Abstract
Lymphoepithelioma-like cholangiocarcinoma (LEL-CC) is a type of lymphoepithelioma-like carcinoma (LELC) and a rare variant of primary liver tumor. Although it is uncommon and only 100 cases have been reported thus far, the number of reports has increased in recent years. LEL-CC reportedly occurs more frequently in Asian women; Epstein–Barr virus (EBV) and hepatitis viruses are both strongly associated with tumor development. Here, we describe a 76-year-old woman who exhibited LEL-CC not associated with EBV or hepatitis virus. She was referred to our department with a 3.0-cm × 2.8-cm tumor in the left lobe of the liver. Based on computed tomography and magnetic resonance imaging findings, the tumor was preoperatively diagnosed as hepatocellular carcinoma. Thus, we performed extended left hepatectomy with caudal lobectomy. Histopathological examinations revealed columnar tumor cells with atypical nuclei that proliferated in a cord-like or glandular tubular pattern with dense lymphocytic infiltration. Immunohistochemical analysis showed negative HepPar-1 and arginase findings, indicating non-hepatocyte origin; however, the biliary-type cytokeratins CK7 and CK19 were detected. Based on these findings, the tumor was identified as LEL-CC. EBV-encoded RNA in situ hybridization findings were negative; the patient’s clinical characteristics were not suggestive of hepatitis virus infection. In conclusion, we suggest that clinicians consider LEL-CC as a differential diagnosis for liver tumors in Asian women, including patients without EBV or hepatitis virus.
Keywords
Introduction
Lymphoepithelioma-like carcinoma (LELC) is a rare tumor defined by histopathological features that comprise undifferentiated epithelial cells with prominent lymphoid infiltration. 1 Although these findings were originally considered characteristic of nasopharyngeal carcinoma, 2 LELC occurs in various organs, including the gastrointestinal tract, lungs, and urogenital system.1,3–6 There are two types of LELC in the liver: lymphoepithelioma-like hepatocellular carcinoma and lymphoepithelioma-like cholangiocarcinoma (LEL-CC). 1 To our knowledge, only 100 cases of LEL-CC have been reported in the English literature, but the number of reports has increased in recent years. 7
LEL-CC is reported to exhibit a strong association with Epstein–Barr virus (EBV).1,8 Most LEL-CC cases not associated with EBV involve viral hepatitis or cirrhosis; very few such cases involve no known risk factors. 9 Herein, we report a case of LEL-CC not associated with EBV and hepatitis virus; we also present a literature review of 100 published cases.
Case presentation
A 76-year-old Japanese woman was referred to our department for liver tumor treatment. The tumor was incidentally detected in an abdominal computed tomography (CT) scan performed during hospitalization for a diagnosis of ischemic enteritis. The patient’s medical history included hypertension, dyslipidemia, diabetes mellitus, and angina (all of which had been diagnosed approximately 15–25 years prior); treatment for these conditions constituted valsartan, pitavastatin, and bisoprolol fumarate. The patient reported no smoking or alcohol consumption; she also reported no family history of any clinically significant medical conditions. Physical examination revealed no clinically significant positive signs, including jaundice and abdominal tenderness. Laboratory examinations indicated no abnormal findings, such as liver function test results or changes in serum tumor markers. All viral serological markers suggestive of active hepatitis (hepatitis B: HBs antigen, HBe antigen, HBV-DNA; hepatitis C: HCV core antigen, HCV-DNA) were negative. The patient’s Child-Pugh score was class A. Simple CT revealed a 3.0-cm × 2.8-cm low-density mass located in the left median lobe of the liver (Figure 1(a)). Dynamic contrast-enhanced CT showed an enhanced mass in the arterial phase (Figure 1(b)), as well as central washout and slight pseudocapsule enhancement in the portal and delayed phases (Figure 1(c) and (d)). Magnetic resonance imaging (MRI) using gadolinium ethoxybenzyl diethylenetriamine penta-acetic acid revealed a low-intensity mass on T1-weighted images (Figure 2(a)) and a high-intensity mass on T2-weighted and diffusion-weighted images (Figure 2(b) and (c)); MRI also revealed slight enhancement in the arterial phase (Figure 2(d)) and contrast medium washout in the portal and liver phases (Figure 2(e) and (f)). The patient was preoperatively diagnosed with hepatocellular carcinoma (HCC) and underwent extended left hepatectomy with caudal lobectomy. The operation time was 422 min, and blood loss was 427 mL. The procedure was completed without any intraoperative complications. Gross examination of the excised specimen revealed a 3.0-cm × 2.8-cm tumor with clear borders. Pathological examination showed columnar tumor cells with atypical nuclei that proliferated in a cord-like or glandular tubular pattern (Figure 3(a)). The tumor cells were surrounded by collagen fibers and dense lymphocytic infiltration, as well as lymphoid follicles (Figure 3(b)). The background liver showed normal histological findings with no fibrosis or inflammation suggestive of cirrhosis or chronic hepatitis. Immunohistochemical analysis showed negative HepPar-1 and arginase findings, indicating non-hepatocyte origin (Figure 4(a) and (b)); however, the biliary-type cytokeratins CK7 and CK19 were positive (Figure 4(c) and (d)). EBV-encoded RNA in situ hybridization (EBER-ISH) using a fluorescein-conjugated probe (Bond Ready-to-Use ISH EBER Probe, Leica Biosystems, Melbourne, Australia) according to the instructions was negative, suggesting no latent EBV infection in the tumor (Figure 4(e)). Programmed death-ligand 1 (PD-L1) findings were partially positive in tumor cells (20–30%) and negative in immune cells (Figure 4(f)). Overall, the above findings supported a final diagnosis of LEL-CC. Postoperative recovery was both good and uneventful. The patient was discharged on the 16th postoperative day and has remained alive for 14 months postoperatively without recurrence.

Simple and dynamic contrast-enhanced CT findings.
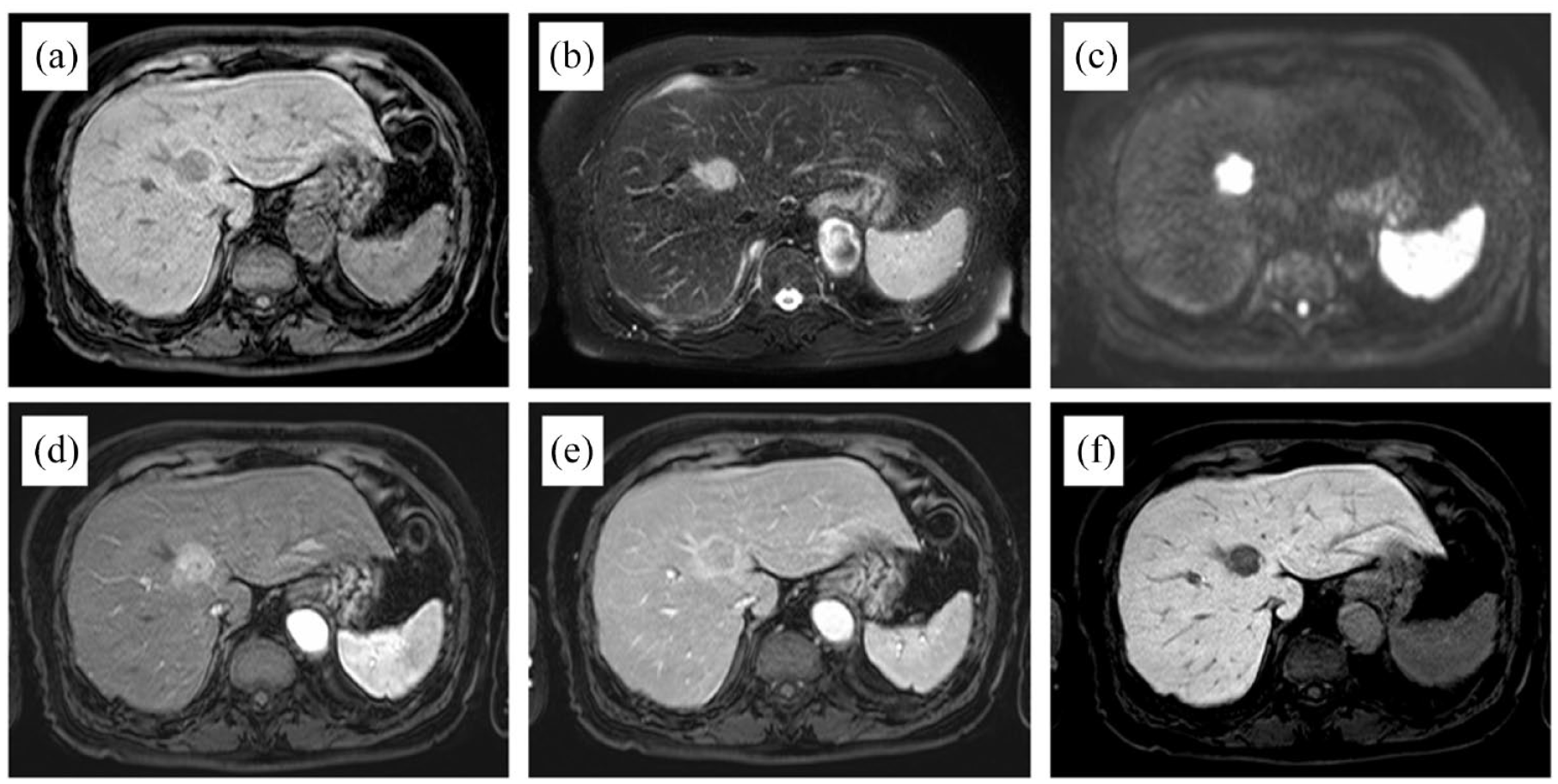
MRI findings using Gd-EOB-DTPA.

Histopathological findings.

Immunohistochemical staining and EBER-ISH findings.
Discussion
LEL-CC was first reported in 1996 by Hsu et al. 10 To our knowledge, 101 cases (including the present case) have been reported in the English literature (Supplemental Table 1)9–37. Of these 101 cases, 74% were published after 2015; the number of reports has increased in recent years. 7 The mean age at diagnosis is 55.3 years (range, 19–79 years). LEL-CC is more common in women (63.3%), and most reported cases have involved individuals with Asian ethnicity (95.0%).
Previous studies have shown that LEL-CC is associated with EBV, as well as the presence of LELC in other organs.1,3–6 Notably, many reported cases (76.2%) have exhibited positive EBER-ISH findings, suggesting an association with EBV. Half of the EBV-negative cases had hepatitis B virus-positive findings, 20.8% had hepatitis C virus-positive findings, and 29.2% exhibited cirrhosis. Most previous reports of LEL-CC were in cases that involved some viral infection or hepatic damage (94.1%); a small proportion of patients (5.9%) had no risk factors, similar to our patient. EBV is generally presumed to have some relationship with LEL-CC tumorigenesis, but the detailed pathogenic mechanism has not been identified. 9 Similar to typical intrahepatic cholangiocarcinoma (iCCA), the development of LEL-CC is hypothesized to involve chronic inflammation caused by a viral infection and immune response. However, recent studies have revealed that the patterns of genetic mutations differ between iCCA and LEL-CC. 9 Genomic analysis of iCCA showed recurrent mutations in KRAS, IDH1, IDH2, BAP1, ARID1A, ARID2, and PBRM1, and defects in translocations involving FGFR2, but these findings were rarely seen in LEL-CC. Mutations in TERT and TP53, which are rarely seen in iCCA, were frequently identified in LEL-CC. 9 The report also suggested that the presence of EBV contributes to distinct histopathological morphology and affects both the frequency and type of genetic mutations. However, another report has suggested that the presence of EBV is irrelevant. 19 Risk factors for classic iCCA include cirrhosis, diabetes mellitus, obesity, alcohol consumption, and smoking. 38 Although our patient had diabetes mellitus, her blood glucose level was adequately controlled by diet alone. It is unclear whether the patient’s diabetes mellitus contributed to the pathogenesis of LEL-CC.
There is no specific symptom that suggests the presence of LEL-CC; it is often detected incidentally during imaging studies of asymptomatic patients. 36 Recently, several studies have been published regarding the imaging findings of LEL-CC. Yang et al. 37 suggested that LEL-CC should be suspected if dynamic contrast-enhanced CT or MRI findings include hypervascularity, washout, delayed intratumoral enhancement, or pseudocapsule enhancement. With respect to contrast-enhanced ultrasonography findings, Ling et al. 33 reported that LEL-CC is characterized by homogeneous hyperenhancement in the arterial phase, as well as gradual washout involving interior hypoenhancement and periphery rim-like slight hyperenhancement in the portal and late phases. In our case, the imaging findings were similar to previously reported results. Typically, dynamic contrast-enhanced CT and MRI findings of iCCA show hypovascular and progressive uptake of contrast during the venous phase. Thus, the typical imaging findings of iCCA differ from those of LEL-CC. 39 However, preoperative diagnosis of LEL-CC may be challenging because the imaging findings are similar to the findings in patients with HCC. 25 Indeed, most patients have been diagnosed by postoperative histopathological findings. 37
LEL-CC is an undifferentiated tumor, resembling nasopharyngeal carcinoma, with syncytial tumor cells arranged in nests, sheets, or cords, rarely glands, with extensive lymphocyte infiltration. It can be seen alone or in combination with areas resembling typical iCCA.1,9 In terms of immunohistochemical staining patterns, the diagnosis of LEL-CC is based on negative hepatocyte marker findings (e.g. HepPar-1 and arginase) and positive cholangiocyte-associated keratin findings (e.g. CK7 and CK19), as well as positive stem cell marker findings (e.g. CD133 and EpCAM). 24
Although there are no guidelines or consensus treatment strategies for LEL-CC, surgical resection is the first choice for patients with resectable lesions.1,9 Although lymph node dissection is generally not indicated, long-term survival was reported in a patient with suspected lymph node metastasis who underwent lymph node dissection and postoperative radiotherapy. 22 For cases that involve distant metastases, chemotherapy is often performed without surgical resection.32,35 On the basis of the view that LEL-CC is a subtype of iCCA, the GC regimen (gemcitabine and cisplatin), the GS regimen (gemcitabine and S-1), and the FOLFOX regimen (folinic acid, fluorouracil, and oxaliplatin) have been selected for LEL-CC chemotherapy.32,35,37 However, these regimens have not been sufficiently effective. Furthermore, considering that LEL-CC is often genetically distinct from iCCA, different management may be needed for LEL-CC. 9 Several studies have shown that the expression of PD-L1 is higher in LEL-CC than in typical iCCA.28,32 Therefore, immune checkpoint inhibitors, including PD-L1 antibodies, are expected to have therapeutic effects. Previous case reports noted that immune checkpoint inhibitors were effective in LELC of the liver and other organs.40,41 The accumulation of cases will presumably lead to the standardization of treatment plans.
The prognosis of LEL-CC is better than the prognosis of typical iCCA. In a retrospective study that compared seven LEL-CC cases with 11 typical stage-matched iCCA cases, Chan et al. 24 found that LEL-CC had a significantly higher overall survival rate than did iCCA (5-year survival: 100% versus 13.2%, p = 0.003). Another report by Tsai et al. 9 compared 27 LEL-CC cases with 250 typical iCCA cases in a single center; LEL-CC was a significantly better independent survival factor in multivariate analysis (hazard ratio, 0.271; 95% confidence interval, 0.127–0.579; p = 0.001). Genetic and non-genetic differences in tumorigenicity and lymphocytic infiltration in the stroma that affect antitumor immune activity are potential reasons for the better prognosis in LEL-CC cases than in typical iCCA cases. 8 Kaplan–Meier survival analysis of 91 patients with available follow-up duration data (median: 27 months; mean: 35.1 months) showed an estimated overall survival rate of 85.9% at 2 years and 62.7% at 5 years. These results compared favorably with other liver malignancies, as in previous reports. The prognosis for LEL-CC may improve further if appropriate drug therapy, including immune checkpoint inhibitors, is developed in the future.28,40
In conclusion, we reported an LEL-CC case without any risk factors such as EBV, viral hepatitis, or cirrhosis. Although such cases are rare, LEL-CC should always be considered in the differential diagnosis of primary liver tumors in middle-aged and Asian women.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221133169 – Supplemental material for Lymphoepithelioma-like cholangiocarcinoma not associated with Epstein–Barr virus or hepatitis virus: case report and literature review of 100 reported cases
Supplemental material, sj-docx-1-tam-10.1177_17588359221133169 for Lymphoepithelioma-like cholangiocarcinoma not associated with Epstein–Barr virus or hepatitis virus: case report and literature review of 100 reported cases by Yuki Adachi, Hideki Yokoo, Masahiro Hagiwara, Hiroyuki Takahashi, Hiroyoshi Iwata, Tomohiro Takeda, Tomohiro Yamamoto, Koji Imai, Sayaka Yuzawa, Mishie Tanino and Naoto Matsuno in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
