Abstract
Background:
Patients with multiple myeloma (MM) are at risk of venous thromboembolism (VTE), worsened by immunomodulatory drugs. Although antithrombotics are recommended for prophylaxis, existing guidelines are suboptimal and treatment outcomes remain unclear.
Objectives:
This study aimed to investigate adverse events, antithrombotic utilization, and their associations with survival outcomes in patients with MM initiating multi-drug immunomodulatory combinations.
Design:
A posthoc analysis of individual-participant level data (IPD).
Methods:
IPD from three daratumumab clinical trials (MAIA, POLLUX, and CASTOR) were pooled. Adverse events incidence and antithrombotic utilization were assessed. Logistic and Cox regression were utilized to examine associations between antithrombotics use with adverse events and survival outcomes at the baseline and 6-month landmark.
Results:
Among 1804 patients, VTE occurred in 10%, bleeding in 14%, ischemic heart disease in 4%, and stroke in 2%. Patients with these adverse events demonstrated elevated rates of any grade ⩾3 events. Antiplatelet (primarily aspirin) and anticoagulant (primarily LMWH and direct oral anticoagulants) prescriptions have seen an increase from baseline (25% and 14%, respectively) to 6 months (35% and 31%). The primary indication for their use was prophylaxis. Anticoagulant use within 6 months was associated with reduced VTE (OR (95% CI) = 0.45 (0.26–0.77), p = 0.004), while antiplatelet use showed no associations with any evaluated adverse events. Antithrombotics and survival outcomes had no significant associations.
Conclusion:
This study underscores the complexities of antithrombotic therapy and adverse events in MM and highlights the need for vigilant and proactive management due to increased grade ⩾3 adverse events. While anticoagulant use was associated with reduced VTE risk, further research is needed to optimize thromboprophylaxis guidelines and explore antithrombotic efficacy and safety in patients with MM.
Trial registration:
MAIA (NCT02252172), POLLUX (NCT02076009), CASTOR (NCT02136134).
Plain language summary
This study aimed to understand how blood-thinning medications are used by patients with multiple myeloma, a type of blood cancer. Specifically, we wanted to find out how often these medications are used, what side effects they might cause, and whether they are linked with how long the patients live.
This study is important because patients with multiple myeloma often have a higher risk of blood clots, especially when they are taking certain anticancer treatments. Blood-thinning drugs are usually recommended to prevent these clots, but it’s not always clear how well these drugs work or what side effects they might cause.
This study looked at data from three clinical trials involving a multiple myeloma drug called daratumumab. We looked at how often side effects occurred and how often blood-thinning drugs were used. Two groups of blood thinning drugs were investigated: antiplatelets and anticoagulants. We used two types of statistical methods, called logistic and Cox regression, to see if there was a connection between the use of these blood-thinning drugs and the occurrence of side effects or survival rates at the start of the study and after six months.
The study found that the use of blood-thinning drugs increased over time and that using anticoagulants within the first six months was linked to a lower risk of blood clots. However, blood-thinning drugs were not linked with how long the patients lived. These results are important because they can help doctors better manage the use of blood-thinning drugs in patients with multiple myeloma. The key message is that more research is needed to improve guidelines for preventing blood clots and to better understand the safety and effectiveness of blood-thinning drugs in these patients.
Keywords
Introduction
In the last decade, significant advancements have transformed the therapeutic landscape for patients with multiple myeloma (MM) with the introduction of immunomodulatory drugs (IMiDs), proteasome inhibitors, and monoclonal antibodies. Despite these strides, the administration of these agents has presented distinct challenges, particularly in managing adverse events such as venous thromboembolism (VTE), cardiac events, or stroke.1–5 Notably, patients with MM face a VTE risk that is 7–9 folds higher than non-MM patients do. 4 Additionally, treatment with IMiDs in conjunction with dexamethasone (DEX) further increases the risk of VTE in MM patients initiating these regimens, resulting in a mean VTE incidence of 21.5% in studies devoid of thromboprophylaxis. 5 VTE and pulmonary embolism in MM are correlated with increased mortality, 4 with death rates being three folds higher compared to patients with MM but without these complications. 6 To address this concern, several scientific panels proposed a VTE risk assessment guide to stratify patients with MM based on patients’ disease, or therapy-related risk factors, and recommend prophylactic use of antithrombotic drugs such as aspirin, warfarin, or low-molecular-weight heparin (LMWH) for patients with ⩾1 risk factors.1,7–9
Despite the availability of clinical guidelines, determining optimal thromboprophylaxis and treatment choice for VTE events in patients with MM remains a clinical challenge.1,8–10 An analysis published in 2020 on patients with MM initiating IMiDs reported a VTE rate of 13.2% despite 80.5% of patients being on thromboprophylaxis. 11 While numerous studies have reviewed the efficacy and safety of antithrombotic drugs including aspirin, warfarin, LMWH, and direct oral anticoagulants (DOAC) in patients with MM, the data is limited. Comparative effects of various antithrombotic drugs concerning VTE, or bleeding events, remain inconclusive and cannot confirm or exclude beneficial or detrimental effects.1,11,12 Hence, existing scientific literature on thromboprophylaxis in MM falls short in providing evidence-based recommendations. 7
The association of antithrombotic drugs with survival outcomes in patients with MM remains uncertain. 13 Several studies on solid tumors, such as head and neck cancers, report significant associations between the use of different anticoagulants and increased overall or cancer-specific survival.14–16 Moreover, literature suggests that widely used anticoagulants, such as LMWH and warfarin, or antiplatelets such as aspirin, may exhibit anticancer effects, hinting at their potential use to improve survival and prevent cancer progression or metastasis.14,17,18 Conversely, other studies report worse survival outcomes or inconclusive results with concomitant anticoagulant use.19–22
The aims of this study were to (1) investigate the incidence of adverse events, including VTE, ischemic heart diseases, stroke, and bleeding, associated with antithrombotic treatment, and assess whether there is a significant difference in the pattern of these adverse events between MM treatment types, (2) assess the incidence of documented use and indications for antithrombotics in patients with MM initiating daratumumab, lenalidomide, or bortezomib combination treatments, and (3) investigate the association of antithrombotic therapy with adverse events and survival outcomes.
Ultimately, this study aims to elucidate the complex interplay between antithrombotics, adverse events, and survival outcomes and provide insights into optimizing treatment strategies and improving outcomes in patients with MM.
Methods
Patient population
Individual patient data were pooled from three randomized, open-label trials: MAIA (NCT02252172, data cut-off: February 19, 2021), 23 POLLUX (NCT02076009, data cut-off: March 7, 2016), 24 and CASTOR (NCT02136134, data cut-off: January 11, 2016). 25
All these studies enrolled patients who were 18 years or older. The efficacy of daratumumab on patients with relapsed or refractory MM who had undergone at least one prior line of therapy was evaluated in the POLLUX and CASTOR trials. The MAIA trial enrolled newly diagnosed MM patients who were not candidates for high-dose chemotherapy or autologous stem cell transplantation due to their age (65 years or older) or the presence of coexisting conditions that could lead to unacceptable side effects.
In the MAIA and POLLUX trials, patients were treated with a combination of daratumumab (administered as 16 mg/kg IV infusion), lenalidomide (25 mg capsule taken orally), and dexamethasone (40 mg taken orally or intravenously; DRd). This was compared to a regimen of lenalidomide plus dexamethasone (Rd). In the CASTOR trial, daratumumab (16 mg/kg IV infusion) was combined with bortezomib (1.3 mg/m2 administered subcutaneously) and dexamethasone (20 mg taken orally; DVd) and compared to a regimen of bortezomib plus dexamethasone alone (Vd).
All trials adhered to the International Conference on Harmonisation Good Clinical Practice guidelines and the Declaration of Helsinki.26,27 Participants provided written informed consent. The secondary analysis of de-identified data reported in this study was considered negligible risk research and has been approved by the University of Sharjah Ethics Committee (Approval reference number: REC-23-11-07-01-F). Data were accessed according to the Johnson & Johnson policy and made available through Vivli, Inc. (www.vivli.org). The reporting of this study adhered to The Strengthening the Reporting of Observational Studies in Epidemiology Statement Guidelines (Supplemental Table 2). 28
Outcomes
The primary objectives were as follows: (a) the incidence of adverse events that encompassed VTE, ischemic heart disease, bleeding, and stroke occurring throughout treatment duration and (b) the incidence of documented use and indications for antithrombotics (anticoagulants and antiplatelets) at baseline (i.e., prior to treatment initiation) and within a landmark of 6 months from treatment initiation. Adverse events were reported in MAIA, POLLUX, and CASTOR trails using the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTAE) version 4.0. 29
The secondary objective was to investigate the association of antithrombotic use with overall survival (OS), progression-free survival (PFS), and adverse events at baseline and 6-month landmark. OS was defined as the time from the date of randomization to the date of the participant’s death, and PFS was defined as the time from patient randomization to either disease progression according to the International Myeloma Working Group (IMWG) response criteria or death, whichever occurred first.
Missing data were imputed via the Transcan function in the Hmisc (version 5.1-0) R package. 30 Transcan is a nonlinear additive transformation and imputation function.
Statistical analysis
Data were assessed for statistical significance and tabulated with descriptive statistics. Statistical comparisons between categorical variables were performed using the Chi-square test, while the Kruskal–Wallis test was employed to evaluate the statistical significance of continuous variables.
Logistic regression was employed to examine the association between anticoagulant or antiplatelet use and the occurrence of adverse events. Results were reported as odds ratios (ORs) with their corresponding 95% confidence intervals (95%CI). For the assessment of associations between anticoagulant and antiplatelet use with OS and PFS, a Cox proportional hazards regression was employed. Results were reported as hazard ratios (HR) with 95% CI. Analyses were done at baseline and at the 6-month landmark. For the landmark analyses, patients experiencing an event or censored before the 6-month cutoff time was excluded. 31 Statistical significance was set at p-value <.05. All analyses were adjusted for age, gender, weight, MM international staging system (ISS) stage, Eastern Cooperative Oncology Group performance status (ECOG-PS) score, and comorbidity count and stratified by clinical trial and treatment arms. 32 All analyses were performed using R version 4.2.
Results
Overall incidence of adverse events across the study population
Data from a total of 1804 patients was pooled from the three clinical trials. A summary of patients’ baseline characteristics and adverse events incidence by study cohort is provided in Table 1. The duration of anticancer treatment was (median (IQR)) 32.7 (41.1) months for MAIA, 12.6 (5.5) months for the POLLUX, and 5.3 (2.4) months for CASTOR. The median follow-up time was 56.2, 7.43, and 13.5 months for MAIA, CASTOR, and POLLUX, respectively.
A summary of patients’ baseline characteristics by study cohort.
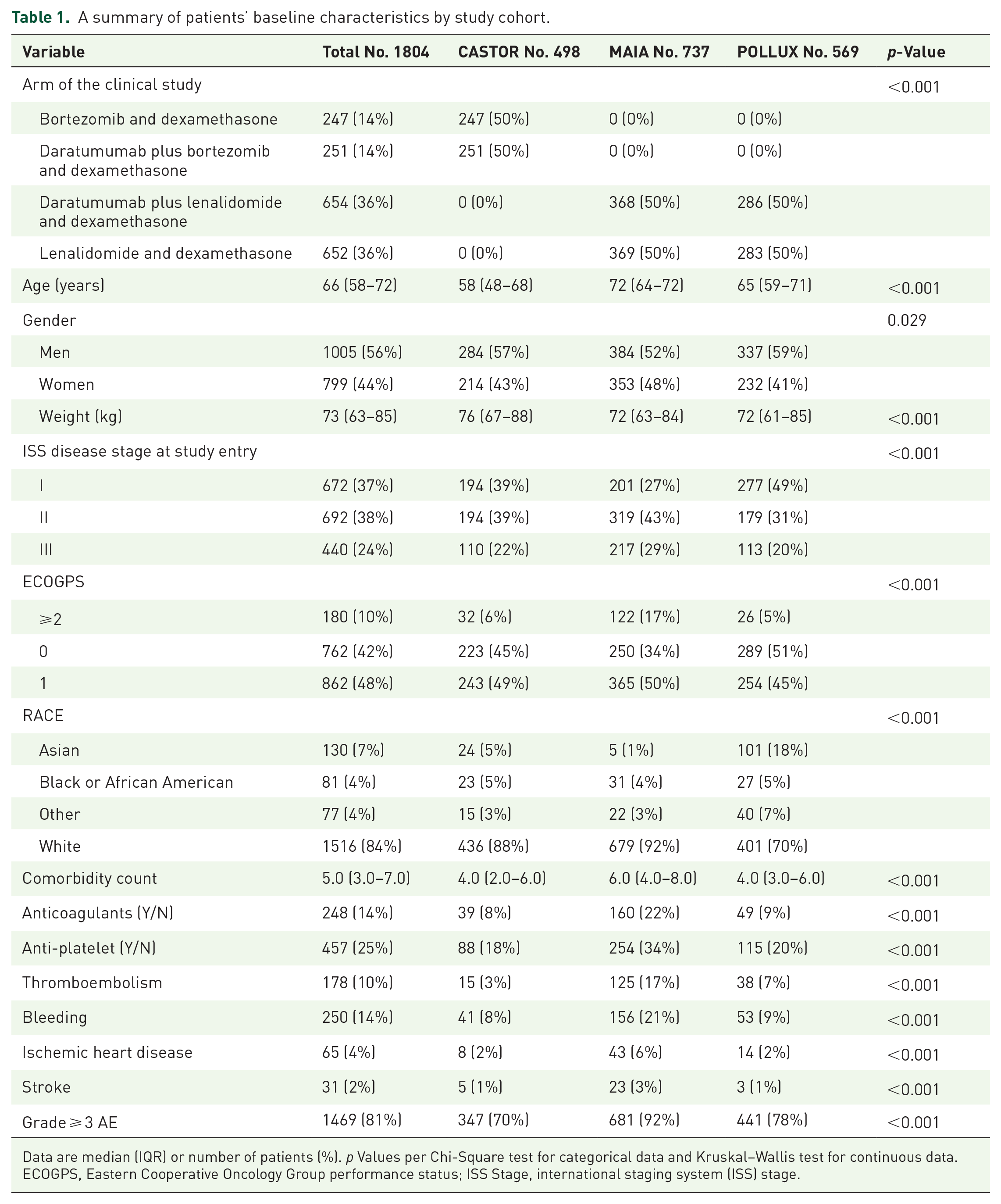
Data are median (IQR) or number of patients (%). p Values per Chi-Square test for categorical data and Kruskal–Wallis test for continuous data.
ECOGPS, Eastern Cooperative Oncology Group performance status; ISS Stage, international staging system (ISS) stage.
Of the 1804 patients, antiplatelets were the most frequently utilized antithrombotic class (457/1804 patients, 25%) followed by anticoagulants (248/1804 patients, 14%) across all trials. The utilization of antithrombotics (i.e., antiplatelet and anticoagulants) was significantly prevalent in the MAIA and POLLUX trials compared to CASTOR. Antiplatelets were prescribed for (254/737 patients, 34%) in MAIA, (115/569 patients, 20%) in POLLUX, and (88/498, 8%) in CASTOR. whereas anticoagulants were prescribed for (160/737 patients, 22%), (49/569 patients, 9%), and (39/498, 8%), in MAIA, POLLUX, and CASTOR trials, respectively.
VTE occurred in 178 (10%) patients, with higher rates in MAIA (125/737 patients, 17%) and POLLUX (38/569 patients, 7%), compared to CASTOR (15/498 patients, 3%). As for bleeding, a total of 250 (14%) events occurred, with significantly higher rates in MAIA (156/737 patients, 21%), followed by POLLUX (53/569 patients, 9%), and CASTOR (41/498 patients, 8%).
Ischemic heart disease and stroke were observed in (65/1804 patients, 4%) and (31/1804 patients, 2%), respectively. A significantly higher incidence of these events occurred in MAIA compared to the POLLUX and CASTOR trials. Notably, the MAIA trial had significantly higher age, ISS stage, ECOG score, and comorbidity count.
During the course of treatment, a large proportion of patients (1469/1804, 81%) experienced any grade ⩾3 adverse event, with a notable prevalence in the MAIA and POLLUX cohorts.
In the pooled cohort, 905 (50%) patients received a daratumumab-containing regimen. Daratumumab arms had significantly higher incidences of grade ⩾3 adverse events (780 patients, 53%). However, there were no significant differences in the incidence of VTE, bleeding, ischemic heart disease, and stroke between the two treatment types (Supplemental Table 1).
Anticoagulants’ use and indications
Table 2 provides an overview of the documented use and indications of anticoagulants and antiplatelets at baseline and within 6 months of treatment initiation.
Documented use and indications of anticoagulants and antiplatelets at baseline and within 6 months of treatment initiation.
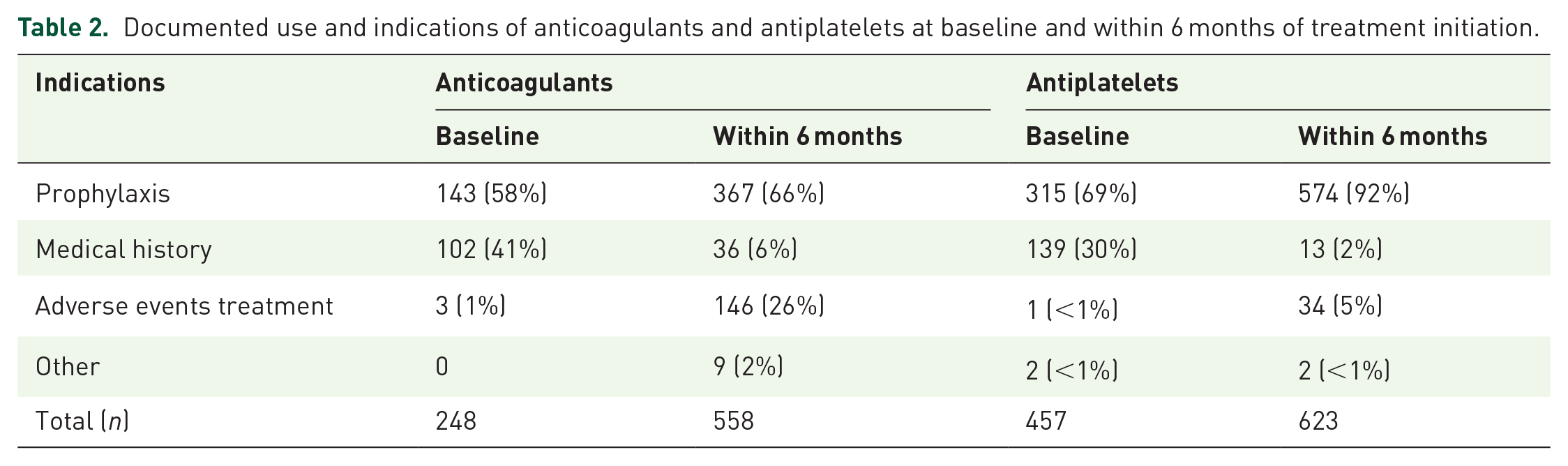
Within the pooled cohort, a total of 248 (14%) patients were on anticoagulants at baseline, with the most common indications being prophylaxis (143 patients, 58%) and comorbidity management (102 patients, 41%). Heparin (135 patients, 54%) and warfarin (61 patients, 26%) were the most frequently prescribed anticoagulant classes. Other anticoagulant classes used included direct factor XA inhibitors (43 patients, 17%), indirect factor XA inhibitors (5 patients, 2%), and direct thrombin inhibitors (4 patients, 2%).
Table 3 summarizes the incidence of AE by anticoagulant use at baseline. Among the 248 patients receiving anticoagulant therapy at baseline, the vast majority (225 patients, 91%) experienced grade ⩾ 3 AE. The incidence of VTE did not show a significant difference when compared with patients on anticoagulants (34 patients, 14%) and those without (144 patients, 9%). Furthermore, there was no remarkable difference in bleeding rates between patients using anticoagulants (39 patients, 16%) versus nonusers (211 patients, 14%).
Incidence of adverse events based on the anticoagulant use at baseline.

Data are median (IQR) or number of patients (%). p-Values per Chi-Square test for categorical data and Kruskal–Wallis test for continuous data.
Transitioning to the first 6 months of the study period, 558 (31%) patients were initiated on anticoagulants. Prophylaxis (367 patients, 66%) and treating active adverse events (146 patients, 26%) were the predominant indications. The anticoagulant classes initiated included heparin (480 patients, 86%), direct factor XA inhibitors (38 patients, 7%), warfarin (18 patients, 3%), indirect factor XA inhibitors (18 patients, 3%), and direct thrombin inhibitors (4 patients, <1%).
Antiplatelets use and indications
A total of 457 (25%) patients received antiplatelets at baseline, primarily for prophylaxis (315 patients, 69%) and comorbidity management detailed in medical history (139 patients, 30%; Table 2). The antiplatelets prescribed fell into two classes: salicylic acid derivatives, primarily aspirin (428 patients, 94%), and platelet aggregation inhibitors (29 patients, 6%), which comprised clopidogrel and ticagrelor.
Table 4 summarizes the incidence of AE by antiplatelet use at baseline. Bleeding events (88 patients, 19%), VTE (58 patients, 13%), ischemic heart disease (30 patients, 7%), stroke (15 patients, 3%), and grade ⩾3 adverse events occurred at significantly higher rates in patients with antiplatelets.
Incidence of adverse events based on antiplatelet use at baseline.

Data are median (IQR) or number of patients (%). p Values per Chi-Square test for categorical data and Kruskal–Wallis test for continuous data.
A total of 623 patients initiated antiplatelet use within 6 months of the study period. The predominant indication for antiplatelet use was prophylaxis 574 (92%), followed by addressing adverse events 34 (5%; Table 2). The antiplatelets prescribed consisted of salicylic acid derivatives (604 patients, 97%) and platelet aggregation inhibitors (19 patients, 3%).
Association of antithrombotic drugs use with adverse events
Table 5 summarizes the associations between anticoagulant and antiplatelet use at baseline and the 6-month landmark.
Association of anticoagulants or antiplatelet use with adverse events at baseline and the 6-month landmark.

Analyses stratified by study and arms.
Analysis adjusted for age, gender, ISS disease stage, weight, ECOG score, and comorbidity count.
At baseline, the use of anticoagulants did not show any significant associations with any of the adverse events evaluated. Antiplatelet use at baseline was significantly associated with higher odds of ischemic heart disease (OR (95% CI) = 1.86 (1.09–3.14), p = 0.022) but did not show significant associations with other adverse events.
At the 6-month landmark, the use of anticoagulants within 6 months was significantly associated with decreased odds of VTE events (OR (95% CI) = 0.45 (0.26–0.77), p = 0.004). There were no significant associations between the use of anticoagulants and the other adverse events. For antiplatelets, no significant associations were observed (Table 5).
Association of antithrombotic use with survival outcomes
Table 6 presents the prognostic associations between anticoagulant and antiplatelet use with OS and PFS. Anticoagulant and antiplatelet use was not significantly associated with OS or PFS at both baseline and the 6-month landmark. At the 6-month landmark, there were 1568 and 1348 of 1804 patients included in the analysis for OS and PFS, respectively.
Association of anticoagulant and antiplatelet use with survival outcomes.
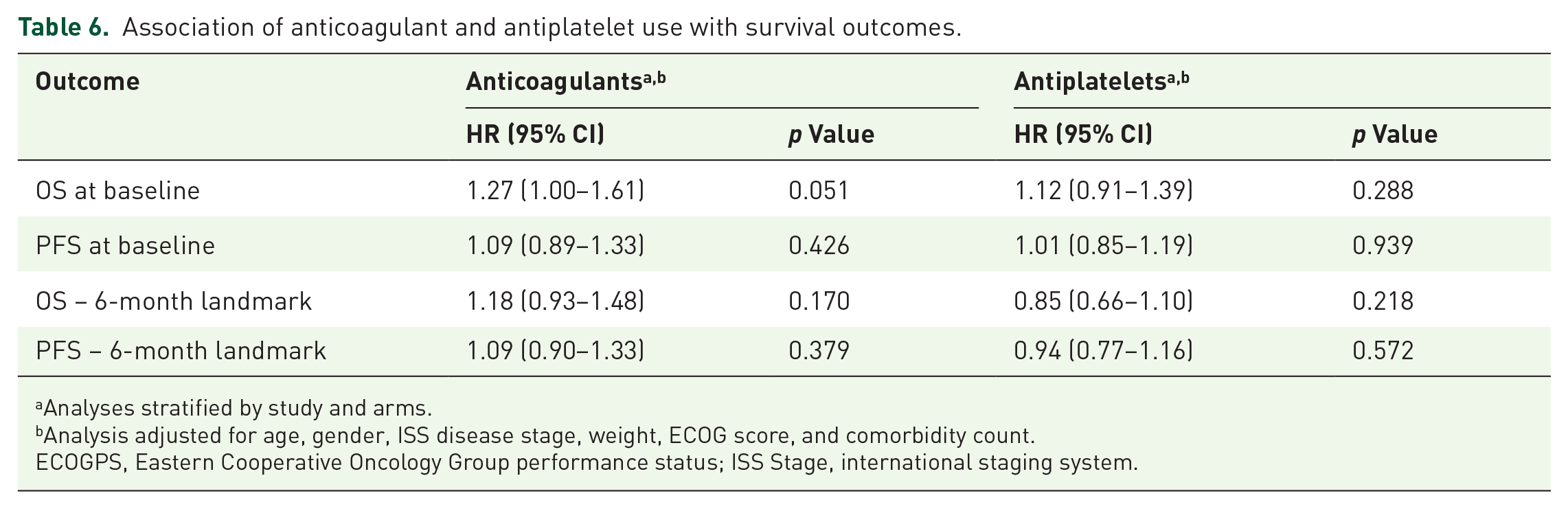
Analyses stratified by study and arms.
Analysis adjusted for age, gender, ISS disease stage, weight, ECOG score, and comorbidity count.
ECOGPS, Eastern Cooperative Oncology Group performance status; ISS Stage, international staging system.
Discussion
This study aimed to provide comprehensive insights into the utilization of antithrombotic drugs and their potential associations with adverse events and survival outcomes in patients with MM initiating contemporary immunomodulatory regimens. The analysis revealed a notable incidence of VTE, ischemic heart disease, bleeding, and stroke among the studied population cohort. Patients experiencing any of the evaluated adverse events demonstrated elevated rates of any grade ⩾3 adverse events. The study also highlighted a substantial increase in the prescription of both antiplatelets (mainly aspirin) and anticoagulants (primarily LMWH and direct oral anticoagulants) from baseline to the 6-month landmark, with prophylaxis being the primary indication. Notably, while anticoagulant use was associated with reduced odds of VTE, antiplatelet therapy did not show a similar association with adverse events. Furthermore, there were no significant associations between antithrombotic use and OS or PFS. To our knowledge, this is the first study to comprehensively examine the interplay between antithrombotics, adverse events, and survival outcomes in patients with MM initiating multi-drug immunomodulatory combinations.
In the pooled cohort, a total of 705 (39%) patients were on anticoagulants or antiplatelets at baseline, while 1181 (65%) patients initiated these medications within the first 6 months of the study period. The primary indication for starting antithrombotics at both time points was prophylaxis, with 25% of patients receiving antithrombotics at baseline and 52% within the initial 6 months. Interestingly, the rate of VTE incidence was 10% despite preventative measures. This finding aligns with updated studies involving patients with MM who receive ImiDs and thromboprophylaxis per existing guidelines.5,33,34 For instance, in the recent GRIFFIN clinical trial on patients with MM initiating daratumumab + lenalidomide/bortezomib/dexamethasone, the efficacy endpoint was not met due to high venous VTE rates reaching 12.9%.5,35 These findings underscore that VTE continues to pose critical complications and reaffirm that thromboprophylaxis guidelines may be suboptimal in these patient populations.
Recognizing the potential risks associated with VTE, the International Myeloma Working Group (IMWG), European Myeloma Network, and National Comprehensive Cancer Network (NCCN) proposed a risk stratification algorithm based on expert opinion and available data from clinical trials.9,10,36–40 These consensus-based guidelines recommended aspirin for low-risk patients and low-molecular-weight heparin (LMWH) or a therapeutic dose of warfarin for high-risk patients. In our study, a higher proportion of patients were on antiplatelets at baseline (25%) and within 6 months (35%), compared to anticoagulants (14% at baseline and 31% within 6 months). Notably, the association analysis revealed a significant reduction in the odds of VTE at the 6-month landmark compared to baseline for patients using anticoagulants. However, antiplatelet use was not associated with reduced risk of any of the evaluated adverse events. Several studies have reported on the superiority of anticoagulants such as LMWH or DOAC over aspirin in preventing VTE.38,39,41,42 For instance, the MELISSE observational study on patients with MM initiating thalidomide or lenalidomide found a VTE incidence of 7% with aspirin and only 3% with LMWH prophylaxis. 43 Furthermore, in a study comparing DOAC rivaroxaban to aspirin, the VTE rate surpassed 16% for patients initiating aspirin and was 4.8% in the DOAC group. 44 Recently, the American Society of Clinical Oncology (ASCO) recommended the use of DOACs for VTE treatment for cancer patients. 45 However, DOAC use was not updated in MM guidelines for the prevention or treatment of VTE and remains uncertain and requires further clinical evidence. 1 Our findings, alongside existing literature, underscore the pressing need for additional studies and updated guidelines offering more effective thromboprophylaxis alternatives to aspirin. 46
Our findings demonstrate a significantly higher prevalence of all reported adverse events and antithrombotic utilization in the MAIA trial, reflecting the distinct characteristics inherent in the patient population who were ineligible for autologous stem cell transplantation or high-dose chemotherapy and were significantly older than the patient population in CASTOR and POLLUX (p < 0.001). The “transplant-ineligible” patients enrolled in the MAIA trial were characterized by a significantly higher comorbidity burden and poor performance status (p < 0.001), which are frequently associated with older age, thereby placing them at an elevated risk of treatment-related adverse events. 47
Patients with MM initiating regimens from MAIA and POLLUX exhibited a higher incidence of VTEs, ischemic heart disease, bleeding, and stroke. The heightened risk of VTE associated with IMiDs plus dexamethasone, reflected in the study cohort, has been well established in the literature. 48 Notably, patients in MAIA and POLLUX trials exhibited a higher incidence of VTE despite initiating antithrombotic drugs more frequently than those in the CASTOR trial. Interestingly, the lower rates in CASTOR might suggest a potential protective role of bortezomib against VTE, as demonstrated in a previous study. 38 Further, an investigation into thalidomide-based regimens and antithrombotic drugs revealed a 1.38 times higher risk of VTE among patients receiving thalidomide without bortezomib. 49 This points toward potential treatment regimens with anticoagulants. As for daratumumab arms in the pooled cohort, there was no significant difference between the incidence of VTE between daratumumab versus non-daratumumab containing regimens, aligning with findings reported in prior studies.7,35
While antithrombotic drugs are primarily prescribed for thromboprophylaxis, they also play a role in managing comorbidities and treatment-related adverse events in patients with MM, including CVD and stroke. 50 Therefore, investigating their impact on survival and other adverse events, such as bleeding, which may lead to a worse prognosis, is important, as these associations are poorly explored. In a recent systematic review, the efficacy and safety of aspirin, DOAC, LMWH, and warfarin were evaluated for preventing VTE in 1042 patients with MM. Notably, no significant associations between all-cause mortality and bleeding risks were reported for the prophylactic use of these drugs. 13 Similarly, in our findings, there were no significant associations between antithrombotics and OS or PFS. As for bleeding, patients on antiplatelets exhibited a higher frequency of bleeding events, although antiplatelet use was not statistically associated with increased odds. A recent publication evaluated bleeding rates in a real-world computed analysis of around 1300 MM patients and reported that antiplatelets are associated with bleeding risk (HR, 2.4; 95% CI, 1.03–5.68). 51
In our study population, patients who had a VTE event were predominantly of white race (89%). However, it’s worth noting that the majority of the study population included in the analysis was white (84%) with a limited representation of Blacks, Hispanics, and other ethnicities. This is in keeping with published data about Blacks and Hispanic patients being underrepresented in global MM clinical trials. 52
The limitations of this study include the lack of detailed information on the specific types, durations, and dosages of the antithrombotic drugs administered, which impedes our capacity to directly investigate the association of distinct antithrombotic types, their varying doses and regimens, with the treatment outcomes. Moreover, the strict eligibility criteria applied in clinical trials can limit generalizability. For instance, the examination of specific subsets like refractory or relapsed MM in POLLUX and CASTOR, and newly diagnosed MM in MAIA. Another limitation is the completeness of the data. Despite having low missing data percentages and the use of imputation methods for minimizing uncertainty, some potential bias possibility remains. Finally, we acknowledge that the identified associations observed in our study do not establish causality. The statistical relationships identified between antithrombotic use and adverse events should be interpreted as correlations, not causal links. Future studies are warranted to further investigate these associations.
Conclusion
In conclusion, this study comprehensively examined the intricate association between antithrombotic therapy and adverse events in patients with MM initiating contemporary immunomodulatory regimens. The higher occurrence of grade ⩾3 adverse events in patients facing these complications emphasizes the necessity for monitoring and proactive management approaches in clinical practice. While anticoagulant use was associated with a reduction in VTE risk, antiplatelet therapy did not exhibit a similar association. These findings emphasize the need for continued research to optimize thromboprophylaxis guidelines and improve patient care.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241275387 – Supplemental material for Antithrombotic utilization, adverse events, and associations with treatment outcomes in multiple myeloma: pooled analysis of three clinical trials
Supplemental material, sj-docx-1-tam-10.1177_17588359241275387 for Antithrombotic utilization, adverse events, and associations with treatment outcomes in multiple myeloma: pooled analysis of three clinical trials by Sara A. Almansour, Mohammad A. Y. Alqudah, Ziad Abuhelwa, Humaid O. Al-Shamsi, Ahmad Alhuraiji, Mohammad H. Semreen, Yasser Bustanji, Karem H. Alzoubi, Natansh D. Modi, Ross A. Mckinnon, Michael J. Sorich, Ashley M. Hopkins and Ahmad Y. Abuhelwa in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This study, carried out under YODA Project # 2022-5048, used data obtained from the Yale University Open Data Access Project, which has an agreement with JANSSEN RESEARCH & DEVELOPMENT, L.L.C. The interpretation and reporting of research using these data are solely the responsibility of the authors and do not necessarily represent the official views of the Yale University Open Data Access Project or JANSSEN RESEARCH & DEVELOPMENT, L.L.C.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
