Abstract
Background:
Pegylated liposomal doxorubicin (PLD), epirubicin and pirarubicin are the main anthracyclines widely used in China. PLD demonstrates therapeutic response comparable to epirubicin and pirarubicin in neoadjuvant chemotherapy (NAC) of breast cancer.
Objectives:
The objectives of our study were to retrospectively assess the real-world effectiveness and prognostic characteristics of PLD as NAC for HR ⩽ 10%/human epidermal growth factor receptor 2 (HER2)-negative breast cancer.
Design:
This was a retrospective study.
Methods:
Our study enrolled patients with HR ⩽ 10%/HER2-negative breast cancer who received PLD-, epirubicin- or pirarubicin-based NAC from three centres in Hunan Province, China, between 2015 and 2022. We employed inverse probability of treatment weighting to balance the differences in patients’ characteristics among the PLD, epirubicin, and pirarubicin groups. The endpoints were pathological complete response (pCR), event-free survival (EFS), and overall survival (OS).
Results:
A total of 267 patients were included. After NAC, the pCR rates in PLD group were superior to epirubicin group (PLD, 34.1%; epirubicin, 20.8%, p = 0.038). The differences in EFS (log-rank p = 0.99) and OS (log-rank p = 0.33) among the three groups were not statistically significant. Among the three groups, non-pCR patients had worse EFS than pCR patients (log-rank p = 0.014). For patients with pCR, the differences in EFS (log-rank p = 0.47) and OS (log-rank p = 0.38) were not statistically significant among the three groups, and the EFS (log-rank p = 0.59) and OS (log-rank p = 0.14) of non-pCR patients in the PLD group were similar to those in the epirubicin and pirarubicin groups.
Conclusion:
PLD had a similar therapeutic response and prognosis compared to epirubicin or pirarubicin in NAC for patients with HR ⩽ 10%/HER2 negative breast cancer, which means that PLD represents a potential NAC option.
Keywords
Introduction
Breast cancer (BC), the most commonly diagnosed cancer worldwide, ranks first among the causes of cancer deaths in women. 1 Triple-negative breast cancer (TNBC) is defined as being negative for estrogen receptors (ERs) and progesterone receptors (PRs) and human epidermal growth factor receptor 2 (HER-2)2,3 and is characterized by limited therapeutic options and poor survival outcomes due to the lack of effective targeted drugs and its high metastatic potential.1,4–6 Currently, anthracycline- and taxane-based neoadjuvant chemotherapy (NAC), which effectively decreases the recurrence rates and enhances survival outcomes of early and locally advanced BC patients,7–9 has become the clinically preferred option for TNBC. 10
Pegylated liposomal doxorubicin (PLD), epirubicin and pirarubicin are the main anthracyclines widely used in China. Epirubicin and pirarubicin have been frequently used in NAC for TNBC. 11 It is reported that numerous significant clinical trials advocated epirubicin as a fundamental chemotherapeutic agent in neoadjuvant and adjuvant chemotherapies for invasive BCs.12–15 Nevertheless, the long-term clinical application of these anthracyclines is limited by severe adverse effects, including myelosuppression, gastrointestinal reactions, neutropenia, thrombocytopenia and cardiotoxicity.16–19 Furthermore, anthracycline-induced cardiotoxicities, which are closely associated with the dose-dependent increases in these drugs, are progressive and irreversible and can lead to death.20,21
PLD, a liposomal formulation of doxorubicin, exhibits similar effectiveness to conventional anthracycline and a markedly lower incidence of cardiotoxicity. 22–25 The National Comprehensive Cancer Network (NCCN) guidelines advocated PLD as the first-line treatment for advanced BC. Recently, some studies have found that the effectiveness of PLD in NAC is not inferior to that of traditional anthracyclines, such as epirubicin and pirarubicin. One study found that in the TNBC group, patients who received PLD as NAC had higher pathological complete response (pCR) rates than those who received epirubicin. 26 Another study revealed that, compared with patients receiving pirarubicin as NAC, those who received PLD demonstrated a significantly higher proportion achieving a major pathologic response according to the Miller–Payne grading (p = 0.047) and a reduced incidence of adverse cardiac events (p = 0.014). 27 In a retrospective study of neoadjuvant treatment, the PLD group presented lower incidences of myelosuppression, gastrointestinal reactions, and cardiotoxicity than the epirubicin group, and the difference in the pCR rate (p = 0.4578) was not statistically significant.10,28 Moreover, Manguso reported satisfactory effectiveness and a low incidence of toxicity when employing NAC that involved a low dose of PLD in combination with weekly paclitaxel in operable and locally advanced BC. 29 However, the efficacy and prognosis of PLD as NAC for patients with TNBC still lack real-world clinical evidence.
The current classification of HER2 status focuses on whether HER2 is overexpressed, and only patients with HER2-overexpressing BC receive anti-HER2 therapy. HER2-overexpressing BCs constitute 15%–20% of all BCs,30,31 while a much larger group is categorized as HER2-negative and is deemed unlikely to benefit from conventional anti-HER2 treatments. Many studies are now exploring the efficacy of anti-HER2 treatments in HER2-low BC, which includes those with HER2 IHC 1+ and IHC 2+ without gene amplification.32,33 Trastuzumab deruxtecan (T-Dxd), for instance, has been proven effective in HER2-low metastatic BC.34–36 However, current trials have focused primarily on metastatic BC,37–40 and research on neoadjuvant and adjuvant treatments remains under development.
According to previous studies, HR-low/HER2-negative BCs, defined as low positive expression of ER (1%–10%) with low positive or negative expression of PR, low positive expression of PR (1%–10%) with low positive or negative expression of ER, and HER2-negative status, are more likely to be similar to TNBCs in terms of molecular characteristics, gene expression patterns, rate of BRCA 1/2 mutations, effectiveness of NAC and survival outcomes.41–43 Thus, we also included and analysed HR-low/HER2-negative BC patients who received NAC.
In our real-world study, we compared the efficacy of PLD, epirubicin and pirarubicin as NAC and survival outcomes among these three groups in patients with HR ⩽ 10%/HER2-negative BCs.
Materials and methods
Patients
A total of 267 BC patients were collected in this retrospective study. They were from the Department of General Surgery of the Second Xiangya Hospital, the Department of Breast Surgery of Xiangya Hospital, and the Department of Breast Surgery of the First People’s Hospital of Xiangtan City in Hunan Province, China, from 2015 to 2022. Inclusion criteria: (1) female patients; (2) 20–80 years of age; (3) histologically proven primary HER2-negative BC (immunohistochemistry category 0 or 1, or 2+ with negative FISH results) 44 ; (4) stage II or stage III; (5) ER and PR ⩽10% (pathological reports that included a numerical value for percent ER and PR immunohistochemical staining were used); and (6) received PLD or epirubicin or pirarubicin as NAC. Exclusion criteria: (1) advanced and metastatic BC; (2) inflammatory or bilateral BC; (3) unclear or missing NAC regimens; and (4) incomplete follow-up data (Figure 1).

Trial design.
We extracted data regarding age, molecular subtype, menopausal status, lymph node status, clinical tumour stage, histological tumour type (IDC/other), tumour grade, NAC regimen (AC-T/TAC/other) and cycles, platinum-based NAC regimen, treatment with PD-1 blockade, pathological complete response (pCR), event-free survival (EFS) and overall survival (OS) from the database. The common chemotherapy regimens included PLD or epirubicin or pirarubicin combined with cyclophosphamide (C) followed by taxane (T); PLD or epirubicin or pirarubicin combined with T and C; PLD or epirubicin or pirarubicin combined with T, etc. (Table 1). The therapeutic dose was 30–35 mg/m2, 75–90 mg/m2 and 50 mg/m2 for PLD, epirubicin and pirarubicin, respectively. The American Joint Committee on Cancer (AJCC) system was employed as the basis to assess staging.
Patient characteristics.

IDC, invasive ductal carcinoma; Low-HR/HER2-negative BC, low hormone receptor/human epidermal growth factor receptor 2 negative breast cancer; NAC, neoadjuvant chemotherapy; pCR, pathologic complete response; PD-1, programmed death-1; PLD, pegylated liposomal doxorubicin; TNBC, triple-negative breast cancer.
Endpoints
pCR, EFS and OS were regarded as the clinical endpoints in this study. After neoadjuvant therapy, the absence of residual invasive BC tumours in the breast can be defined as pCR (ypT0/is). The definition of EFS was the time interval between the date of the initiation of NAC treatment and the first occurrence of the following events: local or distant recurrence, death from any cause, etc. The definition of OS was the time interval between the date of the initiation of NAC treatment and death from any cause. We assessed the disease with ultrasonography or magnetic resonance imaging (MRI) at baseline, once every two or three NAC cycles and once prior to surgery.
Statistical analysis
The mean is used to describe continuous variables, and the frequency is used to describe categorical variables. We employed T tests to evaluate age differences, and the clinicopathological features and pCR rates were assessed by Chi-square test among the PLD, epirubicin and pirarubicin groups. The correlation between each variable and pCR was observed by employing a logistic regression analysis, and Cox regression was employed to model OS and EFS among the three groups. The difference in baseline characteristics of the PLD, epirubicin and pirarubicin groups was balanced by using inverse probability of treatment weighting (IPTW). We used Kaplan‒Meier survival curves to calculate OS and EFS, and differences were assessed by the log-rank test. Statistical significance was defined as a two-sided p value < 0.05. All statistical analyses were conducted using R version 4.2.2.
Reporting guideline
The reporting of this study conforms to the Strengthening the reporting of observational studies in epidemiology (STROBE) statement (Supplemental Table 1). 45
Results
Patients’ characteristics
The PLD, epirubicin and pirarubicin groups were consisted of 85, 106 and 76 patients, respectively. The mean age at first diagnosis was 47.96 years in the PLD group, 46.38 years in the epirubicin group, and 47.80 years in the pirarubicin group. Premenopausal status (PLD, 54.1%; epirubicin, 66.0%; pirarubicin, 59.2%), TNBC (PLD, 72.9%; epirubicin, 81.1%; pirarubicin, 71.1%), T1 or T2 tumours (PLD, 75.3%; epirubicin, 65.1%; pirarubicin, 63.2%), positive pathological nodal status (PLD, 60.0%; eepirubicin, 65.1%; pirarubicin, 69.7%) and invasive ductal carcinoma (PLD, 91.8%; epirubicin, 92.5%; pirarubicin, 88.2%) accounted for a higher proportion in all three groups. In the PLD group, grade II and III tumours were both 43.5% of the total number of patients. A total of 46.2% and 40.6% of the patients in the epirubicin group were grade II and III, respectively, and 55.3% and 31.6% of the patients in the pirarubicin group were grade II and III, respectively. A total of 80.0% of the patients in the PLD group, 54.7% of the patients in the epirubicin group and 65.8% of the patients in the pirarubicin group were treated with no less than six cycles of NAC. Almost all patients had received TAC or AC-T as an NAC regimen (PLD, 100.0%; epirubicin, 95.3%; pirarubicin, 96.1%). A minority of the patients received platinum (PLD, 11.8%; epirubicin, 10.4%; pirarubicin, 17.1%) and PD-1 blockade (PLD, 5.9%; epirubicin, 3.8%; pirarubicin, 5.3%) combined with NAC. These three groups, as shown in (Table 1), were consistent with each other in the clinical characteristics after IPTW.
Clinical response and survival
In this study, 34.1% of patients in the PLD group achieved pCR, whereas the pCR rates were 20.8% and 30.3% among patients in the epirubicin group and pirarubicin group, respectively. Notably, compared with the epirubicin group, the PLD group demonstrated a higher pCR rate (p = 0.038; Figure 2). The univariate logistic regression analysis showed that patients from the PLD group were significantly more likely to achieve pCR than those from the epirubicin group (OR = 0.51, 95% CI: 0.26–0.97, p = 0.040), and NAC treatment cycles (OR = 2.09, 95% CI: 1.13–3.85, p = 0.019) and the combination of PD-1 blockade with NAC (OR = 6.54, 95% CI: 1.95–21.96, p = 0.002) were also significantly associated with pCR. The multivariate logistic regression analysis demonstrated that the combination of PD-1 blockade with NAC (OR = 5.46, 95% CI: 1.59–18.74, p = 0.007) remained independently associated with pCR, but PLD group was not significantly associated with pCR (epirubicin: OR = 0.58, 95% CI: 0.29–1.13, p = 0.110; pirarubicin: OR = 0.90, 95% CI: 0.45–1.79, p = 0.760) (Table 2). In multivariate Cox regression analysis, pathological nodal status (HR = 3.34, 95% CI: 1.68–7.00, p < 0.001), histological tumour type (HR = 2.33, 95% CI: 1.26–4.30, p = 0.007) and pCR (HR = 0.31, 95% CI: 0.13–0.73, p = 0.007) were independently associated with EFS (Table 3). In addition, clinical tumour stage (HR = 3.24, 95% CI: 1.25–8.44, p = 0.016) and NAC treatment cycles (HR = 0.25 95% CI: 0.08–0.73, p = 0.011) were independently associated with OS (Table 4).

pCR (ypT0/is) among the PLD, epirubicin and pirarubicin groups.
Univariate and multivariate logistic regression analyses of pCRa.
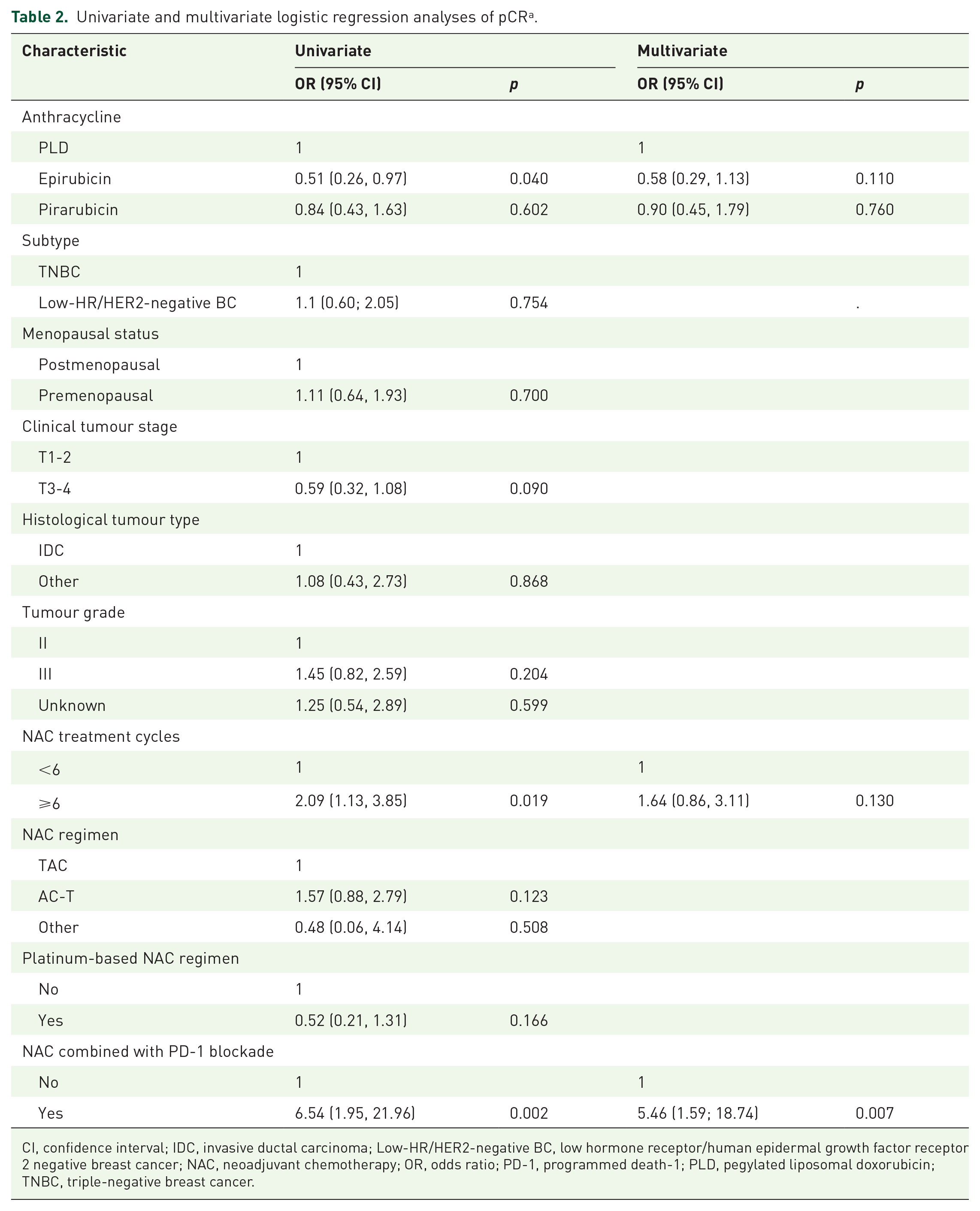
CI, confidence interval; IDC, invasive ductal carcinoma; Low-HR/HER2-negative BC, low hormone receptor/human epidermal growth factor receptor 2 negative breast cancer; NAC, neoadjuvant chemotherapy; OR, odds ratio; PD-1, programmed death-1; PLD, pegylated liposomal doxorubicin; TNBC, triple-negative breast cancer.
Univariate and multivariate Cox regression analyses of EFS.

CI, confidence interval; EFS, event-free survival; HR, hazard ratio; IDC, invasive ductal carcinoma; Low-HR/HER2-negative BC, low hormone receptor/human epidermal growth factor receptor 2 negative breast cancer; NAC, neoadjuvant chemotherapy; pCR, pathologic complete response; PD-1, programmed death-1; PLD, pegylated liposomal doxorubicin; TNBC, triple-negative breast cancer.
Univariate and multivariate Cox regression analyses of OS.

CI, confidence interval; HR, hazard ratio; IDC, invasive ductal carcinoma; Low-HR/HER2-negative BC, low hormone receptor/human epidermal growth factor receptor 2 negative breast cancer; NAC, neoadjuvant chemotherapy; OS, overall survival; pCR, pathologic complete response; PD-1, programmed death-1; PLD, pegylated liposomal doxorubicin; TNBC, triple-negative breast cancer.
EFS and OS were used to evaluate patient prognosis. The difference regarding either EFS (log-rank p = 0.99) or OS (log-rank p = 0.33) was not statistically significant among the three groups (Figure 3). Further survival analyses were conducted for those with pCR or non-pCR among the three groups. When EFS and OS were analysed with categories of pCR (yes/no), no significant differences were observed among the three groups for patients who achieved a pCR after NAC (EFS, log-rank p = 0.47; OS, log-rank p = 0.38). Similarly, EFS (log-rank p = 0.59) and OS (log-rank p = 0.14) were not significantly different in non-pCR patients among the three groups. In addition, among the three groups, the difference in OS (log-rank p = 0.095) between patients with pCR and non-pCR was not statistically significant. However, non-pCR patients in the three groups showed a worse EFS than those with pCR (log-rank p = 0.014; Figure 4).

(a) EFS before IPTW of all patients from the PLD, epirubicin and pirarubicin groups. (b) OS before IPTW of all patients from the PLD, epirubicin and pirarubicin groups. (c) EFS after IPTW of all patients from the PLD, epirubicin and pirarubicin groups. (d) OS after IPTW of all patients from the PLD, epirubicin and pirarubicin groups.
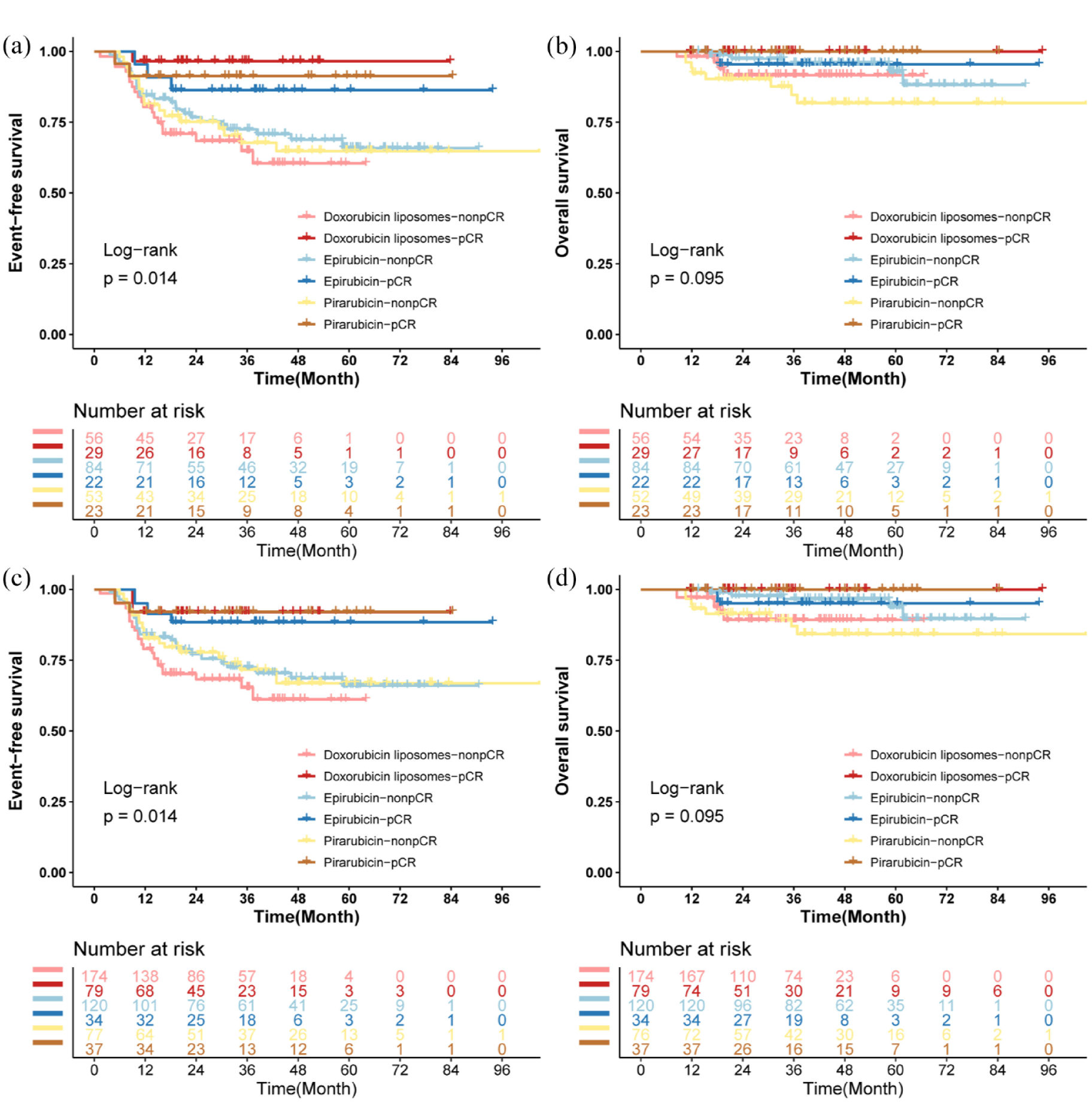
(a) EFS before IPTW in patients with pCR and non-pCR from the PLD, epirubicin and pirarubicin groups. (b) OS before IPTW in patients with pCR and non-pCR from the PLD, epirubicin and pirarubicin groups. (c) EFS after IPTW in patients with pCR and non-pCR from the PLD, epirubicin and pirarubicin groups. (d) OS after IPTW in patients with pCR and non-pCR from the PLD, epirubicin and pirarubicin groups.
Discussion
Anthracycline- and taxane-based NAC regimens, such as AC-T and TAC, contributed to an increase in the pCR rate and improved survival in TNBC patients. Doxorubicin was the first anthracycline drug used to treat TNBC, but its cardiotoxicity has led to great limitations. 46 PLD is an increasingly frequently used anthracycline drug given its minimal toxicity while ensuring treatment effectiveness for advanced BC.24,25,47 However, the clinical evidence supporting PLD as NAC in the treatment of TNBC is insufficient. In addition, due to the similar efficacy and survival outcomes exhibited by HR-low/HER2-negative BC patients and TNBC patients after NAC,41–43 we focused simultaneously on the treatment of HR-low/HER2-negative BC. Consequently, we conducted a real-world multicentre cohort study to evaluate the effectiveness of PLD as NAC in HR⩽10%/HER2-negative BC patients.
A retrospective study comparing treatment response in BC patients receiving NAC showed that the pCR rates (16.3% vs 11.6%, p = 0.317) in the PLD group was 4.5% superior to those in the epirubicin group, while no significant differences were observed. 48 Another study also found that, compared with those in the epirubicin group, patients in the PLD group achieved higher overall pCR rates (p = 0.034) and radiological complete response rates (p = 0.044); regarding TNBC patients, the pCR rates were higher in the PLD group (p = 0.013). 26 However, the fact that the TNBC subgroup contains only 60 individuals is a limitation of the study. In our study, univariate logistic regression analysis revealed that the PLD group was significantly more likely to achieve pCR compared with the epirubicin group (OR = 0.51, 95% CI: 0.26–0.97, p = 0.040; Table 2), with a significantly higher pCR rate (p = 0.038; Figure 2). However, in the multivariate logistic regression analysis, the PLD group was not significantly associated with pCR, which may be related to NAC treatment cycles, the combination of PD-1 blockade with NAC and the limited sample size. Therefore, expanding the sample size is essential in future studies. The pCR rates in the three groups were relatively low in our study, but the rates aligned with those from other real-world studies of the effectiveness of NAC in TNBC.49–52 In the Cox regression analysis, the negative pathological nodal state and IDC were independently associated with better EFS, and the lower clinical tumour stage (T1 and T2) was independently associated with better OS, which aligned with the results of previous studies.53,54 pCR was independently associated with better EFS but not with OS, which may have been affected by the limited follow-up time. More NAC treatment cycles were independently associated with better OS. This may be explained by the fact that patients who respond better to NAC are more inclined to complete the full treatment course, whereas those with poorer treatment responses tend to opt for early surgical intervention.
In terms of survival outcomes, a retrospective study found no significant differences between the PLD group and epirubicin group in 3-year DFS (81.5% vs 88.9%, p = 0.363) or OS (88.9% vs 90.7%, p = 0.85). 47 Our findings corroborate these findings that there was no significant difference in EFS (log-rank p = 0.99) or OS (log-rank p = 0.33) after IPTW among the three groups, indicating that the efficacy of PLD as NAC was comparable to that of conventional anthracyclines in BC. Further analysis of the three groups of patients with pCR and non-pCR was conducted. There was no significant difference in EFS (log-rank p = 0.47) or OS (log-rank p = 0.38) for patients with HR ⩽ 10%/HER2-negative BC who achieved pCR among the PLD, epirubicin and pirarubicin groups. Similar results were observed in patients with non-pCR among these three groups (EFS, log-rank p = 0.59; OS, log-rank p = 0.14). No statistically significant difference in OS (log-rank p = 0.095) was observed between all patients with pCR and non-pCR, whereas EFS in patients with non-pCR was significantly worse compared to that in patients with pCR (log-rank p = 0.014).
By comparing the therapeutic response and prognosis among patients receiving PLD, epirubicin, and pirarubicin as NAC, we observed that PLD demonstrated superior efficacy to epirubicin and comparable efficacy to pirarubicin. These findings suggest that PLD may serve as an effective NAC regimen. Reports from several clinical studies have illustrated similar viewpoints to our work and expressed safety benefits from PLD, such as lower incidences of myelosuppression, decreased appetite, gastrointestinal reaction, and cardiotoxicity.24,55 However, most of the previous similar studies were single-centre cohort studies. Moreover, to our knowledge, this is the first real-world multicentre study comparing the effectiveness of PLD, epirubicin, and pirarubicin as NAC simultaneously for HR < 10%/HER2-negative BC, which may be more representative. The current results suggest that PLD-based NAC is a potential option for early BC patients, and the long-term benefit of this neoadjuvant treatment regimen warrants further investigation.
Our retrospective study has several limitations. First, the small sample size and limited follow-up time lead to a possible lack of representativeness of the results to some extent; thus, a sufficient sample size and long-term follow-up are necessary. In addition, we acknowledge the potential ethnic bias in patient enrolment, as all our participants were from Hunan Province, China, and the sample size was relatively limited. Therefore, expanding the study population and increasing the sample size are necessary for future studies. Moreover, due to its retrospective nature, this study lacked complete data on the side effects of these three drugs, which could inevitably produce selection bias and confounding factors. Furthermore, because PD-1 inhibitors have been applied in NAC in recent years and the time span of our inclusion was long, only 13 patients had received NAC with PD-1 inhibitors in our study.
Conclusion
In this real-world multicentre cohort study, compared to epirubicin and pirarubicin, PLD showed a similar therapeutic response and prognosis in NAC treatment for patients with HR ⩽ 10%/HER2-negative BC, revealing the potential of PLD to be an effective NAC option despite the need for further validation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241279695 – Supplemental material for Comparison of neoadjuvant chemotherapy response and prognosis among pegylated liposomal doxorubicin, epirubicin and pirarubicin in HR ⩽ 10%/HER2-negative breast cancer: an exploratory real-world multicentre cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359241279695 for Comparison of neoadjuvant chemotherapy response and prognosis among pegylated liposomal doxorubicin, epirubicin and pirarubicin in HR ⩽ 10%/HER2-negative breast cancer: an exploratory real-world multicentre cohort study by Yue Hong, Jing Peng, Qitong Chen, Qin Zhou, Feng Xu, Jia Yao, Qiongyan Zou, Liqin Yuan, Lun Li, Qian Long, Liqiu Liao, Mingwen Liu, Xuan Liu, Danhua Zhang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
