Abstract
Background:
Complete resection followed by adjuvant chemotherapy is the gold standard for patients with localized cholangiocarcinoma (CC) or gallbladder cancer (GBC). However, this is not always feasible, and recurrence rates remain high.
Objectives:
To understand the real-world proportions and reason for treatment failure in resected biliary tract cancers.
Design and methods:
We performed a retrospective population-based review of patients with GBC or CC [intrahepatic (IHCC) or extrahepatic (EHCC)] resected between 2005 and 2019 using the BC Cancer provincial database. A chart review was conducted to characterize demographics, treatments received and outcomes.
Results:
In total, 594 patients were identified of whom 416 (70%) had disease recurrence. Most GBCs (96%) were diagnosed incidentally, and repeat oncologic resection was performed in 45%. Adjuvant chemotherapy was received in 51% of patients diagnosed after 2017 (mostly capecitabine). Patient co-morbidities, disease progression and patient preference were the commonest reasons for not proceeding with adjuvant chemotherapy. One-third of patients did not complete all planned cycles. Median overall survival was significantly higher in those with complete (R0) versus incomplete (R1) resection [31.6 versus 18 months, hazard ratio (HR): 0.43, 95% confidence interval (CI): 0.35–0.53] and in those with versus without re-resection for GBC [29.4 versus 19 months, HR: 0.55, 95% CI: 0.41–0.73]. There was a trend towards improved survival with versus without adjuvant therapy (HR: 0.79, 95% CI: 0.61–1.02). Only 25% in the more contemporary cohort (2017–2019) had an R0 resection and completed adjuvant chemotherapy.
Conclusion:
Complete resection, including reresection for incidentally diagnosed GBCs, and adjuvant chemotherapy were associated with improved outcomes in this retrospective cohort, yet many patients were not able to complete these treatments. Neoadjuvant strategies may improve treatment delivery and ultimately, outcomes.
Introduction
Biliary tract cancers (BTCs) are an uncommon group of malignancies comprising cholangiocarcinoma (CC), gallbladder cancer (GBC) and ampullary tumours. 1 CCs can be further subdivided based on their location into intrahepatic cholangiocarcinoma (IHCC) and extrahepatic cholangiocarcinoma (EHCC). It is estimated that around 12,000 people will be diagnosed with BTCs in the United States in 2023, although prevalence rates are higher in other geographical regions such as parts of Asia. 2 The incidence of BTCs in Western countries has been rising recently, likely related to improved imaging and detection techniques and a higher burden of predisposing risk factors such as gallstone disease.3,4 Unfortunately, only 20–30% of BTCs are diagnosed at an early stage, and prognosis remains poor for those with locally advanced and metastatic disease.
Radical surgical resection with negative tumour margins (R0 resection) is the only curative treatment option for patients diagnosed with BTC. However, surgery is often complex, with positive microscopic or macroscopic tumour margins (R1 or R2 resection) found, and recurrence rates remain high. In addition, patients with GBC are often incidentally diagnosed at the time of cholecystectomy for another reason, such as presumed gallstone disease, and are recommended to undergo repeat oncologic resection (ROR) to achieve the best possible margin clearance and nodal assessment.5,6 Adjuvant chemotherapy with 6 months of capecitabine is the standard of care for patients following an R0 or R1 resection, based on the results of phase III randomized BILCAP trial published in 2019. 7 Postoperative radiotherapy may also have a role in select patients, particularly for patients with EHCC and GBC with R1 resection or node-positive disease.1,8
This multi-modality approach to resectable BTCs is not always feasible in clinical practice. Understanding the real-world proportions and reason for treatment failure is important for improving treatment outcomes for patients and designing new approaches. Data regarding this remain scarce, and we aimed to review the real-world population outcomes and patient experience for patients with resectable BTCs in British Columbia, Canada.
Materials and methods
Study design and patient cohort
A retrospective population-based review was undertaken using the BC Cancer provincial database, which is a registry of all new cancers diagnosed in the province of British Columbia, Canada. Inclusion criteria were any patient 18 years of age or older who had a resectable CC or GBC diagnosed between 2005 and 2019. Exclusion criteria included the presence of unresectable or metastatic disease at the time of diagnosis, out-of-country diagnosis, no histological confirmation available, diagnosis of cancer or unknown primary or a confirmed other primary or no available information regarding staging or treatments received. Data collection of the eligible patients was performed using the hospital’s electronic health record. The study was approved by the local Research Ethics Board (approval number H21-02936) and all patient data were anonymized.
A chart review was conducted to collect demographic data including patient age, gender and health authority where resection was performed. Baseline tumour characteristics including primary site, date of diagnosis and mismatch repair status were recorded. Surgical details such as the date of surgery, re-resection date or the reason for not undergoing re-resection, pathological stage and margin status were recorded. Details regarding adjuvant therapy, such as what treatment was given, reasons for not proceeding and completion of planned treatment, were collected. Treatment outcomes including date of recurrence or progression and date of death were recorded.
The reporting of this study conforms to the European Society of Medical Oncology (ESMO) guidance for reporting oncology real-world evidence (see completed checklist in Supplemental Appendix). 9
Statistical analysis
Demographic data, baseline characteristics and treatments received were summarized as frequencies (%) for categorical variables and as medians with range for continuous variables and compared using χ2 tests. Median overall survival (OS) was calculated according to the Kaplan–Meier method, OS was defined as the time from pathologic diagnosis to death with patients censored at their last follow-up if no date of death was available. Treatment groups were compared using a log-rank test and univariate Cox proportional hazard model to generate hazard ratios (HRs) with corresponding 95% confidence intervals (CIs), with p values less than 0.05 considered significant. Multivariate Cox proportional hazard models were subsequently performed using a stepwise forward selection procedure (p value for entry 0.05) with all univariable significant demographic, tumour- or therapy-related parameters. Statistical analysis was performed using IBM SPSS version 28 (IBMI, Armonk, NY, USA).
Results
Patient characteristics
The BC Cancer provincial database identified 1672 patients with a diagnosis of CC or liver adenocarcinoma diagnosed between 2005 and 2019. Those with a confirmed other primary or cancer of unknown primary (150 patients) were excluded, as well as those lacking information regarding diagnosis, staging or treatment (250 patients) (Figure 1). The most common reason for exclusion from the analysis was the presence of unresectable or metastatic disease (n = 676).
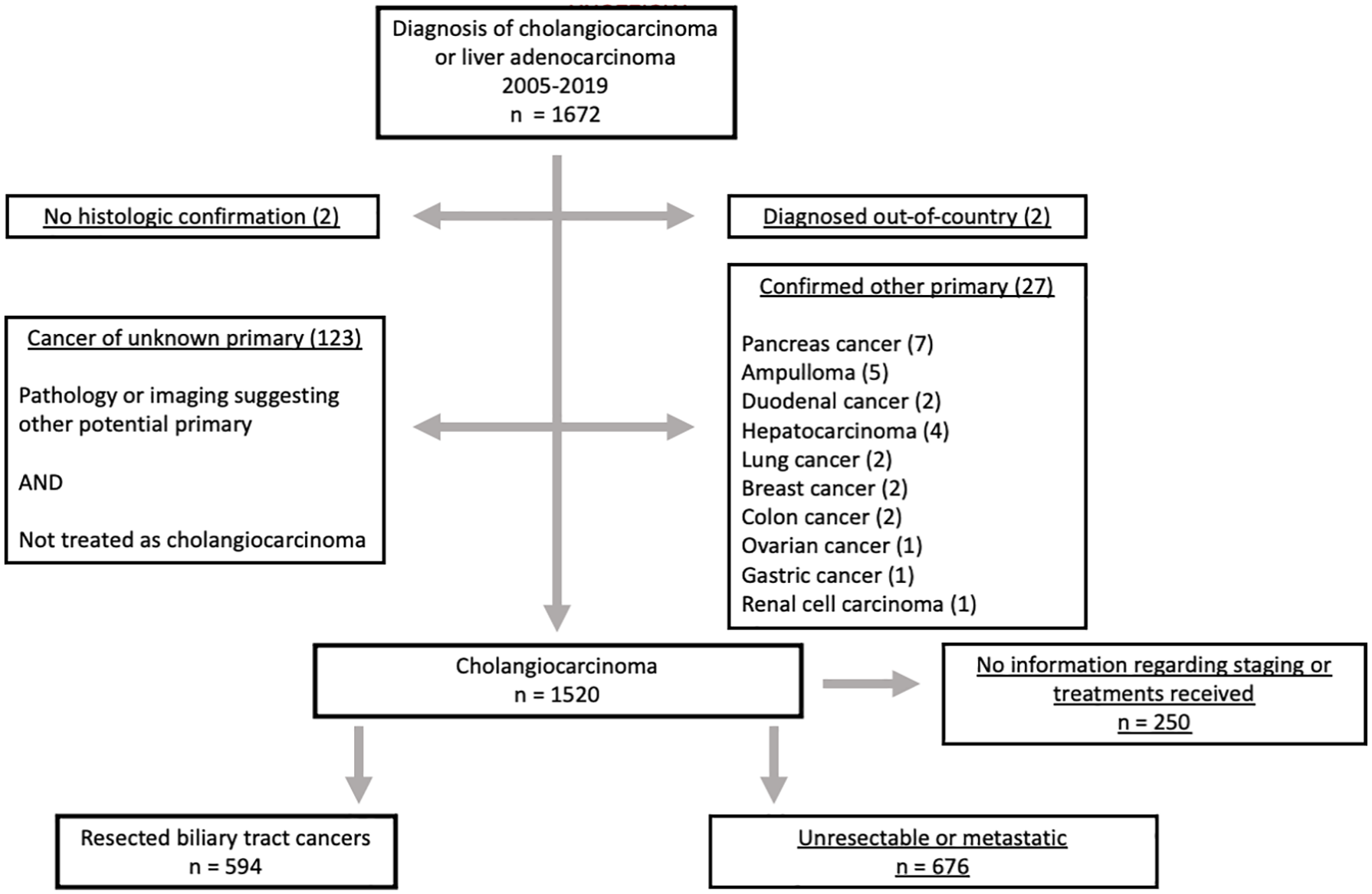
Disposition of patients with a diagnosis of cholangiocarcinoma or liver adenocarcinoma diagnosed between 2005 and 2019.
In total, 594 patients were identified of whom 416 (70%) had disease recurrence. The baseline characteristics are summarized in Table 1. A total of 52.4% of the total cohort were female, and 66.9% of those with GBC were female. Patients diagnosed with IHCC were younger (p = 0.01) and the proportion of stage 1 disease was higher (p < 0.01). Almost half of the included patients (44.3%) were treated between 2015 and 2019. IHCC, GBC and EHCC were found in 21.9%, 44.1% and 34.0%, respectively. Most patients had stage 2 or 3 disease (42.2% and 40.8%, respectively).
Baseline characteristics of the 594 patients in the full study cohort, those with IHCC, GBC and EHCC.
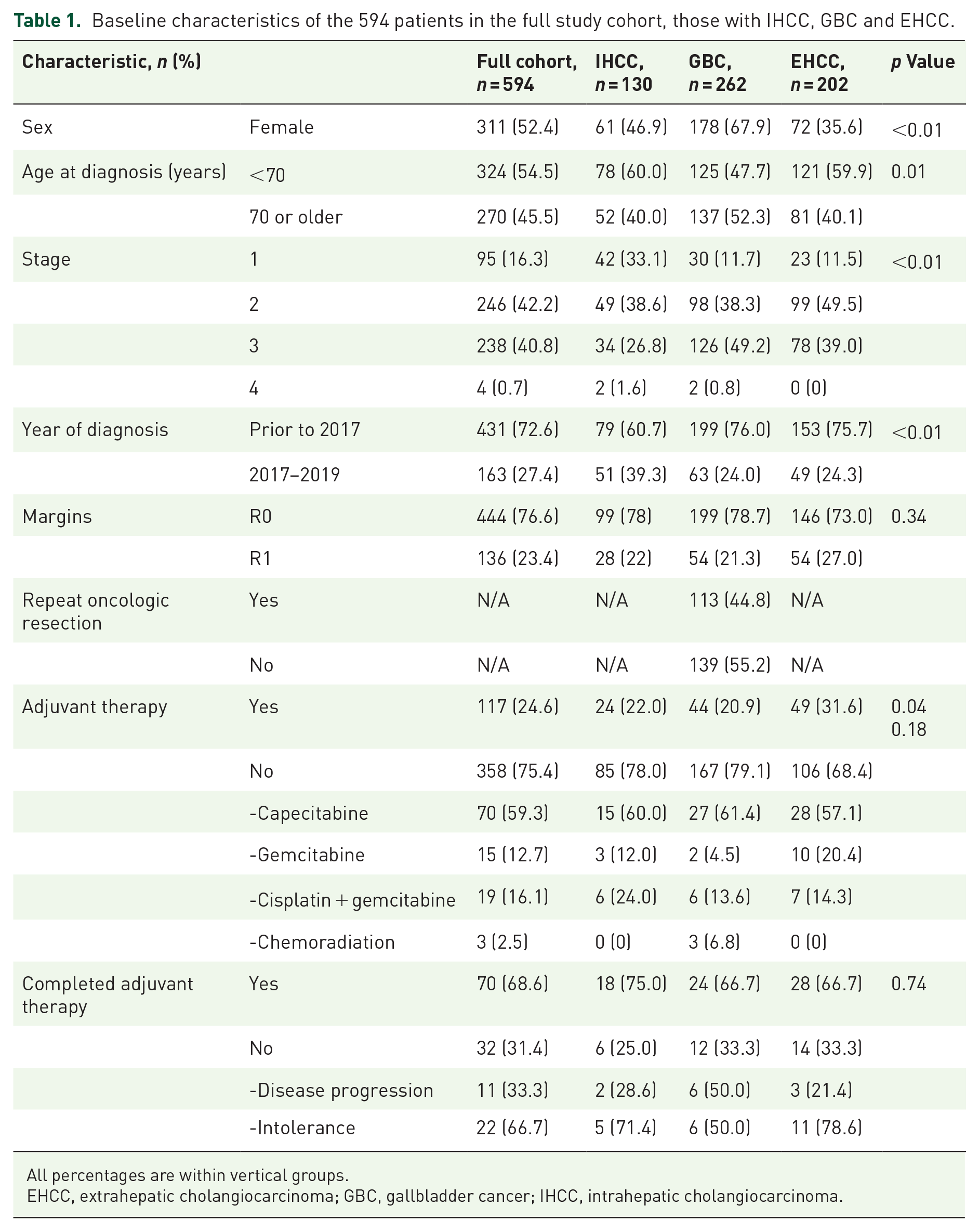
All percentages are within vertical groups.
EHCC, extrahepatic cholangiocarcinoma; GBC, gallbladder cancer; IHCC, intrahepatic cholangiocarcinoma.
ROR in patients with GBC
Most GBCs (96%) were diagnosed incidentally and were stage 2 (38.3%) or stage 3 (49.2%). R0 resection occurred in 78.7% of patients. ROR was performed for 45% of patients with GBCs. The proportion of patients undergoing ROR increased over the years from 36.1% from 2005 to 2009 and 39.7% from 2010 to 2014 to 53.8% from 2015 to 2019 (p = 0.04). Patients undergoing ROR were younger (median age 65 and 75, respectively, p < 0.01) and had more advanced disease on final pathological staging than patients who did not undergo ROR (stage 3 in 61.3% and 41.0%, respectively, p = 0.02). The most common reason for not proceeding with ROR was interval disease progression between initial cholecystectomy and planned re-resection (27.4%). Of those undergoing ROR, 64.4% were found to have residual disease. A higher proportion of patients undergoing ROR achieved R0 resection (86.0% and 72.5%, respectively, p = 0.01) and received adjuvant therapy (34.4% and 7.0%, respectively, p < 0.01). Baseline characteristics are shown in Supplemental Appendix Table 2.
Adjuvant therapies
In the total cohort from 2005 to 2019, 24.6% of patients received adjuvant chemotherapy. However, a larger proportion of patients received adjuvant chemotherapy after presentation of the BILCAP trial results in 2017 (51% of 163 patients). Of these patients, adjuvant therapy consisted of capecitabine (86%), gemcitabine (4%), cisplatin and gemcitabine (5%) or chemoradiation (4%). Common reasons for not receiving adjuvant therapy were age or co-morbidities (23.9%), postoperative complications (18.3%), disease progression (16.9%) and patient’s preference (16.9%). Only three patients in the entire cohort received adjuvant chemoradiation, all of whom had GBC.
Of those receiving adjuvant chemotherapy, 31.3% of the 78 patients treated after 2017 did not complete all planned cycles. The most common reasons for not completing chemotherapy were intolerance (55%) or disease progression (45%). In this more contemporary cohort, only 25% of patients had both complete (R0) resection and completed adjuvant chemotherapy.
Survival outcomes
The median OS in the entire cohort was 27.6 months (95% CI: 25.2–30.0 months). Median OS was longest for those with IHCC (37.3 months, 95% CI: 21.8–52.9 months), and was 25.9 months for those with GBC (95% CI: 22.3–29.5 months) and 25.6 months for those with EHCC (95% CI: 22.0–29.2 months). There was no significant difference in survival between those treated prior to versus after 2015 (p = 0.932). Median OS was 56.6, 26.5, 22.6 and 6.0 months for those with stage 1, 2, 3 and 4 disease, respectively. Survival for patients with pathological stages 1–3 after resection is shown in Figure 2.

Overall survival of the included patients after resection by pathological stage.
In the total cohort, 77% of patients achieved an R0 resection, and there was no significant difference in the R0 resection rate between the IHCC, GBC and EHCC groups (p = 0.34) (Table 1). OS was significantly better in those with a negative surgical margin (p < 0.01). Median OS after resection was 31.6 versus 18.0 months for those who underwent R0 versus R1 resection (HR: 0.43, 95% CI: 0.35–0.53).
In patients with GBC, the median OS was significantly longer (29.4 versus 19.0 months) with ROR versus without re-resection (HR: 0.55, 95% CI: 0.41–0.73) (Supplemental Appendix Figure 1). Furthermore, both those who did (28.4 months, HR: 0.57, 95% CI: 0.37–0.87) and did not (48.4 months, HR: 0.53, 95% CI: 0.29–0.94) have residual disease after ROR had an improved survival compared to those who did not undergo ROR (19.0 months) (Figure 3). Pathological stage, R0 resection and ROR remained associated with improved median OS in a multivariate analysis.

Residual disease and overall survival after repeat oncologic resection for gallbladder carcinoma.
In the total cohort, the median OS was 29.4 versus 25.9 months with and without adjuvant therapy (HR: 0.79, 95% CI: 0.61–1.02), although this did not reach statistical significance (Figure 4). Pathological stage, R0 resection and adjuvant chemotherapy remained associated with improved median OS in a multivariate analysis.

Overall survival with versus without adjuvant therapy.
Discussion
This study provides real-world evidence regarding treatment patterns and outcomes for patients with resectable BTCs. Radical surgery followed by adjuvant therapy offers the best chance of cure. Recurrence rates in this study were high (70%) in keeping with previous evidence; however, only about half of patients treated after 2017 received adjuvant chemotherapy (mostly capecitabine as per the BILCAP study). Survival was significantly higher in those who had a complete resection, and who underwent ROR for GBC. The administration of adjuvant chemotherapy seemed to delay recurrence and improve median survival (although did not reach statistical significance in this study). Overall, only one-quarter of patients in this cohort had both a complete resection and finished adjuvant chemotherapy, demonstrating the need for improved care pathways for patients with BTCs.
With regard to surgical management, specific challenges exist due to the location of these tumours. Accurate diagnosis and staging can be complex, and a substantial proportion of patients with ‘resectable’ disease may have occult metastatic disease detected at the time of surgery or soon after, likely contributing to the high recurrence rates seen. 10 Complete oncologic resection is another challenge, with positive microscopic margins frequently observed despite an increase in the aggressiveness of the surgical approach.11,12 Surgery may not be feasible in over half of cases for a variety of other reasons including vascular invasion not amenable to reconstruction, bilobar involvement, inability to reconstruct the bile ducts, insufficient volume or function of the liver remnant, liver cirrhosis or poor performance status.13,14 Potentially resectable cases should be discussed with a multidisciplinary team of dedicated radiologists, pathologists, oncologists and hepatobiliary surgeons with both surgical oncology and vascular or transplant skills. 15 The European Society of Medical Oncology (ESMO) guidelines recommend increased centralization with surgery performed in specialist hepatobiliary centres due to the relative infrequency and often complex nature of the surgery required. 16
GBCs are often diagnosed incidentally at the time of cholecystectomy for another reason such as presumed gallstone disease. 5 Tumours greater than pT1b are recommended to undergo re-resection with a radical cholecystectomy, regional lymphadenectomy and resection of adjacent liver parenchyma as required to achieve an R0 resection. 6 This has been shown to improve survival compared to no-repeat surgery in retrospective cohorts.17,18 Re-resection allows for more accurate staging of the disease, which, in turn, helps to prognosticate and identify those most likely to benefit from adjuvant therapies. 19 Tumour biology and stage remain the major determinants of GBC prognosis rather than the extent and type of surgical resection, but improved outcomes are seen with R0 resections. 20 In this study, we found that nearly two-thirds of patients undergoing ROR had residual disease. Re-resection was associated with improved pathological staging, higher R0 resection rate and improved OS, confirming findings reported above.
Despite improvements in the surgical management of BTCs over time, recurrence rates remain high. Prior to the BILCAP study, the benefit of adjuvant chemotherapy was uncertain, with two earlier randomized controlled trials being negative. Both the French PRODIGE-12 study of gemcitabine plus oxaliplatin versus surveillance and a Japanese study of gemcitabine versus observation failed to show an improvement in recurrence-free survival or OS after resection.21,22 The BILCAP study was a randomized phase III study of 6 months of adjuvant capecitabine versus observation in patients with resected BTCs. Although this study did not meet its primary endpoint of improved OS in the intention-to-treat (ITT) population, there was an OS benefit seen in the prespecified ITT analysis adjusted for nodal status, disease grade and sex, as well as in the per-protocol analysis. The general consensus is that the data suggest a clinically meaningful benefit of adjuvant capecitabine for patients with BTCs. This quickly became the standard of care for suitable patients and is recommended by multiple treatment guidelines.8,23 In our cohort, approximately two-thirds of patients completed all planned cycles of chemotherapy, which is higher than in the BILCAP study where only 55% completed chemotherapy. There was also a trend seen in this study towards improved survival with versus without adjuvant therapy.
Adjuvant chemoradiotherapy may be offered to patients with EHCC or GBC with a positive resection margin (R1 resection).8,23 This is based on nonrandomized evidence and informal expert consensus opinion, with a significant positive association seen with adjuvant radiotherapy in the R1 and R2 subgroups but not the R0 subgroup.24–26 The single-arm phase II SWOG0809 study established the feasibility of adjuvant chemoradiotherapy with promising survival results in the 79 included patients. Patients received four cycles of chemotherapy (gemcitabine plus capecitabine) followed by chemoradiotherapy to the regional lymphatics and tumour bed. Survival rates were similar between those with R0 and R1 resections. In our study, only a small number of patients received adjuvant radiotherapy, so it is difficult to draw specific conclusions about the outcomes of this cohort.
Alternative patient care pathways should be explored to further improve outcomes in BTC. For example, neoadjuvant chemotherapy or chemoradiation followed by liver transplantation may be a potential option for select patients with unresectable perihilar cholangiocarcinoma.27–29 Locoregional therapies such as radiofrequency ablation, chemoembolization, radioembolization, chemotherapy hepatic arterial infusion, external beam radiotherapy and stereotactic body radiation have some evidence in unresectable BTC, and further studies should explore their potential role in resectable disease. 30
Neoadjuvant and perioperative strategies may also improve outcomes. In particular, patients with GBC comprised less than 20% of those enrolled in the BILCAP study, and it is difficult to conclude for this small subgroup. We confirmed that those patients not undergoing ROR or with residual disease after ROR have the poorest survival outcomes. Even in the more contemporary cohort of 2015–2019, many patients do not undergo ROR most frequently because of interval disease progression, which may partly account for the differences noted in survival. One potential approach to address this is being studied in the OPT-IN/EA2197 trial (currently underway) exploring the role of perioperative cisplatin plus gemcitabine chemotherapy versus upfront ROR and then adjuvant chemotherapy for patients with T2 or T3 GBC found incidentally at cholecystectomy. 31
Limitations of this study include the retrospective design with associated selection bias. Included patients spanned a broad range of time from 2005 to 2019, during which period the management of BTCs has evolved considerably, both with regard to adjuvant therapy recommendations and surgical techniques. The reasons behind the choice of whether or not to offer adjuvant treatment were not examined in this study, and this introduces bias to the results. The specific advances in the surgical management of patients with BTCs were outside the scope of this study. Other factors that may impact survival for patients include lifestyle factors, such as smoking status and alcohol intake, and these should be explored in future analyses.
Conclusion
Overall, this study highlights the high recurrence rates seen with BTCs despite optimal surgical resection, and the need to find improved treatment pathways for patients. There is now evidence for re-resection in GBC, adjuvant chemotherapy with capecitabine and adjuvant chemoradiation for select patients. Decisions regarding post-resection management require shared decision-making in a multidisciplinary team, preferably at a high-volume centre. This study re-affirms the evidence and current recommendations that R0 resection and adjuvant chemotherapy are key components to improve survival for patients with resectable BTCs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241247008 – Supplemental material for Real-world experience supporting the role of oncologic resection and adjuvant chemotherapy in biliary tract cancers
Supplemental material, sj-docx-1-tam-10.1177_17588359241247008 for Real-world experience supporting the role of oncologic resection and adjuvant chemotherapy in biliary tract cancers by Melina Boutin, Tharani Krishnan, Maria Safro, Jenny Yang, Helia Jafari, Janine M. Davies and Sharlene Gill in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241247008 – Supplemental material for Real-world experience supporting the role of oncologic resection and adjuvant chemotherapy in biliary tract cancers
Supplemental material, sj-pdf-2-tam-10.1177_17588359241247008 for Real-world experience supporting the role of oncologic resection and adjuvant chemotherapy in biliary tract cancers by Melina Boutin, Tharani Krishnan, Maria Safro, Jenny Yang, Helia Jafari, Janine M. Davies and Sharlene Gill in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
