCL.7–14 To design efficacious chemotherapy regimens, various combinations of cytotoxic drugs have been explored, mainly depending on the consideration of pharmacological effect and mechanism of action.17–25 Previous studies have demonstrated that non-ANT-based chemotherapy can improve survival outcomes compared with ANT-based regimens.8–10,17–25 However, numerous non-ANT-based regimens,8–10,17–30 mainly asparaginase (ASP)-based regimens,8–10,19–21,24,25 have been recommended as first-line treatment. The optimal chemotherapy regimen has yet to be determined. Evidence supporting the clinical use of various chemotherapy regimens is limited to small series, variable enrolment criteria, heterogeneous stages, and treatment strategies. The introduction of highly efficacious agents has prompted significant considerations regarding the optimal combination of drugs and integration of RT.
We hypothesized that the efficacy of chemotherapy regimens was derived originally from a single agent, and a combination of several highly efficacious single agents is needed. 31 To fill a critical gap in knowledge between selection of efficacious drugs and combination therapies, we conducted a comprehensive analysis based on three aims. The first aim was to retrieve all available clinical data from the China Lymphoma Collaborative Group (CLCG) study observing ENKTCL patients treated with various chemotherapy regimens and RT. Second, we undertook a multidrug screening process to identify the most efficacious cytotoxic drugs. Third, we appraised the efficacy of various combinations of drugs and RT. Our study can provide evidence on the appropriate clinical utilization of new agents to improve the long-term outcomes of ENKTCL patients.
Patients and methods
Study population and treatment
We performed a retrospective analysis of patients with newly-diagnosed ENKTCL registered in the CLCG database between 2000 and 2017. Patients were eligible for study inclusion if they received definitive treatments. Patients treated with palliation or unknown chemotherapy regimens were excluded. In total, 3105 patients who met these criteria constituted the study population. The institutional review board approved the study and waived the need of informed consent because of deidentified patient data.
Patients were staged according to the Ann Arbor staging system, and stratified further by the NRI model. 6 Patients received chemotherapy, RT, or a combination of chemotherapy and RT according to stage and risk-stratified groups.11,12 In total, 2718 patients received chemotherapy with 40 regimens (Supplemental Table 1) and 2489 patients received RT (Table 1). Involved-site radiation therapy was given following the International Lymphoma Radiation Oncology Group guideline for ENKTCL. The median dose was 50 Gy for RT alone and 52 Gy for combined modality therapy (interquartile range, 50–56 Gy for both). The clinical target volume included the entire involved primary site and high-risk organs or structures of potential contiguous spread. Of 2102 patients who received combined modality therapy, 1913 patients (91.0%) received sequential chemoradiotherapy, while 189 patients (9.0%) received concurrent chemoradiotherapy. The median number of chemotherapy cycles was four for both sequential and concurrent chemoradiotherapy.
Clinical characteristics at baseline and treatment of 3105 patients.

ANT, anthracycline; ASP, asparaginase; CTX, cyclophosphamide; ECOG, Eastern Cooperative Oncology Group score; GCS, corticosteroids; GEM, gemcitabine; LDH, lactate dehydrogenase; MTX, methotrexate; NOS, not otherwise specified; PLA, platinum; PTI, primary tumor invasion; UADT, upper aerodigestive tract; VCR, vincristine; VP-16, etoposide.
Classification of cytotoxic drugs and chemotherapy regimens
Despite the heterogeneity of chemotherapy regimens, the most commonly used drugs can be classified into nine classes according to chemical composition (Figure 1): ANT; oxazaphosphorine-type of alkylating agents (cyclophosphamide (CTX), ifosfamide (IFO)); vinca alkaloids (vincristine (VCR), vindesine); ASP (L-asparaginase (L-ASP), pegaspargase (P-ASP)); pyrimidine analog (gemcitabine (GEM)); platinum (PLA; oxaliplatin (OXA), cisplatin (DDP), carboplatin); topoisomerase-II inhibitor (etoposide (VP-16)); anti-folate metabolite (methotrexate (MTX)); and glucocorticoids (GCS; prednisolone (PDN), dexamethasone (DXM), methylprednisolone). The nine drug classes are represented by agents of ANT, CTX, VCR, ASP, GEM, PLA, VP-16, MTX, and GCS, respectively, in the next analyses.

Classifier of single agents and chemotherapy regimens. The first panel presents the nine classes of drug and the most commonly used single agent. The Sankey diagram visualizes the flow of drugs from agent compounds through to regimen classifier. The second panel presents the drug combination based on hierarchical clustering after drug screening. Detailed chemotherapy regimens are presented under the classifier of drug combination. The third panel presents the regimen group of interest for survival comparison with the aim of finding the optimal drug combination or chemotherapy regimen.
Cytotoxic drugs could be classified into six combinations based on hierarchical clustering (Figure 1): ANT-based (CHOP (CTX, doxorubicin, VCR, PDN) or CHOP-like), ASP/ANT-based (ASP added to CHOP-like regimens), ASP/GEM-based (usually with PLA), ASP/MTX-based (usually with VP-16 and IFO), ASP/NOS-based (usually with VCR and CTX/IFO, without ANT; NOS, not otherwise specified), and PLA-based regimens (without ASP and ANT, usually with GEM, VP-16, or IFO). The corresponding chemotherapy regimens were summarized in Figure 1 and Supplemental Table 1. We included a CHOP-like regimen as a reference regimen to screen for efficacious drugs and compare survival outcomes.
Endpoints and statistical analyses
The primary endpoint was OS, defined as the time from first treatment to death from any cause. Given that the time of cure of ENKTCL patients can be within 5 years,32,33 5-year OS was considered the primary benchmark to assess the increased risk of mortality.
We mimicked a well-known omics analysis for multidrug screening process. The least absolute shrinkage and selection operator (LASSO) method was used to screen for an efficacious single drug from primary data, 34 because analyzing an individual variate with correlation and high dimensions is a powerful method. Cross-validation was used to confirm suitable tuning parameters (λ) for LASSO logistic regression.
Survival data were analyzed using the Kaplan–Meier method and compared with the log-rank test. The difference in clinical features between treatment arms was adjusted using inverse probability treatment weighting (IPTW). Multivariable Cox proportional hazards regression was carried out to identify independent prognostic factors, efficacious drugs, and treatment regimens. Statistical analyses were undertaken with SPSS 23.0 (IBM, New York, USA) and “survival,” “WeightIt,” and “RISCA” packages in R 4.1.2 (www.rproject.org/). The report of this study conforms to the STROBE statement (Supplemental Table 2).
Results
Clinical features
The clinical characteristics are summarized (Table 1). The male:female ratio was 2.4:1; median age was 44 years (range, 1–87). Most patients had the Eastern Cooperative Oncology Group (ECOG) score of 0–1 (92.7%) and primary site in the UADT (94.2%). A total of 55.6% of patients had primary tumor invasion (PTI), and 30.1% had an elevated lactate dehydrogenase (LDH). Most patients (88.8%) had early-stage ENKTCL, and 76.6% of them were classified into intermediate- and high-risk early-stage groups according to the NRI model. 6
Screening and selection of efficacious drugs using LASSO logistic regression
In the LASSO regression algorithm (Figure 2(a) and (b)), analysis was conducted on 18 clinical and treatment variables including 8 clinical features (sex, age, B symptoms, stage, PS, primary site, PTI, and LDH), 9 classes of drugs (ANT, CTX, VCR, ASP, GEM, PLA, VP-16, MTX, GCS), and RT for feature selection and drug screening. RT, ASP, and GEM were the most important treatment variables for OS (Figure 2(b)). Following exclusion by the LASSO analysis due to a coefficient of 0, RT, ASP, GEM, age, ECOG, LDH, stage, and PTI were recognized as independent factors (Figure 2(b)). The model yielded a C-statistic of 0.71. In multivariate Cox regression analysis, RT (hazard ratio (HR) = 0.45, 95% confidence interval (CI) = 0.39–0.53, p < 0.001), ASP (0.65, 0.55–0.75, <0.001), and GEM (0.58, 0.47–0.73, <0.001) were identified as independent predictive factors for OS (Figure 2(c)). In addition, old age, ECOG ⩾2, elevated LDH, PTI, and stage-II or III–IV were independent adverse disease-related factors (Figure 2(c)), consistent with previous finding in the NRI model. 6

Screening of cytotoxic agents and prognostic factors using LASSO logistic regression and their validation as independent factors of disease and treatment in multivariable Cox regression. (a) LASSO logistic regression in cross validation (1000 fold) was done with 18 texture features (i.e., eight clinical features (sex, age, B symptoms, stage, PS, primary site, PTI, LDH) and nine classes of agents (ANT, CTX, VCR, ASP, GEM, PLA, VP-16, MTX, GCS)) and RT. Two dotted vertical lines mark lambda (λ) at minimum and 1 standard error mean-squared error, respectively. A λ value of 0.034, with log (λ) of −3.381, were chosen according to cross validation (1000 fold). (b) LASSO coefficient profiles of the 18 texture features. A vertical line to the right was drawn at the value selected using cross validation, where optimal λ resulted in eight nonzero coefficients. (c) A forest plot indicated the independent prognostic effects of treatment and clinical variables on OS. The HRs (95% CIs) for OS in eight selected clinical and treatment features are presented using multivariable Cox regression analysis.
Role of RT as a single modality in risk- and stage-stratified patients
First, we evaluated the role of RT as a single modality in risk-stratified patients. RT alone resulted in favorable 5-year OS (~87.5%) in low-risk early-stage patients. The median follow-up time was 48.5 months. Adding an ANT-based regimen or non-ANT-based regimen to RT did not improve outcomes (p > 0.05; Figure 3(a)) even after adjustment for clinical covariates with IPTW (p > 0.05; Figure 3(b)).
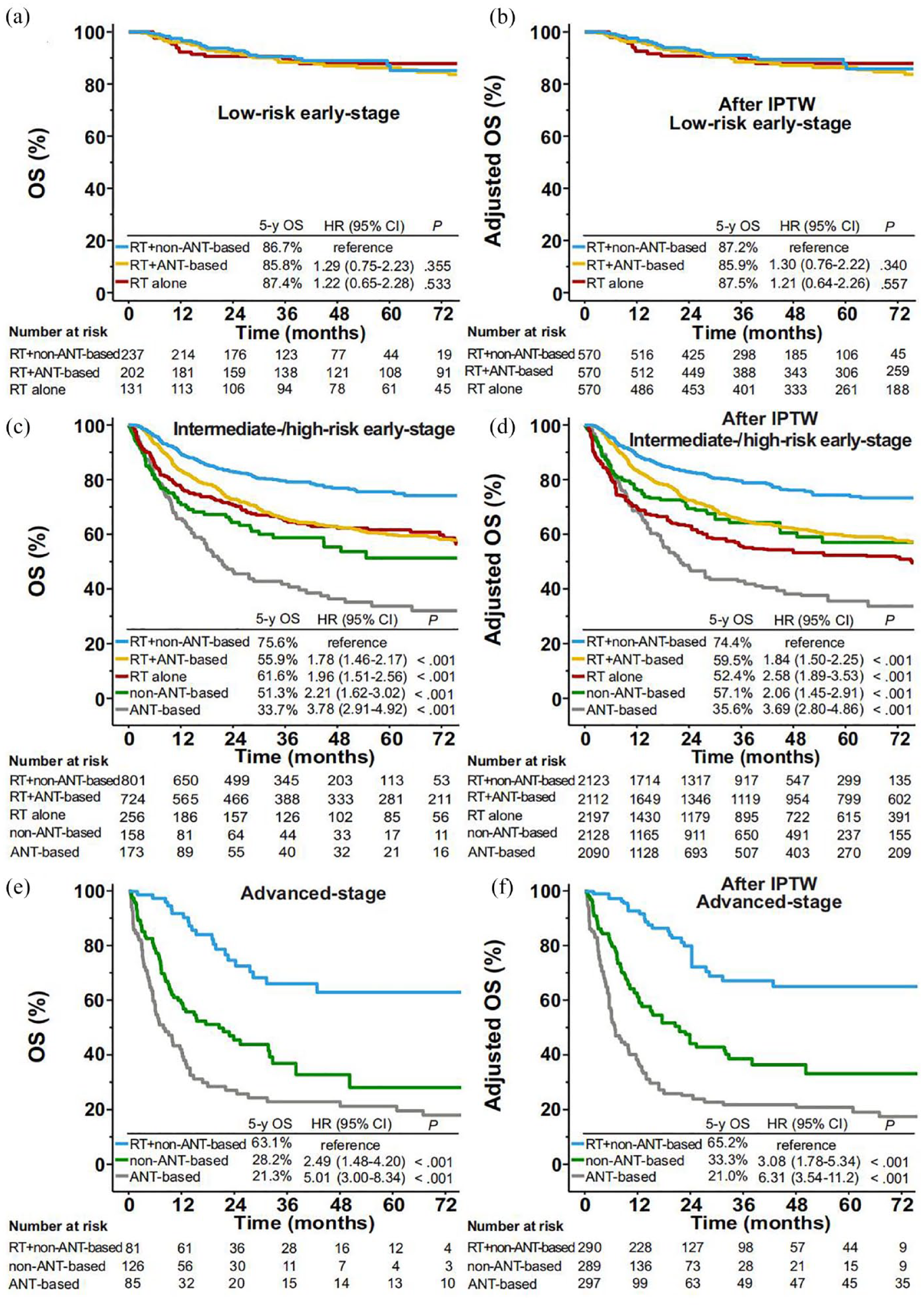
OS stratified by treatment groups in risk- and stage-stratified patients. OS for patients with low-risk early-stage ENKTCL stratified by RT alone, RT plus non-ANT-based regimens, and RT plus ANT-based regimens before (a) and after (b) adjustment with IPTW. OS for patients with intermediate and high-risk early-stage ENKTCL stratified by RT with or without ANT-based or non-ANT-based chemotherapy, chemotherapy alone with ANT-based or non-ANT-based regimens before (c) and after (d) adjustment with IPTW. OS for patients with advanced-stage ENKTCL stratified by RT plus non-ANT-based regimens and chemotherapy alone with ANT-based or non-ANT-based regimens before (e) and after (f) adjustment with IPTW.
In patients with intermediate-/high-risk early-stage ENKTCL, RT plus non-ANT-based regimens had significantly higher 5-year OS (~75%) than chemotherapy alone, RT alone, or RT plus ANT-based regimens (p < 0.001; Figure 3(c) and (d)). In advanced-stage patients, non-ANT-based regimens plus RT had significantly higher OS (~60%) than chemotherapy alone (p < 0.001) (Figure 3(e) and (f)). These results suggested that RT was an efficacious single modality regardless of stage or chemotherapy regimen.
Efficacy of ASP as a single drug in two-arm comparative analyses
Next, we assessed the efficacy of ASP as a single drug in a direct comparison. Adding ASP to CHOP-like or GEM/PLA-based regimens significantly improved survival. Five-year OS in the entire cohort was 69.3% for ASP-CHOP-like regimens compared with 57.6% for CHOP-like regimens (p < 0.001; Supplemental Figure 1(A)). After adjustment of covariates and RT, IPTW-adjusted 5-year OS was 69.7% for ASP-CHOP-like regimens compared with 57.4% for CHOP-like regimens (p < 0.001; Supplemental Figure 1(B)). A significant difference in OS between the two groups was also observed in patients with intermediate-/high-risk early-stage ENKTCL and advanced-stage ENKTCL (p < 0.05; Supplemental Figure 2).
Adding ASP to GEM/PLA-based regimens for the entire cohort had significantly higher 5-year OS (80.8% vs 70.7%, p = 0.002; Supplemental Figure 1(C)). IPTW-adjusted 5-year OS was 81.0% for ASP/GEM/PLA-based regimens compared with 59.3% for GEM/PLA-based regimens (p < 0.001; Supplemental Figure 1(D)). A significant difference in OS between the two groups was observed in patients with intermediate-/high-risk early-stage and advanced-stage ENKTCL (p < 0.05; Supplemental Figure 3). These results suggested the important role of a single ASP drug.
Survival benefit of ASP in combination with other drugs in multivariable analysis
A heatmap based on the correlation coefficients between nine classes of drugs was generated (Figure 4(a)). ANT exhibited positive correlations with CTX and VCR, but negative correlations with ASP, GEM, and PLA. ASP exhibited positive correlations with GEM, PLA, and MTX. Based on hierarchical clustering (Figure 4(b)), we identified six distinct drug combinations: ANT-based, ASP/ANT-based, ASP/GEM-based, ASP/NOS-based, ASP/MTX-based, and PLA-based regimens (Table 1 and Figure 1).

Optimizing the drugs combination and clustering of chemotherapy regimens. (a) Heatmap indicating the correlation between single drugs and RT using Kendall’s Tau correlation. Values in the grid present the correlation coefficients (blue = positive correlation; red = negative correlation). (b) Hierarchical clustering of nine classes of agents according to their chemical composition. (c) OS curves between different drug combinations were analyzed using a multivariable Cox regression model after adjustment for the variables of sex, age, B symptoms, ECOG score, primary site, PTI, LDH, stage, and RT. (d) A forest plot indicated the independent prognostic effects of treatment and clinical variables on OS. The HRs (95% CIs) for OS in five selected clinical features, RT, and drug combinations are presented using multivariable Cox regression analysis.
After adjustment for all covariates and RT in multivariable analysis, ASP-based regimens independently predicted superior OS (Figure 4(c)). Compared with ANT-based regimens, the HRs for OS with ASP/GEM-based, ASP/MTX-based, ASP/ANT-based, and ASP/NOS-based regimens were, respectively, 0.35 (95% CI: 0.27–0.45, p < 0.001), 0.61 (0.41–0.91, 0.018), 0.62 (0.50–0.78, <0.001), and 0.76 (0.60–0.96, 0.023; Figure 4(d)). These findings merited further evaluate survival differences between ASP-based regimens.
Survival benefit of ASP/GEM-based versus other ASP-based regimens
OS between ASP-based regimens was compared to identify optimal drug combinations. ASP/GEM-based regimens for the entire cohort had significantly higher 5-year OS (79.9%) compared with ASP/ANT-based (69.2%, p = 0.001), ASP/MTX-based (63.5%, p = 0.011), or ASP/NOS-based regimens (63.2%, p < 0.001; Figure 5(a)). After adjustment for covariates and RT with IPTW, similar differences in OS were observed between treatment groups (p < 0.05 for all; Figure 5(b)).

Comparison of OS between ASP-based regimens in stage- and risk-stratified patients. OS was compared between ASP/GEM-based regimens and three ASP-based regimens (based on ASP/MTX, ASP/ANT, and ASP/NOS) before (a) and after (b) IPTW in the entire cohort of patients. OS was compared between ASP/GEM-based regimens and other ASP-based regimens merged before (c) and after (d) IPTW in patients with intermediate-/high-risk early-stage ENKTCL. OS was compared between ASP/GEM-based regimens and other ASP-based regimens merged before (e) and after (f) IPTW in patients with advanced-stage ENKTCL. OS was compared between RT + ASP/GEM-based and ASP/GEM-based regimens alone before (g) and after (h) IPTW in the entire cohort of ENKTCL patients.
In patients with intermediate-/high-risk early-stage ENKTCL, ASP/GEM-based regimens had significantly higher 5-year OS than other ASP-based regimens (80.7% vs 68.0%, p < 0.001; Figure 5(c)). IPTW-adjusted 5-year OS was 81.1% for ASP/GEM-based regimens versus 68.4% for other ASP-based regimens (p < 0.001; Figure 5(d)). In advanced-stage patients, unadjusted and IPTW-adjusted 5-year OS were 63.0% and 57.6% for ASP/GEM-based regimens, compared with 36.9% (p = 0.012; Figure 5(e)) and 35.8% (p = 0.045; Figure 5(f)) for other ASP-based regimens. These results suggested the survival advantage of ASP/GEM-based regimens over ASP-drug conjugate MTX, ANT, or other drugs.
In an exploratory analysis, we evaluated the role of adding RT to ASP/GEM-based regimens. Five-year OS was 83.5% for RT + ASP/GEM-based regimens, significantly higher than that for ASP/GEM-based regimens: 62.7% (p = 0.04; Figure 5(g)). A similar result was observed after IPTW-adjustment for all covariates (83.7% vs 70.9%, p = 0.05; Figure 5(h)). This result confirmed the benefit of using RT in combination with efficacious ASP/GEM-based chemotherapy.
Survival benefit of ASP/GEM-based regimens in combination with RT
We evaluated the survival benefit of ASP/GEM-based chemotherapy in combination with RT. Patients receiving ASP-based chemotherapy alone were excluded. For the entire cohort, unadjusted and IPTW-adjusted 5-year OS were 83.5% and 84.3% for RT + ASP/GEM-based regimens compared with 74.7% (p < 0.001) and 74.2% (p < 0.001) for RT plus other ASP-based regimens. For intermediate-/high-risk early-stage patients, unadjusted and IPTW-adjusted 5-year OS were 83.3% and 83.5% for RT + ASP/GEM-based regimens compared with 72.7% (p = 0.005) and 72.8% (p = 0.006) for RT plus other ASP-based regimens.
Discussion
Due to the rarity of ENKTCL, identification of efficacious cytotoxic drugs and optimizing their combinations with RT as first-line treatment are challenging. In this real-world dataset, a broad combination of cytotoxic drugs and abundant sample size of patients permitted powerful multidrug screening process using LASSO logistic regression analysis. We identified RT, ASP, and GEM to be the most efficacious single modality/drugs in first-line treatment of ENKTCL. Adding RT to non-ANT-based or ASP/GEM-based regimens, or adding ASP to CHOP-like or GEM/PLA-based regimens, significantly improved OS. These results suggested that RT and ASP are the most important components of combination therapy for ENKTCL. Furthermore, ASP-drug conjugate (GEM, MTX, or ANT) resulted in superior OS compared with ANT-based regimens. The survival benefit of ASP/GEM-based regimens over other ASP-based regimens was substantial across risk-stratified and advanced-stage subgroups. These findings have therapeutic implications for selecting efficacious cytotoxic drugs and guiding the design of combination therapies in prospective studies.
Our study represents an important step toward understanding the differential impact of drug combinations on survival outcomes for ENKTCL. Over the past decade, chemotherapy regimens for ENKTCL have expanded rapidly,8–10,17–30 in parallel with increased understanding of the multidrug resistance (MDR) mechanisms and genomic heterogeneity that impact disease biology and clinical outcomes.16,35–37 Physicians provide recommendations on the selection and combination of investigative drugs for treatment of ENKTCL. Chemotherapy regimens varied widely between institutions and across countries.8–10,17–30 In this large cohort study from the CLCG database, 40 regimens with different combinations of 9 drug classes were applied as first-line chemotherapy for ENKTCL. After screening with LASSO logistic regression and validation by multivariable Cox regression, RT, ASP, and GEM were identified as the most efficacious single modality/drugs for newly-diagnosed ENKTCL.
Independent of MDR, the cytotoxic effects of RT and GEM (also including PLA and VP-16) are mediated by the interaction with and damage to DNA.38,39 A combination of RT and GEM shows a synergistic antitumor effect in vitro, 40 and reveals an immunomodulatory role in vivo.41,42 In contrast, an ASP drug is an anti-metabolic agent and shows promising anti-tumor effects on ENKTCL associated with aberrant metabolism of glutamine in tumor cells. 37 Although the precise mechanism underlying the drug combinations remains unclear, the combinations of ASP with either GEM and/or PLA, such as P-GEMOX (P-ASP, GEM, OXA) and DDGP (DXM, DDP, GEM, P-ASP), may exhibit a synergy or additive benefit, due to the lack of cross-resistance mechanisms. We showed that RT remained an important component of first-line treatment for ENKTCL. Studies have demonstrated that RT is the most effective single modality for ENKTCL in terms of locoregional control (LRC) and survival.7,11,12,43 In an early CLCG study, RT not only improves LRC but also prolongs survivals in a dose-dependent manner in early-stage patients. 44 There is a linear relationship between improved LRC by RT and prolonged progression-free survival and OS. 44 More importantly, we demonstrated that ASP is a fundamental drug of non-ANT-based regimens. Adding an ASP-drug to CHOP-like or GEM/PLA-based regimens significantly improved OS. However, early-stage patients who achieve a complete response after ASP-based chemotherapy alone have a significantly higher prevalence of disease relapse (5-year disease-free survival, 46.9%), 45 indicating that ENKTCL exhibits secondary resistance to ASP-based regimens. Adding RT to ASP-based chemotherapy significantly reduces the risk of disease relapse, thereby leading to improved disease-free survival and OS in these patients. 45 Modern combination therapies for ENKTCL are based on sound clinical and biological principles: RT is used to eradicate locoregional tumors and thereby prolong survivals44–46; novel ASP-based regimens are employed to overcome the multidrug-resistant P-glycoprotein on NK/T tumor cells 8 and reduce the risk of distant metastasis, 46 and eventually improve OS.13–15 By overcoming primary and secondary MDR or ASP resistance and the genetic heterogeneity of ENKTCL,35–37,45 a spatial combination exhibiting an independent antitumor action of drugs along with RT may confer survival benefit for patients. 31 Those findings, together with our data, offer context for the combined use of RT, ASP, and GEM in first-line treatment for ENKTCL.
Emerging evidence has shown that various ASP-based regimens significantly improve the survival outcomes of ENKTCL patients, but the optimal combinations of ASP with other non-MDR-related drugs have not been well-defined.10,20,21,26,28 In the present study, ASP/GEM combination regimens, most frequently used in China, resulted in 5-year OS of ~80%, which is significantly higher than that (~65%) of other ASP-based regimens, such as ASP/MTX-based, ASP/ANT-based, and ASP/NOS-based regimens. This clinical advantage of ASP/GEM-based regimens was also evident in patients with intermediate- and high-risk early-stage ENKTCL (~80%) and advanced-stage ENKTCL (~60%). Our results are consistent with findings from a recent randomized phase-II trial showing an OS benefit of the DDGP regimen in comparison with the SMILE (DXM, MTX, IFO, P-ASP, VP-16) regimen for 80 patients with Ann Arbor stage II–IV ENKTCL (5-year OS, 74.3% vs 51.7%). 25 Other single-arm or comparative studies have also demonstrated favorable survival outcomes with ASP/GEM-based regimens such as P-GEMOX or GELOX (GEM, OXA, L-ASP).10,20,21,26,28,45 A recent CLCG study revealed that the improved OS after first-line ASP/GEM-based chemotherapy and RT for intermediate- and high-risk early-stage patients was associated with decreased distant metastasis and increased distant metastasis-free survival, but not LRC, 46 indicating that the therapeutic efficacy of ASP drugs may mostly rely on control of distant occult disease. Thus, the promising efficacy and safety profile of ASP/GEM-based regimens reported in those studies support their combined use with RT,20,21,25,26 and suggest the feasibility of de-intensified first-line treatments for ENKTCL, especially in view of the difficulty in conducting relevant randomized trials. ASP/GEM-based chemotherapy regimens varied in this study. However, according to the present published data, 25 we preferred to use P-GEMOX or DDGP in clinical practice, which exhibited significant activity and acceptable toxicity. However, several challenges will need to be resolved through future prospective trials, including optimal doses and combinations of various drugs and immunotherapy, the sequencing of chemotherapy and RT, and identification of predictive biomarkers.
Our study has strengths and therapeutic implications. This is the first study to screen for efficacious single drugs and optimize drug combinations in a nationwide real-world cohort of ENKTCL patients. LASSO logistic regression was undertaken for multidrug screening process; this is the most powerful method for the regression analysis of high-dimensional and correlated predictors. A multidrug comparison provided insight on selection of chemotherapy regimens for ENKTCL patients based on cytotoxic drug- and disease-related factors. Despite the complexity of chemotherapy regimens, RT, ASP, and GEM were identified as the efficacious single modality/drug. The survival benefits of ASP/GEM-based chemotherapy and RT were verified after correcting for multiple covariates with IPTW and multivariable analyses. Additionally, these combinations exhibit independent antitumor action and show an immunomodulatory effect in cancers,39,41 and confer a synergistic anti-tumor effect in combination with anti-programmed cell death protein 1 (PD-1) therapy for ENKTCL. 47 Given the impressive response and durable efficacy of PD-1 blockade in patients with refractory/relapsed or advanced-stage ENKTCL,48–51 the synergistic anti-tumor and immunomodulatory effects of GEM and RT support their combination with immunotherapy in early-stage ENKTCL. Prospective trials evaluating the efficacy of a combination of ASP, GEM, RT, and a PD-1 inhibitor in these patients are ongoing (ClinicalTrials.gov identifiers: NCT04365036 and NCT05254899). 52
This retrospective study had two main limitations. First, treatment regimens were not assigned randomly. We attempted to reduce a selection bias using LASSO logistic regression, IPTW, and multivariable analyses, but the underlying confounders may have influenced treatment outcomes. Patients with low- or intermediate-risk early-stage ENKTCL tended to receive RT or de-intensified regimens, whereas patients with high-risk ENKTCL or those who were younger or with good PS were more likely to receive intensified regimens. Before generalizing a hypothetical survival benefit of the combination of ASP, GEM, and RT proposed in our study, further evidence from prospective clinical trials controlling confounding factors and from experimental studies investigating the mechanism of action are warranted. Second, toxicity profiles were not available in our study; the toxicity of various ASP-based regimens can affect clinical decision-making. However, studies have shown grade-3/4 toxicities (~80%) and treatment-related mortality (as high as 17.5%) with ASP/MTX-based regimens,8,9,19 though mostly in relapse/refractory or advanced-stage ENKTCL. In contrast, ASP/GEM-based regimens are well tolerated in patients with early-stage ENKTCL (if combined with RT),10,20,26 and even in advanced-stage ENKTCL. 25 The optimization of drug combination is essential to ensure that patients receive therapies which maximize efficacy while minimizing toxicity and treatment-related mortality. Given the severe toxicity8,9,19,25 and low risk of involvement of the central nervous system, 53 the role of incorporating MTX into ASP-based regimens is uncertain for first-line combination therapies for early-stage ENKTCL.
Conclusion
Combining ASP/GEM with RT as first-line treatment for ENKTCL appears to be an efficacious and feasible therapeutic option. These findings will provide a rationale and strategy for developing combination therapies in further prospective studies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-docx-2-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-docx-3-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-4-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-pdf-4-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-5-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-pdf-5-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-6-tam-10.1177_17588359241285981 – Supplemental material for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma
Supplemental material, sj-pdf-6-tam-10.1177_17588359241285981 for Optimizing the combination of chemotherapeutic drugs along with radiotherapy for extranodal NK/T-cell lymphoma by Fei Luo, Qiu-Zi Zhong, Xin Liu, Xiao-Rong Hou, Li-Ting Qian, Xue-Ying Qiao, Hua Wang, Yuan Zhu, Jian-Zhong Cao, Jun-Xin Wu, Tao Wu, Su-Yu Zhu, Mei Shi, Hui-Lai Zhang, Xi-Mei Zhang, Hang Su, Yu-Qin Song, Jun Zhu, Yu-Jing Zhang, Hui-Qiang Huang, Ying Wang, Xia He, Li-Ling Zhang, Bao-Lin Qu, Yong Yang, Chen Hu, Min Deng, Shu-Lian Wang, Shu-Nan Qi and Ye-Xiong Li in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank Dr Changfa Xia, PhD, associate researcher of the Office of Cancer Screening, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
