Abstract
Background:
Due to the complex histological type and anatomical structures, there has been considerable debate on the classification of adenocarcinoma of the esophagogastric junction (AEG), especially Siewert II AEG. Furthermore, neither the American Joint Committee on Cancer (AJCC) 7th tumor–node–metastasis (TNM) [esophageal adenocarcinoma (E) or gastric cancer (G)] nor the AJCC 8th TNM (E or G) accurately predicted the prognosis of patients with Siewert II AEG.
Objective:
This study aimed to investigate the factors influencing the survival and prognosis of patients with Siewert II AEG and establish a new and better prognostic predictive model.
Design:
A retrospective study.
Methods:
Patients with Siewert II AEG, retrieved from the Surveillance, Epidemiology, and End Results (SEER) databases, were assigned to the training set. Patients retrieved from a single tertiary medical center were assigned to the external validation set. Significant variables were selected using univariate and multivariate Cox regression analyses to construct the nomogram. Nomogram models were assessed using the concordance index (C-index), a calibration plot, decision curve analysis (DCA), and external validation.
Results:
Age, tumor grade, and size, as well as the T, N, and M stages, were included in the nomograms. For the SEER training set, the C-index of the nomogram was 0.683 (0.665–0.701). The C-index of the nomogram for the external validation set was 0.690 (0.653–0.727). The calibration curve showed good agreement between the nomogram estimations and actual observations in both the training and external validation sets. The DCA showed that the nomogram was clinically useful.
Conclusion:
The new predictive model showed significant accuracy in predicting the prognosis of Siewert II AEG.
Introduction
The incidence of adenocarcinoma of the esophagogastric junction (AEG) has increased rapidly in the past few decades, especially in the United States and the United Kingdom, due to smoking, obesity, and gastroesophageal reflux disease.1–3 According to a retrospective study conducted in China, there has been a notable increase in the prevalence of AEG among surgical patients, from 22.3% to 35.7% over the past 25 years. 4
However, the absence of a cohesive standard classification system for AEG is evident. Siewert et al. proposed a classification scheme that categorizes AEG into three distinct types. Type I lesions are located between 1 and 5 cm above the anatomical esophagogastric junction (EGJ). Type II lesions straddle the EGJ with their epicenter from 1 cm above to 2 cm below the EGJ. Type III cardia tumors extend up to the stomach from 2 to 5 cm below the EGJ, with infiltration proximal to the EGJ. 5 Currently, the American Joint Committee on Cancer (AJCC) tumor–node–metastasis (TNM) staging system is the most commonly used system for assessing solid tumors, including gastric and esophageal cancers. 6 Furthermore, in October 2016, the 8th edition of the staging guidelines was modified to state that for AEG, if the tumor invades the EGJ and the tumor center is located within 2 cm below the EGJ, the staging should follow the criteria for esophageal adenocarcinoma. If the tumor center is located beyond 2 cm below the EGJ, the staging should follow the criteria for gastric cancer. 5 Nevertheless, controversy persists regarding the staging of AEG according to esophageal or stomach adenocarcinoma TNM staging, especially for Siewert II AEG. A retrospective study with a large sample size showed that AEG TNM staging based on the 8th gastric tumor staging scheme was superior to that based on the 8th esophageal adenocarcinoma staging system in predicting the prognosis of AEG, especially in patients with Siewert II AEG who underwent trans-hiatal approaches. 7 However, several studies have shown that neither staging approach is perfect or precise in predicting the prognosis of patients with Siewert II AEG.8,9 Therefore, it is necessary to identify a better prognostic predictive model for this specific subset of patients based on a large population cohort. Nomograms are widely used to predict cancer prognosis because they can simplify statistical prediction models into a single numerical estimate of the probability of an event, such as death or recurrence. 10 Furthermore, nomograms have been applied to various cancer types,11–13 including gastric, colon, and rectal cancers.14–16 Several studies have developed nomograms to predict the prognosis for patients with Siewert II AEG17,18 but they lack effective external validation and have a small sample size. Hence, we conducted this study to investigate the factors influencing the survival and prognosis of patients with Siewert II AEG and to establish a new and more precise prognostic predictive model.
Methods
Study design and data selection
We conducted a retrospective analysis of the Surveillance, Epidemiology, and End Results (SEER) database and prospectively collected Siewert II AEG data from our center. Clinicopathological data and prognostic outcomes for patients with Siewert II AEG were screened from the SEER database [The National Cancer Institute’s SEER*Stat software version 8.4.0.1 (https://seer.cancer.gov/seerstat/)] with the search term ‘EsophgusGEJunction and adenocarcinoma NOS’ (n = 22,418). The study was reported in line with the STROCSS (Strengthening the Reporting of Cohort, Cross-sectional, and Case–Control Studies in Surgery) criteria 19 (Supplemental Material 1).
The inclusion criteria were defined as follows: (1) patients with Siewert II AEG confirmed by histology; (2) patients undergoing surgical treatment irrespective of the degree of lymph node dissection, curative degree, surgical method, reconstruction method, or adjuvant therapy; and (3) patients without other malignant tumors. The exclusion criteria comprised (1) the presence of more than one primary tumor and (2) cases with unknown grade, T stage, N stage, M stage, age, or tumor size. All patients with AEG (n = 1523) accrued from SEER were assigned to the training set. Furthermore, 399 patients with Siewert II AEG from a single tertiary medical center (West China Hospital) were assigned to the external validation set. The factors considered included sex, age, tumor grade, tumor size, AJCC 7th TNM stage based on esophageal adenocarcinoma and gastric cancer, AJCC 8th TNM stage based on esophageal adenocarcinoma and gastric cancer, vital status (dead or alive), and survival time. The flowchart of the selection process for the Siewert II AEG is presented in Figure 1.
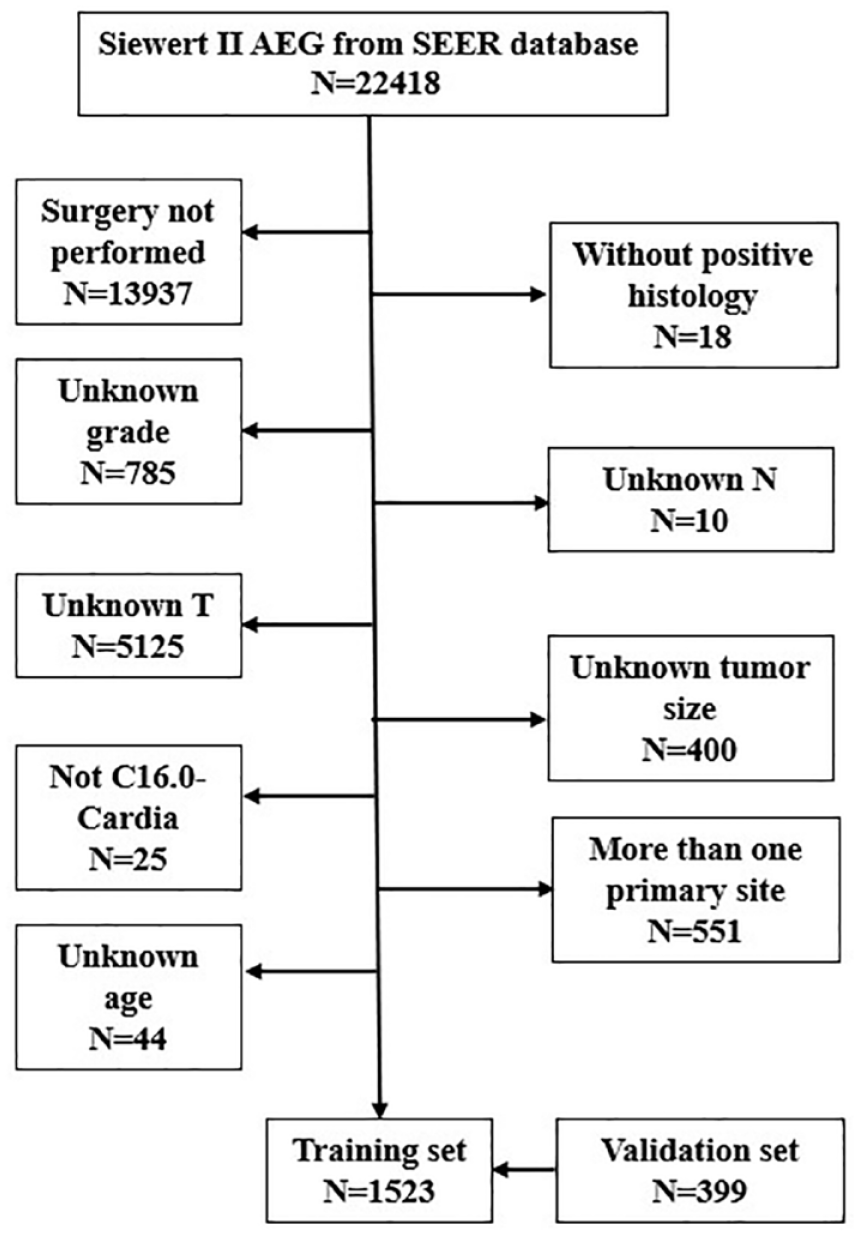
Flow chart diagram of the AEG patients in the training and validation sets.
Construction and validation of the nomogram model
The primary endpoint was overall survival (OS) in patients with Siewert II AEG. Tumor size was categorically divided based on the optimal cutoff value generated by X-tile software version 3.6.1 (Yale University School of Medicine, New Haven, CT, USA) to distinguish the population and assess statistical significance. 20 Differences in patient characteristics were assessed using the Mann–Whitney U test for continuous variables and Fisher’s exact test or χ 2 test for categorical variables. Sex, age, grade, tumor size, AJCC 7th TNM stage based on esophageal adenocarcinoma and gastric cancer, and AJCC 8th TNM stage based on esophageal adenocarcinoma and gastric cancer were selected for nomogram modeling. Univariate Cox analysis was used to select distinct variables (p < 0.05) for further multivariate analysis and nomogram construction. The concordance index (C-index) was used to assess the performance of the nomograms by comparing the measurement between the predicted and actual results. The theoretical value of the C-index is between 0 and 1, with a value larger than 0.5 indicating superior prediction performance than random guessing. 13 When evaluating the performance of different models, the C-indices from different models were compared using analysis of variance. Calibration plots were used to compare the nomogram-predicted and actual outcomes using a 45-degree line as the reference for an optimal model. 10 Considering the group imbalance in the validation cohorts, we carried out 1000 bootstrapping resamples in each group for internal and external validations. Decision curve analysis (DCA) was conducted to evaluate the clinical usefulness of the nomogram by quantifying the net benefit at different threshold probabilities. 21 The R software version 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria) was used for all analyses. A statistically significant cutoff value was set at p < 0.05.
Results
Clinicopathologic characteristics
The baseline clinicopathological characteristics of the SEER training cohort (n = 1523) and the external validation cohort (n = 399) are shown in Table 1. Most patients in our center were diagnosed at an advanced stage, with a larger tumor size (p < 0.001), higher grade (p < 0.001), more lymph node dissections (p < 0.001), and a younger age (p = 0.011) than that of patients from the SEER.
The baseline clinicopathologic characteristics of the SEER training set and external validation set.

E, esophageal adenocarcinoma; G, gastric cancer; IQR, interquartile range; NA, not available; RNE, regional lymph node examined; RNP, regional lymph node positive; SEER, Surveillance, Epidemiology, and End Results; TNM, tumor–node–metastasis; WCH, West China Hospital.
According to univariate Cox analysis, age (p < 0.05), tumor size (p < 0.001), grade (p < 0.001), T stage (p < 0.001), N stage (p < 0.001), and M stage (p < 0.001) were significantly associated with OS. These significant variables were further entered into the multivariate Cox analysis. Multivariate Cox analysis showed that age (p < 0.05), tumor grade (p < 0.001), tumor size (p < 0.05), T stage (p < 0.001), N stage (p < 0.05), and M stage (p < 0.001) were independent prognostic factors for OS (Table 2).
Univariate and multivariate analyses of OS in the training set.

CI, confidence interval; E, esophageal adenocarcinoma; G, gastric cancer; HR, hazard ratio; NA, not available; TNM, tumor–node–metastasis.
Nomogram construction and performance assessment
We developed a nomogram including age, grade, tumor size, T stage, N stage, and M stage for OS based on the results of multivariate Cox analysis (Figure 2), where each variable was assigned a point according to the hazard ratio. Then, by adding the total score of each variable and locating the score on the total point scale, the probabilities of 3- and 5-year OS were obtained. For the SEER training set, the C-index of the nomogram was 0.683 (0.665–0.701). The C-index of the nomogram for the external validation set was 0.690 (0.653–0.727).

OS nomograms for AEG at 3 and 5 years in the training set.
The calibration curve showed good agreement between the estimations with the nomogram and actual observations in the training (Figures 3 and 4) and external validation sets (Supplemental Figures 1 and 2). In addition, DCA revealed that using the nomogram to predict the 3-year and 5-year survival of the patients in the training set would add more net benefit, indicating the clinical usefulness of the nomogram (Figures 5 and 6). Similar results were demonstrated in the external validation set (Supplemental Figures 3 and 4).

Calibration plots of 3-year OS-associated nomograms in the training set.

Calibration plots of 5-year OS-associated nomograms in the training set.

Decision curve analysis plots of 3-year OS in the training set.

Decision curve analysis plots of 5-year OS in the training set.
Discussion
The incidence of AEG has significantly increased in Western and Asian countries, and patients with AEG have experienced poor prognosis over the past decades.4,22–24 The incidence of AEG in the United States has increased nearly 2.5 times since the 1970s, 2 imposing a severe financial burden on individuals and countries. Due to the complicated histological type and anatomical structures, there has been a great debate on the classification of AEG, especially for Siewert II AEG. Furthermore, there is a lack of precise prognostic models with a large sample size and external validation to predict OS for AEG.25,26 Consequently, this study proposed to investigate the factors influencing the survival and prognosis of AEG and to establish a new and more precise model, with a larger sample size and external validation to predict its prognosis.
The external validation set was obtained from West China Hospital, which has the largest specialized disease database for patients with gastric cancer and AEG in China. Based on eligibility criteria, 399 patients with Siewert II AEG were included in the external validation set. Most patients from West China Hospital were diagnosed at a younger age (p = 0.011) and an advanced stage and presented with a larger tumor size (p < 0.001), higher grade (p < 0.001), and had more lymph nodes dissected (p < 0.001) compared with those of patients from the training set (SEER). Six significant prognostic indicators were selected from the variables included in the training set through univariate analysis. Then, using multivariate analysis, we identified six independent prognostic factors in the training set. A study involving 396 patients also indicated that age, grade, and TNM stage were risk factors for patients with Siewert type II/III AEG, 18 which is consistent with our study. In another study, tumor size independently worsened survival and was deemed more important than the T status for esophageal carcinoma.27,28 Furthermore, the inclusion of patients with distant metastases highlighted the poor prognosis. The M stage is another important factor that affects the prognosis of patients with cancer. While surgical treatment is not the standard approach for patients with distant metastases, some individuals with metastatic disease may require emergency surgery to address acute situations including gastrointestinal obstruction and perforation caused by tumors. This intervention aims to circumvent urgent issues in the emergency department potentially reducing in-hospital mortality. Moreover, some patients may not exhibit distant metastasis before surgery but could develop it due to tumor recurrence or progression after the surgical procedure. These patients still met the inclusion criteria. Therefore, after comprehensive consideration, this study included patients with M stage. Eventually, a nomogram was established based on six variables: age, grade, tumor size, T stage, N stage, and M stage. The nomogram achieved C-indices of 0.683 (0.665–0.701) in the training set and 0.690 (0.653–0.727) in the external validation set. Moreover, we calibrated the 3-year and 5-year OS nomograms of the training and validation cohorts, which closely approximated the ideal curve. This demonstrated good consistency between the nomogram predictions and actual observed outcomes in the training and validation cohorts. Furthermore, DCA revealed that using a nomogram to predict individual 3-year and 5-year survival rates would add more net benefits. Overall, the nomogram exhibited clinical usefulness and had a discriminative ability to identify patients.
Furthermore, we performed an external validation of the developed nomogram for predicting OS in patients with Siewert II AEG. The model demonstrated consistent performance when applied to patients treated in a high-volume hospital. Subsequently, a new nomogram was formulated based on available data from both databases. Patients diagnosed at an advanced stage, lower grade, or at an older age showed a poor prognosis.
When applying nomograms in clinical practice, several factors must be considered. First, due to incomplete knowledge of prognostication, survival predictions are not perfect. Using predictive models for individual patients allows a rough estimation of the future outcome events they may experience; however, these events may not necessarily occur. 29 Furthermore, integrating gene and protein expression allows the incorporation of molecular prognosticators to further improve their performance. 30 In addition, deep learning has been widely applied to predict prognosis and lymph node metastasis. Combining clinicopathological factors with radiomics using deep learning could be utilized to predict the prognosis of Siewert II AEG in the future.
Our study has several limitations. First, this was a retrospective study with selection and information biases that may have affected the results. Therefore, prospective studies are warranted to further validate this predictive model. Second, the adequacy of surgery and the use of preoperative chemo-radiotherapy (CRT), perioperative chemotherapy (CT), and postoperative CT have all been shown to influence survival outcomes in curatively treated patients in randomized trials. However, the SEER database has limited data on the adequacy of surgery and the use of preoperative CRT, perioperative CT, and postoperative CT. The SEER database only reports patients who underwent surgery but without the extent of lymph node dissection, surgical method, and type of anastomosis. Third, this study only included patients who underwent surgery. However, this may represent selection bias. Furthermore, other routine clinical data such as routine blood test indices, which could have enhanced the accuracy of the predictive model, were lacking.
Conclusion
We constructed and validated a new model to accurately predict individual survival in patients with Siewert II AEG. The nomogram can effectively predict the OS of patients.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359241229425 – Supplemental material for Development and validation of a nomogram for Siewert II esophagogastric junction adenocarcinoma: a retrospective analysis
Supplemental material, sj-pdf-1-tam-10.1177_17588359241229425 for Development and validation of a nomogram for Siewert II esophagogastric junction adenocarcinoma: a retrospective analysis by Tao Jin, Ze-Dong Li, Ze-Hua Chen, Feng-Jun He, Zheng-Wen Chen, Pan-Ping Liang, Jian-Kun Hu and Kun Yang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
