Abstract
Gastric cancer (GC), one of the tumours with the highest mortality worldwide, is not a homogeneous disease, showing different features according to location, macroscopic aspect, histotype and molecular alterations. Adenocarcinoma is the most frequent epithelial GC (95%), the remaining 5% comprising rare epithelial tumours with their peculiarities, behaviour and incidence <6 cases/100,000/year. Due to the low number of cases, many aspects must be elucidated in this context. In this narrative review, we highlight the importance of a better understanding of rare GCs to personalize the cures in the light of the precision medicine concept. Our main aim is to translate the scarce evidence from the literature into daily clinical practice, never forgetting that all the clinicians dedicated to rare GCs should encourage such patients’ enrolment in clinical trials and promote international collaborations. Hence, we focused on the treatment of the following rare GCs: rare gastric adenocarcinomas (hepatoid adenocarcinoma, medullary carcinoma with lymphoid stroma, Paneth cell carcinoma and Salivary Gland carcinoma); squamous cell carcinoma; adenosquamous carcinoma; neuroendocrine gastric neoplasms; gastroblastoma.
Keywords
Introduction
Gastric cancer (GC) is one of the tumours with the highest mortality worldwide, especially in Western countries. 1 If the screening programmes allow the identification of tumours at a very early stage in Asian countries, the diagnosis is often late in the West. Additionally, in the case of non-metastatic tumours treated with curative surgery, the rate of relapses is high. 2
In this context, the search for prognostic and predictive biomarkers is the main goal to persuade to tailor the treatment for GC and improve the outcomes. 3 Indeed, GC is not a homogeneous disease, showing different characteristics according to location, macroscopic aspect, histotype and molecular alterations. However, if several data showed that GC has its peculiarities according to the location (distal versus proximal tumours) 4 as well as molecular classifications,5,6 the role of histology needs to be defined. In this regard, the World Health Organization (WHO) provided GC’s most comprehensive histological classification. 7 According to that, adenocarcinomas are the most frequent epithelial GC (95%); they can also be classified according to Lauren’s classification from the histological point of view. 8
Nevertheless, 5% of GCs are rare epithelial tumours with peculiarities, behaviour and incidence, as per definition, of <6 cases/100,000/year. In this context, how the histology might affect the prognosis and the response to treatment (predictive role) is unclear, mainly due to the low number of existing cases. Therefore, a better understanding of rare epithelial GCs is crucial to personalizing the cures for these patients in light of the precision medicine concept.
In this narrative review, we report an overview of the state of the art of treatments for rare epithelial GCs, trying to translate evidence from the literature into daily clinical practice. Thus, the description of histological classification and the complexity of those tumours is not the aim of this paper, so please refer to Table 1 and the existing literature for these data.7,9–20
Histological classification of gastric carcinomas.

Locoregional approaches
Endoscopic resections and surgery are the main locoregional treatments in case of localized rare GC, both epithelial and neuroendocrine. However, there is currently no unanimous agreement on managing these neoplasms, and the guidelines are founded on data about GC and neuroendocrine tumours (NETs) in general.21–23 Therefore, managing rare gastric tumours requires a multidisciplinary evaluation in dedicated teams, including gastroenterologists, oncologists and surgeons, to ensure comprehensive care and improved patient outcomes.
Endoscopic resections
Endoscopic resection has emerged as a promising minimally invasive approach for treating rare gastric tumours and gastric NETs, avoiding the need for more invasive surgical procedures.
Rare epithelial GCs
No specific recommendations for rare epithelial GCs are available at the time of writing. Thus, this section will refer to general recommendations for endoscopic resection for gastric adenocarcinoma as per existing guidelines.21,22
According to the American Society of Gastrointestinal Endoscopy and European Society of Gastrointestinal Endoscopy guidelines, endoscopic ultrasound is crucial for rare gastric tumours or lesions without a precise diagnosis. 24 Proper patient selection and preoperative evaluation are essential for the success of endoscopic resection; factors such as tumour size, location and suspected histology play a role in determining the feasibility of this approach.
According to international guidelines, 25 endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) can be considered in case of focal nodules ⩽2 cm with the double aim of diagnosis (large biopsy sample) and treatment. The resection could be considered curative in the case of GC ⩽2 cm in diameter and with good prognostic features [G1–G2, no margins involved, without lymphovascular invasion (LVI)] and in the early GC. 25 A surgical approach is recommended in all the other cases. However, regarding undifferentiated early gastric cancer (UD-EGC), which has a higher risk of lymph node metastasis, 26 JCOG1009/1010 demonstrated excellent technical results and 5-year overall survival (OS) in patients with UD-EGC treated with ESD. 27 Based on these positive outcomes, ESD for UD-EGC (cT1a) of ⩽2 cm without ulceration was deemed technically feasible and an acceptable standard treatment option instead of gastrectomy.
While endoscopic resection is generally considered safe, it is not without risks. Complications may include bleeding and perforation; thus, close postoperative monitoring and appropriate follow-up are essential for early detection and management of adverse events. 28
Lastly, endoscopic resections could be considered a palliative locoregional approach in the case of bleeding.
Gastric NETs
EMR and ESD could also be considered in the case of gastric NET. However, the evidence in this regard is limited and mainly based on clinical cases or case series due to the rarity of those neoplasms. In this context, The European Neuroendocrine Tumor Society (ENET) guidelines recommend the endoscopic approach in the case of localized, type 1 and low-grade gastric NET. 23
Additionally, recently, Chacchi-Cahuin et al. 29 reviewed the available knowledge about the endoscopic management of Gastro-Entero-Pancreatic (GEP) NET, reporting a possible endoscopic approach for gastric NET < 10 mm in size with the double intent of diagnose and cure.
However, there are few data comparing the endoscopic techniques in this field.30–32 A systematic review, including 112 gastric type 1 NETs from six trials, showed similar results between en bloc resections and endoscopic procedures. 23
Regarding type 2 gastric NETs, the ENETS guidelines recommend endoscopic treatment only in a few selected cases after multidisciplinary evaluation, always after excluding multi-endocrine neoplasm syndrome 1. 23
Lastly, due to their behaviour, type 3 gastric NETs should be treated as gastric adenocarcinomas, also regarding the endoscopic resections (see the section ‘Rare epithelial GCs’).23,29
Surgery
Surgery is considered an essential part of the therapy for rare gastric tumours. However, there is currently no unanimous agreement on managing these neoplasms, and the guidelines are founded on limited data with low levels of supporting evidence. Furthermore, surgical guidelines in this field vary depending on the country.21–23
In this context, we can distinguish surgical approaches for rare epithelial GC and NETs. However, in the first case, we will refer to general recommendations for surgery in the case of GC as per existing guidelines (see the section ‘Rare epithelial GCs’ for additional details) since no specific recommendations for rare tumours are available at the time of writing.
Rare epithelial GCs
Surgery is a vital component of the treatment for GC and involves gastrectomy with sufficient margins, peri-gastric and extra-gastric lymph node dissection (LND) and gastrointestinal reconstruction. 21 The primary goal is achieving R0 resection.
However, controversies have emerged regarding the type of gastric resection, the extent of LND, the splenectomy, the approach (laparoscopy or laparotomy) and the reconstruction. 22 Regarding the margins, the recommendations suggest staying between 2 and 5 cm according to the T stage, Borrmann type and the guidelines considered.25,32–34
Concerning the type of gastrectomy, in the case of distal GC, a distal gastrectomy can be performed if safe margins can be achieved21,22,35; otherwise, total gastrectomy is recommended. 21
Regarding LND, although D2 LND is highly recommended in Eastern countries, 36 Western countries have yet to reach a strict consensus. However, long-term results did show higher cancer-related survival for D2 dissection after 15-year follow-up.35–38 D2 lymphadenectomy is the standard surgical approach in Eastern and Western countries. 21 Additionally, it is widely accepted that this should only be performed by experienced surgeons at high-volume centres.21,22
Various randomized trials have investigated the survival benefits of splenectomy since the D2 dissection for upper third and greater curvature tumours includes the removal of the lymph node at station 10 (splenic hilum). 39 Prophylactic splenectomy is not recommended. However, the recommendations vary according to the country and guidelines considered,21,22,25,35,36 and the European Society for Medical Oncology (ESMO) guidelines do not offer specific recommendations for splenectomy. 21
Laparoscopic surgery is still controversial for gastric tumours since there are different recommendations according to the guidelines in this case.21,22,25,35,36 The ESMO guidelines consider laparoscopy as a viable option. 21 The principal evidence is the randomized phase III KLASS-01 and JCOG0912 trials, which showed comparable survival outcomes to open gastrectomy for stage I GC40–42; several other studies, such as KLASS-03, JCOG 1401 and KLASS-02 trials, have shown the safety of laparoscopic total gastrectomy for clinical stage I GC.43–45 Regarding advanced GC, an open gastrectomy remains the recommended approach.21,44,46,47
The reconstruction aims to achieve three primary outcomes: satisfactory nutritional status, good quality of life and low postoperative morbidity. 48 Many reconstruction techniques are described in the literature; the most popular are Billroth-I (B-I), Billroth-II (B-II) with or without Braun anastomosis, Roux-en-Y (RY) and uncut Roux-en-Y. 49 While B-I is highly favoured in Eastern countries, Western surgeons prefer the RY after distal gastrectomy. A recent meta-analysis demonstrated that all these techniques appear equally safe, with comparable rates of anastomotic leak, anastomotic stricture, overall morbidity and short-term outcomes (12 months follow-up). 50 However, RY is associated with a reduced risk of remnant gastritis, lesser bile reflux and esophagitis.
Focusing more on rare epithelial GC, the current scenario has limited published literature on gastric squamous cell carcinoma (GSCC) and gastric adenosquamous carcinoma (GASC), leading to clinical management primarily relying on small single institutional case reports or series (Table 2). Then, a comparison between GSCC, GASC and gastric adenocarcinoma was performed using the National Cancer Database from 2004 to 2013. 51 Surgical resection was conducted in 26.0%, 54.4% and 45.2% of GSCC, GASC and adenocarcinoma patients. Positive surgical margins were found in 2.0%, 11.3% and 6.1% of patients. Approximately half of the patients with GSCC and GASC presented with metastatic disease. The study reported a median OS of 9 months, a 1-year OS rate of 43% and a 5-year OS of approximately 15% after multimodal treatment (surgery + neoadjuvant/adjuvant chemotherapy) for GSCC and GASC. However, there is no established standard of care for surgery in GSCC and GASC, and data regarding the extent of LND needed are unavailable. 52
Ten years reported literature on GSCC.

Alive at follow-up.
CT, adjuvant chemotherapy; CTRT: chemoradiotherapy; DFS, disease-free survival; DG, distal gastrectomy; ESD, endoscopic submucosal dissection; F, female; GSCC, gastric squamous cell carcinoma; LDG, laparoscopic distal gastrectomy; M, male; NA, not available; NCT, neoadjuvant chemotherapy; OS, overall survival; PPG, partial proximal gastrectomy; PG, proximal gastrectomy; RS, radical surgery; RT, adjuvant radiotherapy; SH, splenectomy and hepatectomy; SP, spleno-pancreatectomy; Spl, splenectomy; TG, total gastrectomy.
Despite the type of surgery, the timing for treating those rare tumours is crucial. Indeed, some of them, like gastroblastoma, have a favourable prognosis if promptly treated. Table 3 presents the recent literature on gastroblastoma cases.
Clinical case literature on gastroblastoma (reported by year of publication).
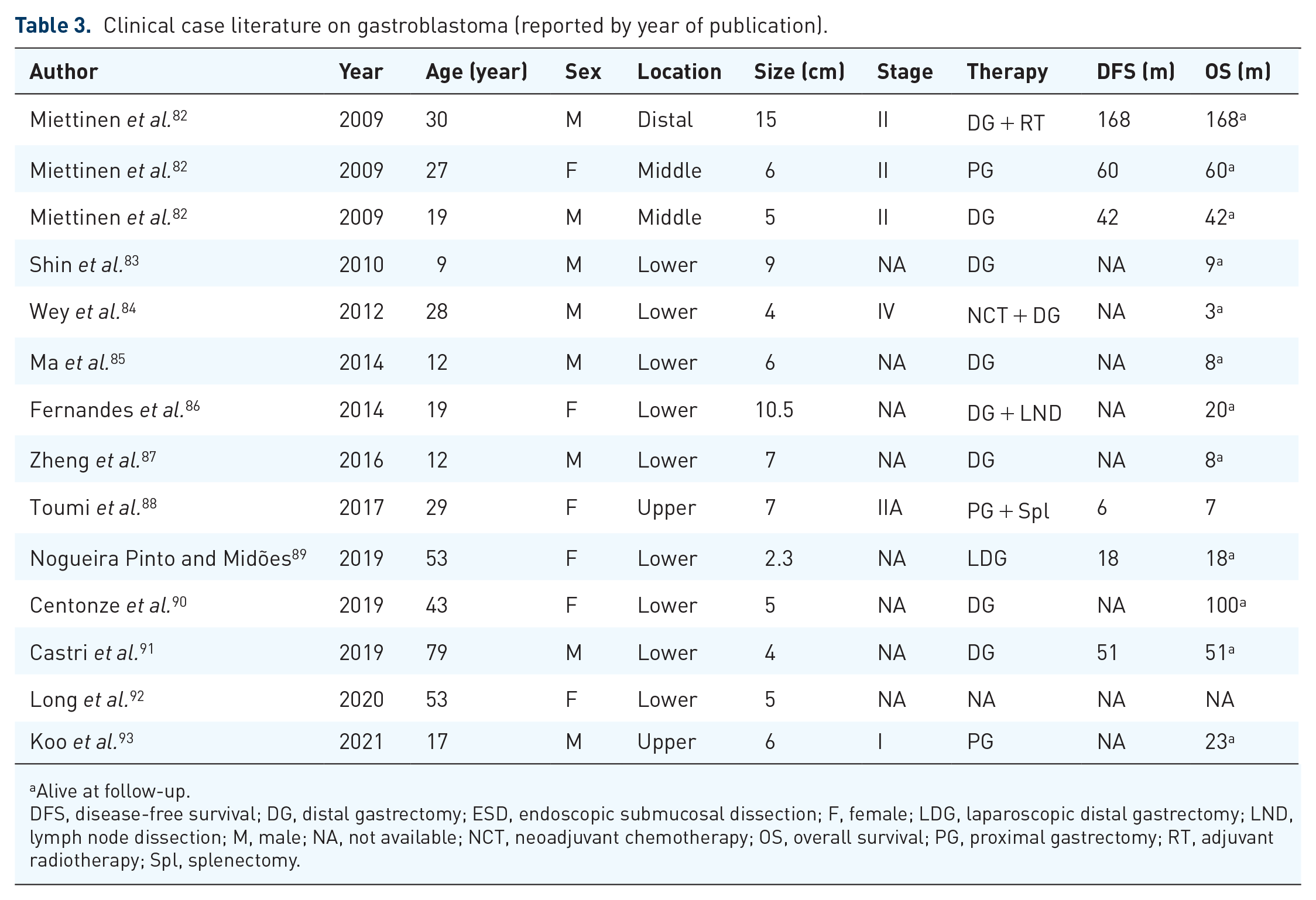
Alive at follow-up.
DFS, disease-free survival; DG, distal gastrectomy; ESD, endoscopic submucosal dissection; F, female; LDG, laparoscopic distal gastrectomy; LND, lymph node dissection; M, male; NA, not available; NCT, neoadjuvant chemotherapy; OS, overall survival; PG, proximal gastrectomy; RT, adjuvant radiotherapy; Spl, splenectomy.
Generally confined to the stomach, gastroblastoma does not exhibit metastatic potential or disease recurrence after successful resection.82,83,85–87,89 However, there have been instances of lymph node involvement and distant metastases in two young adult patients aged 28 and 29 years.84,88 This fact indicates that a standard LND cannot be performed. In 2012, Wey et al. 91 described a gastroblastoma in a 28-year-old man with a similar biphasic architecture, bland cytology and the same histological and immunohistochemical features but with microscopic evidence of regional lymph node metastases. Similarly, Toumi et al. 88 reported the presence of regional nodal and distant metastases in a 29-year-old woman. It is crucial to thoroughly report, describe and discuss each case of gastroblastoma to gain a comprehensive understanding of the tumour from a pathological and morphological perspective.
Undifferentiated GC is a rare histopathological cancer type that does not exhibit any differentiation towards adenocarcinoma or GSCC. It is characterized as highly malignant with a poor prognosis. 94
Gastric-NETs
Surgery plays a significant role in the management of gastric NET. 95 ESMO and the ENETS guidelines recommend considering locoregional indications and selecting the appropriate procedure based on Rindi’s classification 96 for gastric neuroendocrine neoplasms (g-NENs).23,97–103 However, in the case of functional NETs, it is essential to manage clinical symptoms before considering any intervention. A multidisciplinary approach is preferred to ensure that all therapeutic options are considered in these cases (for additional details, refer to the section ‘Medical treatment, Gastric-NETs’).
Medical treatment
Rare gastric adenocarcinomas
Among rare gastric adenocarcinomas, Hepatoid adenocarcinoma (HAS), medullary carcinoma with lymphoid stroma, Paneth cell carcinoma and Salivary Gland carcinoma are the most important. In the following sections, we will focus on the current knowledge regarding the treatment of those rare tumours. For an overview of epidemiology, molecular and clinical features, which are not the aim of this paper, as stated above, please refer to Figure 1.

Rare gastric adenocarcinomas: features and medical treatment.
Hepatoid adenocarcinoma
HAS is a rare tumour with histological characteristics similar to hepatocellular carcinoma (HCC). It is mainly diagnosed as metastatic disease and has a dismal prognosis even after early diagnosis and curative resection [5-year disease-free survival (DFS) rate: 20.7%] 104 ; the worst outcome is reported in cases of HAS expressing high alpha fetoprotein (αFP) levels. 105
Considering the low number of cases worldwide, no specific treatments for HAS exist. Additionally, no data regarding HAS can be found in the landmark trials for GC.21,22 Therefore, a multidisciplinary assessment of these cases is mandatory. However, treatment choice is according to international guidelines for GC.21,22 A multimodal approach with perioperative chemotherapy and surgery is the standard of care in case of non-metastatic disease, 106 while adjuvant chemotherapy (FOLFOX schedule) can be considered after curative surgery.107,108 However, the best option for non-metastatic patients is still debated. Indeed, Zeng et al. 109 showed better outcomes in the case of neoadjuvant treatment. Otherwise, in a retrospective analysis including 100 Chinese patients, Zhou K et al. showed that patients with locally advanced HAS did not benefit from neoadjuvant chemotherapy (29 patients) compared to surgery alone (71 patients). Additionally, they found that patients who received adjuvant chemotherapy for at least 6 months had a better outcome than those who received it for a shorter time. 110 No randomized data regarding the use of radiotherapy, specifically in HAS, are available.
In stage IV, no specific data regarding the efficacy of chemotherapeutic drugs for HAS exists. Nevertheless, like in non-rare GCs, human epidermal growth factor receptor 2 (HER-2) evaluation is mandatory. At the time of writing, in the case of HER-2 negative disease, doublet-based chemotherapy with platin and fluoropyrimidine should be proposed as first-line treatment, with or without Nivolumab (according to programmed death ligand 1 (PD-L1) combined positive score value). 21 However, a case report showed a major response (complete response) in two patients treated with cisplatin and etoposide in the first-line setting. 111 Otherwise, HAS expresses HER-2 in 25% of tumours; in those cases, trastuzumab or trastuzumab plus pembrolizumab, according to the local approval at the time of writing, should be added to doublet-based chemotherapy in the first line of treatment.112–114
Interestingly, local ablative treatments generally used in HCC, such as trans-catheter arterial chemoembolization, have shown to be effective for treating liver metastasis from HAS. Additionally, the ‘HCC profile’ is also revealed in response to antiangiogenetic drugs, such as ramucirumab – alone or in combination with paclitaxel –, which is the best option in the second-line treatment for metastatic and microsatellite stable GC, including HAS. 115 Of note, in the case of HAS producing αFP, this marker could work as an early biomarker of response to treatment. 116 The use of other antiangiogenic drugs, such as sorafenib, which is known to be effective in HCC, is not supported by randomized data in HAS.
Lastly, no randomized data are available in the literature on using immunotherapy in HAS, and only anecdotic case reports exist. 117
Therefore, future evaluations are needed to explore this issue; additionally, the response to antiangiogenic drugs and the ‘HCC profile’ might represent the rationale for testing immune checkpoint inhibitors (ICIs) alone or in combination with antiangiogenic agents in this field.
GC with lymphoid stroma: medullary carcinoma and lymphoepithelioma-like carcinoma
Gastric carcinoma with lymphoid stroma was historically defined as a gastric tumour with a prevalence of intratumoural inflammatory cells. However, this definition included two types of tumour that are considered different entities today: the medullary carcinoma and the lymphoepithelioma-like carcinoma. Both tumours are rare variants of gastric adenocarcinoma and arise mainly in men and the gastroesophageal junction.
The medullary carcinoma might have microsatellite instability (MSI-H), whereas the lymphoepithelioma-like carcinoma is mainly linked with Epstein–Barr virus (EBV) positivity. However, taking into account that not all the medullary and lymphoepithelioma-like carcinomas have MSI-H or EBV positivity and that a detailed description of EBV-positive and MSI-H GC is not in the aim of this review, it is essential to search for the EBV and MSI-H status due to the prognostic and predictive value of these biomarkers. Some experience in GC showed that EBV positivity and MSI-H status are linked with a better prognosis and good response to immunotherapy.118,119 Those results were confirmed in all the landmark trials in the field of GC immunotherapy.21,22 Of note, Corallo S et al. evaluated the prognostic role of EBV in a cohort study of 175 patients with metastatic GC treated at two Italian centres. They showed that only 4% of patients were EBV positive, reporting a significantly better survival in this group of patients when treated with chemotherapy if compared to the EBV negative patients [3-year OS: 80% versus 20.1%; hazard ratio (HR): 0.12]. 120 Therefore, considering the response to immunotherapy of EBV-positive tumours, this study might represent the rationale for testing the association between chemotherapy and immunotherapy in those patients.
However, not all the experiences in this field were related to rare GC histotypes. Therefore, the use of immunotherapy in medullary carcinoma and lymphoepithelioma-like carcinoma depends on the molecular expression of the previously cited biomarkers. Additionally, evidence about the outcomes is derived from the MSI-H and EBV-positive GC trials.
Paneth cell carcinoma and Salivary Gland carcinoma
Paneth cell carcinoma is a rare tumour that develops from the Paneth cell located in the gastrointestinal tract, mainly in the colon and stomach. Considering the rarity of this tumour, few anecdotic case reports exist in the literature, and no randomized trials are available. 121 Additionally, no data regarding the number of cases worldwide are available, nor was data from Paneth cell carcinoma reported in the landmark trial for GC.21,22 Considering the lack of knowledge regarding the pathogenesis and the prognosis of gastric Paneth cell carcinoma, no specific treatment and target agents are available. Therefore, the treatment would be chosen according to GC guidelines.21,22 Interestingly, since some Paneth cell carcinoma showed activation in the Apc/beta-catenin/Tcf pathway with the expression of defensin 5, 122 it might represent a future perspective in the therapeutic landscape for this rare tumour.
According to the RARECARENet database, 39 cases of Salivary Gland carcinoma of the stomach were recorded worldwide. 123 However, no data are available in the literature, including case reports or case series. Thus, when a diagnosis of Salivary Gland carcinoma of the stomach is made, no specific treatments are available, and in this case, we should refer to a multidisciplinary team with expertise in rare tumours.
Other rare gastric epithelial tumours
GSCC, GASC, gastroblastoma and undifferentiated tumours are the most important rare gastric epithelial tumours. Figure 2 summarizes the epidemiology, molecular and clinical features of those tumours.

Other rare gastric epithelial tumours: epidemiology, molecular and clinical features and medical treatment.
Squamous cell carcinoma
GSCC is an infrequent entity, with a reported incidence of less than 0.07% among all GC. Nevertheless, several case reports/series49,51,52,54–56,66,67,70,72,73,75,80,124–128 have been published in the last few years, showing that it is an emerging topic in the field.
Recently, squamous cancer cells were found to be positive for EBV in two case reports56,80; however, a concomitant EBV infection could also have been found by chance, not necessarily being responsible for neoplastic transformation or maybe acting as a co-factor.
GSCC is characterized by aggressive behaviour as compared to adenocarcinoma, with a higher frequency of LVI – which explains its ability of early metastasizing to the lymph nodes and the liver – and also a higher frequency of serosa invasion, translating into the advanced stage at diagnosis and poor OS. 56
There has yet to be a consensus about the optimal approach for GSCC patients regarding the therapeutic algorithm. The use of chemotherapy for advanced stages is less effective than what is observed in adenocarcinoma, especially in terms of median OS. It must be pointed out that 5-fluorouracil-based chemotherapy regimens – the standard first-line therapy for gastric adenocarcinoma – were the most commonly adopted,51,128 but the different biology could be responsible for the lower responses observed. Given the impossibility of conducting ad hoc clinical trials and the rarity of this disease, more efforts are needed to identify potentially predictive biomarkers, especially in the era of precision oncology and immunotherapy.
Another issue is the differential diagnosis with gastric metastases from other squamous cancers. Gastric metastases are rare but possible, as described in some case reports.129–132 Gastric metastases from cutaneous squamous cell carcinoma (SCC) were described in an HIV+ patient, 129 and another starting from cervical carcinoma was also reported. 130 Additionally, two case reports of gastric metastasis from squamous lung cancer have been published.131,132 Therefore, a correct differential diagnosis is crucial for the best therapeutic approach for each patient, especially in those with a history of previous SCC from another district.
Adenosquamous cell carcinoma
GASC is an infrequent variant of GC, accounting for less than 0.5–1% of GC cases. 133 It shows an aggressive phenotype with a poorer outcome than classical gastric adenocarcinoma. 134
Unfortunately, considering the rarity of GASC, there is only a little data about the optimal therapeutic management of these patients.135,136 Then, most GASCs are locally advanced at diagnosis (according to their poor prognosis) and have local lymph node metastases or secondary lesions in visceral sites or peritoneal involvement. 135 Thus, the standard of care for this entity in the advanced setting has yet to be established: chemotherapy with platinum and 5-fluorouracil (as for gastric adenocarcinoma) could be used in the first-line setting. However, the squamous component may render their cancers less responsive to the available chemo-regimen (with a rapid selection of resistant subclones). Additionally, the pivotal phase III clinical trials that evaluated different regimens in different lines of therapy in advanced disease did not include specifically GASC.21,22 Therefore, treatment is mediated from GC, even if with less encouraging outcomes.
Gastroblastoma
Gastroblastoma is a rare epithelial–mesenchymal gastric tumour. To date, only a few cases of gastroblastoma are present in literature, of which only four occurred in adulthood (
The correct treatment strategies have yet to be defined. In general, the disease is almost always confined to the stomach, without metastatic spread potential or risk of recurrence after curative resection. Due to its rarity and virtually absent metastatic potential, which is characterized by a very good prognosis, there is no data regarding systemic treatment strategies for gastroblastoma. Thus, surgery is the primary therapeutic approach in this rare organ-confined disease (see the section ‘Rare epithelial GCs’ for additional details).
Undifferentiated rare carcinomas
Undifferentiated, not otherwise specified (NOS) GC is a completely separate chapter. It includes very rare tumours of epithelial origin that lack any features of differentiation beyond an epithelial phenotype. They generally have a bad prognosis, 137 and no subtype-specific treatment consensus has been reached yet.
About other rare GC subtypes, rhabdoid tumour accounts for 0.1–0.2% of all.138,139 They are extremely rare outside of the kidney (extrarenal rhabdoid tumours or ERRT), and only a few cases are described in the literature 140 affecting the stomach. GC with these features have a dismal prognosis, 141 similar to primary renal rhabdomyosarcoma, and they are aggressive both biologically and clinically, with poor response to available therapies.
Another rare particular histology is undifferentiated sarcomatoid GC, an aggressive biphasic tumour with a combination of malignant epithelial and mesenchymal cells, and to date, only six cases have been reported in the literature. 142 From a prognostic point of view, sarcomatoid carcinoma is metastatic by nature, and the prognosis is strongly related to systemic spread itself, with no possibility of tumour growth control with available therapies. There is very little data about the chemotherapy response of this entity: in general, we know that chemo- and radio-refractoriness is common in gastric sarcomatoid carcinoma, and the best treatment remains a controversial issue. The median survival is about 6 months. 143 Only surgery in localized disease (with tumour resection and LND) could be considered a standardized treatment.
Lastly, the osteoclast-like giant cell (OGC) is a rare undifferentiated GC. This entity is infrequent in the stomach (about 20 patients in the literature 144 ). It could be stratified into at least three distinct types: lymphoepithelioma-like carcinoma, non-lymphoepithelioma-like carcinoma and giant cell tumour (GCT). For lymphoepithelioma-like carcinoma, see the section ‘Gastric cancer with lymphoid stroma: medullary carcinoma and lymphoepithelioma-like carcinoma’. The non-lymphoepithelioma-like subtype is not related to EBV infection; inflammatory infiltrates characterize it, and it is possible that viral EBV DNA was gradually lost in the cancer progression process. The GCT tumour is a neuroendocrine carcinoma enriched in OGC with metaplastic bone formation. There is still debate about the prognostic role of OGC presence within the tumour. Some authors have suggested that lymphoepithelioma-like carcinoma is associated with a better prognosis, 144 while others observed a mixed prognosis, 143 especially in the other two histologies.
The therapy of undifferentiated GC (both NOS and specific subtypes) has yet to be defined. Surgery is the pivotal treatment in early disease. In advanced/metastatic settings, no specific chemo-regimens are to be used (because these histologies are absent in clinical trials), and these tumours are treated with systemic therapies according to gastric adenocarcinoma international guidelines.21,22
Gastric-NETs
Gastric NENs (g-NENs) are rare cancers as per epidemiological definition; however, their incidence has recently increased in most countries, mainly due to the development in diagnostic work-up, the improvement in the knowledge of the disease among clinicians and the frequent use of upper gastrointestinal endoscopy.20,145–147 In summary, the management of g-NENs depends on three key parameters: tumour subtype, histological grade and tumour stage.148,149 Despite recent advances in the molecular profiling of other malignancies, due to the research difficulties for rare cancers and the exact low incidence of g-NENs, no substantial pathways implicated in the development of these tumours have been recognized yet, bringing to a slowed growth of personalized therapeutic options. Finally, the latest clinical studies have revealed that less aggressive treatment strategies such as medical therapy or endoscopic approach may be indicated in patients with smaller and indolent g-NETs. However, more clinical studies should be carried out to clarify the indication for each treatment option better and improve patient outcomes in poorly differentiated g-NENs.
According to Rindi’s classification, 96 g-NENs can be divided into three subtypes and a newly defined fourth type, which may be summarized in Table 4.
Characteristics of the subtypes of neuroendocrine neoplasms of the stomach.

ECL, enterochromaffin-like cells; g-NEN, gastric neuroendocrine neoplasms.
Type I–II g-NENs
Current ENETS guidelines recommend conservative management for type I g-NENs 23 since the risk of metastatic dissemination is directly linked to tumour dimension. 150 As a result, endoscopic surveillance programmes with contextual histological evaluation are the most used approaches for small tumours. Indeed, annual or 2-yearly endoscopic surveillance of type I g-NENs with biopsy sampling and endoscopic resection is recommended for lesions ⩾10 mm in diameter. 23 Nevertheless, several data report that during the biological course of type I g-NET, about 30% of patients undergo endoscopic resection or surgery.151,152 Primary tumour resection is, instead, recommended when endoscopic resection is not indicated, and antrectomy is advisable to remove the source of gastrin production 98 ; indeed, after gastric resection, gastrin levels usually normalize and recurrences are less likely if a radical resection is achieved. 98 A regional lymphadenectomy should be performed for staging purposes.
Regarding medical treatment options, netazepide, a gastrin/Cholecystokinin-2 (CCK2) receptor antagonist, has recently been branded as a potential new treatment for hypergastrinaemia-associated conditions, with evidence from both preclinical and clinical studies in healthy volunteers.153–155 Therapy with netazepide over 12 weeks brought a decrease in the number and size of type I g-NETS, plasma chromogranin A levels and gastric mucosal biomarkers, such as chromogranin A,156,157 with a good long-term safety profile. 158 Despite numerous clinical trials being needed to endorse this initial evidence, netazepide may be a suitable treatment for some type I g-NETs, alone or in combination with endoscopic surveillance.
Recently, somatostatin analogues (SSAs) have been used as an alternative option in type I g-NETs, as these block gastrin release, inhibiting endocrine cell proliferation.159,160 In a recent meta-analysis incorporating data from patients treated with SSAs due to multiple or frequently recurring diseases, SSA therapy achieved an excellent response rate after 12 months of treatment (complete response rate: 84.5%, including six prospective studies).
The favoured treatment for type 2 g-NEN is local excision 161 and surgical resection of the identified gastrinoma. 162 No evidence is available for the treatment of type 2 g-NEN with netazepide. Surgery or endoscopic resection are curative treatments for resectable g-NETs. However, a significant proportion of patients present a recurrence after curative-intent resection, and up to 20–28% have metastatic disease at the time of diagnosis.163–166 Since there are no data to encourage postoperative therapy in type I–II g-NENs, as for all G1–2 GEP-NETs, adjuvant treatment is not routinely recommended.167–169
Treatment options for advanced well-differentiated NENs have substantially increased in recent years, considering the primary tumour site, the extent of disease, tumour expression of somatostatin receptors, proliferation and growth rates as significant prognostic factors to define the treatment strategy.23,170 Most remarkable drugs recently integrated into the treatment scenario of well-differentiated advanced g-NENs include SSAs, everolimus for non-functioning NETs and 177 Luoxodotreotide for somatostatin-receptor-positive GEP-NETs.
In detail, in the phase III CLARINET trial, lanreotide autogel was associated with better progression-free survival (PFS) in advanced GEP-NET patients [median not reached versus 18.0 months; HR, 0.47; 95% confidence interval (CI): 0.30–0.73]. 171 Due to high tolerability and proven clinical outcomes in randomized trials and real-world clinical settings, SSAs are recommended as initial treatment options. The role of everolimus in treating advanced non-functioning NETs of the lung or GEP was explored in the RADIANT-4 trial. Patients receiving everolimus showed a significantly improved median PFS compared to placebo (11.0 versus 3.9 months; HR, 0.48; 95% CI: 0.35–0.67). 172 NETTER-1 was a randomized controlled trial, which compared 177Lu-DOTATATE plus octreotide long-acting release (LAR) (30 mg every 4 weeks) with high-dose octreotide LAR (60 mg every 4 weeks) in patients with advanced, SSA-progressed, well-differentiated (grade 1–2), somatostatin receptor (SSTR)-positive, midgut NETs. 173 177Lu-DOTATATE showed an improved PFS (20-month PFS rates: 65.2% versus 10.8%; HR, 0.21; 95% CI: 0.13–0.33) and higher response rates (18% versus 3%). Since the NETTER-1 trial only comprised midgut NET patients, the efficacy of 177Lu-DOTATATE in patients with pancreatic NETs or non-midgut gastrointestinal NETs has yet to be demonstrated.
Ongoing randomized trials, including the COMPETE trial (grade 1–2 GEP-NETs) for 177Lu-EDOTREOTIDE (NCT04919226) and the NETTER-2 trial (grade 2–3 GEP-NETs with a Ki-67 of 10–55%) for 177Lu-DOTATATE (NCT03972488), are waiting to provide data about the outcomes of 177Lu-based peptide receptor radionuclide therapy (PRRT) in an overall GEP-NET patient cohort.
Lastly, the role of cytotoxic chemotherapy in well-differentiated g-NENs has yet to be well explored, so its use cannot be recommended routinely. Chemotherapy may only have a potential function in selected patients with rapidly progressive tumours after failure of other more effective therapeutic options, including SSAs, everolimus and PRRT.23,174 Robust data to endorse any particular cytotoxic regimen in this setting are lacking; however, the most commonly administered regimens are Streptozocin-based, Temozolomide-based or platinum-based. Hence, enrolment of patients in clinical trials, whenever available, should be highly encouraged in this context.
Type III–IV g-NENs and g-MiNENs
As reported above, for localized type III and IV g-NENs, a surgical approach should be proposed according to the ENETS consensus guidelines.
23
Four recent studies, three from Asia175–177 and one from Europe,
178
advised that local excision as surgical wedge resection or endoscopic resection is a feasible treatment for type III g-NENs characterized by well-differentiated histology, low grade (grade 1 and potentially some grade 2 NETs with Ki-67 index
The mainstay of treatment for non-metastatic Mixed neuroendocrine non-neuroendocrine neoplasms (MiNENs) remains surgical resection. The type of chemotherapy, as adjuvant treatment, depends on the histological characteristics and the dominance of each component tumour. 179 Recently, a comparison study of long-term survival and patterns of recurrence among g-neuroendocrine carcinomas (NECs), g-MiNENs and gastric adenocarcinomas concluded that patients with a g-NEC or g-MiNEN had poorer prognoses than those with gastric adenocarcinoma; therefore, the presence of a g-NEC or g-MiNEN was independently associated with poorer DFS; recurrence-free survival; increased risk of distant tumour recurrence. 180 Chemotherapy is a fundamental component of the multimodality treatment for localized NECs, though no randomized trials are available for G3 extrapulmonary NENs. Considering the high risk of systemic relapse after surgery, all experts and guidelines recommend adjuvant systemic platinum-based chemotherapy after surgery in patients with localized NECs.23,181,182 Cisplatin/carboplatin and etoposide for four to six cycles are generally the most used schemes. Equally, more evidence is needed to support treatment recommendations for advanced, metastatic extrapulmonary G3 NECs, and this is mainly derived from limited retrospective series or tumour registries and very few small, non-controlled clinical trials. 183 A multimodality approach may be considered in the treatment algorithm for metastatic poor differentiated g-NENs, in which surgery, locoregional treatments and systemic medical therapies are included. 184 However, even so, the estimated OS, with the best available therapy, is less than 12 months and barely 1 month in patients who only receive supportive care. 185
Regarding advanced disease, chemotherapy is the mainstay of care.
186
To date, platinum–etoposide combinations are the preferred regimens based on retrospective data, tumour registries and a few small non-controlled clinical trials, while response rates reported in extrapulmonary NECs are lower (
Second-line chemotherapy is not routinely recommended in NECs. Fluoropyrimidine-based chemotherapy, such as FOLFOX, FOLFIRI or CAPTEM (capecitabine plus temozolomide), may be considered in subsequent lines of therapy in patients with good performance status after careful discussion of potential risks and benefits. Temozolomide or Streptozocin-based regimens are generally preferred in G3 NETs. However, it must be highlighted that evidence to support this recommendation is insufficient, and the efficacy of these regimens in this subset of patients should be prospectively investigated.197–205
Recently, immunotherapy has been added to the therapeutic scenario for NENs, starting from the efficacy of ICIs in some difficult-to-treat NENs, such as small-cell lung cancer and Merkel cell carcinoma.206–210 However, unlike those tumours, ICIs such as pembrolizumab and spartalizumab revealed an overall limited antitumoural activity in GEP-NEN patients, highlighting the minimal activity of single-agent immunotherapy and, thus, the need to investigate the efficacy of immunotherapy in GEP-NENs. Overall, even if some evidence suggests the potential for good outcomes with ICIs in high-grade, poorly differentiated GEP-NENs, immunotherapy in unselected GEP-NENs seems unachievable. 211 Further efforts should focus on identifying GEP-NEN patients who respond best to immunotherapy and which combinations will be most effective.
Conclusion
The heterogeneity of rare GCs poses unbreakable limits in diagnosis, data collection, treatment, collaboration and clinical trial development. Thus, understanding and managing these diseases is very challenging, and no specific treatments are available today, especially for rare epithelial GC. Despite recent advances in the molecular profiling of cancers, due to the rarity and the difficulties of proposing research in this subset of rare malignancies, there are still limiting attempts to molecularly sub-classify, and more efforts to develop personalized therapeutic strategies are needed. Therefore, more preclinical research, clinical sharing and collaborations involving patients with these rare cancers are needed. The best way currently available to offer the best treatment opportunities to our patients is the multidisciplinary collaboration in dedicated reference centres for rare cancers with a higher number of cases and higher expertise to increase the acknowledged potential of networking across institutions at a national and international level. 212 Indeed, only international collaborative efforts, as promoted by the European Reference Network for Rare Adult Solid Tumours (EURACAN), can ensure a continuous exchange of scientific and clinical knowledge, making precision medicine a reality in these neglected diseases.
