Abstract
Background:
Currently, there is no recommended standard third-line chemotherapy for metastatic gastric cancer.
Objectives:
In this study, we aimed to evaluate irinotecan’s efficacy and safety in treating metastatic gastric cancer after the failure of first- and second-line chemotherapy.
Design:
Prospective single-arm, two-center, phase II trial.
Methods:
Patients were aged 18–70 years, with histologically confirmed gastric adenocarcinoma and an Eastern Cooperative Oncology Group performance status of 0–1, progressed during or within 3 months following the last administration of second-line chemotherapy and had no other severe hematologic, cardiac, pulmonary, hepatic, or renal functional abnormalities or immunodeficiency diseases. Eligible patients received 28-day cycles of irinotecan (180 mg/m2 intravenously, days 1 and 15) and were assessed according to the RECIST 1.1 criteria every two cycles. Patients who discontinued treatment for any reason were followed up every 2 months until death. The primary endpoint was overall survival (OS), and the secondary endpoints were progression-free survival (PFS), objective response rate (ORR), disease control rate (DCR), and toxicity.
Results:
A total of 98 eligible patients were enrolled in this study. In the intention-to-treat population, the median OS was 7.17 months, the median PFS was 3.47 months, and the ORR and DCR were 4.08% and 47.96%, respectively. In the per-protocol population, the median OS was 7.77 months, the median PFS was 3.47 months, and the ORR and DCR were 4.82% and 50.60%, respectively. The incidence of grade 3 or 4 hematological and non-hematological toxicities was 19.4%, and none of the patients died owing to adverse events. Cox regression analysis revealed neutropenia and baseline thrombocyte levels were independently correlated with PFS and OS.
Conclusion:
Irinotecan monotherapy is an efficient, well-tolerated, and economical third-line treatment for patients with metastatic gastric cancer as a third-line treatment.
Trial registration:
ClinicalTrials.gov identifier: NCT02662959.
Introduction
Gastric cancer is among the most common cancers worldwide. In 2020, more than 1 million new gastric cancer cases and 769,000 deaths were noted globally, making it the fifth most common cancer and fourth leading cause of cancer-related deaths worldwide. 1 The incidence and mortality of gastric cancer in Eastern Asia are the highest globally, almost twice as high as those in the second-highest region. 1 Gastric cancer is the third most common cancer and the third leading cause of cancer death in China. 2 Although gastric incidence and mortality rates have decreased, the disease burden has remained significant. 2
Surgery or endoscopic resection is recommended as a curative treatment for early gastric cancer. 3 However, gastric cancer is highly heterogeneous and malignant.4,5 Many patients would experience recurrence after curative resection and approximately 50% of patients already have locally advanced or metastatic gastric cancer at diagnosis, for whom systemic chemotherapy is recommended.6–8 Chemotherapy can improve the survival and quality of life of patients with metastatic gastric cancer.9–11 The median overall survival for patients with advanced gastric cancer who received combination chemotherapy was approximately 1 year (Asian patients tended to have slightly longer survival) compared to 3–4 months for patients receiving best supportive care alone. 7 Therefore, patients with good performance status and organ function should receive chemotherapy. Chemotherapeutics widely used in gastric cancer treatment include fluorouracil, oxaliplatin, cisplatin, capecitabine, S-1, paclitaxel, irinotecan, etc. 12
Irinotecan is a semi-synthetic, water-soluble camptothecin derivative inhibiting DNA topoisomerase I and an S-phase-specific drug. The complex formed by irinotecan or its active metabolite, SN-38, together with topoisomerase I and DNA, can break the single-strand DNA, preventing DNA replication and inhibiting RNA synthesis. 13 Although irinotecan directly binds to DNA and prevents DNA replication, similar to platinum-based drugs, it does not cause cross-resistance to other types of antitumor drugs because of its unique mechanism of action. Irinotecan has almost no nephrotoxicity or cardiotoxicity and causes little local tissue irritation. The main side effects are cholinergic syndrome, delayed diarrhea, and neutropenia, which are predictable and relatively easy to handle. 14
In the first-line chemotherapy, a platinum–fluoropyrimidine doublet is preferred for patients with metastatic gastric cancer. 12 Preferred regimens for the second-line therapy include docetaxel, paclitaxel, and irinotecan. In China, paclitaxel and docetaxel are commonly used as second-line chemotherapy for metastatic gastric cancer. However, whether irinotecan monotherapy could be used as a salvage treatment after the failure of second-line treatment lacks evidence. In this trial, we prospectively explore the effect of irinotecan as a third-line regimen.
Methods
Study design and participants
This prospective, single-arm, two-center, phase II trial was performed at the Fudan University Shanghai Cancer Center and Shanxi Cancer Center in China.
Eligible patients were aged 18–70 years, with histologically confirmed gastric adenocarcinoma and an Eastern Cooperative Oncology Group performance status of 0–1, progressed during or within 3 months following the last administration of second-line chemotherapy, and had no severe hematologic, cardiac, pulmonary, hepatic, or renal functional abnormalities or immunodeficiency diseases. Laboratory tests required to meet included the following: hemoglobin level ⩾90 g/L; absolute neutrophil count ⩾1.5 × 109/L; platelet count ⩾100 × 109/L; bilirubin level <1 × the upper limit of normal (ULN); aspartate aminotransferase and alanine aminotransferase levels <2.5 × ULN; serum creatinine level ⩽ 1 × ULN. The key exclusion criterion was previous exposure to irinotecan.
Procedures
Eligible patients received 28-day cycles of irinotecan (180 mg/m2 intravenously on days 1 and 15). The infusion time was 0.5–1.5 h with antiemetics for premedication. Treatment was continued until documented progression of the disease (PD), death, intolerable toxicity, or unwillingness to continue treatment.
Patients received a baseline assessment, including the collection of information on demographics, medical history, and disease characteristics before enrollment, and they underwent systematic physical examination and relevant laboratory and imaging (chest computed tomography (CT), abdominal and pelvic CT, or magnetic resonance imaging (MRI)) tests before treatment. Tumors were evaluated using CT or MRI every two cycles. Response to treatment was assessed as complete response (CR), partial response (PR), stable disease (SD), or PD according to the RECIST 1.1 criteria.15 Patients who discontinued treatment for any reason were followed up every 2 months until death.
Outcomes
The primary endpoint was overall survival (OS), defined as the time from inclusion to death due to any cause. The secondary endpoints included progression-free survival (PFS) (defined as the time from inclusion to PD/death), ORR (defined as the rate of CR and PR), DCR (defined as the rate of CR, PR, and SD), and toxicity. Adverse events (AEs) recorded during treatment were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0.
Statistical analysis
The median OS (mOS) of patients with metastatic gastric cancer who failed second-line treatment was 4 months, which was extended to 6 months in this study. The α value was 0.05, and the 1−β value was 0.8. Therefore, the sample size was calculated as 85 cases. The dropout rate was 10%, and the overall sample size was estimated to be 93.
Statistical analyses were conducted using SPSS software (version 23.0; SPSS Inc., Chicago, IL, USA), R software 4.0.1 (The R Foundation, https://www.r-project.org/), and GraphPad Prism 9 (GraphPad Software, Inc., San Diego, CA, USA). Continuous variables are summarized using medians and ranges, and categorical variables are described using frequencies and percentages. The patients who provided informed consent were included in the intention-to-treat (ITT) population. The per-protocol (PP) population included patients who met the eligibility criteria, completed at least two chemotherapy cycles, and underwent one measurement according to the study protocol. Kaplan–Meier curves were used to calculate the PFS and OS. Continuous variables were transformed into categorical variables using optimal cutoff values. Pearson’s chi-square test was used to compare the ORR and DCR between subgroups. Univariate and multivariate analyses were performed using Cox regression models. Variables with a p value <0.1 in the univariate analysis were considered for multivariable analysis. Statistical significance was set at p < 0.05.
Results
Patient characteristics
From 26 May 2015 to 8 December 2020, 98 eligible patients were enrolled. All the patients were pathologically diagnosed with gastric adenocarcinoma. All patients had previously received two-line chemotherapy without irinotecan and had no severe contraindications to chemotherapy. According to the study design, patients who completed at least two chemotherapy cycles and one measurement out of the 98 enrolled patients were 83 (84.7%). In all, 15 patients received the regimen but did not complete at least two chemotherapy cycles or undergo one measurement due to AEs (2 patients), disease progression after the first cycle (1 patient), or unwillingness (12 patients).
Patient characteristics are summarized in Table 1. Half the patients underwent resection of the primary tumor lesion. Approximately one-third of the patients had mucinous adenocarcinoma or adenocarcinoma containing mucinous adenocarcinoma. Most patients have multiple metastases, with the liver being the most common site. For previous chemotherapy, 90.8% and 90.4% of patients received oxaliplatin/cisplatin combined with 5-fluorouracil (5-FU) as first-line treatment in the ITT and PP populations, respectively; the rest of them received docetaxel/paclitaxel-based regimen or cisplatin + 5-FU + anti-human epidermal growth factor receptor 2 (HER-2) regimen. For second-line treatment, 65.3% and 61.4% of patients received docetaxel/paclitaxel-based regimens in the ITT and PP populations, respectively; the rest of them received paclitaxel + 5-FU + anti-vascular endothelial growth factor (VEGF) regimen, paclitaxel + anti-VEGF regimen, oxaliplatin/cisplatin-based chemotherapy, or paclitaxel + poly ADP-ribose polymerase inhibitor (PARPi) regimen.
Patient characteristics.
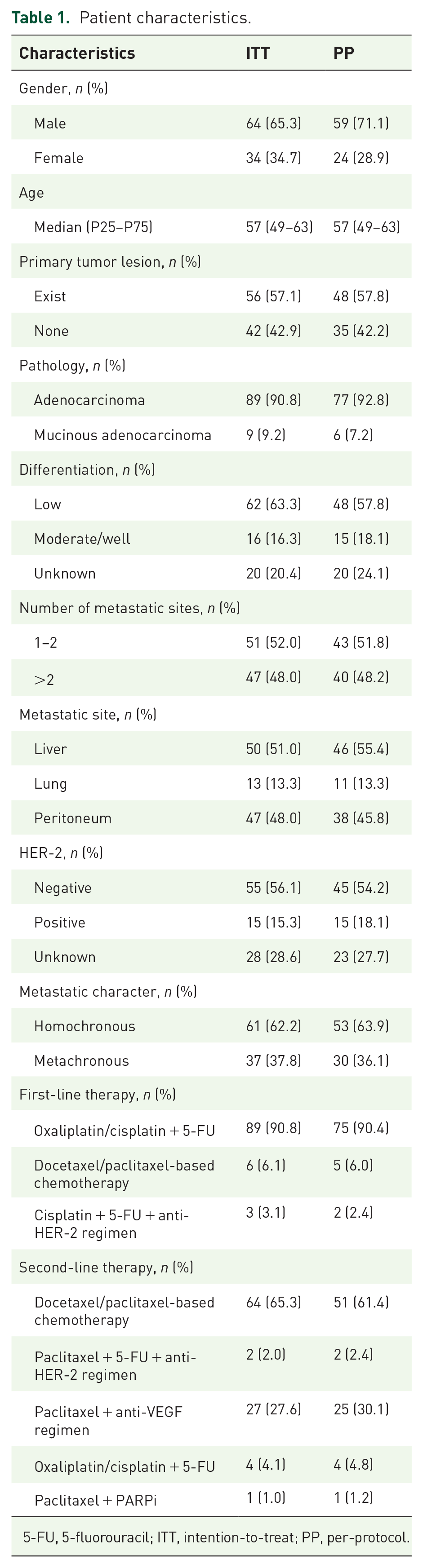
5-FU, 5-fluorouracil; ITT, intention-to-treat; PP, per-protocol.
Effects
In the ITT population (98 patients), 4 and 43 patients (4.08% and 43.88%, respectively) achieved PR and SD, the ORR was 4.08%, and the DCR was 47.96%. Median PFS and OS were 3.47 [95% confidence interval (CI): 2.80–4.14] and 7.17 (95% CI: 5.89–8.45) months, respectively (Figure 1). After disease progression or the end of the study treatment, 33 (33.67%) patients underwent further systemic anticancer treatment.

PFS and OS Kaplan–Meier survival curves in the ITT population. (a) PFS Kaplan–Meier survival curves in the ITT population (N = 98). (b) OS Kaplan–Meier survival curves in the ITT population (N = 98).
In the PP population (83 patients), 4 and 42 patients (4.82% and 50.60%, respectively) achieved PR and SD, respectively; the ORR was 4.82%, and the DCR was 55.42%. Median PFS and OS were 3.47 (95% CI: 2.81–4.13) and 7.77 (95% CI: 6.77–8.77) months, respectively (Figure 2). After disease progression or at the end of the study, 30 patients (36.14%) underwent further systemic anticancer treatment.

PFS and OS Kaplan–Meier survival curves in the PP population. (a) PFS Kaplan–Meier survival curves in the PP population (N = 83). (b) OS Kaplan–Meier survival curves in the PP population (N = 83).
Safety
Treatment-related AEs are summarized in Table 2. The treatment-emergent AEs included neutropenia, anemia, thrombocytopenia, abnormal hepatic function, elevated bilirubin levels, hypoalbuminemia, elevated creatinine levels, and diarrhea. The most common treatment-emergent AEs of all grades were anemia (57.1%, 56/98), neutropenia (38.8%, 38/98), and diarrhea (12.2%, 12/98). Most of these toxicities were grade 1–2. The incidence of grade 3 or 4 hematological and non-hematological toxicities was 19.4% (19/98). The most common grade 3 or 4 treatment-emergent AEs were neutropenia (16.3%, 16/98) and diarrhea (2.0%, 2/98). Dose reductions due to AEs occurred in 12 of the 98 patients. None of the patients died of AEs.
Treatment-emergent AEs.

AE, adverse event.
Multivariate analysis
In univariate analysis, the presence of neutropenia during irinotecan treatment, peritoneal metastasis, the presence of a primary tumor lesion, the baseline albumin level, baseline alkaline phosphatase level, baseline neutrophil level, and baseline thrombocyte level were significantly associated with OS (Table 3). Then, characteristics with a p value <0.1 are included in a multivariable model. The presence of neutropenia (no versus yes, hazard ratio (HR): 2.32; 95% CI: 1.35–3.97), peritoneum metastasis (no versus yes, HR: 0.57; 95% CI: 0.35–0.92), and the baseline platelet (PLT) level (⩽288 * 109/L versus >288 * 109/L, HR: 0.42; 95% CI: 0.23–0.76) remained independent prognostic factors after multivariable adjustment (Figure 3).
Univariate and multivariable analyses for OS.

ALB, albumin; AKP, alkaline phosphatase; CI, confidence interval; HGB, hemoglobin; LDH, lactic dehydrogenase; LYM, lymphocyte; NE, neutrophil; WBC, white blood cell.

Subgroup OS Kaplan–Meier survival curves in the ITT population. (a) OS Kaplan–Meier survival curves of the population without neutropenia (N = 60) and with neutropenia (N = 38). (b) OS Kaplan–Meier survival curves of the population without peritoneum metastasis (N = 51) and with peritoneum metastasis (N = 47). (c) OS Kaplan–Meier survival curves of population with low baseline PLT level (N = 79) and with high baseline PLT level (N = 18).
A subgroup analysis of DCR in the ITT population is shown in Table 4. Patients who experienced neutropenia during treatment had a better DCR.
Subgroup analysis of DCR.

As shown in Table 5, the presence of neutropenia during irinotecan treatment, pathology type, peritoneal metastasis, baseline alkaline phosphatase levels, and baseline thrombocyte levels were significantly associated with PFS in univariate analysis. Then, characteristics with a p value <0.1 are included in a multivariable model. The presence of neutropenia during the irinotecan treatment (no versus yes, HR: 1.86, 95% CI: 1.02–3.40), the pathology type (adenocarcinoma versus mucinous adenocarcinoma, HR: 0.17; 95% CI: 0.05–0.59), and the baseline PLT level (⩽288 * 109/L versus >288 * 109/L, HR: 0.37; 95% CI: 0.18–0.75) remained the independent factors of PFS after multivariable adjustment (Figure 4).
Univariate and multivariable analyses for PFS.

CI, confidence interval.

Subgroup PFS Kaplan–Meier survival curves in the ITT population. (a) PFS Kaplan–Meier survival curves of the population without neutropenia (N = 60) and with neutropenia (N = 38). (b) PFS Kaplan–Meier survival curves of the population with adenocarcinoma (N = 89) and with mucinous adenocarcinoma (N = 9). (c) PFS Kaplan–Meier survival curves of the population with low baseline PLT level (N = 79) and with high baseline PLT level (N = 18).
Discussion
For patients with metastatic gastric cancer, systemic therapy could improve both survival and quality of life compared to the best-supporting care alone.9,16–18 For the initial treatment of patients with metastatic gastric cancer, two-drug cytotoxic regimens composed of fluorouracil and oxaliplatin/cisplatin are preferred over the triplet regimen because it has been demonstrated that the doublet regimen is as effective as the triplet regimen and has a better toxicity profile.3,12,19 Second-line or subsequent therapy should depend on prior therapy and performance status. Standard second-line therapies include irinotecan-based and taxane-based (docetaxel or paclitaxel) chemotherapies. 7 A randomized phase III trial demonstrated no statistically significant difference between paclitaxel and irinotecan for OS. 20 Researchers have also explored the potential of substituting platinum and taxane in the first-/second-line treatment. 21
Based on the results of prospective clinical trials, anti-angiogenesis therapeutic agents and immune checkpoint inhibitors have been approved globally for the third-line treatment of gastric cancer. A phase III clinical trial showed that apatinib mesylate treatment could prolong median PFS compared with placebo (2.6 versus 1.8 months, p = 0.016) and improve the disease control rate (42.05% versus 8.79%, p < 0.001). 22 In a phase II trial, regorafenib effectively prolonged PFS (regorafenib for 2.6 months and placebo for 0.9 months) in the second-line or third-line refractory advanced gastric adenocarcinoma. 23 As anti-angiogenesis-targeted drugs, apatinib and regorafenib have less toxicity than traditional chemotherapy drugs and less anticancer activity. Therefore, they are suitable for patients with poor physical fitness, poor bone marrow function after chemotherapy, and low tumor burden. In addition, it is unsuitable because of its unique mechanism for patients with uncontrolled hypertension, thrombosis, or a high risk of bleeding or perforation. The ATTRACTION-02 study in an Asian population showed that nivolumab in the third-line treatment of recurrent or metastatic gastric or esophagogastric junction adenocarcinoma significantly reduced the risk of death compared with placebo and increased the 1-year overall survival rate. 24 The KEYNOTE-059 showed that the OS of third-line pembrolizumab treatment for recurrent/metastatic gastric or esophago-gastric junction (EGJ) adenocarcinoma with programmed cell death 1 ligand 1 (PD-L1) combined positive score (CPS) ⩾1 was 6 months, and the ORR was 12%. 25 Treatment with anti-PD-1 antibodies such as pembrolizumab/nivolumab is based on testing for microsatellite instability (MSI)/mismatch repair (MMR), PD-L1 expression, or high tumor mutation burden (TMB) because they are effective in only a small number of people identified by molecular markers (dMMR/MSI-H, TMB-H, high CPS, etc.). Moreover, TAS102 is an oral chemotherapeutic drug approved for previously treated recurrent or metastatic gastric and EGJ adenocarcinomas based on the TAGS trial. 26 It is convenient but produces a few partial or CRs and substantial toxicities. Therefore, TAS102 should be considered for patients with normal bone marrow function, low tumor burden, or contraindications for targeted therapies.
However, as a traditional chemotherapy drug in the second-line treatment, whether irinotecan could be used as a salvage treatment after the failure of second-line chemotherapy has not provided powerful evidence. Several retrospective studies showed that the overall response rate of irinotecan monotherapy as third-/fourth-line therapy was 3–18.4%, the disease control rate was 22–55.2%, the median PFS was 1.9–3.5 months, and the median OS was 4.0–11.3 months.27–32
This study is the first to propose a prospective third-line chemotherapy regimen using irinotecan in patients with metastatic gastric cancer. In 98 patients following this regimen, the ORR and DCR were 4.08% and 47.96%; the mPFS and mOS were 3.47 and 7.17 months, which in numbers were better than the effects of apatinib with mPFS of 2.6 months and mOS of 6.5 months, 22 and nivolumab with mOS of 5.26 months. 24 Therefore, irinotecan monotherapy may be an effective and economically viable treatment option.
Compared with other third-line treatments, such as TAS102, apatinib, regorafenib, and nivolumab, irinotecan, as a type of chemotherapy drug, has relatively more toxicity, especially hematological toxicity. Thus, it is suitable for patients with good physical fitness, good bone marrow function, and no advantage in using anti-PD-1 antibodies, or contraindications for targeted therapies.
We found that patients who experienced neutropenia during treatment had better DCR according to subgroup analysis and benefited more from PFS and OS according to multivariable analysis. Several studies have confirmed the correlation between neutropenia and regimen efficacy. We assumed that neutropenia might be a surrogate marker, indicating that the dose intensity was adequate to provide an antitumor effect.33–35 Intolerable toxicities such as grade 4 neutropenia often lead to dosage reductions. The toxicities of irinotecan, especially severe ones, are often related to UGT1A1 polymorphism. Heterozygous or homozygous UGT1A1 mutations are associated with more severe AEs caused by irinotecan, including diarrhea and neutropenia, owing to the accumulation of SN38. 36 According to the univariate Cox analysis, dosage reduction did not affect efficacy and prognosis, indicating that patients who experienced dosage reduction would not benefit less from irinotecan monotherapy. For patients who experience severe AEs, a reduced dosage may be appropriate. The appearance of neutropenia might indicate that SN38 had reached the effective dosage in these patients. By contrast, some of those without neutropenia may have received an insufficient dosage and need an increase. These findings show that personalized dosage adjustment might be a useful strategy for treatment and should be further investigated.
In the multivariable model, we also found that baseline platelet level was an independent factor for both OS and PFS. Correlations between increased platelet levels and shorter survival times have been described for most common solid tumors, including gastric cancer, verified in our study. 37 We speculated that the tumor affected the baseline PLT level through cytokines. Thus, patients with higher baseline PLT levels, equivalent to a larger tumor burden, had poorer prognoses.
In conclusion, our study proved that irinotecan monotherapy could be an attractive regimen for patients with metastatic gastric cancer after the failure of second-line therapy because of its efficacy, safety, and low cost.
