Abstract
Background:
MET overexpression represents the most MET aberration in advanced non-small-cell lung cancer (NSCLC). However, except MET exon 14 (METex14) skipping mutation was recognized as a clinical biomarker, the role of MET overexpression as a predictive factor to MET inhibitor is not clear.
Objectives:
The purpose of the pooled analysis is to explore the safety and efficiency of gumarontinib, a highly selective oral MET inhibitor, in drive-gene negative NSCLC patients with MET overexpression.
Design and methods:
NSCLC patients with MET overexpression [immunohistochemistry (IHC) ⩾3+ as determined by central laboratory] not carrying epidermal growth factor receptor mutation, METex14 skipping mutation or other known drive gene alternations who received Gumarontinib 300 mg QD from two single arm studies were selected and pooled for the analysis. The efficacy [objective response rate (ORR), disease control rate (DCR), duration of response, progression-free survival (PFS) and overall survival (OS)] and safety [treatment emergent adverse event (TEAE), treatment related AE (TRAE) and serious AE (SAE) were assessed.
Results:
A total of 32 patients with MET overexpression were included in the analysis, including 12 treatment naïve patients who refused or were unsuitable for chemotherapy, and 20 pre-treated patients who received ⩾1 lines of prior systemic anti-tumour therapies. Overall, the ORR was 37.5% [95% confidence interval (CI): 21.1–56.3%], the DCR was 81.3% (95% CI: 63.6–92.8%), median PFS (mPFS) and median OS (mOS) were 6.9 month (95% CI: 3.6–9.7) and 17.0 month (95% CI: 10.3–not evaluable), respectively. The most common AEs were oedema (59.4%), hypoalbuminaemia (40.6%), alanine aminotransferase increased (31.3%).
Conclusion:
Gumarontinib showed promising antitumour activity in driver-gene negative locally advanced or metastatic NSCLC patients with MET overexpression, which warranted a further clinical trial.
Trial registration:
ClinicalTrials.gov identifier: NCT03457532; NCT04270591.
Introduction
Lung cancer remains a leading cause of cancer-related mortality worldwide. 1 About 85% of lung cancer is non-small cell lung cancer (NSCLC), and about 70% lung cancer patients are stage IIIB or IV at first diagnosis and have lost the opportunity for radical surgery or radical radiation therapy, 2 palliative radiotherapy and/or chemotherapy are the main treatments for these patients, but the prognosis is poor, with a 5-year survival rate of only about 15%. 3 Currently, the choice of second-line or subsequent treatment for driver-gene negative advanced NSCLC is almost similar in National Comprehensive Cancer Network, American Society of Clinical Oncology, European Society for Medical Oncology, Chinese Society of Clinical Oncology and Japanese Lung Cancer Society guidelines, mainly include docetaxel, pemetrexed, albumin-paclitaxel or immune checkpoint inhibitors (if not used in front-line therapy).4–6 The overall objective response rate (ORR) is less than 10%, and the median progression-free survival (PFS) and median overall survival (OS) are no more than 4 and 7–9 months, respectively.5,7 So, there is still an unmet clinical need for the subsequent treatment of driver-gene negative advanced NSCLC patients.
Cellular-mesenchymal to epithelial transition factor (c-Met), an important member of a subfamily of receptor tyrosine kinases, also known by the alias hepatocyte growth factor (HGF) receptor, plays a critical role in tumour proliferation, invasive growth and angiogenesis. 8 MET aberration mainly include three types: MET exon 14 (METex14) skipping mutation, MET amplification (METamp) and MET overexpression.9,10 Among these, METex14 skipping mutations occur in approximately 3% of NSCLC cases, 11 METamp occurs at a rate of 1–5% in treatment-naïve NSCLC patients, 12 but 18% after osimertinib treatment of epidermal growth factor receptor (EGFR) mutated patients. 13 MET overexpression ranges from 15% to 70%, which depends on the antibody assay and positive threshold. 14 METex14 skipping mutation is believed to be an independent driver mutation in NSCLC and is usually mutually exclusive from other drivers. 15 Currently, METex14 skipping mutation was recognized as a clinical biomarker for stratifying NSCLC patients based on their predicted response to MET inhibitors. Four selective type Ib small molecule MET inhibitors targeting METex14 skipping mutations, including Tepotinib, 16 Capmatinib, 17 Savolitinib 18 and Gumarontinib, 19 were approved or conditionally approved and have become a new standard of care in NSCLC. The combination of MET inhibitor with EGFR inhibitor targeting METamp also showed promising efficacy for those METamp-driven EGFR inhibitor resistant NSCLC patients.20,21 However, MET overexpression, its role as a predictive factor was not clear although high MET expression rather than METamp may be associated with a poor prognosis.22,23
Gumarontinib (company code SCC244) is a highly selective, oral MET inhibitor. Gumarontinib demonstrated a favourable safety profile in preclinical study 24 and in preliminary clinical research. 25 Gumarontinib has been conditionally approved by National Medical Products Administration for METex14 skipping mutation positive, locally advanced or metastatic NSCLC. 19 Given the crucial significance of MET overexpression in NSCLC population, we pooled data of driver-gene negative NSCLC patients with MET overexpression and with or without METamp from two single arm studies to (1) explore the efficacy and safety of Gumarontinib; (2) evaluate whether MET overexpression [immunohistochemistry (IHC)3+] could serve as a useful predictor for identifying patients who are likely to benefit from anti-MET therapy.
Materials and methods
Patient eligibility
The analysis is performed based on the pooled data from the phase Ib of two single-arm studies, SCC244-104 study (NCT03457532) and SCC244-108 study (NCT04270591). The SCC244-104 study evaluates the safety, tolerability, pharmacokinetics characters and preliminary efficacy of gumarontinib in patients with MET alterations in histologically or cytologically confirmed locally advanced or metastatic solid tumours. The SCC244-108 study evaluates the efficacy and safety of gumarontinib in patients with locally advanced or metastatic NSCLC including pulmonary sarcomatoid carcinoma (stage IIIb, IIIc or IV) harbouring MET alterations. Both studies included the patients were adult ⩾18 years old at the time of informed consent; ECOG performance status: 0–1 and adequate bone marrow reserve, renal and liver function; at least one measurable lesion according to RECIST 1.1. All the patients were administered gumarontinib 300 mg orally continuous once daily in 21-day treatment cycles until disease progression, unacceptable adverse events (AEs), withdrawal of consent or other criteria for termination of study treatment, whichever is earlier. There are no breaks in dosing between cycles.
In this pooled analysis, only advanced NSCLC patients who failed the standard treatment or no standard treatment (including patients who were intolerant to chemotherapy, who were unsuitable for chemotherapy as judged by the investigator or who refused chemotherapy) with MET overexpression (IHC ⩾ 3+ as determined in central laboratory) not carrying EGFR mutation, METex14 skipping mutation or other known drive gene mutation, and without previous EGFR-tyrosine kinase inhibitor (TKI) treatment were included.
The studies were conducted in accordance with the Declaration of Helsinki and the International Conference on Harmonization Guidelines for Good Clinical Practice. The protocol and all its amendments were approved by the ethics committees from each participating centre. All participants provided written informed consent before enrolment.
Assessments
Safety
The severity of AEs was judged in accordance with the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03 (SCC244-104 study) and 5.0 (SCC244-108 study). Patients was monitored by physical examination, vital signs (blood pressure and heart rate), Electrocardiogram (ECG) and laboratory test (haematology, coagulation function, biochemistry and urinalysis), and results worsened compared with baseline was reported during the study. And AEs were monitored continuously during the study from the time when the informed consent was signed to 28 days after the last dose of gumarontinib.
Efficacy
Radiographic tumour size assessment was evaluated by investigators were performed at baseline and every 6–8 weeks using computed tomography and magnetic resonance imaging scans, and evaluation method, instrument and technical parameters were consistent throughout the study. Tumour responses were assessed according to the tumour response criteria in the RECIST version 1.1.
The primary end point was to assess the preliminary efficacy and safety of gumarontinib in patients with MET alternation. ORR was defined as the proportion of patients with a best overall response of complete response (CR) or partial response (PR) from the first dose to the first disease progression (as per RECIST 1.1). CR/PR may be confirmed by another tumour assessment with at least 4 weeks apart. PFS was defined as the time from start treatment to disease progression or death for any cause (whichever is earlier).
Biomarker analysis
Archived tumour tissue was analysed for MET protein expression level by IHC. Membrane MET expression level was determined centrally via IHC using CONFIRM anti-Total MET SP44 rabbit monoclonal primary antibody (Clinical Trial Assays, CTA1) on Ventana Benchmark Ultra Platform in CAP controlled area within the Oncology and Immunology Unit of WuXi AppTec (China, Wuxi) for the patients from SCC244-104 study, whereas Abcam rabbit monoclonal (EP1454Y) to MET-N-terminal (CTA2) was used to detect MET protein expression level within the AmoyDx BioTec for those patients from SCC244-108 study. The CTA1 demonstrated 90.9% sensitivity, 91.3% specificity and 91.1% accuracy in the methodology validation report; and the CTA2 exhibited 100% sensitivity, specificity and accuracy in the methodology validation report. MET overexpression was defined as positive if tumours with a score of 3+ (⩾50% of all tumour cells staining with strong intensity) in this pooled study.
METamp was detected by fluorescence in situ hybridization (FISH). FISH assays were performed using MET SpectrumRed and CEP7 SpectrumGreen DNA probes (Vysis; Abbott Laboratories) (China, Shanghai) in central lab of WuXi AppTec (SCC244-104 study) and AmoyDx (SCC244-108 study). METamp was defined either as the threshold of MET gene copy number (GCN) per nucleus greater than or equal to 4.0 signals (GCN ⩾ 4) or as the ratio of MET to CEP7 greater than or equal to 2.0 (MET/CEP7 ⩾ 2).
Statistical analysis
No formal statistical hypothesis testing was conducted. ORR and disease control rate (DCR) results were summarized by treatment naïve or pre-treated group. ORR was also summarized by patients with METamp and without METamp. Ninety-five percent confidence intervals (CIs) were calculated using the Clopper–Pearson method based on the exact binomial distribution based on investigator assessment. For time-to-event endpoints [duration of response (DoR), PFS and OS], the median DoR, median PFS, median OS and their survival curves were estimated by Kaplan–Meier method by patients previously treated or not.
The reporting of this study conforms to the STROBE statement 26 (Supplemental File named ‘STROBE-cohort_checklist’).
Results
Patients’ demographics and baseline characteristics
At the data cutoff date of 11 July 2023, a total of 32 NSCLC patients from 22 centres in China were filtered and pooled in the analysis, and each study included 16 patients (Supplemental Figure 1). The ORR was 43.8% (95% CI: 19.8–70.2%) for the 16 patients from SCC244-104 study, and 31.3% (95% CI: 11.7–58.7%) for the 16 patients from SCC244-108 study. The SCC244-104 study and SCC244-108 study did not show substantial differences in ORR due to different antibodies used in CTAs. Furthermore, considering the similar performance of the two CTAs in their respective methodology validation reports, we indirectly infer that the two assays are equivalent in methodology. Ultimately, the pooled dataset included 12 treatment naïve patients who refused or unsuitable for chemotherapy, and 20 pre-treated patients who received ⩾1 lines of prior systemic anti-tumour therapies (Table 1). These patients were all with MET overexpression (IHC ⩾ 3+ as determined by central laboratory) not carrying METex14 skipping or EGFR mutation and no previous EGFR-TKI treatment. When MET GCN ⩾ 4, 10 of the 32 patients had only MET overexpression without METamp, and the remaining 22 patients were with METamp in addition to MET overexpression (Table 1). These 32 patients were also used for the safety and efficacy analysis. Overall, most patients were male (26/32), the median age was 61 years (range: 44–83) and more patients had smoking history (20/32). Patient demographic and baseline characteristics were summarized in Table 1.
Demographics and baseline characteristics.

ECOG, Eastern Cooperative Oncology Group; SD, standard deviation.
Safety
Among the 32 patients, all had at least one AE that emerged or worsened after initiation of a trial drug until 28 days after the last dose of the trial drug, the most common treatment emergent adverse events (TEAEs) occurred in at least 30% of patients included oedema (62.5%), hypoalbuminaemia (53.1%), alanine aminotransferase increased (37.5%), anaemia (34.4%), aspartate aminotransferase increased (34.4%) and decreased appetite (31.3%). At least one treatment-related TEAEs were reported in all patients who received gumarontinib. The most frequent treatment-related AEs (occurring in ⩾20% of patients) were oedema (59.4%), hypoalbuminaemia (40.6%), alanine aminotransferase increased (31.3%), aspartate aminotransferase increased (25.0%), nausea (25.0%) and vomiting (25.0%). Grade ⩾ 3 treatment-related AEs occurred in 11 patients (34.4%), with oedema (6.3%) being the most common (Table 2), others are only happened in one patient. Among the 32 patients, 11 (34.4%) patients reported serious TEAEs with 7 (21.9%) patients judged to be related to the treatment. Except for decreased appetite, which was reported in 2 (6.3%) patients, all other SAEs were reported in 1 patient each. The common treatment-related AEs that occurred were similar to those observed in other gumarontinib studies, 19 most are grade 1 or 2, and most are manageable and controllable.
Summary of treatment-related TEAEs ⩾10% by grouped PT and maximum CTCAE grade.
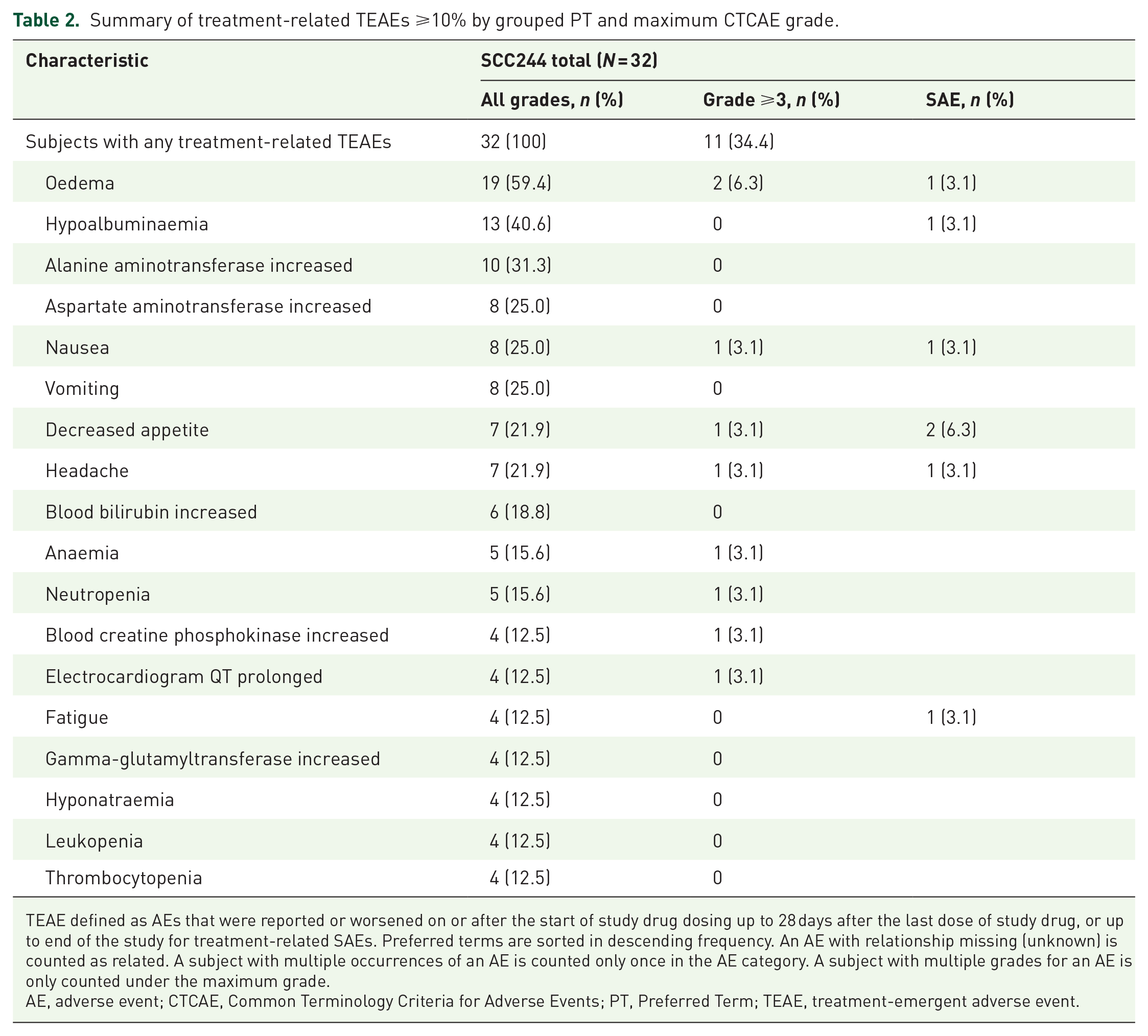
TEAE defined as AEs that were reported or worsened on or after the start of study drug dosing up to 28 days after the last dose of study drug, or up to end of the study for treatment-related SAEs. Preferred terms are sorted in descending frequency. An AE with relationship missing (unknown) is counted as related. A subject with multiple occurrences of an AE is counted only once in the AE category. A subject with multiple grades for an AE is only counted under the maximum grade.
AE, adverse event; CTCAE, Common Terminology Criteria for Adverse Events; PT, Preferred Term; TEAE, treatment-emergent adverse event.
Efficacy
The efficacy results were summarized in Table 3. Among the 32 patients, none achieved a CR, while 12 patients had a PR, 7 in the pre-treated group, 5 in the treatment naïve group; and 14 patients exhibited stable disease. The confirmed ORR was 37.5% (95% CI: 21.1–56.3%) in all patients, 41.7% (95% CI: 15.2–72.3%) in treatment-naïve patients and 35.0% (95% CI: 15.4–59.2%) in pre-treated patients (Table 3). The DCR was 81.3% (95% CI: 63.6–92.8%) in all patients, 91.7% (95% CI: 61.5–99.8%) in treatment naïve and 75.0% (95% CI: 50.9–91.3%) in pre-treated patients (Table 3). Figure 1(a) displayed the maximum percentage change from baseline in target lesions for each patient. Fifteen (53.6%) patients showed target lesions shrank by more than 30% from baseline. Tumour response was still maintained ongoing in 4 of 12 responders by the data cutoff date [Figure 1(b)]. The median follow-up time was 14.7 months. Among the total 12 PR patients, median DoR was 10.3 months [95% CI: 2.8–not evaluable (NE)] (Supplemental Figure 2 and Table 3).
Efficacy summary in MET overexpression NSCLC patients treated with gumarontinib.
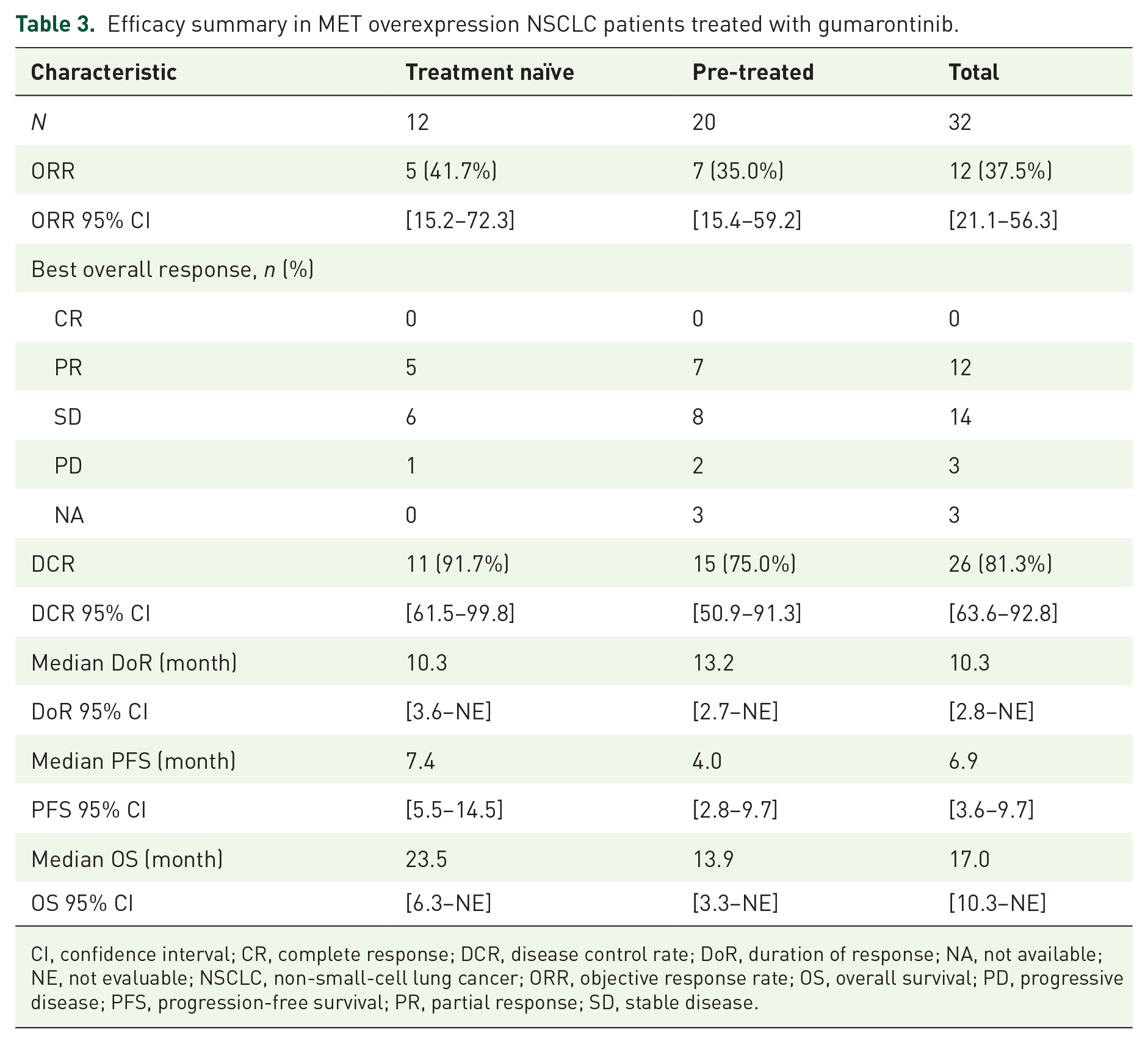
CI, confidence interval; CR, complete response; DCR, disease control rate; DoR, duration of response; NA, not available; NE, not evaluable; NSCLC, non-small-cell lung cancer; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
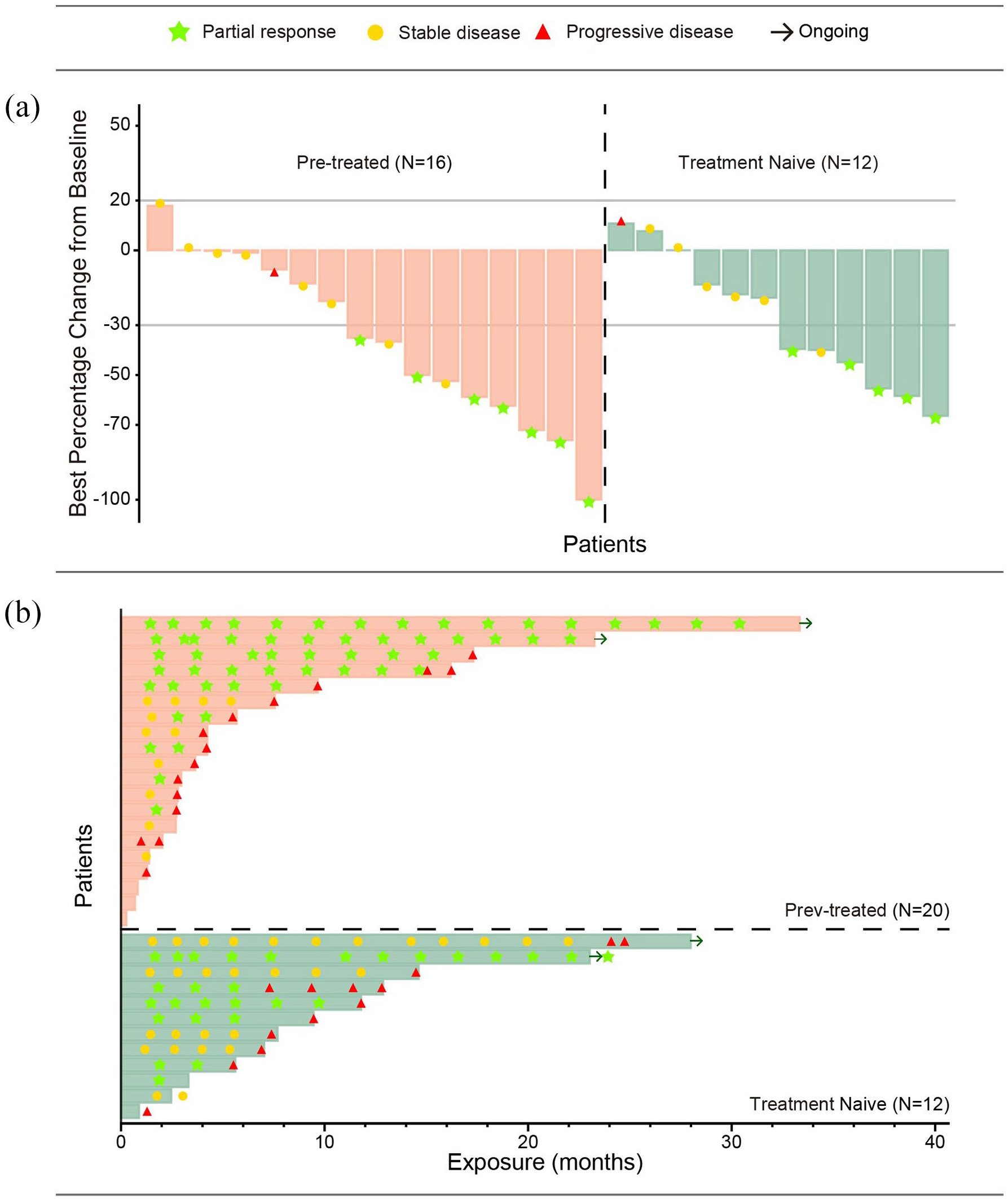
(a) Waterfall plot displaying the maximum percentage change from baseline in target lesions for each NSCLC patient with MET overexpression (IHC3+). (b) Swimmer plot illustrating the duration of response for each NSCLC patient with MET overexpression (IHC3+).
Median PFS was 6.9 months (95% CI: 3.6–9.7) in all patients, 7.4 months (95% CI: 5.5–14.5) in treatment naïve and 4.0 months (95% CI: 2.8–9.7) in pre-treated patients [Figure 2(a) and Table 3]. Median OS was 17.0 months (95% CI: 10.3–NE) in all patients, 23.5 months (95% CI: 6.3–NE) in treatment naïve and 13.9 months (95% CI: 3.3–NE) in pre-treated patients [Figure 2(b) and Table 3].

Kaplan–Meier plot of (a) PFS assessed by investigator and (b) OS assessed by investigator.
Biomarker analysis
At a cutoff of MET GCN ⩾4, the ORR was 30% (95% CI: 6.7–65.2%) (3 responders in 10 patients) for the patients with MET overexpression only (METamp-negative group, hereafter negative group) and 40.9% (95% CI: 20.7–63.6%) (9 responders in 22 patients) for patients with concurrent MET overexpression and METamp (METamp-positive group, hereafter positive group).
To evaluate whether MET IHC3+ can serve as a potential independent representative of MET overexpression as a clinical biomarker, we conducted a more in-depth analysis. Figure 3(a) and (b) utilized two different METamp measurements, MET GCN and MET/CEP7 ratio, respectively, to observe the distribution of patients in negative and positive groups at various integer cutoff values. For instance, in Figure 3(b), at a MET/CEP7 ratio cutoff of 2, there were 17 patients in the positive group, with 9 showing CR or PR, whereas 15 patients were in the negative group, with 3 showing CR or PR. At a MET/CEP7 ratio cutoff of 4, only 4 patients were in the positive group, with 3 showing CR or PR, whereas 28 patients were in the negative group, with 9 showing CR or PR. Overall, with increasing cutoff values, the number of patients in the positive group decreased, whereas the number in the negative group increased. Additionally, an analysis of the relationship between patients’ frontline treatment and efficacy was added. In Figure 3(c), for the pre-treated subgroup, when the MET/CEP7 ratio cutoff was ⩾2 and <4, the curves for METamp-negative and METamp-positive intersected, but they no longer intersected after a cutoff of 4. In Figure 3(d), for the treatment-naïve subgroup, there was no observed intersection of curves when the MET/CEP7 ratio cutoff was ⩾2. Furthermore, in Figure 3(c) and (d), similarly, with increasing cutoff values, the number of patients in the positive group decreased, whereas the number in the negative group increased.

Biomarker analysis. (a, b) The distribution of NSCLC patients with MET IHC3+ in MET amplification negative and MET amplification positive groups at various integer cutoff values based on MET GCN and MET/CEP7 ratio measurement, respectively, and the ORR at different cutoff values. (c, d) Analysis of the relationship between patients’ frontline treatment and efficacy based on MET/CEP7 (Chromosome Enumeration Probe 7) ratio measurement in the pre-treated subgroup (c) and in the treatment-naïve subgroup (d).
Discussion
Currently, the second-line or subsequent treatment for driver-gene negative advanced NSCLC is mainly chemotherapy or immune checkpoint inhibitors (if not used in front-line therapy).4–6 However, the overall ORR is less than 10%, and the median PFS and median OS are no more than 4 and 7–9 months, respectively.4,5 There are still greatly clinical unmet needs for these NSCLC patients. Currently, METex14 skipping mutation is recognized as an independent driver mutation in NSCLC and is usually mutually exclusive from other drivers. 15 It is used as a clinical biomarker for stratifying NSCLC patients based on their predicted response to MET inhibitors. However, MET overexpression, the most occurred among the MET alterations, its role as oncogenic driver events remains under debate.
Very little clinical trials were conducted based on MET inhibitor monotherapy in driver-gene negative NSCLC population with MET overexpression, BPI-9016M, a novel TKI targeting c-Met. A clinical trial performed in locally advanced or metastatic NSCLC showed BPI-9016M monotherapy exhibited limited efficacy in MET overexpression or METex14 skipping mutation patients. Only one PR patient was observed. ORR was 2.6% (1/38, 95% CI: 0.1–13.8%) in all 38 patients. The median PFS and OS were 1.9 months (95% CI: 1.9–3.7) and 10.3 months (95% CI: 7.3–NE) in all 38 patients, respectively. 27 Two previous phase III trials assessed the efficacy of the combination of MET inhibitor/antibody plus Erlotinib in previously treated NSCLC patients with chemotherapy regimens.28,29 Tivantinib is a potent non-ATP competitive-selective MET-TKI. The MARQUEE phase III study (NCT01244191), the exploratory subgroup analysis showed OS was prolonged [median OS: 8.8 months (Erlotinib + tivantinib) versus 5.0 months (Erlotinib + placebo); Hazard ratio (HR), 0.56; 95% CI, 0.35–0.89 for Erlotinib plus Tivantinib in the EGFR wild-type, KRAS wild-type and MET high (defined as high if membranous staining intensity was ⩾2 in ⩾50% of tumour cells) subgroup. 28 Onartuzumab is a monoclonal antibody targeting c-Met. The phase III METLung trial (NCT01456325) examined the efficacy and safety of Onartuzumab plus Erlotinib in patients with locally advanced or metastatic NSCLC. Erlotinib with or without Onartuzumab in previously treated advanced NSCLC patients with MET overexpression (defined as ⩾50% of tumour cells with MET IHC scores of 2+ or 3+) showed worse survival rate. 29
In our pooled analysis, gumarontinib 300 mg showed promising antitumour activity in the 32 patients. The overall ORR was 37.5% (95% CI: 21.1–56.3%), 41.7% (95% CI: 15.2–72.3%) in treatment naïve and 35.0% (95% CI: 15.4–59.2%) in pre-treated patients. Median PFS was 6.9 months (95% CI: 3.6–9.7) overall, 7.4 months (95% CI: 5.5–14.5) in treatment naïve and 4.0 months (95% CI: 2.8–9.7) in pre-treated patients. Median OS was 17.0 months (95% CI: 10.3–NE) overall. Compared with the efficacy of standard chemotherapy, the other MET inhibitors, like BPI-9016M and Tivantinib, and the antibody Onartuzumab above, the preliminary results of gumarontinib 300 in NSCLC patients with MET overexpression were favourable both in pre-treated and treatment naïve patients, suggesting MET overexpression may be a potential molecular predictive biomarker for the driver-gene negative NSCLC patients, might help precisely expand the NSCLC population that would benefit from the clinical use of gumarontinib. Thus, an open-label, multicentre, randomized, phase III study (CTR20230451; http://www.chinadrugtrials.org.cn/) of gumarontinib versus docetaxel in driver-gene negative locally advanced or metastatic NSCLC patients with MET overexpression who have progressed after immunotherapy and platinum-based chemotherapy is ongoing to confirm this finding.
So far, MET overexpression is not sufficient as an independent predictive biomarker to guide clinical practice, meaning even if MET overexpression is detected, an assessment of METamp status is still necessary. If METamp is confirmed, consideration may be given to the use of MET-TKI. This is because not all MET overexpression is caused by METamp, and METamp indeed serves as a specific predictive biomarker to guide the application of MET-TKI. 30 However, what is the probability of METamp in the case of MET overexpression? The correlation between MET overexpression and de novo METamp is relatively low. 31 A study involving 181 cases of non-targeted therapy-treated lung adenocarcinoma patients showed that among MET overexpression positive patients (H score ⩾ 200), only 1% had METamp (detected by FISH, MET/CEP7 ⩾ 2.2), METamp could still be detected in samples that were negative for MET overexpression. 31 Nevertheless, in the TATTON study, among EGFR-mutated NSCLC patients with MET overexpression (⩾50% tumour cells with MET IHC scores 3+) who developed acquired resistance after a prior EGFR-TKI treatment, 80% (16/20) of them were concurrently found to have METamp (MET GCN ⩾ 5 or MET/CEP7 ⩾ 2). 32 In advanced NSCLC patients lacking driver-gene mutations and exhibiting MET overexpression (IHC3+), the correlation between METamp fold and efficacy remains unknown. Therefore, this study also deeply investigated this question. Figure 3 illustrates that, whether based on the MET GCN [Figure 3(a)] or MET/CEP7 ratio [Figure 3(b)], across different cutoff values, the longitudinal distribution of patients treated with gumarontinib within the MET IHC3+ population changed between METamp-negative and METamp-positive subgroups, with increasing cutoff values, the number of patients in the METamp-positive group decreased, whereas the number in the METamp-negative group increased. Overall, the ORR in the METamp-positive subgroup is higher than that in the METamp-negative subgroup. Specifically, within the METamp-positive subgroup, the trend of ORR increases with the amplification fold. In the METamp-negative subgroup, the ORR to gumarontinib remains relatively stable, ranging from 30% to 37.5%. Taken together, it can be inferred that the efficacy data are predominantly influenced by MET overexpression. This inference is supported by the increasing number of patients included in the METamp-negative subgroup as the cutoff values increase, indicating that the patients with only MET overexpression, no amplification demonstrates the promising efficacy, and MET IHC3+ could potentially serve as an independent representative of MET overexpression as a clinical biomarker.
However, we must acknowledge the limitations of this study. Firstly, this study based on two single arm studies, the retrospective design was therefore prone to selection bias. Secondly, the patient number was limited (n = 32) in our study.
Conclusion
In summary, in this pooled study, gumarontinib showed promising clinical efficacy in driver-gene negative locally advanced or metastatic NSCLC patients with MET overexpression and warranted a phase III study to further evaluated.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241264730 – Supplemental material for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib
Supplemental material, sj-docx-1-tam-10.1177_17588359241264730 for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib by Yongfeng Yu, Wen Dong, Yanxia Shi, Rong Wu, Qitao Yu, Feng Ye, Chengzhi Zhou, Xiaorong Dong, Xingya Li, Yongsheng Li, Zhen Li, Lin Wu, Yueyin Pan, Hong Shen, Dehua Wu, Zhongyuan Xu, Jinsheng Wu, Nong Xu, Yanru Qin, Aimin Zang, Jingdong Zhang, Jianya Zhou, Xiaotao Zhang, Yanqiu Zhao, Fugen Li, Huizhen Wang, Qi Liu, Zhenyong Han, Jin Li and Shun Lu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241264730 – Supplemental material for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib
Supplemental material, sj-docx-2-tam-10.1177_17588359241264730 for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib by Yongfeng Yu, Wen Dong, Yanxia Shi, Rong Wu, Qitao Yu, Feng Ye, Chengzhi Zhou, Xiaorong Dong, Xingya Li, Yongsheng Li, Zhen Li, Lin Wu, Yueyin Pan, Hong Shen, Dehua Wu, Zhongyuan Xu, Jinsheng Wu, Nong Xu, Yanru Qin, Aimin Zang, Jingdong Zhang, Jianya Zhou, Xiaotao Zhang, Yanqiu Zhao, Fugen Li, Huizhen Wang, Qi Liu, Zhenyong Han, Jin Li and Shun Lu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241264730 – Supplemental material for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib
Supplemental material, sj-docx-3-tam-10.1177_17588359241264730 for A pooled analysis of clinical outcome in driver-gene negative non-small cell lung cancer patients with MET overexpression treated with gumarontinib by Yongfeng Yu, Wen Dong, Yanxia Shi, Rong Wu, Qitao Yu, Feng Ye, Chengzhi Zhou, Xiaorong Dong, Xingya Li, Yongsheng Li, Zhen Li, Lin Wu, Yueyin Pan, Hong Shen, Dehua Wu, Zhongyuan Xu, Jinsheng Wu, Nong Xu, Yanru Qin, Aimin Zang, Jingdong Zhang, Jianya Zhou, Xiaotao Zhang, Yanqiu Zhao, Fugen Li, Huizhen Wang, Qi Liu, Zhenyong Han, Jin Li and Shun Lu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
