Abstract
Objectives
Malignant pleural mesothelioma is a rare and aggressive thoracic tumor with a poor prognosis, wherein distant metastasis is associated with the lowest survival rates. It is imperative and emergent to construct nomograms based on risk factors and prognostic factors for distant metastasis in patients with malignant pleural mesothelioma.
Methods
We extracted data for the duration between 2010 and 2015 from the Surveillance, Epidemiology, and End Results (SEER) database and randomly categorized the patients into the training (70%) and validation (30%) cohorts. Risk factors for distant metastasis in patients with malignant pleural mesothelioma were identified using univariate and multivariate logistic regression analyses, and prognostic factors for patients with distant metastasis were determined using univariate and multivariate Cox regression analyses. Two nomograms were established based on the training cohort and evaluated using the validation cohort. The C-index, receiver operating characteristic curve, calibration curve, and decision curve analysis were used to assess the performance of the two nomograms.
Results
In total, 2056 primary malignant pleural mesothelioma patients were included, and 341 patients were initially diagnosed with metastatic malignant pleural mesothelioma. Histology, laterality, grade, tumor stage, and node stage were independent risk factors for distant metastasis in patients with malignant pleural mesothelioma. Chemotherapy and metastasis to the lung, bone, and brain were independent prognostic factors for patients with malignant pleural mesothelioma and distant metastasis. The C-index values of the risk nomogram in the training and validation cohorts were 0.723 and 0.782, respectively. The C-index values of the prognostic nomogram in the training and validation cohorts were 0.678 and 0.712, respectively. The receiver operating characteristic curves, calibration curves, and decision curve analysis also demonstrated good predictive performance for the two nomograms in the training and validation cohorts.
Conclusions
Nomograms are useful and reliable tools for predicting distant metastatic risk in patients with malignant pleural mesothelioma and overall survival in patients with malignant pleural mesothelioma who had distant metastasis. These nomograms can provide strong references to clinicians to facilitate clinical decisions.
Introduction
Malignant pleural mesothelioma (MPM), a rare and aggressive tumor, arises from the pleura and is typically associated with high mortality and poor overall survival (OS). Annually, ≥2000 and ≥5000 MPM cases are diagnosed in the US and Europe, respectively.1,2 The recognized predominant risk factor is asbestos exposure, which accounts for more than 90% of MPM cases.3,4 The incidence of MPM is expected to increase continuously because of the long latency period between asbestos exposure and the development of MPM. 5 The median survival is <10 months, and the 5-year survival rate remains at 5%–10%.6,7 MPM exhibits histological heterogeneity; 50%–60% of the cases are of the epithelial subtype, approximately 10% are sarcomatoid, and the remainder are biphasic, with the sarcomatoid histology being associated with the lowest survival rates. 8 Currently, a multimodal treatment regimen including chemotherapy, surgery, and radiotherapy provides the best long-term outcomes in patients with early-stage MPM; however, patients with MPM who have distant metastasis (DM) have poorer median survival and OS compared with those without DM.9,10
Previously, MPM was described as a local, invasive disease most commonly affecting the lungs, pericardium, chest wall, and vertebrae. 11 Although DM in MPM is uncommon, evidence of metastatic spread is gradually increasing. 12 In a postmortem study of 318 patients with MPM, extrapleural dissemination was observed in 87.7% of the patients, whereas extrathoracic metastasis occurred in 55.4%, affecting nearly all organs, including the liver (31.9%), spleen (10.8%), thyroid (6.9%), and brain (3.0%). 13 Another retrospective observational study of patients with MPM reported that 69 patients (18%) developed DM, including 14 who had DM at diagnosis and 55 who were diagnosed during follow-up. 9 Metastasis to other organs, such as the tongue, salivary gland, and breast, have been documented in patients with MPM.11,14,15 Thus, extrathoracic dissemination of MPM may be much more widespread than currently reported, and its impact on survival could be underestimated. Previous studies suggest that age, sex, tumor differentiation, histology type, tumor (T) stage, metastasis (M) stage, surgery, and chemotherapy are independent prognostic factors in patients with MPM.16–18 However, studies investigating the risk factors and prognostic factors in patients with MPM and DM remain limited.
As a widely used novel method, nomograms can effectively predict lymph node metastasis, DM, and OS in patients with cancer. Most existing nomograms are derived from patients with MPM without DM; however, a nomogram for patients with MPM and DM has not yet been published.
Therefore, we aimed to construct a novel nomogram to predict DM in patients with MPM and a novel nomogram to predict OS in patients with MPM and DM based on the demographic and clinicopathologic variables from the Surveillance, Epidemiology, and End Results (SEER) database. Consequently, this approach is expected to facilitate individualized patient care as well as medical therapy.
Materials and methods
Patients and data collection
The SEER database was searched from 2010 to 2015, which comprised approximately 28% of the US population and a wide range of ethnic groups. The inclusion criteria were as follows: (a) diagnosis of primary MPM (Histology code: 9050–9055); (b) diagnostic confirmation based on positive histology, positive laboratory test/marker study, or positive microscopic confirmation; (c) OS of at least 1 month after diagnosis; and (d) complete T, node (N), and M stages according to the 7th edition American Joint Committee on Cancer (AJCC) staging system. In addition, we extracted data on demographic and clinocopathologic variables, including age, sex, race, grade, laterality, histology, tumor size, surgery, radiotherapy, chemotherapy, and metastasis to the lung, brain, bone, and liver. After inclusion, we excluded patients with unknown or incomplete data on age, sex, race, and laterality. Finally, 2056 patients met the criteria for the primary cohort, of whom 341 were diagnosed with DM. To further evaluate prognostic factor in patients with DM, we excluded patients with unavailable data on surgery, radiotherapy, chemotherapy, and metastasis to the lung, bone, liver, and brain. Therefore, only 327 patients with DM were selected to establish a prognostic nomogram. We have de-identified all patient details. This study was retrospective in nature and conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. All the data extraction processes for the primary and prognostic cohorts are shown in Figure 1.

The selection process of the present study. MPM: malignant pleural mesothelioma; DM: distant metastasis.
Age at diagnosis was categorized as <60, 60–69, 70–80, and >80 years. Tumor grade was grouped into grades I–II and grades III–IV. Tumor size was classified into the following four groups: <5 cm, 5–10 cm, ≥10 cm, and unknown. Patients were randomized into the training and validation cohorts in a ratio of 7:3 for the diagnostic and prognostic cohorts. The nomogram was constructed based on the training cohort and was validated using the validation cohort. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 19
Statistical analyses
In the present study, R software (version 4.0.3) was used to analyze all data, and p-values of <0.05 (two-sided) were considered statistically significant. The training and validation cohorts were extracted using R software; meanwhile, the distribution and differences between the two cohorts were examined using the chi-square or Fisher’s exact test.
In the diagnostic study, we first identified risk factors for DM using univariate logistic regression analysis in the training cohort. Next, DM-related factors with a p-value of <0.05 were further analyzed using multivariate logistic regression analysis. Finally, independent DM-related factors were determined, and a diagnostic nomogram was created using the ‘rms’ package. The receiver operating characteristic (ROC) curves of the nomogram were generated, and the corresponding area under the curve (AUC) and C-index were measured to describe the discrimination ability of the nomogram in the training and validation cohorts. Calibration curves and decision curve analysis (DCA) were used to assess the performance of the diagnostic nomogram.
In the prognostic study, univariate Cox proportional hazards regression analysis was performed to determine OS-related factors in the training cohort. Variables with p-values of <0.05 were assessed using multivariate Cox regression analysis to screen for independent OS-related factors. A prognostic nomogram was generated based on the independent prognostic factors. Time-dependent ROC analysis was employed to predict the 6-, 12-, and 18-month OS rates, and the AUC was calculated to show the discrimination. The predictive accuracy, calibration, and clinical usefulness of the nomogram were reflected by the C-index, calibration curves, and DCA, respectively. To compare the OS status of patients with and without DM, Kaplan–Meier (K–M) survival curves were generated using the log-rank test.
Results
Patients’ baseline characteristics
Table 1 summarizes the demographic and clinicopathological characteristics of 2056 patients with MPM between 2010 and 2015, of which 341 patients (16.6%) were initially diagnosed with DM and 1715 patients (83.4%) were diagnosed without DM. The median OS for all patients was 10 (1–81) months. Patients without DM had a median survival of 11 months, whereas those with DM had a median survival of 6 months. K–M analysis showed that patients with DM had significantly lower OS than those without DM (Figure 2(a)). The majority of the population was White, whereas individuals of other ethnicities accounted for 8.9% of the study population. The median age of the patients was 72 (19–97) years. Most patients were male (77.9%). Excluding the unknown group, the most common tumor grades of differentiation and tumor size were III–IV and <50 mm, respectively. Epithelioid mesothelioma was the most common histology type, followed by mesothelioma (not otherwise specified, NOS), fibrous mesothelioma, and biphasic mesothelioma. The majority of primary MPM cases (59.5%) originated from the right pleura, whereas 39.5% originated from the left pleura and 1.5% from the bilateral pleura. The most common T and N stages were T1 and N0, respectively. No significant differences were found between the training and validation sets in the diagnostic cohort (Table 1).
Characteristics of primary MPM patients in the diagnostic cohort.

MPM: malignant pleural mesothelioma; NOS: not otherwise specified; T: tumor; N: node; M: metastasis.

(a) Kaplan–Meier survival analysis of patients with MPM with and without DM and (b) the diagnostic nomogram for predicting DM in patients with MPM. MPM: malignant pleural mesothelioma; DM: distant metastasis.
DM-related risk factors
As shown in Table 2, grade, laterality, histology, T stage, and N stage were identified as DM-related risk factors using univariate logistic regression analysis. There were no significant differences in the metastatic probability among the T1, T2, and T3 stages; therefore, we combined them into a single group for analysis. Subsequently, multivariate logistic regression analysis further confirmed that bilateral, higher grade, higher T stage, higher N stage, and non-biphasic histology are independent risk factors for predicting DM in patients with MPM.
Univariate and multivariate logistic regression analyses of risk factors associated with distant metastasis in patients with MPM.

MPM: malignant pleural mesothelioma; HR: hazard ratio; CI: confidence interval; NOS: not otherwise specified; T: tumor; N: node.
Diagnostic nomogram construction and validation
A diagnostic nomogram was constructed based on the results of multivariate logistic regression analysis (Figure 2(b)). The C-index values were 0.723 (0.688–0.759) and 0.782 (0.731–0.832) in the training and validation cohorts, respectively. ROC curves were plotted, and the AUC values were 0.723 (0.687–0.759) and 0.781(0.730–0.832) for the training and validation cohorts, respectively, which demonstrated good discrimination, similar to the C-index (Figure 3). The calibration curves showed good consistency between predicted and actual probability of DM (Figure 4(a) and (b)). Additionally, DCA demonstrated excellent net benefits of the nomogram among most threshold probabilities, indicating its potential as an effective and precise tool for DM prediction (Figure 4(c) and (d)).

The receiver operating characteristic curves of the diagnostic nomogram in the (a) training and (b) validation cohorts.
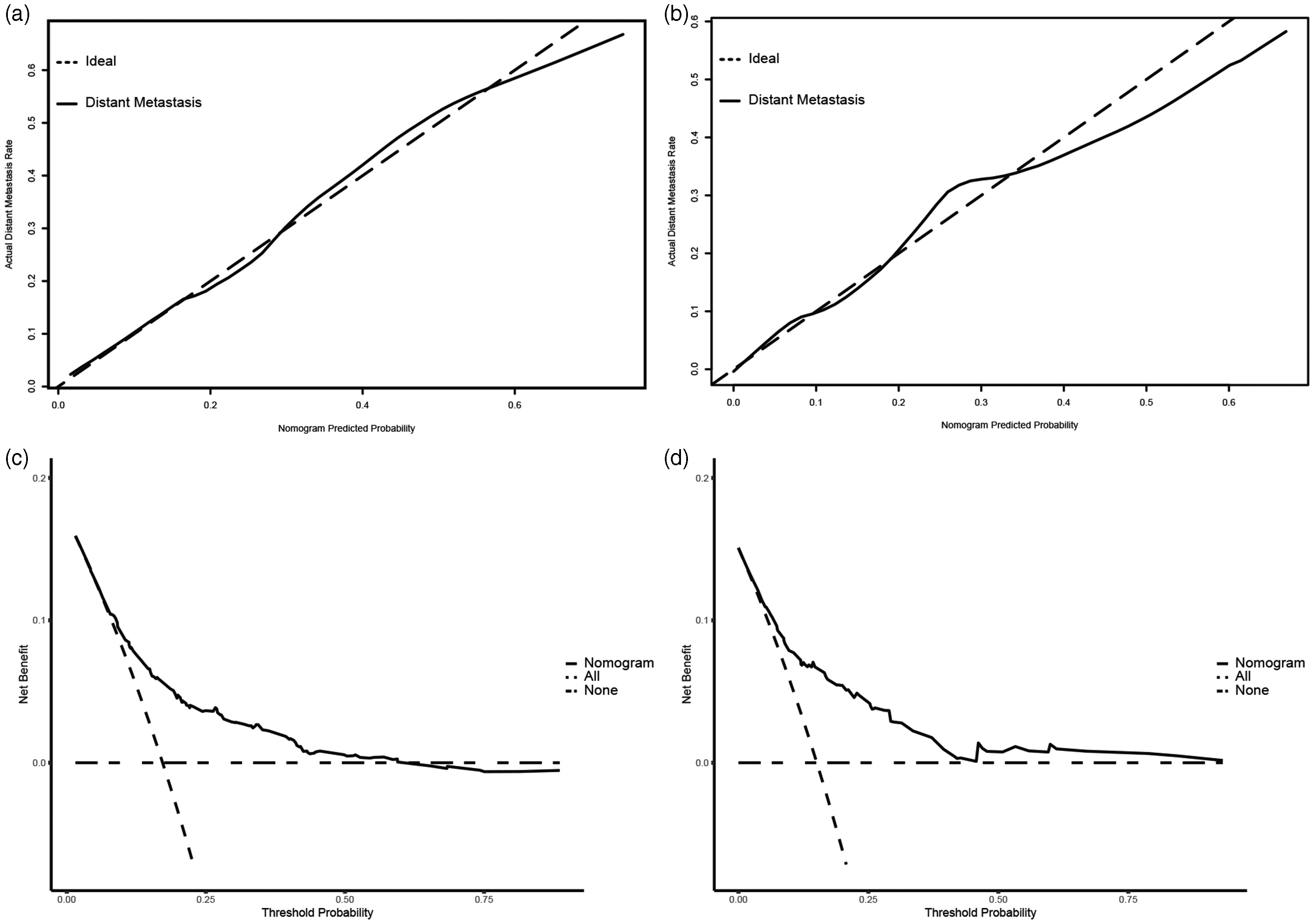
Validation of the diagnostic nomogram. (a) Calibrating curve and (c) decision curve analyses of the training cohort. (b) Calibrating curve and (d) decision curve analyses of the validation cohort.
Prognostic factors for patients with DM
In total, 327 patients with DM who met the inclusion criteria were identified for the evaluation of prognostic factors. The median OS for all patients with DM was 6 (1–80) months. As summarized in Table 3, 61 patients (18.7%) underwent surgery, 43 (13.1%) underwent radiotherapy, and 211 (64.5%) underwent chemotherapy (Table 3). The most common metastatic organ was the lung, followed by the bone, liver, and brain. There was no significant difference between the training and validation prognostic cohorts. Chemotherapy and metastasis to the lung, bone, and liver were identified as independent prognostic factors for patients with DM using univariate and multivariate Cox regression analyses (Table 4).
Characteristics of patients with MPM and DM in the prognostic cohort.
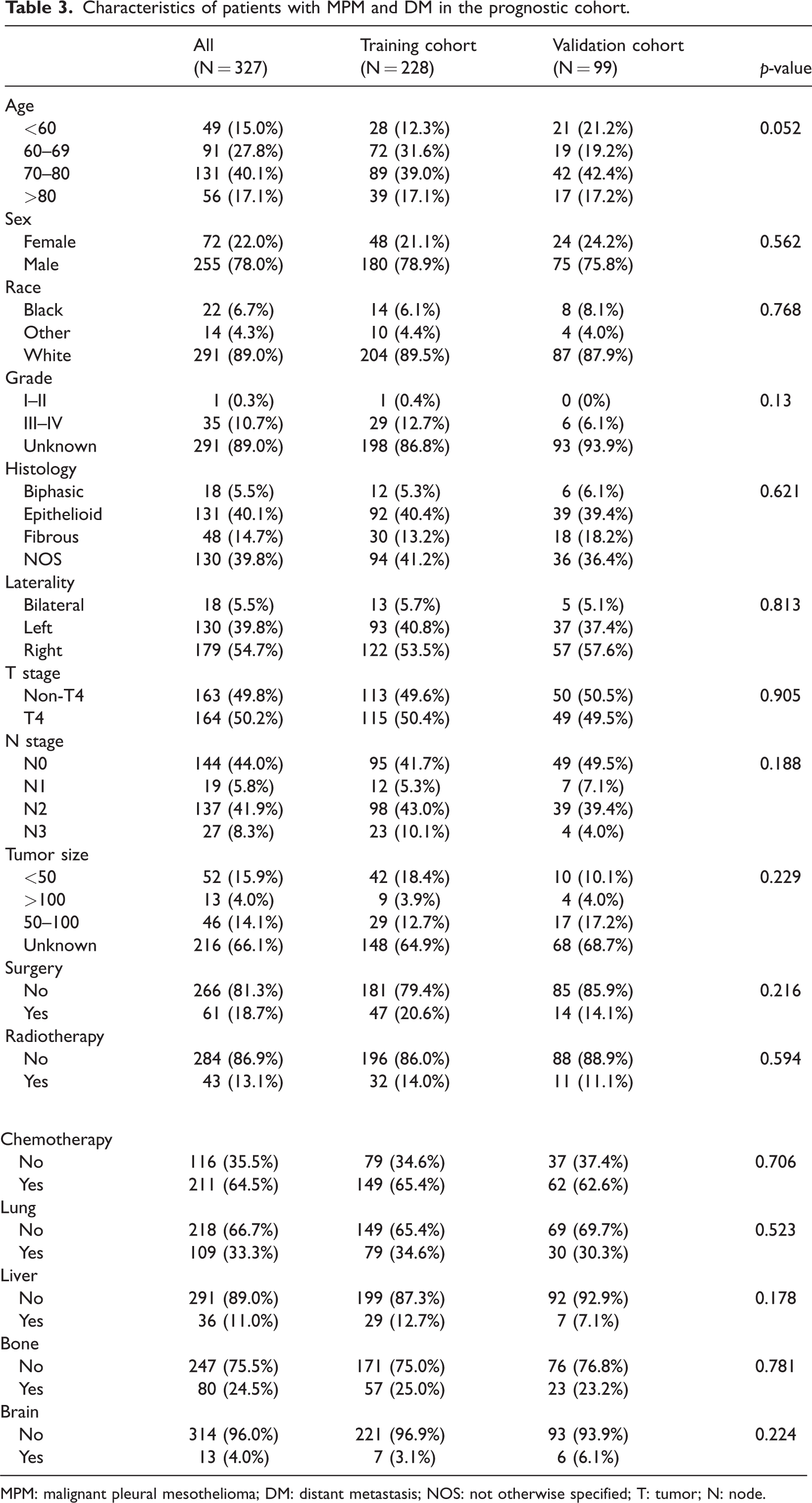
MPM: malignant pleural mesothelioma; DM: distant metastasis; NOS: not otherwise specified; T: tumor; N: node.
Univariate and multivariate Cox regression analyses of prognostic factors correlated with OS inpatients with MPM and DM.

OS: overall survival; MPM: malignant pleural mesothelioma; DM: distant metastasis; HR: hazard ratio; CI: confidence interval; NOS: not otherwise specified; T: tumor; N: node.
Prognostic nomogram construction and validation
A prognostic nomogram was established based on the four robust independent prognostic factors (Figure 5). The C-index was 0.678 in the training cohort, demonstrating a relatively good discrimination ability of the nomogram, and 0.712 in the validation cohort. Additionally, the AUC values of the nomogram at 6, 12, and 18 months were 0.722, 0.728, and 0.759 for the training cohort and 0.804, 0.711, and 0.812 for the validation cohort, respectively (Figure 6). To further clarify the predictive ability of the prognostic nomogram, the AUC values calculated at 24 months were 0.762 in the training and 0.862 in the validation cohorts. Moreover, calibration plots demonstrated outstanding consistency between the predicted and actual OS at 6, 12, and 18 months in the training and validation cohorts (Figure 7). DCA at the 6, 12, and 18 months indicated that the nomogram had high predictive accuracy in both the cohorts (Figure 8).
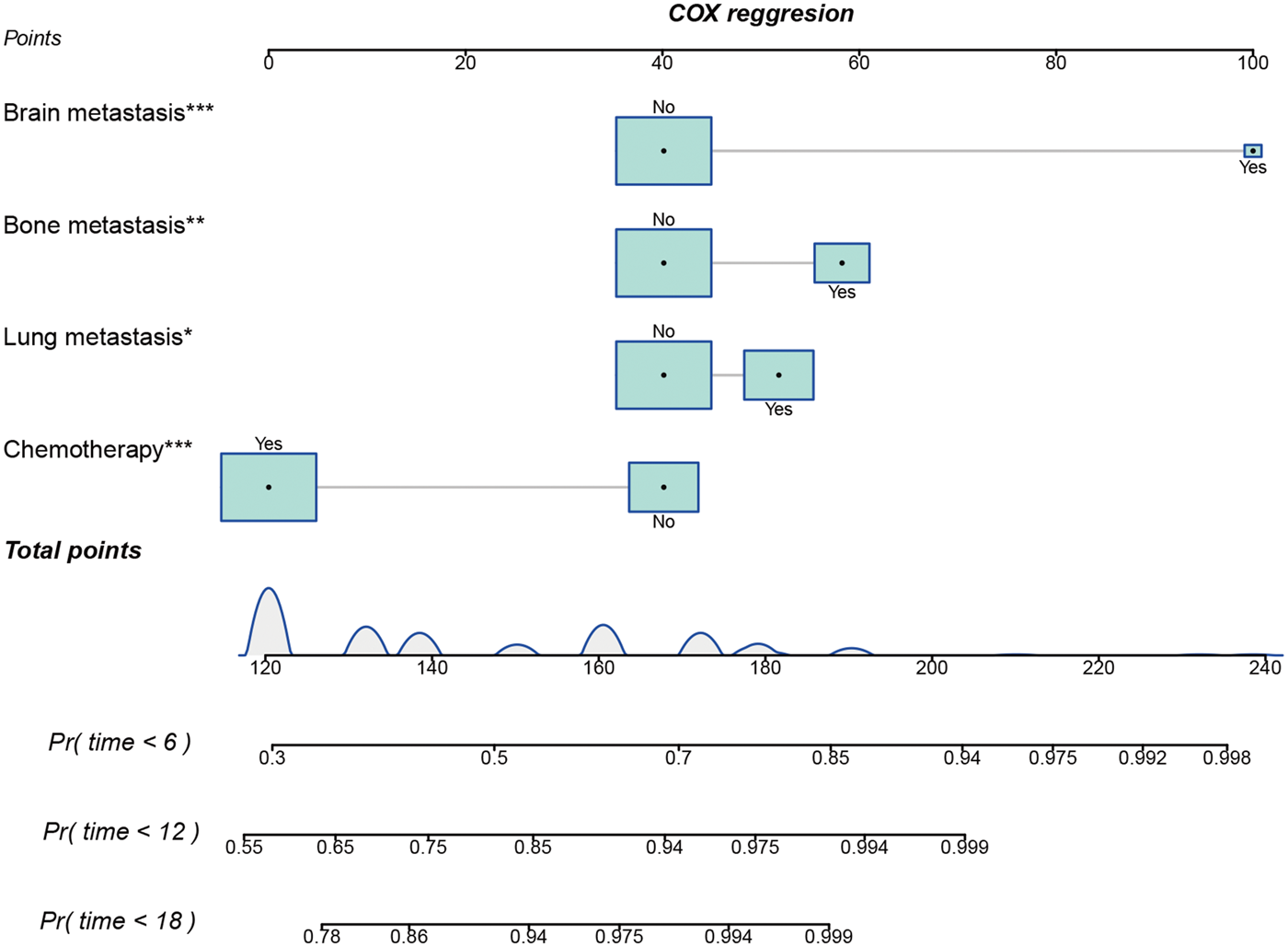
Prognostic nomogram for predicting the 6-, 12-, and 18-month overall survival rates in patients with MPM and DM. MPM: malignant pleural mesothelioma; DM: distant metastasis.

Time-dependent ROC and AUCs of the prognostic nomogram for 6-, 12-, and 18-month overall survival rates in the (a) training and (b) validation cohorts. ROC: receiver operating; AUC: area under the curve.

Calibration curves of the prognostic nomogram for (a) 6, (c) 12, and (e) 18 months in the training cohort and for (b) 6, (d) 12, and (f) 18 months in the validation cohort.
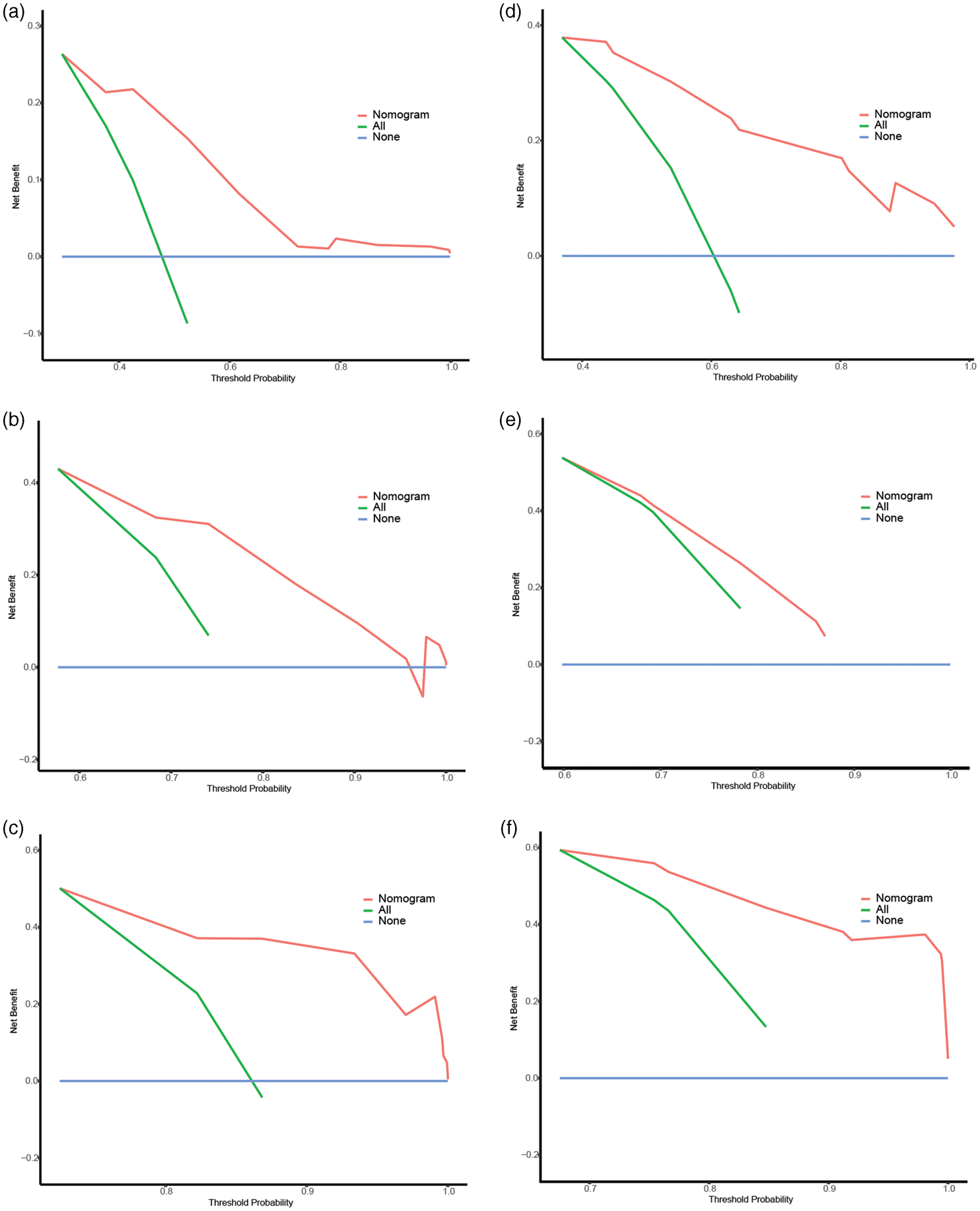
DCA of the prognostic nomogram for (a) 6, (c) 12, and (e) 18 months in the training cohort and for (b) 6, (d) 12, and (f) 18 months in the validation cohort. DCA: decision curve analysis.
Discussion
To the best of our knowledge, this is the first study that used a large-scale, population-based, national cancer registry database to identify independent risk and prognostic factors for patients with MPM who had DM. Simultaneously, diagnostic and prognostic nomograms were developed with reliable accuracy and discriminative ability, which were validated using ROC, calibration, and DCA curves. These two nomograms can serve as a practical tool for clinicians to identify patients with a high risk of DM and determine the optimal clinical interventions for patients diagnosed with MPM.
Recently, some studies have focused on DM in MPM; however, most of them are case reports, literature reviews, or molecular-level studies. A case review summarized that male patients and patients with epithelioid mesothelioma accounted for 79% and 64% of the study population, respectively; the average age was 60 (45–73) years for all patients with oral region metastasis. 20 Law et al. found that direct extension occurred more frequently with epithelioid than with mixed and sarcomatous pleural mesothelioma, including extension to the pericardium, collateral pleura, and peritoneum. 21 They also reported DM in 39 of the 60 cases on postmortem and revealed that DM occurred significantly more frequently with sarcomatous mesotheliomas. In another series of 1092 patients with MPM, 159 patients were diagnosed with DM, and OS was worse in these patients than in those without DM. 17 Epithelial–mesenchymal transition was associated with MPM development and metastasis because of increased oxidative stress and elevated levels of transforming growth factor-beta (TGF-β) induced by asbestos fibers. 22 Mir-137 was closely associated with the invasion and migration of MPM by targeting YBX1 23 as well as CD70, 24 ADAM10, 25 and BNDF. 26 However, these parameters and methods were invasive and impractical for clinicians to apply due to lack of efficacy or convenience. Nomograms can predict DM efficiently in other tumors. Shou et al. combined age, primary site, grade, T stage, and surgery to predict metastasis in osteosarcoma. 27 In breast cancer, a nomogram was established based on age, tumor size, race, location, grade, histology, and subtype. 28 Nonetheless, no study has focused on DM in MPM based on the clinical characteristics despite its poor prognosis.
In this study, logistic regression analysis was used to determine five risk factors for DM in MPM, namely, grade, laterality, histology, T stage, and N stage. Meanwhile, out of the ≥2000 patients with MPM, we observed DM in 341 patients (16.6%), which was lower than that reported previously. Similar to other tumors, grade, T stage, and N stage were confirmed to be associated with DM in MPM.29,30 Compared with bilateral tumors, we speculated that unilateral tumors have a low tumor burden and reduced opportunity for direct extension and DM. Although epithelioid MPM is associated with higher survival rates than biphasic MPM, patients with epithelioid MPM, sarcomatous MPM, and NOS MPM are more likely to develop DM, consistent with previous findings. 21 Further research is necessary to examine this phenomenon in detail.
To date, many studies have analyzed the prognosis of MPM; however, unlike these studies, our study focused on the prognosis of DM in patients with MPM. Although age was regarded as a prognostic factor in patients with MPM, it was not included in our prognostic nomogram. MPM predominantly affects older individuals and is rare in individuals <50 years, with incidence sharply rising after the age of 50 years; the median age at diagnosis is 76 years. 31 The impact of age on the prognosis of patients with MPM is controversial, with some studies stating that younger age is associated with higher OS and other studies reporting contradictory findings.32,33 It is generally believed that advanced MPM, particularly with DM, is more common in older patients than early-stage MPM. Moreover, the OS in patients with MPM and DM is extremely short, and accurately determining its impact in a timely manner is nearly impossible. As reported by other studies, the TNM stage was not associated with prognosis in patients with advanced-stage DM.34,35 Our nomogram excluded surgery, which indicated that surgery could not improve the prognosis in patients with DM. Although the median survival and OS reportedly improved in patients with early-stage disease after radical surgery, 36 patients with DM typically have advanced-stage DM with poor physical conditions, making them unsuitable for surgery. Most experts recommended the use of radiation therapy for MPM in the following three specific scenarios: (a) hemithoracic radiation therapy to be used before or after extrapleural pneumonectomy; (b) hemithoracic radiation as an adjuvant to lung-sparing procedures (i.e. without pneumonectomy); and (c) palliative radiation therapy for focal symptoms caused by the disease. 37 Nonetheless, radiotherapy was not a prognostic factor in our prognostic nomogram. Trimodality therapy significantly improved OS in patients with locally advanced cancer; however, no benefit was observed for patients with regional or distant cancer. 4 Thus, the benefits of radiotherapy in patients with MPM and DM are unclear and require further clinical trials for clarification. In our study, chemotherapy was the only treatment identified using Cox regression analysis, consistent with the findings in other tumors. Platinum-pemetrexed combined chemotherapy is the first-line therapy recommended in the guidelines.31,38 Additionally, for patients with resectable and unresectable MPM, chemotherapy is recommended. 39 The most frequently involved organs in patients with MPM are reported to be the liver (55.9%), adrenal glands (31.1%), kidneys (30.1%), contralateral lung (26.8%), and brain (3%). 40 Nonetheless, the most common metastatic organ was the lung, followed by the bone, liver, and brain in the present large population. Furthermore, distant invasions to the lung, bone, and brain were identified as independent prognostic factors, whereas liver metastasis exerted no impact on the survival of patients with DM. Unlike previous prognostic nomograms focusing on patients with MPM who underwent surgery, 16 those diagnosed with malignant peritoneal mesothelioma, 41 and those with invasive procedure metrics of MPM, 5 our study primarily aimed to identify risk factors for DM development and prognostic factors following metastasis.
Therefore, it is recommended that clinicians take appropriate and timely measures to prevent DM in patients with MPM and to monitor the development of metastasis in these patients.
In contrast, Wendy et al. reported that ERCC1 and RRM1 were useful biomarkers for predicting improved survival outcomes in patients with advanced MPM who received chemotherapy. 42 In another series of 122 patients with advanced MPM, a relatively high prevalence of BAP1 gene mutations, particularly after second-line treatment, was associated with disease progression and increased mortality risk. 43 Based on routinely available variables from the time of diagnosis, a simple yet clinically relevant model was developed and validated to reliably classify patients with high and low risks of death; however, hemoglobin and serum albumin levels were still included. 44 Genetic examination and laboratory tests are expensive and invasive. Therefore, there is a need to develop an economical model for evaluating metastasis, similar to our nomogram.
However, there are certain limitations in this study. First, prospective cohort studies are required to confirm our results because of the potential selection bias in this retrospective study. Second, given that the SEER database is derived exclusively from the US population and lacks external validation in this study, inherent biases may persist in the model’s generalizability. Third, because of the high mortality and low survival rates, the small number of patients with MPM and DM may have contributed to a possible inaccuracy. Finally, the data on tumor size and tumor grade were incomplete, which may have led to an underestimation of their impact on metastasis and survival.
Conclusions
We have comprehensively demonstrated that grade, laterality, histology, T stage, and N stage are independent risk factors of DM in patients with MPM. Furthermore, chemotherapy and metastasis to the lung, bone, and brain were found to be independent prognostic factors in patients with MPM and DM. These two nomograms may be clinically useful in the assessment of risk and prognosis in patients with MPM and DM, potentially facilitating clinical decision-making.
Footnotes
Acknowledgment
We are grateful to all the developers of R package who shared their code freely. We also acknowledge the use of the Surveillance, Epidemiology, and End Results (SEER) database for the data used in this study.
Author contributions
QZL and CL conceptualized and designed the study. QZL and MXX generated the figures and tables. CL and HZ performed the statistical analyses. QZL and CL wrote the first draft of the manuscript. JY and HY scrutinized every aspect of this study and critically reviewed the original draft of the manuscript. QZL and CL contributed equally to this work.
Data availability statement
Data used and/or analyzed in the present study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
None declared.
Ethics approval
As the publicly available and accessible SEER database was used and no individual patient information was used, written informed consent or ethical approval was not required for the present study.
Funding
This work was supported by the Hunan Provincial Natural Science Foundation of China (2025JJ70211), Shaoyang City Science and Technology Plan Guiding Project (2024PT6135) and the Scientific Research Project of the First Affiliated Hospital of Shaoyang University (24FY1001).
Patient consent for publication
Not applicable.
