Abstract
Background:
Few epidemiological data are available on surgically treated Caucasian patients with non-small-cell lung cancers (NSCLCs) harboring epidermal growth factor receptor (EGFR) mutations. The main objective of this study was to describe, in the real-world setting, these patients’ incidence, clinical, and tumoral characteristics.
Methods:
The participating centers included all consecutive localized non-squamous NSCLC patients undergoing surgery between January 2018 and December 2019 in France. EGFR status was determined retrospectively when not available before surgery.
Results:
The study includes 1391 no squamous NSCLC patients from 16 centers; EGFR status was determined before surgery in 692 (49.7%) of the cases and conducted as part of the study for 699 (50.3%); 171 (12.3%) were EGFR mutated; median age: 70 (range: 36–88) years; female: 59.6%; never smokers: 75.7%; non-squamous histology 97.7%, programmed death ligand-1 expression 0%/1–49%/⩾50 in 60.5%/25.7%/13.8%, respectively. Surgery was predominantly lobectomy (81%) or segmentectomy (14.9%), with systematic lymph node dissection in 95.9%. Resection completeness was R0 for 97%. Post-surgery staging was as follows: IA: 52%, IB: 16%, IIA: 4%, IIB: 10%, IIIA: 16%, and IIIB: 0.05%; EGFR mutation exon was Del19/exon 21 (L858R)/20/18 in 37.4%/36.8%/14%, and 6.4% of cases, respectively; 31 (18%) patients received adjuvant treatment (chemotherapy: 93%, EGFR tyrosine kinase inhibitor: 0%, radiotherapy: 20%). After a median follow-up of 31 (95% confidence interval: 29.6–33.1) months, 45 (26%) patients relapsed: 11/45 (24%) locally and 34 (76%) with metastatic progression. Median disease-free survival (DFS) and overall survival were not reached and 3-year DFS was 60%.
Conclusion:
This real-world analysis provides the incidence and outcomes of resected EGFR-mutated NSCLCs in a European patient cohort.
Keywords
Introduction
In France, non-small-cell lung cancer (NSCLC) represents the most common lung tumor (80–85% of cases) of which approximately 20% are localized or 20% locally advanced at diagnosis. 1 The standard treatment for localized NSCLCs is surgery, including complete lymph node dissection.2,3 Adjuvant chemotherapy is recommended for tumors >4 cm in diameter and/or with lymph node involvement.2,3 Adjuvant chemotherapy has achieved only minor benefits with a limited 5-year overall survival (OS) gain of 5.4%. 4 Since the publication of the Lung Art study results, the place of postoperative radiotherapy seems marginal. 5
However, significant progress in knowledge about NSCLC oncogenesis has been made in recent years, particularly the discovery of a specific oncogene playing a role in the oncogenic addiction responsible for NSCLC development.6–10 Activating mutations in the epithelium growth factor receptor (EGFR) gene have been found in 10–15% of patients with metastatic NSCLCs in Europe, and 80–85% of them are activating mutations in exons 19 and 21 L858R (common mutations), sensitive to targeted EGFR-tyrosine kinase inhibitors (TKIs). 11
The benefit of adjuvant EGFR-TKI (first generation) therapy for localized EGFR-mutated NSCLCs was evaluated in several studies.11–14 These studies seemed to show an improvement in disease-free survival (DFS) for patients treated with adjuvant EGFR-TKI.
Recently, the ADAURA study tested the efficacy of adjuvant osimertinib treatment for 3 years versus placebo for patients with resected stage IB–IIIA, EGFR-mutated NSCLCs. 15 That study’s results showed a significantly longer DFS for osimertinib-treated stage II–IIIA patients [hazard ratio (HR): 0.17 (99.06% confidence interval (CI): 0.11–0.26); p < 0.001]. In stage II–IIIA disease, the 4-year DFS rate was 70% (osimertinib) and 29% (placebo). In the overall population, the 4-year DFS rate was 73% (osimertinib) and 38% (placebo). 15 A recent update of the study showed a significant improvement in OS for patients treated with adjuvant osimertinib versus placebo. 16 In the overall population (stage IB–IIIA), OS HR was 0.49 (95% CI: 0.34–0.70; p < 0.0001); the 5-year OS rate was 88% with osimertinib versus 78% with placebo, with a median follow-up for OS of 60.4 months (osimertinib) and 59.4 months (placebo). 16
Osimertinib adjuvant therapy for resected EGFR-mutated NSCLCs would appear to be a new standard for treatment but we still have a few data on the epidemiology of the EGFR mutation in these patients, especially Europeans. The primary objective of this study was to describe, in the real-world setting, the incidence, clinical characteristics, the mode of progression in patients with resected EGFR-mutated NSCLCs.
Methods
This retrospective multicenter study included all consecutive patients >18 years old, with resected non-squamous NSCLCs or non-smokers with squamous NSCLCs, stages I–IIIA (neoadjuvant or adjuvant chemotherapy was allowed) from 1 January 2018 until 31 December 2019. The main exclusion criteria were patients under guardianship or inability to retrieve collected data, and no available tissue for retrospective testing.
The following information was recorded: sociodemographic parameters, smoking status, medical history; NSCLC characteristics: histology, stage, mutation search before surgery or not; perioperative treatments. When EGFR status was not available, it was determined retrospectively, either locally or by a central laboratory. EGFR mutation was assessed by Cobas, Sanger sequencing, and/or next-generation sequencing. Ex19del, L858R, and other uncommon EGFR mutations were included. All patients were staged according to the American Joint Committee on Cancer eighth edition.
Patient follow-up was consistent with the participating center’s usual practices.
Sample size
In light of the descriptive design of the study, the number of patients was not calculated. It is estimated that 50,000 new lung cancers are diagnosed and 10,000 curative resections are performed annually in France; among them, 65% are patients with non-squamous histology. 17 Inclusion of 1300 patients over 2 years would suffice as a representative sample.
Statistical methods
Qualitative parameters are expressed as numbers (%) and (95% CIs) and quantitative parameters as mean ± standard deviation or median [interquartile range (IQR)]. Numbers of missing data are reported. The primary outcome was EGFR-mutated NSCLC incidence. Secondary outcomes included DFS, defined as the date from surgery until disease relapse or death (whichever occurred first); surviving patients without relapse were censored from the date of their last follow-up visit. OS was defined as the time from initial surgery (date) to date of death, with surviving patients censored at the date of their last follow-up visit. Statistical analyses were computed with SAS® software.
The study conformed to the principles of the Declaration of Helsinki and Good Clinical Practice Guidelines. The study received Ethics Committee approval (Comité de Protection des Personnes, Sud-Est) on 06/04/2021 (no. ID RCB: 2020-A03528-31). The Groupe Francais PneumoCancerologie (GFPC) has committed to the Commission Nationale Informatique et Libertés (CNIL) to respect the MR-003 reference methodology in the prospective module for research involving human subjects category 3 studies. A written informed consent was obtained from all patients.
The reporting of this study conforms to the STROBE guidelines statement (Supplemental Table S1). 18
Results
From 1 January 2018 to 31 December 2019, 1391 consecutive patients were included in 16 centers (Supplemental Table S1). Histological diagnoses were known before surgery for 49.7% of the patients. This study provided the EGFR status for 100% of them: that is, 171/1391 (12.3%) patients harbored an EGFR-activating mutation: 102/171 (59.6%) women, 126/171 (73.7%) no-smokers, with a median age of 70 (36–88) years. Almost all NSCLCs (97.7%) were non-squamous. Tumor-cell expression of programmed death ligand-1 (PD-L1) was negative for 60.5% of them. The molecular assay used for EGFR testing was Next Generation Sequencing (132/171: 77%, Sanger sequencing 7%, unknown 16%). Exon 19 deletion (37.4%) and exon 21 L858R (36.3%) EGFR mutations were the most common (Table 1). Uncommon mutations were as follows: exon 21 L861Q, n = 2, exon 20 insertion, n = 22, with T790M de novo alone, n = 3 and with exon 21 L858 R mutation, n = 1, exon 18, n = 9. Six patients had detectable co-mutations: three Kirsten rat-sarcoma viral oncogene, one human epidermal growth factor receptor-2, one anaplastic lymphoma kinase, and one proto-oncogene tyrosine-protein kinase-1 (ROS-1). Seven patients had complex EGFR mutations.
Clinical characteristics of all Caucasian patients with resected, EGFR-mutated NSCLCs.

Results are expressed as n (%) unless stated otherwise.
One patient’s stage is unknown and one patient is stage IV.
Exons 18–20: 3, exons 19–20: 2, exons 20–21: 1.
Exons 19–18: 1.
EGFR, epidermal growth factor receptor gene; NSCLC, non-small-cell lung cancer; PD-L1, programmed death protein-1 ligand.
Surgical techniques (n = 171) were video-assisted thoracoscopy (45%), robot-assisted thoracoscopy (12%), and thoracotomy (38%) (unknown 5%). Lobectomy (81%) was the predominant surgical procedure and 96% of patients underwent systematic lymph node dissection (Table 1). The completeness of the surgical resection was R0 for 97%. The most frequent, postoperatively determined stages were IA (89; 52%) and IB (28; 16%). Only 9 (5%) patients received neo-adjuvant treatment (chemotherapy for 7 and EGFR-TKI for 2) and 31 (18%) were prescribed adjuvant treatment (chemotherapy for 29 and radiotherapy for 6); none were given EGFR-TKI.
After a median follow-up of 31 [95% CI: 29.6–33.1] months, 45 (26%) patients relapsed: 12/45 (24%) locally and 33 (74%) with metastatic progression (Table 2). The most frequent metastatic sites were brain 24% (n = 11; patients stage: IIIa: n = 3, stage II: n = 4, stage I: n = 4), bone 24% (n = 11), and lung 22% (n = 10). Relapse patients stage I, stage II, and stage III relapse locally (n = 5, n = 5, and n = 2, respectively), relapse with metastatic progression (n = 7, n = 6, and n = 11, respectively), and both (n = 2, n = 2, and n = 5, respectively) (Supplemental Table S2).
Characteristics of patients with resected, early-stage, EGFR-mutated NSCLCs according to relapse status.
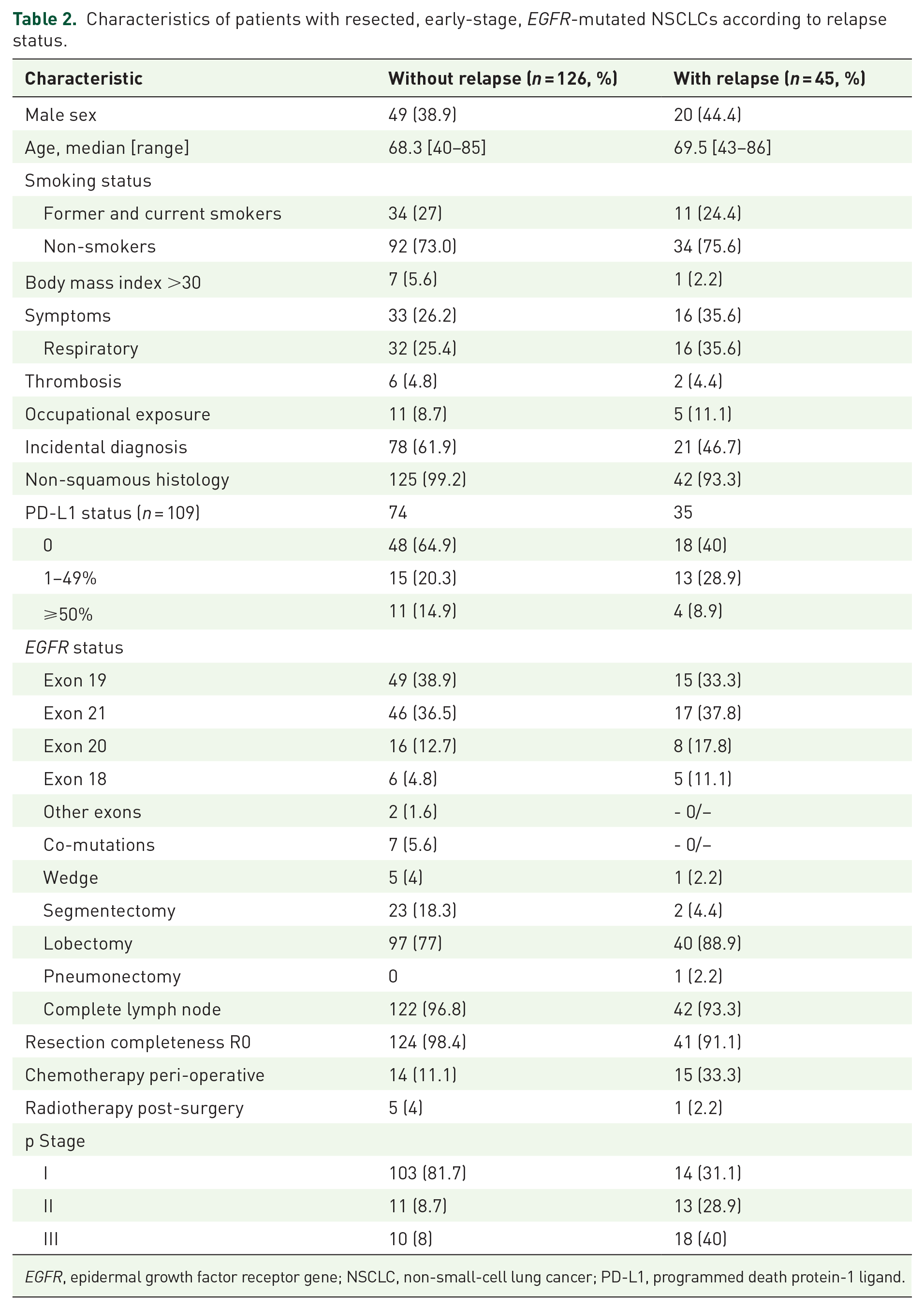
EGFR, epidermal growth factor receptor gene; NSCLC, non-small-cell lung cancer; PD-L1, programmed death protein-1 ligand.
Among the patients whose disease progressed, 18/45 (40%) had a new biopsy (tissues and liquid biopsies for 15, only liquid biopsies for 3). EGFR mutation findings were the same for 14/18 (77.8%) of them (an EGFR mutation was not found in one patient and the results are not available for three others). Almost all patients with exclusively local progressive disease (10/11, 91%) received local treatment. When used, systemic agents were most often EGFR-TKI (24/34, 71%), chemotherapy (4/34, 11.8%), and immune checkpoint inhibitor (4/34, 11.8%). Median DFS and OS were not reached (Table 3). The 3-year DFS and OS were 60% and 84.7%, respectively (Table 3).
DFS and OS according to NSCLC p stage.

CI, confidence interval; DFS, disease-free survival; NSCLC, non-small-cell lung cancer; OS, overall survival.
DFS and OS appear similar for exons 18, 19 deletion, exon 21 L585R EGFR mutations DFS and OS at 3 years of 71%/85%, 67%/87%, and 59%/85%, respectively), but worse for exon 20 insertion mutations (49–69%) (Table 4).
Patients’ follow-up status according to the NSCLC EGFR-mutated exon.

CI, confidence interval; DFS, disease-free survival; EGFR, epidermal growth factor receptor; NR, not reached; NSCLC, non-small-cell lung cancer; OS, overall survival.
Discussion
In this retrospective, multicenter analysis of consecutive resected non-squamous NSCLCs (2018–2019), the EGFR mutation rate was 12.3%. Albeit slightly higher than for metastatic forms in France, it is consistent with previously published findings. 19 That analysis of the France biomarker database yielded a 10.2% EGFR mutation rate for the 854 surgically treated patients. Even though that rate is not strictly an incidence because patients were not included consecutively, it is consistent with ours.
Using Pan-cAnadian Lung cancEr Observational Study data (2012–2019), authors reported the results of a retrospective, multicenter, observational cohort study on stage IB–IIIA resected NSCLCs that had not received neoadjuvant therapy. Among the 535 patients whose tumor samples underwent reflex EGFR mutation testing, 23% were carriers, 15.9% with common, and 5.6% with uncommon mutations. 20 That higher rate could be explained by a larger Asian-ancestry population (35%) than ours.
The clinical characteristics of patients with localized EGFR-mutated NSCLCs are similar to those of metastatic forms, with a predominance of women and non-smokers. The high percentage of patients with PD-L1-negative tumor cells (68%) should also be noted. The activating mutations (in exons 19 and 21, L858R) represented the majority of the alterations, as in metastatic forms. Exon 20 mutation seemed to be overrepresented in our population and the Canadian series. 20 The findings also agreed with data from Asian populations.21,22 Similar results were reported based on a retrospective analysis of an Asian cohort (389 patients managed between 1 January 2010 and 30 June 2018, at a specialized cancer center in Singapore) for distributions of stages, surgical procedures, R0 rates, relapse rates, and DFS results. 22 The results were also very close for the Canadian series for stage distribution, surgery, and DFS. 20 At 2 years, the probability of being disease-free was 75% (95% CI: 65–87), 48% (95% CI: 36–66), and 38% (95% CI: 24–61), respectively, for stages IB, II, and IIIA, and 45% (95% CI: 35–58) for stages II–IIIA combined. 20 Finally, our results are also in agreement with the control arm of the ADAURA study, with 2-year DFS at 44% (95% CI: 37–51) for stage II–IIIA patients. 15
We emphasize, as did the Asian study, that outcomes for stage IA and IB NSCLCs were similar. That observation is worth noting, given that patients with stage IA NSCLCs were not included in the ADAURA study and are otherwise not eligible for osimertinib. Even among patients with very early-stage disease, nearly 20% of them still experience early relapse or death. This high rate of relapse in early-stage patients (stage I) was also observed in the Canadian study (stage Ia 19% and stage Ib 21%), but no difference was found between mutated and wild-type EGFR patients. 22
The prognostic impact of EGFR alterations on early-stage NSCLC outcomes is conflicting, and factors associated with relapse risk in patients with EGFR-mutated NSCLCs need further elucidation.23,24 In an Asian cohort, prognostic EGFR mutation impact on DFS for patients with pT1a and pT1b invasive lung non-squamous cancers after surgical resection did not differ from that of patients whose tumors expressed wild-type EGFR; however, among all patients, OS was significantly longer for those with EGFR-mutated NSCLCs, and N0 and N1–2-stage subgroups. 21 That observation is probably explained by the number of effective targeted treatment options for metastatic EGFR-mutated NSCLCs. Uni- and multivariate analyses of wild-type EGFR-expressing NSCLCs found only more advanced stage and lymphovascular invasion to be significantly associated with relapse. By contrast, univariate and multivariate analyses of patients with EGFR-mutated NSCLCs identified more advanced stage, non-acinar and non-lepidic non-squamous subtype, sub-lobar resection, positive resection margins, and lymphovascular invasion were significantly associated with relapse. 24 Exploratory whole-exome and transcriptome-sequencing analyses of a subgroup of 85 patients with EGFR-mutated NSCLCs found alterations in rhophilin-2 (RHPN2) and β-catenin-1 (CTNNB1), and micropapillary subtype increased the risk of relapse, while loss of retinoblastoma-1 (RB1) was associated with a lower risk. 25 Confirmation of those preliminary results would contribute to better identification of the patients at the highest risk of relapse.
This analysis has several unavoidable limitations given its retrospective nature. One of the main limitations is that of the interpretation of DFS results in a real-world study. Indeed, disease relapse was defined here by the investigators according to their usual clinical practices, which does not imply systematic achievement of the objective Response Evaluation Criteria In Solid Tumor 1.1. In this study, the proportion of early-stage patients (stage I) is high but correctly reflects surgical activity. 26 Also, not all patients had a complete NGS which may underestimate the proportion of co-mutations.
Conclusion
The incidence of surgically resected, early-stage, EGFR-mutated NSCLCs in France is slightly higher than for advanced NSCLC stages. The findings based on this consecutive series agree with previous research findings that relapse rates of those EGFR-mutated NSCLCs are high, including for stage IA. Identifying individual risk features could facilitate tailored surveillance and adjuvant treatment strategies for early-stage EGFR-mutated NSCLCs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241236451 – Supplemental material for Resected EGFR-mutated non-small-cell lung cancers: incidence and outcomes in a European population (GFPC Exerpos Study)
Supplemental material, sj-docx-1-tam-10.1177_17588359241236451 for Resected EGFR-mutated non-small-cell lung cancers: incidence and outcomes in a European population (GFPC Exerpos Study) by Jean-Bernard Auliac, Pascal-Alexandre Thomas, Olivier Bylicki, Florian Guisier, Hubert Curcio, AlainVegnenègre, Aurelie Swalduz, Marie Wislez, Jacques Le Treut, Chantal Decroisette, Victor Basse, Lionel Falchero, Gonzague De Chabot, Diane Moreau, Eric Huchot, Audrey Lupo Mansuet, Helene Blons, Christos Chouaïd and Laurent Greillier in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
