Abstract
Background:
Several studies have systematically assessed the efficacy and safety of progressive or recurrent glioblastoma multiforme (GBM). However, the discernible limitations of efficacy and the elevated costs of interventions instigate an investigation into the cost-effectiveness of these treatments.
Objectives:
This study aimed to evaluate cost-effectivenesses of 11 pharmacotherapeutic interventions for recurrent GBM from the perspective of healthcare payers in the United States (US) and China.
Design:
A model-based pharmacoeconomic evaluation.
Methods:
A partitioned survival model was employed to evaluate the cost-effectiveness of 11 distinct drug-based treatments. The clinical efficacy and safety data were obtained from a network meta-analysis, while the medical expenditure and health utility were primarily derived from published literature. One-way sensitivity analyses, scenario analyses, and probabilistic sensitivity analyses (PSA) were performed to scrutinize the impact of potential uncertainties to ensure the robustness of the model. The primary endpoint was the incremental cost-effectiveness ratio.
Results:
Among the therapeutic interventions evaluated, lomustine emerged as the cheapest option, with costs amounting to $78,998 in the United States and $30,231 in China, respectively. Regorafenib displayed the highest quality-adjusted life years at 0.475 in the United States and 0.465 in China. The one-way sensitivity analyses underscored that drug price was a key factor influencing cost-effectiveness. Both scenario and PSA consistently demonstrated that, considering the willingness-to-pay thresholds, lomustine was a cost-effective treatment with probability of more than 94%.
Conclusion:
In comparison to the alternative antitumor agents, lomustine was likely to be a cost-effective option for relapsed GBM patients from the perspective of healthcare payers in both the United States and China.
Introduction
Glioblastoma multiforme (GBM), originating from glial cells or their precursors represents a formidable challenge in the realm of neuro-oncology. The World Health Organization categorizes GBM as a grade 4 diffuse glioma, 1 characterized by a fervent cellular division, heightened vascular proliferation, central necrosis within the tumor core. 2 The incidence of GBM approximates 3–5 cases per 100,000 individuals. 3 Alarmingly, this malignancy has a dismal 5-year survival rate of 7%. Even worse, GBM exhibits an exceptionally high recurrence rate, with a median time to relapse of fewer than 7 months. 4
After a progression or recurrence of GBM, it remains bereft of efficient treatments. The drugs including bevacizumab, nitrosoureas, temozolomide (TMZ), regorafenib, multiple drug combination regimens based on bevacizumab, and so on, can be used for recurrent GBM (rGBM).5–8 In the previous investigation, a network meta-analysis of randomized controlled trials including 11 pharmacotherapies for patients with rGBM was conducted. 9 The findings manifested that the patients with rGBM who underwent regorafenib or bevacizumab plus lomustine exhibited relative favorable survival outcomes, despite the backdrop of suboptimal objective response rates. The treatment of rGBM poses multiple challenges compared to primary GBM. First, rGBM heightened malignancy and frequently demonstrates resistance to standard treatments such as TMZ or radiotherapy, rendering conventional protocols less effective. 10 Second, the absence of clear and uniform treatment recommendations in different guidelines compounds the complexity of treating rGBM. The lack of recommendation arises from the limited efficacies of numerous regimens through clinical trials. Additionally, the economic burden associated with antitumor drugs further complicates the management of the disease. Although several studies have analyzed the cost-effectiveness of treatments for primary GBM based on surgery, TMZ, or radiotherapy,11–15 the therapeutic tolerance of rGBM, coupled with the diverse treatment options available, necessitates a comprehensive analysis of the cost-effectiveness of potential therapies for rGBM to assist in health care decision-making.
There are two studies that have investigated the cost-effectiveness of drugs for rGBM, which focused on three treatments including TMZ, a combination of procarbazine, lomustine, and vincristine (PCV), and bevacizumab plus irinotecan.16,17 However, the pharmacoeconomic evaluation of several novel antitumor agents for rGBM, including small molecule targeted drugs and programmed death receptor 1 inhibitors, remains insufficient. To address the gaps, we developed a partitioned survival model to assess the cost-effectiveness of 11 pharmacotherapies for rGBM in the perspectives of both United States (US) and Chinese healthcare. These 11 regimens encompass bevacizumab monotherapy, lomustine monotherapy, regorafenib monotherapy, nivolumab monotherapy, and 7 distinct bevacizumab-based combination regimens. Notably, these therapeutic choices align closely with the recommendations provided by United States and Chinese clinical practice guidelines.5,7
Materials and methods
This research adhered to the Consolidated Health Economic Evaluation Reporting Standards 2022 checklist (Supplemental Table 1). 18
Model structure
This study employed a partitioned survival model in Microsoft Excel 2021 to analyze the clinical and economic outcomes of 11 pharmacotherapeutic interventions based on the perspectives of United States and Chinese payers. The framework of the model comprised three mutually exclusive states including the progression-free (PF) state, the progression-disease (PD) state, and death (Supplemental Figure 1). All patients were uniformly situated within the PF state at the onset of the modeling process. A modeling cycle of 4 weeks was adopted, which was consistent with prior cost-effectiveness investigations focused on GBM.11,15,19 The temporal scope of the model was set at a 5-year horizon, as survival rates beyond this temporal threshold were extremely rare based on the simulated survival curves, with fewer than 1% of patients. Key model outputs encompassed life years, quality-adjusted life years (QALYs), and costs. QALYs and costs were discounted at rates of 3% per year in the United States 20 and 5% per year in China. 21 The incremental cost-effectiveness ratios (ICERs) were assessed to ascertain the cost-effectiveness of various treatments.
Clinical inputs
Clinical efficacy data of the 11 treatments were obtained from a network meta-analysis. 9 Notably, certain drugs, like epidermal growth factor receptor inhibitor family, were excluded due to the failure for forming a head-to-head comparison with the drugs in the network meta-analysis.
Bevacizumab, commonly employed in clinical practice for rGBM and frequently utilized as a control group in head-to-head clinical trials of rGBM,22–24 was designated as the benchmark for the derivation of clinical data for the remaining treatments. Given the study design, sample size, locations, and time to publication, the survival data of bevacizumab from CheckMate 143, a multinational phase III randomized controlled clinical trial enrolling 369 patients with recurrent glioblastoma, was utilized to reconstruct the individual patient data. To extract information from the kaplan-meier (KM) curves, the GetData Graph Digitizer 2.26 was employed. 25 Subsequently, employing the methodologies delineated by Guyot et al., 26 the pseudo-patients-level overall survival (OS) and progression-free survival (PFS) data were generated by the R 4.2.2 software.
To deduce lifetime health outcomes for individuals with rGBM, an array of mathematical models including exponential, Weibull, Gompertz, log-logistic, lognormal, and Gamma models were developed to extrapolate and fit the KM curves for OS and PFS of patients within the bevacizumab cohort from the CheckMate 143 trial (Supplemental Figures 2 and 3).27,28 The goodness-of-fit of these models was assessed using Bayesian Information Criterion and Akaike information criterion (Supplemental Table 2).
Lognormal distribution model was ultimately selected to estimate and calculated the probabilities of survival, PD, PF, and death per cycle. At any given temporal point denoted as ‘t’, the survival probability was defined as
Survival and PF probabilities corresponding to the remaining 10 treatments were calculated based on the hazard ratios obtained from the network meta-analysis. 9
Costs and health utility
This investigation was based on the perspective of the healthcare system, focusing exclusively on the direct medical costs (Table 1). All monetary values were converted to US dollars (USD) at an exchange rate of 1 USD = 6.76 China Yuan.
Cost and utility inputs to the models.

Vorinostat was not approved in China at the time of this study.
Medical services include outpatient, hospitalizations, nursing care, and meals.
AEs, adverse events; ASP, average sales price of 2022 Medicare part B drug; PD, progression-disease; PF, progression-free; TMZ, temozolomide.
The therapeutic regimens evaluated in the study were bevacizumab monotherapy, lomustine monotherapy, nivolumab monotherapy, regorafenib monotherapy, bevacizumab plus carboplatin, bevacizumab plus dasatinib, bevacizumab plus irinotecan, bevacizumab plus lomustin, bevacizumab plus TMZ, and bevacizumab plus vorinostat. A thorough exposition of the specific drug administrations was presented in Supplemental Table 4. For precision in dosage calculations, it was presumed that the body surface area would be 1.72 m2 in Chinese and 1.8 m2 in American.11,32 The body weights were set at 65 kg for Chinese patients and 70 kg in American patients.11,32
Prices of drugs in the United States were obtained from the 2022 Medicare Part B Drug Average Sales Price published by the US Centers for Medicare and Medicaid Services. 33 Non-part B drug prices were derived from Drugs.com, a website provided accurate and up-to-date information on prescription drugs in the United States. 34 The drug prices in China were sourced from YAOZHI Database, a reputable provider of pharmaceutical and healthcare industry data. 35 The costs of other aspects were derived from previously published literature.
The health utilities for PF and PD status in patients with rGBM were derived from the work of Martikainen et al. 16 The assessment of health utilities was conducted using a visual analog scale, with the questionnaires being completed by neuro-oncologist who served as proxy respondents.
Cost-effectiveness analyses and sensitivity analyses
Within the established framework, the parameter ranges were set at 95% confidence intervals or deviation within a ±20% margin from standardized values. The cost data adhered to a gamma distribution, while effect size data followed beta distribution. 36 The cost-effectiveness of 11 treatments was evaluated by ICERs and sequential ICERs based on the cost-effectiveness frontiers. In the US context, the willingness-to-pay (WTP) threshold was set at $100,000–$150,000 per QALY. 37 As per the World Health Organization’s recommendation, the WTP threshold in China was defined as ranging from 1 to 3 times the per capita gross domestic product (GDP), 38 equivalent to $12,677–$38,032 per QALY.
Accounting for the inherent uncertainty in model parameters, one-way sensitivity analyses were employed to explore the impact of individual parameter variations within a range of ±20% on the model results. 39 The results of one-way sensitivity analyses were visually demonstrated in tornado diagrams, where the horizontal axis represented incremental net monetary benefit (INMB) and the vertical axis represented parameter names. The INMB is calculated as follows: INMB = λΔE − ΔC, where λ is the WTP boundary value, ΔE represents the difference in the QALY between two interventions, and ΔC represents the cost difference. A positive INMB indicates a cost-effectiveness advantage for the intervention over the control group, conversely, a negative INMB suggests that the intervention is not deemed cost-effective. 40 Calculating INMB effectively addressed positive and negative transitions in ICERs due to parameter changes.
In addition, two scenario analyses were conducted. In Scenario 1, cost-effectiveness was assessed using log-logistic distribution model, which was the second most appropriate distribution for fitting survival curves following the lognormal distribution. In Scenario 2, health utilities from Garside et al. 41 of 0.89 for PF and 0.73 for PD were employed. These health utilities were from the National Health Service Health Value Panel estimated for patients with primary GBM using standard gambling methods.
Probabilistic sensitivity analyses (PSA) were employed through Monte Carlo simulation to evaluate the robustness of the results. In PSA, all parameters were simultaneously varied over the specified range, rather than relying solely on the mean or median values for outcome calculations. 42 Subsequently, cost-effect acceptability curves (CEACs) were constructed based on the 1000 simulations, enabling an assessment of the probability that different treatment options attained cost-effectiveness relative between the WTP thresholds.
Results
Base-case results
In terms of health outcomes, the QALY for the 11 treatments ranges from 0.251 to 0.475 in the United States, and from 0.248 to 0.465 in China (Table 2). Regorafenib monotherapy for rGBM exhibited the highest QALY in both the United States and China followed closely by the combination of bevacizumab with lomustine (90 mg/m2). Conversely, nivolumab monotherapy for rGBM displayed the least favorable QALY outcome.
Results of the basic analysis of 11 treatment schemes.
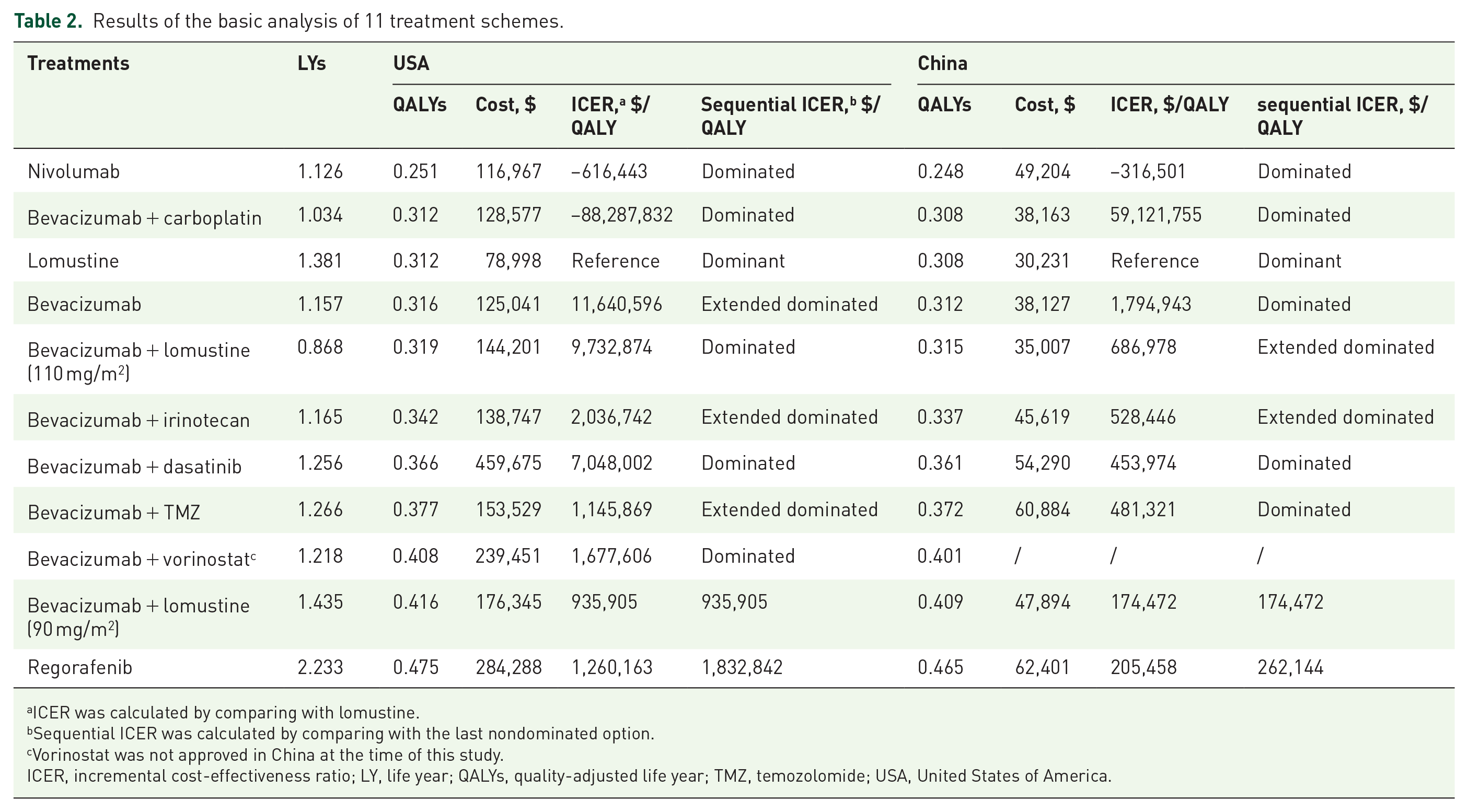
ICER was calculated by comparing with lomustine.
Sequential ICER was calculated by comparing with the last nondominated option.
Vorinostat was not approved in China at the time of this study.
ICER, incremental cost-effectiveness ratio; LY, life year; QALYs, quality-adjusted life year; TMZ, temozolomide; USA, United States of America.
In terms of cost, lomustine monotherapy demonstrated the most economical cost, amounting to $78,998 in the United States. Bevacizumab in combination with dasatinib stood out as the most expensive treatment option, with a cost of $459,675. In China, lomustine monotherapy remained as the cheapest regimen with a cost at $30,231. The regimen with the highest cost was regorafenib monotherapy, costing $62,401.
In terms of cost-effectiveness, three therapeutic regimens, lomustine, bevacizumab plus lomustine (90 mg/m2), and regorafenib comprised the cost-effectiveness frontier in both United States and China, suggesting that the three treatments had potential cost-effectiveness (Figure 1). The remaining treatments were either dominated or extended dominated. Taking the least costly option, lomustine, as the reference, the ICERs for all treatments exceeded WTP thresholds in the United States and China, respectively (Table 2). In the United States, the ICER was $935,905/QALY for bevacizumab plus lomustine (90 mg/m2) and $1,260,163/QALY for regorafenib. In China, the ICER amounted to $174,472/QALY for the combination of bevacizumab with lomustine (90 mg/m2), while for regorafenib, it was $205,458/QALY.
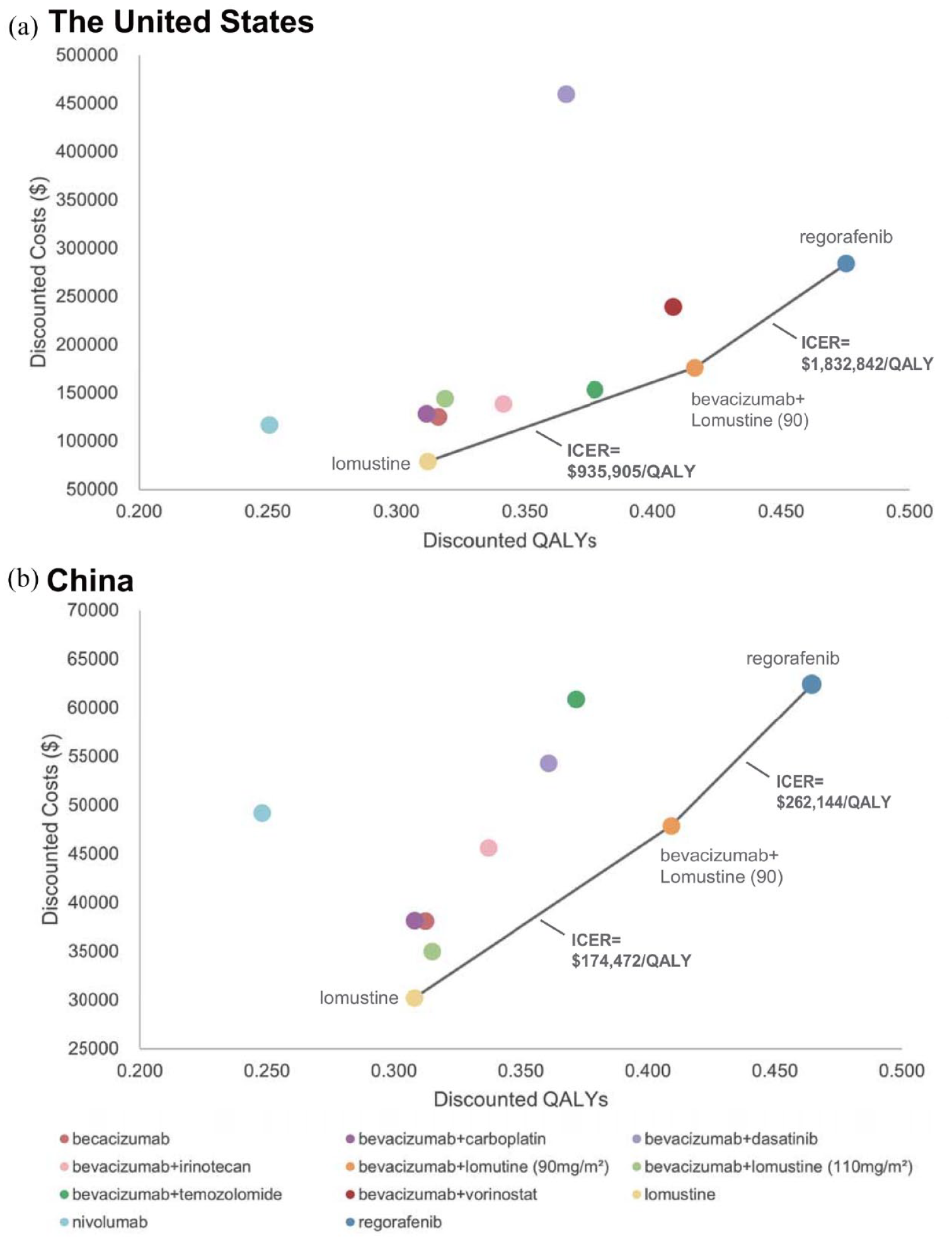
Cost-effectiveness frontier of treatments for patients with rGBM. (a) Cost-effectiveness frontier of 11 treatments for patients with rGBM in the US setting. (b) Cost-effectiveness frontier of 10 treatments for patients with rGBM in Chinese setting. The ICER was compared with next best nondominate treatments.
Sensitivity analyses
The results of the one-way sensitivity analyses for all comparators are presented in Supplemental Figure 5. In the United States, the parameter with the most substantial impact on INMB was the drug cost per cycle. Similarly, in China, the INMBs of most treatments were primarily influenced by drug price, except for the bevacizumab monotherapy, bevacizumab combined with carboplatin, and regorafenib monotherapy, which are predominantly affected by the hazard ratio of OS. Within the maximum WTP threshold, the INMBs for all treatments are negative in both the United States and China, indicating that none of the treatments are cost-effective compared to lomustine.
In the scenario where the model was constructed using a log logistic survival distribution, the distribution of ICERs aligns with that observed in the base-case analysis. Lomustine, bevacizumab + lomustine (90 mg/m2), and regorafenib constitute the cost-effectiveness frontiers. ICERs of bevacizumab + lomustine and regorafenib surpass the WTP threshold in the United States and China, respectively (Supplemental Table 5). Lomustine maintains its status as the cost-effective option within the WTP threshold, consistent with the base-case result.
In Scenario 2, incorporating utilities of patients with primary GBM estimated based on the standard gamble method into the analysis, all regimens exhibit a significant increase in QALYs, resulting in notable changes in ICERs (Supplemental Table 6). In both the United States and China, only bevacizumab + lomustine (90 mg/m2) and regorafenib have positive ICERs compared to the lowest-cost treatment, lomustine, with the rest of the treatments being dominated. Contrary to the base-case results, bevacizumab + lomustine (90 mg/m2) no longer lies at the cost-effectiveness frontier but is instead extended dominated. A further PSA under this model reveals that only lomustine remained cost-effective under WTP thresholds in both countries (Supplemental Figure 6).
The results of the PSA showed that lomustine exhibited a 100% probability of being cost-effective within a WTP range of $100,000–$150,000 in the United States. Notably, even when the WTP escalated to $800,000/QALY, lomustine maintained the highest probability of being cost-effective [Figure 2(a)]. In China, lomustine monotherapy was deemed cost-effective with a probability ranging from 93.8% to 94.3% when the WTP thresholds were between 1 to 3 times the GDP in China [Figure 2(b)]. Bevacizumab combined with lomustine (90 mg/m2) presented the highest probability of being cost-effective when the WTP was between $180,000 and $245,000/QALY. When WTP exceeded $245,000/QALY, regorafenib monotherapy demonstrated the optimal cost-effectiveness. Scatter plots (Supplemental Figure 4), capturing the distributions of QALYs and associated costs, were crafted based on the 1000 simulations, which showed consistent results as observed in the base-case analysis.

Cost-effectiveness acceptability curves of treatments for patients with recurrent glioblastoma. (a) CEAC of 11 treatments in the US setting. (b) CEAC of 10 treatments in the Chinese setting.
Discussion
A conspicuous scarcity of economic investigations pertaining to rGBM has prevailed. To our knowledge, this is the first cost-effectiveness analysis of treatments for rGBM from the perspectives of the healthcare payers in the United States and China. In this study, we employed a partitioned survival model, and obtained the distributions of patients in different states derived from KM curves, thereby avoiding assumptions about natural mortality rates, as often required in conventional Markov model. 43 Moreover, our study utilized appropriate methods to ensure that the reconstructed survival curves closely align with the original curves, thus allowing for a more precise simulation of event occurrence.
The results of cost-effectiveness analyses suggested that lomustine emerged as the most cost-effective treatment within the WTP thresholds in the United States and China. This presented an alternative option that went beyond the judgment based solely on clinical outcomes. If the primary considerations were survival and QALY, the most recommended regimens were regorafenib monotherapy and bevacizumab in combination with lomustine (90 mg/m2).5,7 Despite the fact that lomustine’s LYs and QALY ranked lower among all 11 treatments, and its clinical efficacy appeared to be inferior to regorafenib monotherapy and bevacizumab plus lomustine, lomustine was the most cost-effective regimen due to its low costs. Bevacizumab in combination with lomustine (90 mg/m2) and regorafenib monotherapy were likely to be cost-effective under the circumstances of high WTP thresholds, exceeding approximately sixfold the maximum WTP in the United States and more than fivefold the maximum WTP in China.
It was observed that the QALYs of each treatment are quite similar, confirming the evidence-based study that demonstrated the absence of significant differences in the clinical efficacy of most drug regimens, thus leading to similar health outputs. The ICERs are proportional to the differences in costs and inversely proportional to the difference in QALYs. In the calculation of ICER, a minimal difference in QALYs results in an excessively large value of ICERs. QALYs, in turn, are proportional to the health utilities. Consequently, when the health utilities, PF rates, and PD rates (determined by the objective properties of the drug) are fixed, the difference in treatment cost becomes one of the predominant factors influencing ICERs. This point was further supported by the results of the one-way sensitivity analyses. In the United States and China, drug costs per cycle emerged as the primary factors influencing shifts in INMB. Therefore, controlling drug costs was likely to become one of the available options in order to narrow the gap between ICER and WTP, thereby enhancing the cost-effectiveness of drugs.
The outcomes of Scenario 2 exhibit some divergence from the base-case analysis. Bevacizumab plus lomustine (90 mg/m2) no longer occupies the cost-effectiveness frontiers and becomes an extended dominated intervention. However, this alteration does not impact the final decision, that is, the certainty that lomustine is cost-effective at the WTP threshold remains at 100%. In this scenario, the utility values for PF significantly grew from 0.41 to 0.89, while the utility for PD increased from 0.14 to 0.73. The one-way sensitivity analyses emphasize the pivotal role of the utilities as an important parameter influencing the cost-effectiveness of the treatment. Consequently, it is not surprising that drugs on the cost-effectiveness frontier changed. However, it’s essential to acknowledge that both sources of health utilities in the base-case and Scenario 2 analyses have limitations that affect their applicability in the analytical model. First, these values were assessed a long time ago, originating in 2005 and 2007, respectively. Second, they were interviewed from academics rather than patients, introducing a potential bias into the results. Furthermore, in terms of methodology, while there is no inherent superiority between visual analog scale and standard gamble, contemporary preference is often given to scales like the EuroQol five dimensions questionnaire and 36-item short form, answered directly by patients, to assess their health utility. 44 Unfortunately, the field of glioblastoma research on health-related quality of life and health utility remains scarce. Moreover, the utilities in the Scenario 2 analysis were in the perspective of primary patients, showcasing better vital status compared to recurrent patients. Consequently, these utilities may be higher than those encountered in real-world situations.
Comparing the difference in treatment costs between the United States and China, it was indicated that the cost of treating rGBM in the United States was higher than that in China, regardless of the drugs used. Correspondingly, the WTP in the United States was higher than in China. The treatments that constituted the cost-effectiveness frontier of both countries were the same, namely lomustine, bevacizumab plus lomustine (90 mg/m2), and regorafenib. Between the WTP thresholds, lomustine stood out as cost-effective. In comparison to lomustine, the ICERs of the treatments were higher in the United States than in China, leading to the curve representing lomustine floating above almost unchanged in the US CEAC for WTP values less than $500,000/QALY. In contrast, in the Chinese CEAC curves, the cost-effective regimen switched when the WTP grew to the vicinity of ICERs for bevacizumab plus lomustine (90 mg/m2) (the sequential ICER was $174,472/QALY) and regorafenib (the sequential ICER was $262,144/QALY).
The strength of this study lies in the comprehensive economical evaluation of diverse drug treatments for rGBM, including two targeted agents, one immune checkpoint inhibitor, one chemotherapeutic agent, and seven drug combination regimens based on the targeted agent, most of which are recommended by the clinical practice guidelines in the United States and China based on the efficacy.5,7 However, beyond disparities in therapeutic effects, these regimens exhibit varying cost profiles, and economic considerations are one of the primary drivers in clinical decision-making. It is necessary to analyze the cost-effectiveness of these regimens to evaluate whether the treatments are worth the substantial monetary inputs.
Prior to our investigation, there have been two cost-effectiveness studies addressing rGBM. Martikainen et al. 16 analyzed the cost-effectiveness of TMZ and PCV in patients with rGBM in the context of Finnish healthcare landscape, employing a Markov model. The results of the study revealed an ICER of TMZ compared to the PCV regimen amounting to €32,471 per QALY. Importantly, the probability of cost-effectiveness was substantiated at 80% when the threshold of WTP was established at €20,000 per QALY. Ruiz-Sánchez et al. conducted a cost-effectiveness analysis based on a retrospective cohort of patients with rGBM in Spain and showed that, compared with patients who did not receive second-line treatment after relapse or who received other second-line regimens, patients who received bevacizumab plus irinotecan paid an additional €46,401.99 per life year, with an increase in costs that would be difficult to be accepted by the public health system. These earlier studies are limited in their scope, involving a constrained number of treatment options. 17 With the large number of clinical trials, more treatment options and evidence have emerged. Our study responds to the imperative for a comprehensive and updated pharmacoeconomic in the dynamic landscape and provides evidence for the clinical decision-making.
Despite the above advantages, it is important to acknowledge certain limitations of this study. Specifically, the health utilities for PF and PD states are not derived directly from American or Chinese patients but are instead based on an estimation by concerned scholars. Additionally, the original study of utilities dates back to quite some time ago. Therefore, the utilities may be somewhat unrepresentative. This highlights the imperative need for more investigations into the quality-of-life of GBM patients to better evaluate clinical outcome and to facilitate health rehabilitation and health decision-making.
As for clinical costs, certain parameter values are derived from cost studies for primary glioblastoma or brain tumor, potentially introducing bias in model results. Nonetheless, one-way sensitivity analyses and PSA robustly indicate that healthcare costs have minimal impact on the final results. Moving forward, it is essential to place greater emphasis on healthcare costs associated with rGBM. Doing so will contribute to the generation of high-quality data, ultimately facilitating more rigorous studies of health economics, specifically focusing on antitumor agents. In the future, it is necessary to increase the focus on healthcare costs for patients with rGBM to provide high-quality data for health economics studies of antitumor agents.
In terms of treatment regimens, some potentially effective therapies were excluded from this study due to their original clinical trials as single-arm studies 45 or their inability to establish head-to-head comparisons with the drugs cited in the network meta-analysis.46–48 It is anticipated that the forthcoming publication of clinical trials for rGBM will contribute to the ongoing enhancement and updating of cost-effectiveness studies.
Conclusion
Lomustine is the most cost-effective therapeutic option for patients with rGBM at a WTP threshold of $100,000–$150,000 per QALY from the perspective of US healthcare payer. Similarly, from the Chinese payer’s perspective, with a WTP threshold range of $12,677–$38,032 per QALY, lomustine maintains its resolute status as the prominent cost-effective choice. Although lomustine may not yield the highest QALY among the available options, its cost-effectiveness profile renders it an advantageous choice for consideration in the clinical management of rGBM patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241264727 – Supplemental material for Cost-effectiveness analysis of 11 pharmacotherapies for recurrent glioblastoma in the USA and China
Supplemental material, sj-docx-1-tam-10.1177_17588359241264727 for Cost-effectiveness analysis of 11 pharmacotherapies for recurrent glioblastoma in the USA and China by Yanan Xu, Boya Xu, Haijing Guan and Zhigang Zhao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
