Abstract
Background and objectives:
Dimensional response is an unmet need in second lines of advanced soft tissue sarcomas (STS). Indeed, the three approved drugs, pazopanib, trabectedin, and eribulin, achieved an overall response rate (ORR) of less than 10%. This fact potentially hinders the options for fast symptomatic relief or surgical rescue. The combination of trabectedin plus low-dose radiation therapy (T-XRT) demonstrated a response rate of 60% in phase I/II trial, while real-life data achieved 32.5% ORR, probably due to a more relaxed timing between treatments. These results were obtained in progressing and advanced STS. In this study, the merged databases (trial plus real life) have been analyzed, with a special focus on leiomyosarcoma patients.
Design and methods:
As responses were seen in a wide range of sarcoma histologies (11), this study planned to analyze whether leiomyosarcoma, the largest subtype with 26 cases (30.6%) in this series, exhibited a better clinical outcome with this therapeutic strategy. In addition, four advanced and progressing leiomyosarcoma patients, all with extraordinarily long progression-free survival of over 18 months, were collected.
Results:
A total of 847 cycles of trabectedin were administered to 85 patients, with the median number of cycles per patient being 7 (1–45+). A trend toward a longer progression-free survival (PFS) was observed in leiomyosarcoma patients with median PFS (mPFS) of 9.9 months [95% confidence interval (CI): 1.1–18.7] versus 5.6 months (95% CI: 3.2–7.9) for the remaining histologies, p = 0.25. When leiomyosarcoma and liposarcoma were grouped, this difference reached statistical significance, probably due to the special sensitivity of myxoid liposarcoma. The mPFS for L-sarcomas was 12.7 months (95% CI: 7–18.5) versus 4.3 months (95% CI: 3.3–5.3) for the remaining histologies, p = 0.001. Cases with long-lasting disease control are detected among leiomyosarcoma patients.
Conclusion:
Even when extraordinarily long-lasting responses do exist among leiomyosarcoma patients treated with T-XR, we were unable to demonstrate a significant difference favoring leiomyosarcoma patients in clinical outcomes.
Introduction
Soft tissue sarcomas (STS) are mesenchymal tumors with more than 70 different histological types representing 1% of adult malignancies. 1 Leiomyosarcomas (LMS) account for 5–10% of STS and are malignant mesenchymal neoplasms composed of cells that exhibit smooth muscle differentiation. 2 LMS common primary sites include limbs, retroperitoneum, abdomen, and uterus. Clinically, LMS shows biological aggressiveness with approximately 90% of LMS having a moderate to high grade. 3 Metastatic LMS have a poor prognosis and, currently, doxorubicin alone is used as the standard first-line treatment. 4 However, if fast relief is pursued or surgical rescue is foreseen, then the combination of anthracyclines plus dacarbazine could be the best upfront systemic treatment. 5 Considering the approved drugs for second lines in advanced STS: pazopanib, trabectedin, and eribulin, all of them exhibit response evaluation criteria in solid tumors (RECIST) responses below 10%, with disease control rate and progression-free survival (PFS) being the most important added values of these drugs. Therefore, fast symptomatic control and tumoral shrinkage are unmet needs in advanced STS patients beyond the first line. The LMS subset is not an exception to this reality, and the second-line options are trabectedin and pazopanib, derived from randomized phase III trials, or gemcitabine combinations, supported by randomized phase II trials.
Trabectedin (Yondelis®, PharmaMar, Madrid, Spain) is a semisynthetic marine-derived alkylating antineoplastic drug originally isolated from the sea squirt Ecteinascidia turbinata. 6 This drug has several potential mechanisms of action, including its ability to modulate the extracellular matrix. 7 Trabectedin has activity in anthracycline and alkylating agent-resistant STS and represents a valuable second-line option in a range of STS and ovarian cancer.8–11 Moreover, the efficacy of trabectedin is confirmed in terms of clinical benefit and low toxicity showing significant activity in liposarcoma and LMS subtypes.12,13 Real-world evidence suggests that trabectedin has effectiveness in disease control and a satisfactory safety profile in STS, indicating it to be a suitable long-term treatment drug associated with a good quality of life. 14 Combinations of trabectedin with other agents, that is, doxorubicin, have demonstrated significant increases of PFS in the first line of LMS, in comparison to doxorubicin alone. 4 In the second line, the combination of trabectedin and concomitant low-dose radiation therapy has been demonstrated to be synergistic in preclinical investigations while in the clinical settings, it achieved an overall response rate (ORR) of 60% by central radiology assessment in a phase I/II trial. 15 In a unicentric compilation of the real-life data, the authors reported an ORR of 32.5% with the combination of trabectedin and low-dose radiation therapy in progressing and advanced STS. 16 The sixfold and threefold increase in ORR with trabectedin and radiation therapy when compared to the expected response for trabectedin, detected in the clinical trial and real life, translated into longer PFS: 9.9 and 7.5 months, respectively. The real-life scheme was more relaxed regarding radiotherapy initiation times, and this could explain the differences in the outcomes. Interestingly, responses with this therapeutic approach have been detected in up to 11 different histological subtypes, representing a wide range of the STS spectrum. However, the impact of the combination of trabectedin plus low-dose radiation therapy among different histologies could be different. This analysis focuses on advanced LMS patients treated with this protocol and is based on the merged databases (trial and real life), as well as the report of four LMS patients, treated with this regimen and achieving PFS of longer than 18 months.
Patients and methods
Databases of patients treated with the combination of trabectedin plus low-dose radiation therapy derived from the phase I/II TRASTS trial (45 patients) 15 and real-life from Virgen del Rocio University Hospital (HUVR) (40 patients) 16 were considered for the first analysis. Patients derived from TRASTS trial cohort A (GEIS 37) were enrolled in six Spanish, two French, and one Italian hospitals. Patients derived from the second database were treated in one institution, and the investigation has been approved by the local Ethics Committee. Patients included in the clinical cases signed informed consent agreeing to this publication. Trabectedin in the first series was administered at 1.3 mg/m2 (12 patients) and 1.5 mg/m2 (33 patients) via a 24 h infusion every 21 days. Radiation therapy delivered a total dose of 30 Gy in 10 fractions of 3 Gy/day. Radiation therapy started within 1 h after completion of the first trabectedin infusion (cycle 1, day 2). Trabectedin in the second series was administered at 1.2–1.5 mg/m2 via a 24 h infusion every 21 days. Radiation therapy was administered with palliative intention with several regimens, the preferred regimen being 30 Gy at a rate of 3 Gy/day (28 patients). In the second series, the timing was more flexible, and radiation therapy sometimes started with the second cycle and in a few cases within 1 h after finishing the trabectedin infusion. In both series, 4 mg of dexamethasone was recommended at 12 and 24 h before trabectedin was initiated. Trabectedin was continued until progressive disease or intolerance, whichever occurred first. The protocol in both series required the irradiation of one lesion, usually the largest or the fastest-growing lesion, irrespective of the number of metastases or the organs involved in metastatic lesions. It was not mandatory to include all the lesions in the irradiation fields. Radiological assessments were centrally performed every 6 weeks in the first series and every 6–8 weeks in the second series. All but one patient, a total of 84 patients, were assessable for RECIST response while 85 were evaluable for survival. In addition, four case reports of advanced LMS patients treated with trabectedin and concomitant radiation therapy, with an extraordinarily long PFS, stemming from four hospitals are also included here.
For the statistical analysis, variables following binomial distributions (i.e. response rate) were expressed as frequencies and percentages. Comparisons between qualitative variables were expressed using the Fisher exact test or chi-square. Comparisons between quantitative and qualitative variables were performed through nonparametric tests (U of Mann–Whitney or Kruskal–Wallis). Time-to-event variables [overall survival (OS) and PFS] were measured from the date of therapy onset and were estimated according to the Kaplan–Meier method. Comparisons between the variables of interest were performed by the log-rank test. All p values reported were two-sided, and statistical significance was defined at p < 0.05.
Results
The median age was 51 years and, in most cases, the general performance status was ECOG 0 (57.6%). Leiomyosarcoma was the largest histological subtype of this series with 26 patients (30.6%). Other demographics are depicted in Table 1. A total of 847 cycles of trabectedin were administered to 85 patients, with the median number of cycles per patient being 7 (1–45+). The toxicity profile was reported elsewhere, 15 and it was almost superimposable to the usual toxicity exhibited by trabectedin. The only exception was grade-3 pneumonitis seen in 4 out of 85 patients. In two of these cases, an overlapping with previous radiation fields was detected. In any case, no toxic death was reported with this scheme. For the whole series, the median PFS and OS were 6.4 (95% CI: 3.9–8.8) and 21.5 (95% CI: 19.5–23.5) months, respectively. However, if only patients included in the phase II part and real life were selected, the median PFS and OS were 8.3 (95% CI: 5.5–11.0) and 21.6 (95% CI: 13.7–29.6) months, respectively.
Demographics and clinical characteristics of patients.

ECOG, eastern cooperative group UKN unknown; NOS, not other specified sarcoma; UPS, undifferentiated pleomorphic sarcoma.
The median PFS of LMS patients was 9.9 months (95% CI: 1.1–18.7) and it was not significantly different compared with all the remaining histologies as a group, 5.6 months (95% CI: 3.2–7.9), p = 0.25, but it did show a trend toward longer PFS (Table 2 and Supplemental Figure S1). However, when leiomyosarcoma and liposarcoma are considered in the same group, as L-sarcomas, the median PFS was significantly longer compared with others, 12.7 (95% CI: 7–18.5) versus 4.3 (95% CI: 3.3–5.3) months, respectively, p = 0.001 (Table 2 and Figure 1). Similarly, L-sarcomas had a statistically significant longer OS than non-L-sarcomas, 23.5 (95% CI: 11.1–35.8) versus 19.7 (95% CI: 7.1–32.3) months, p = 0.035 (Table 2 and Figure 1).
Univariate analysis of clinicopathologic variables.
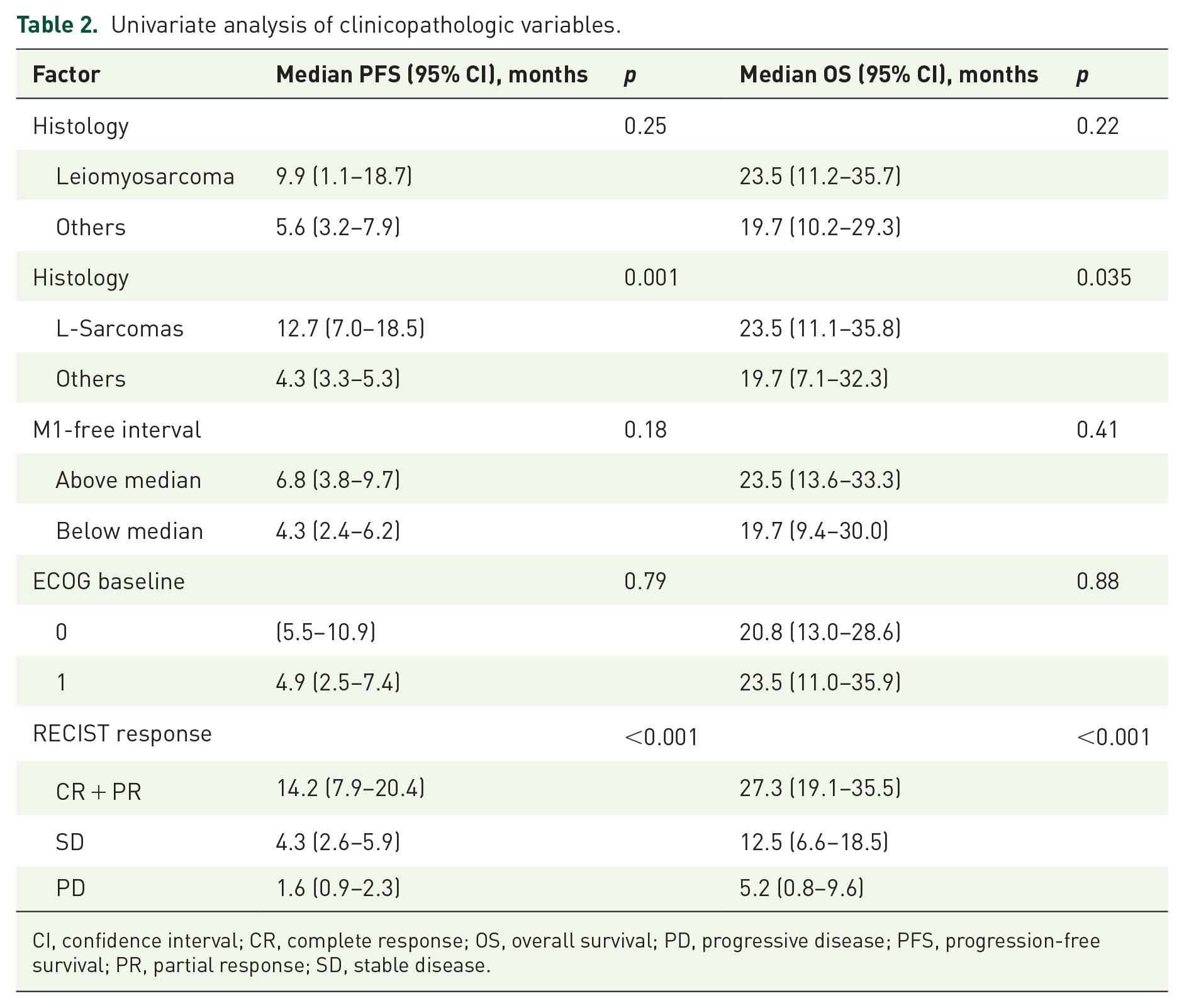
CI, confidence interval; CR, complete response; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.

Survival analysis. Kaplan–Meier curves for (a) PFS between L-sarcomas and non-L-sarcomas and (b) OS between L-sarcomas and non-L-sarcomas.
RECIST responses were distributed as follows: complete responses 5 (6%), partial responses 35 (42%), stabilizations 30 (36%), and progressions 14 (16%), out of 84 evaluable patients. The obtaining of a response (complete and partial) significantly correlated with a longer PFS and OS (Table 2 and Figure 1). Even when LMS patients showed a longer median PFS of 18.7 (95% CI: 8.4–28.9) months in responder patients, this did not reach a statistical difference with respect to non-LMS responder patients, who obtained a median PFS of 12.4 (95% CI: 5.1–19.8) months, p = 0.414 (Table 2 and Supplemental Figure S2). Likewise, the rate of LMS patients receiving at least 12 cycles of trabectedin was 46.2%, while for non-LMS it was 28.8% (p = 0.09). When L-sarcomas were considered, 50% received at least 12 cycles of trabectedin, while for non-L-sarcoma patients this proportion was 20% (p = 0.003). The liposarcoma subset that most contributed to increasing the proportion of patients with at least 12 cycles of trabectedin was myxoid liposarcoma.
Considering the RECIST response rate, there was no difference between LMS (46.2%) and non-LMS (48.3%), p = 0.52, nor between L-sarcomas and non-L-sarcomas, 47.5% for both.
To emphasize the special sensitivity of LMS histology to the combined strategy of trabectedin plus radiotherapy, we have collected four cases of advanced LMS cases (two somatic in extremities, one uterine, and one retroperitoneal that emerged from the inferior vena cava) with PFS of at least 18 months from initiation of trabectedin and concomitant radiotherapy.
Case report #1
An 83-year-old female patient was diagnosed with stage-IIA LMS of the right ankle (T1b N0 M0) in October 2017. The patient was treated with neoadjuvant radiotherapy (50 Gy/25 fx) and surgery. Disease progression in the lungs, liver, paravertebral space, and retroperitoneum was detected in February 2021. In March 2021, the patient started systemic treatment with trabectedin 1.5 mg/m2 every 3 weeks (increased to every month from the sixth cycle due to some bone marrow claudication). After six cycles of trabectedin, pulmonary progression was observed. Then, treatment with radiotherapy (30 Gy/10 fx), according to TRASTS scheme 15 was proposed. Radiotherapy was administered to the left upper lobe (LUL) lung nodule in October 2021. The computerized tomography (CT) scan performed after the 11th cycle of trabectedin (January 2022) showed a partial response of the disease, not only in the irradiated lesion (LUL) but also in other lesions that did not receive radiotherapy, such as the nodule in the upper right lobe, previously in progression with trabectedin, which decreased from 3.7 to 2.2 cm (Figure 2). Moreover, the paravertebral and liver lesions disappeared and the hepatorenal space implant shrank. In September 2022, after the 18th cycle of trabectedin, the upper right lobe (URL) lung nodule showed a further dimensional reduction (1.7 cm) (Figure 2). Other lesions remained stable at this point. The patient presented disease progression in January 2023.

Case report #1. (a) LUL nodule-irradiated nodule. From left to right: baseline PET/CT scan in February 2021. CT imaging previous to cycle 9 of trabectedin and radiotherapy (August 2021). CT straight after the 11th cycle of trabectedin and concomitant radiotherapy, already showing a reduction in the LUL nodule. CT in September 2022 after the 18th cycle of trabectedin showed a maintained partial response. (b) Evolution of the nodule in URL, showing partial response to therapy. (c) Evolution of paravertebral nodule (above) and hepatorenal space implant (below).
Case report #2
A 56-year-old female patient was diagnosed with a International Federation of Gynaecology and Obstretrics (FIGO) stage-IB uterine LMS. Surgical treatment was performed in November 2017. After 9 months, the patient was referred to the Gynecology Clinic at Instituto Português de Oncologia do Porto FG (IPO Porto), due to pelvic and lung recurrence, with a bulky unresectable pelvic mass (7.5 × 6.0 × 6.8 cm), lateralized to the right (conditioning right hydronephrosis) and bilateral pulmonary sub-centimetric micronodules. The patient was treated with palliative anthracycline-based first-line chemotherapy until August 2019, with stable disease [Figure 3(a)].

Case reports #2, #3, and #4. Pelvic CT scan of case report #2: (a) right paramedian retrovesical pelvic mass, 7.0 × 6.1 × 8.8 cm, in July 2019 and (b) pelvic mass, 3.7 × 4.7 cm, after trabectedin and radiotherapy to pelvic mass, followed by 31 cycles of trabectedin, in October 2021. CT scan of case report #3: (c) at baseline and (d) showing stable disease after three cycles. Density change was achieved. Axial MRI of case report #4: (e) at baseline and (f) best response. The response (f) was achieved and maintained, without the need for further treatment.
In October 2019, the patient started trabectedin 1.5 mg/m2, 24-h infusion, and radiotherapy to the pelvic mass (30 Gy, 3 Gy/day, started 1 h after the end of the first cycle), followed by trabectedin every 21 days. The patient achieved a long-lasting partial response in the pelvic mass (dimensions in October 2021 after 31 cycles were 3.7 × 4.7 cm) [Figure 3(b)], with stable pulmonary nodules. In February 2022, the patient stopped trabectedin due to congestive heart failure [left ventricular ejection fraction (EF) 41%]. In March 2022, after recovery and without cardiac contraindication, trabectedin treatment was resumed with a dose reduction to 1.2 mg/m2 (36th cycle). Sixteen days later, the patient was hospitalized due to a cardioembolic stroke. Due to dilated cardiomyopathy with severe depression of systolic function (EF 24%) and left bundle branch block, she was implanted with a cardiac resynchronization therapy device.
In April 2022, the patient maintained a response. In May 2022, treatment with trabectedin was resumed (cycle 37). At cycle 43, in September 2022, the patient presented progressive disease (pulmonary nodules 13 mm, pelvic mass 8.2 × 7.3 cm, and a new pelvic tumor implant measuring 3.5 × 2.0 × 4 cm). Consequently, a new treatment with gemcitabine/docetaxel was proposed.
Case report #3
A 50-year-old female patient was diagnosed with a 4-cm mass dependent on the inferior vena cava during a routine gynecological examination in July 2010. The tumor was resected in September 2010. The pathology report revealed a 5 cm LMS and periodic follow-up was started. In April 2014, a lesion was identified on the scalp, which was biopsied and confirmed to be LMS. Further evaluation with a positron emission tomography (PET) scan showed hepatic metastatic spread. In June 2014, the hepatic disease was resected, and the patient continued to be monitored until December 2016. A PET scan performed at that time identified the disease in the right thigh and left inguinal region. In January 2017, the patient began treatment with doxorubicin and olaratumab as part of a clinical trial, with stability observed as the best response. In September 2018, the disease progressed with lesions involving the right quadriceps, liver, and lungs. The patient was enrolled in the TRAST cohort A clinical trial (trabectedin + radiotherapy) and showed stability after three cycles, although with a tendency to reduce without reaching RECIST partial response [Figure 3(c) and (d)]. A change in density was observed after 38 cycles in December 2020, leading to the cessation of the treatment. After an extended period of hospitalization due to intestinal perforation requiring surgery, a CT scan in March showed disease progression in the liver and thigh. The patient resumed treatment in March 2021 with gemcitabine/dacarbazine, as has so far received 28 cycles with partial response in the liver and lungs, with no appearance of new lesions.
Case report #4
A 59-year-old male patient was referred to the Sarcoma Unit of HUVR. In May 2016, the patient reported self-palpation of a <1 cm mass in the deltoid, which grew rapidly over the next 3 months, reaching a size of 12 cm. CT scans showed multiple pulmonary and regional nodal metastases. Biopsy confirmed Grade 3 LMS. As the disease presented at stage IV, the patient was included in a clinical trial receiving treatment with doxorubicin ± olaratumab with progressive disease as his best response. In December 2017, the patient started treatment with trabectedin 1.5 mg/m2 via continuous 24-h infusion, with concomitant radiotherapy. On the day of the trabectedin infusion pump removal, the patient began radiotherapy (30 Gy in 10 sessions of 3 Gy per fraction) on the main lesion. After three cycles, the disease was stable, and an evaluation of the radiated lesion showed a reduction in size from 16 × 13 × 14 to 12 × 7 × 10 cm, with tumor density changes toward necrosis [Figure 3(e) and (f)].
In June 2019, the patient presented with a superficial T1aN0M0 urothelial cancer, which was endoscopically resected and treated with intravesical instillations of mitomycin C. The patient continued to receive trabectedin treatment without any significant toxicity. In July 2019, after 30 cycles of treatment, the pulmonary nodules disappeared, and only an 8 mm stable lesion in the lower right apical region and a 10 mm axillary lymph node remained. The primary tumor continuously shrank with each cycle of trabectedin. In May 2021, due to the need for a urethroplasty, the patient stopped the trabectedin treatment. After a 3-month break, the patient was closely monitored, and follow-up imaging showed stable disease in February 2023.
Discussion
The analysis of the merged series, derived from clinical trials and real-life data, underlines the efficacy of the combination of trabectedin and low-dose radiation therapy for advanced STS. The efficacy measurements were RECIST response of 48% ORR, or median PFS of 8.3 months and OS of 21.5 months, combining the phase II trial and real-life data. These are all outstanding facts suggesting that this strategy should be seriously taken into account as a prominent option in the advanced setting of STS. These figures are valuable not only for durable disease control but also for attaining other aims that seemed restricted for first-line combo schemes: rapid palliation and perhaps even potential surgical rescues in selected cases. Chemotherapy alone offers an ORR of less than 10% in second lines if we focus on the drugs approved after randomized phase III trials.13,17,18 When eribulin was tested in 17 metastatic breast cancer patients along with palliative radiation therapy, grade 3 neutropenia reached 56% and some advantages in mean pain score (from 2 to 0.7), without changes in analgesic score, were reported. The ORR was 29%, which seems to be in the low range of that obtained by eribulin alone (from 12% to 54%) in breast cancer.19,20 Pazopanib and low-dose radiation therapy (total 45 Gy) resulted in clinical benefit in a patient with angiosarcoma of the scalp. 21 In the renal carcinoma context, pazopanib was concomitantly administered with stereotactic body radiotherapy (SBRT) in a phase I trial, but the combination seemed not to be superior when compared with SBRT or pazopanib alone according to the authors. 22 Currently, there are no data regarding SBRT in combination with systemic therapy in advanced sarcoma. Gemcitabine alone or concomitant with radiation therapy (50.4 Gy) was compared in a randomized phase II trial in 74 unresectable pancreatic carcinomas. A significant benefit was obtained in OS, 9.2 versus 11.1 months, favoring the combination, despite no advantages being detected in ORR, 5% versus 6%, or in median PFS, 6.7 versus 6 months for gemcitabine alone or the combination, respectively. 23
Despite there being a wide range of sarcoma histologies achieving ORR (at least 11 different histologies) with trabectedin and radiotherapy, this analysis showed that this strategy resulted in a longer PFS in L-sarcomas, 12.7 versus 4.3 months (p = 0.001), and a longer OS, 23.5 versus 19.7 months (p = 0.035). However, no difference in terms of ORR was seen in 47.5% for both L- and non-L-sarcomas.
LMS showed a trend toward longer PFS, compared with the remaining histologies: 9.9 versus 5.6 months, but this difference did not reach statistical significance, probably due to the relatively low number of cases. Having detected long-lasting disease control in advanced LMS patients treated with this strategy, we were impelled to additionally collect some cases with an unusually long PFS. The common denominator in the four cases was a PFS of at least 18 months. Case #1 illustrates an abscopal-like effect since the TRASTS protocol was initiated in the context of extensive progression during trabectedin treatment. The relevant shrinkage in non-irradiated pulmonary nodules, as well as the disappearance of paravertebral and liver lesions, and a fast response, are striking observations. The fact that disease control was achieved beyond the irradiated nodules was already detected in the phase II trial 15 and it deserves to be studied more deeply. This finding suggests an immunomodulatory effect, probably through a T-cell-mediated immune response, as has been suggested by some authors.24,25 In addition, case #1 exemplifies good tolerance of trabectedin and TRASTS protocol in elderly patients. 26 Cases #2 and #3 show PFS of 35 and 21 months in uterine and retroperitoneal recurrent and advanced LMS, respectively. Even though randomized trials did not demonstrate survival benefit either in localized uterine LMS 27 or in localized retroperitoneal LMS, 28 it is unquestionable that trabectedin and radiotherapy synergistically elicited not only a partial response but, most importantly, also a maintained and prolonged response that seemed to be only restricted to myxoid liposarcoma. 29 Case #4 displays a fast response in an LMS of a somatic-muscle compartment of a limb in the context of an early fast-growing progression on doxorubicin. The magnitude of the shrinkage and the long-lasting disease control (63+ months) in this case support the message that TRASTS protocol can pursue the aim of attaining fast relief in the second-line STS setting, and this can be translated into long-term disease control.
Some of the study’s weaknesses lie in the retrospective nature of the analysis, the relatively low number of cases for histological sub-analyses, and the lack of translational research in the whole series. High expression of (the final character is the number one): High-Mobility Group Box 1 (HMGB1) correlated with longer OS in the trial 15 which suggests a potential link with immunogenic cell death 30 that deserves attention.
In conclusion, concomitant trabectedin and low-dose radiation therapy represent a valuable strategy of treatment for advanced progressing STS patients. Predictive biomarkers that will be able to select the best candidates and explain the mechanism of action should be elucidated in future studies.
Supplemental Material
sj-tif-1-tam-10.1177_17588359231225044 – Supplemental material for Trabectedin and low-dose radiation therapy in patients with advanced leiomyosarcoma
Supplemental material, sj-tif-1-tam-10.1177_17588359231225044 for Trabectedin and low-dose radiation therapy in patients with advanced leiomyosarcoma by Javier Martin-Broto, Alicia Olarte-García, Ricardo Fernandéz-Rodríguez, Marta Ribeiro Ferreira, Irene Carrasco-García, Fernando Galvez-Montosa, Inmaculada Rincon-Perez, Javier Peinado-Serrano, Ignacio Azinovic, Laura Guzman-Gomez, Lurdes Salgado, Alberto Sevillano, Justo Ortega, Cristina Alvarez, Antonio Gutierrez, David S. Moura and Nadia Hindi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-2-tam-10.1177_17588359231225044 – Supplemental material for Trabectedin and low-dose radiation therapy in patients with advanced leiomyosarcoma
Supplemental material, sj-tif-2-tam-10.1177_17588359231225044 for Trabectedin and low-dose radiation therapy in patients with advanced leiomyosarcoma by Javier Martin-Broto, Alicia Olarte-García, Ricardo Fernandéz-Rodríguez, Marta Ribeiro Ferreira, Irene Carrasco-García, Fernando Galvez-Montosa, Inmaculada Rincon-Perez, Javier Peinado-Serrano, Ignacio Azinovic, Laura Guzman-Gomez, Lurdes Salgado, Alberto Sevillano, Justo Ortega, Cristina Alvarez, Antonio Gutierrez, David S. Moura and Nadia Hindi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors express gratitude to Meisys (Madrid, Spain) for writing assistance. DSM is a recipient of a Sara Borrell postdoctoral fellowship funded by the National Institute of Health Carlos III (ISCIII) (CD20/00155). The authors would also like to thank the SELNET project. SELNET has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement no. 825806. JM-B is an Associate Editor of Therapeutic Advances in Medical Oncology; therefore, the peer review process was managed by alternative members of the board and the submitting editors were not involved in the decision-making process.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
