Abstract
Background:
Substitution of methionine for threonine at codon 790 (T790M) of epidermal growth factor receptor (EGFR) represents the major mechanism of resistance to EGFR tyrosine kinase inhibitors (TKIs) in EGFR-mutant non-small-cell lung cancer. We determined the prognostic impact and association of secondary T790M mutations with the outcomes of osimertinib and chemotherapy.
Methods:
Patients (n = 460) progressing from first-line EGFR-TKI treatment were assessed. Tissue and/or liquid biopsies were used to determine T790M status; post-progression overall survival (OS) was analyzed.
Results:
Overall, 143 (31.1%) patients were T790M positive, 95 (20.7%) were T790M negative, and 222 (48.2%) had unknown T790M status. T790M status [T790M positive versus T790M negative: hazard ratio (HR) 0.48 (95% confidence interval (CI), 0.32–0.70); p < 0.001, T790M unknown versus T790M negative: HR 1.97 (95% CI, 1.47–2.64); p < 0.001] was significantly associated with post-progression OS. T790M positivity rates were similar for tissue (90/168, 53.6%) and liquid (53/90, 58.9%) biopsies (Fisher’s exact test, p = 0.433). Tumor T790M-positive patients had significantly longer post-progression OS than tumor T790M-negative patients (34.1 versus 17.1 months; log-rank test, p = 8 × 10−5). Post-progression OS was similar between plasma T790M-positive and -negative patients (17.4 versus not reached; log-rank test, p = 0.600). In tumor T790M-positive patients, post-progression OS was similar after osimertinib and chemotherapy [34.1 versus 29.1 months; log-rank test, p = 0.900; HR 1.06 (95% CI, 0.44–2.57); p = 0.897].
Conclusion:
T790M positivity predicts better post-progression OS than T790M negativity; tumor T790M positivity has a stronger prognostic impact than plasma T790M positivity. Osimertinib and chemotherapy provide similar OS benefits in patients with T790M-positive tumors.
Plain language summary
The study demonstrates that patients with EGFR-mutant lung cancer who develop resistance due to a secondary T790M mutation, defined by tumor or blood T790M positivity, achieve better survival than patients without secondary T790M mutation; this association was mainly contributed by tumour T790M positivity. Oismertinib and chemotherapy led to similar survival in tumour T790M-positive patients. However, compared to osimertinib, chemotherapy was associated with longer survival in blood T790M-positive patients.
Novelty and impact statement
The study demonstrates that patients with EGFR-mutant advanced non-small-cell lung cancer who develop resistance due to a secondary T790M mutation, defined by tumor or plasma T790M positivity, achieve better post-progression survival than patients without secondary T790M mutation; this association was mainly contributed by tumor T790M positivity. Osimertinib and chemotherapy led to similar post-progression survival in tumor T790M-positive patients. However, compared to osimertinib, chemotherapy was associated with longer post-progression survival in plasma T790M-positive patients.
Introduction
Epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) have tremendously improved the survival of patients with advanced non-small-cell lung cancer (NSCLC) who have sensitizing EGFR mutations. 1 Although the front-line use of third-generation EGFR-TKIs has become a recommended standard practice for this group of patients,2,3 there are gaps in this practice across different countries. The high cost of third-generation EGFR-TKIs is largely responsible for this gap. However, other significant issues remain, such as questions of whether the efficacy of third-generation EGFR-TKIs outperforms second-generation EGFR-TKIs for front-line use; particularly as several real-world studies have demonstrated similar efficacy for afatinib and osimertinib 4 and favorable survival outcomes in patients who received sequential afatinib and osimertinib treatment.5,6
Approximately 50% of patients treated with front-line first- or second-generation EGFR-TKIs develop drug resistance related to the on-target secondary T790M mutation.7,8 Other off-target non-T790M resistance mechanisms account for the remainder of cases of resistance, including (but not limited to) mutations in MET and PIK3CA, HER2 amplification, and small-cell histological transformation.9,10 Previous studies, mostly tissue-based analyses, have demonstrated that patients with tumors that acquire the EGFR T790M mutation usually have a better prognosis than patients with tumors that acquire resistance that bypasses the EGFR pathway. 11 This finding is partly attributed to the availability of the T790M-active agent osimertinib, in addition to chemotherapeutic drugs, for the treatment of tumors with the T790M mutation. The AURA 3 trial of patients who progressed from gefitinib/erlotinib treatment and whose tumors were positive for T790M revealed equal overall survival (OS) between the groups treated with osimertinib and platinum-based chemotherapy. 12 Nevertheless, similar clinical trials for this group of patients are lacking and insufficiently reported from practice-based settings.
Clinically, post-progression tissue biopsies are not always feasible for every patient and thereby determination of T790M status from plasma is frequently used as an alternative.13,14 However, the prognostic value of plasma T790M in patients with unknown tumor T790M status is poorly explored. Previously, the detection of T790M status in plasma was comprehensively studied in patients with T790M-positive tumors, in which the capture of plasma T790M was positively correlated with tumor burden. 15 In that study, patients with T790M-positive plasma demonstrated poorer outcomes after both osimertinib treatment and chemotherapy compared to patients with T790M-negative plasma. 15 Nevertheless, the therapeutic relevance of plasma T790M in patients with T790M-negative tumors has received less research. The AURA phase I study demonstrated that the T790M mutation could be detected in the plasma in 30% of patients with T790M-negative tumors, suggesting that clonal heterogeneity comes along with tumor development and drug resistance.16,17 In that trial, osimertinib had similar efficacy in patients with T790M-positive plasma compared to those with T790M-positive tumors. 16 However, it remains largely unknown whether the predictive performance of plasma T790M is similar in clinical practice.
Recently, a prospective phase II study of patients with drug resistance to first-line gefitnib/erlotinib or afatinib screened the T790M mutation in plasma without prior knowledge of tumor T790M status. Second-line osimertinib treatment demonstrated acceptable efficacy in these plasma T790M-positive patients, with a response rate of 55% and progression-free survival (PFS) of 8.3 months. 18 However, the efficacy of osimertinib is also affected by the presence of other co-mutations, such as TP53 mutations. 19 Thus, whether osimertinib actually outperforms chemotherapy in plasma T790M-positive patients is largely unknown. As yet, no prospective randomized trials have addressed this issue, and evidence from practice-based settings remains relatively lacking.
In this study, we retrospectively analyzed a group of patients with advanced EGFR-mutant NSCLC who were treated with, and subsequently developed drug resistance to, first-line gefitnib/erlotinib or afatinib. The prognostic impact of the secondary T790M mutation, detected by tissue and/or liquid biopsies, was investigated and the survival outcomes of osimertinib and chemotherapy were compared.
Methods
Patients and treatment
Patients with advanced NSCLC who had EGFR-sensitizing mutations (exon 19 deletion or exon 21 L858R) and suffered disease progression after first-line EGFR-TKI treatment (gefitinib, erlotinib, or afatinib) were retrospectively included. Tissue and/or liquid biopsies were used to determine T790M status; tissue and liquid samples were assayed using the therascreen EGFR RGQ PCR kit (Qiagen, Valencia, CA, USA) and the RainDrop Digital PCR System (RainDance Technologies Lexington, MA, USA), respectively. Post-progression OS was defined as the interval between the date of radiologically or clinically determined progression during first-line EGFR-TKI treatment and the date of death. This study retrospectively assessed data from the Chang Gung Research Database and the Ethics Committee of Chang Gung Memorial Hospital approved the study protocol and provided a waiver of informed consent (No. 201801967B0).
Statistical analysis
The Student’s t-test was used to determine the significance of the differences in continuous variables between two groups, and Fisher’s exact test was applied to compare categorical variables among groups. Kaplan–Meier survival curves and Cox regression model-based hazard ratios (HRs) were generated and determined using the R package survival; all other analyses were performed using SPSS (version 10.1; SPSS, Chicago, IL, USA). All reported p values are two-sided and p < 0.05 was considered statistically significant.
Results
Baseline patient characteristics stratified by T790M status
A total of 460 patients who suffered disease progression after/during front-line treatment with an EGFR-TKI were retrospectively analyzed; 143 (31.1%) patients were T790M positive, 95 (20.7%) patients were T790M negative, and 222 (48.2%) patients had unknown T790M status (Table 1). Patients who had known T790M status were significantly younger (T790M positive versus T790M negative versus unknown: 64.8 ± 11.0 versus 63.9 ± 11.4 versus 69.3 ± 12.6 years; p < 0.001) and had better performance status [Eastern Cooperative Oncology Group performance status (ECOG) PS 0–1: 86.7% versus 89.5% versus 74.3%, p = 0.004] than patients with unknown T790M status.
Baseline patient characteristics.

ECOG PS, Eastern Cooperative Oncology Group performance status; PD-L1, programmed death-ligand 1; SD, standard deviation.
Cox regression analysis of post-progression OS
We determined the impact of various clinical factors on post-progression OS. Univariate Cox regression analyses demonstrated that age ⩾65 [HR 1.28 (95% confidence interval (CI), 1.01–1.62); p = 0.039], ECOG PS 0–1 [HR 0.41 (95% CI, 0.31–0.74); p < 0.001], and T790M status [T790M positive versus T790M negative: HR 0.48 (95% CI, 0.32–0.70); p < 0.001, T790M unknown versus T790M negative: HR 1.97 (95% CI, 1.47–2.64); p < 0.001, Table 2] were significantly associated with post-progression OS. In addition, patients with the EGFR L858R mutation also tended to achieve poorer post-progression OS [HR 1.27 (95% CI, 1.00–1.60); p = 0.052, Table 2]. Multivariate regression analyses subsequently demonstrated that ECOG PS 0–1 [HR 0.42 (95% CI, 0.32–0.55); p < 0.001] and T790M status [T790M positive versus T790M negative: HR 0.44 (95% CI, 0.30–0.65); p < 0.001, T790M unknown versus T790M negative: HR 1.81 (95% CI, 1.35–2.43); p < 0.001, Table 2] remained significant predictive factors for post-progression OS. Kaplan–Meier analysis revealed that T790M-positive patients achieved significantly longer post-progression OS (30.4 months) compared to T790M-negative patients (15.6 months) and patients with unknown T790M status (4.5 months; log-rank test p = 2 × 10−16, Figure 1).
Cox regression analysis of post-progression survival.

T790M negative as reference.
PD-L1 high as reference.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; PD-L1, programmed death-ligand 1.

Post-progression OS for T790M-positive patients (blue), T790M-negative patients (red), and patients with unknown T790M status (green).
Tumor T790M positivity has a stronger prognostic value for post-progression OS than plasma T790M positivity
Overall, 168 patients underwent a tissue biopsy, 90 patients underwent a liquid biopsy, and 20 patients received both tissue and liquid biopsies to determine T790M status. T790M positivity was observed in the tissue biopsies of 90 (53.6%) patients and liquid biopsies of 53 (58.9%) patients; the T790M positivity rate was not significantly different between the tissue and liquid biopsies (Fisher’s exact test, p = 0.433). Kaplan–Meier analysis demonstrated that patients with T790M-positive tumors had significantly longer post-progression OS (34.1 months) compared to patients with T790M-negative tumors [17.1 months; log-rank test p = 8 × 10−5, Figure 2(a)]. By contrast, post-progression OS was similar between patients with T790M-positive plasma (17.4 months) and patients with T790M-negative plasma [not reached; log-rank test p = 0.600, Figure 2(b)].

Post-progression OS for (a) tumor T790M-positive and -negative patients and (b) plasma T790M-positive and -negative patients.
Patterns of post-progression treatment
The subsequent post-progression treatment was further analyzed in the groups of T790M-positive and T790M-negative patients. Among the 143 patients with T790M-positive status, 100 (69.9%) patients received a third-generation EGFR-TKI, 26 (18.2%) patients received chemotherapy, 2 (1.4%) patients received an antiangiogenetic agent, 5 (3.5%) patients received a first- or second-generation EGFR-TKI, and 12 (8.4%) patients received only best supportive care. Of the 95 patients with T790M-negative status, 18 (18.9%) patients received a third-generation EGFR-TKI, 53 (55.8%) patients had chemotherapy, 5 (5.3%) patients received an antiangiogenetic agent, 4 (4.2%) patients received immunotherapy, 3 (3.2%) patients received a first- or second-generation EGFR-TKI, and 19 (20.0%) patients received only best supportive care (Table 3).
Post-progression subsequent treatment.

EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor.
Post-progression OS of third-generation EGFR-TKI treatment or chemotherapy stratified by tumor T790M status
Next, we further analyzed the association between tumor T790M status and the subsequent outcomes of third-generation EGFR-TKIs versus chemotherapy. Of the 90 patients with tumor T790M-positive status, 63 (70.0%) patients underwent treatment with a third-generation EGFR-TKI and 16 (17.8%) patients received chemotherapy. Post-progression OS was similar in the third-generation EGFR-TKI group (34.1 months) compared to the chemotherapy group [29.1 months; log-rank test p = 0.900, Figure 3(a)], as well as the reduction in the risk of death [HR 1.06 (95% CI, 0.44–2.57); p = 0.897]. Of the 78 patients with tumor T790M-negative status, 50 (64.1%) patients underwent chemotherapy and 13 (16.7%) patients received a third-generation EGFR-TKI. The chemotherapy group demonstrated similar post-progression OS (17.8 versus 13.3 months; log-rank test, p = 0.200) and reduction in the risk of death [HR 0.57 (95% CI, 0.23–1.40); p = 0.220] compared to the third-generation EGFR-TKI group [Figure 3(b)].
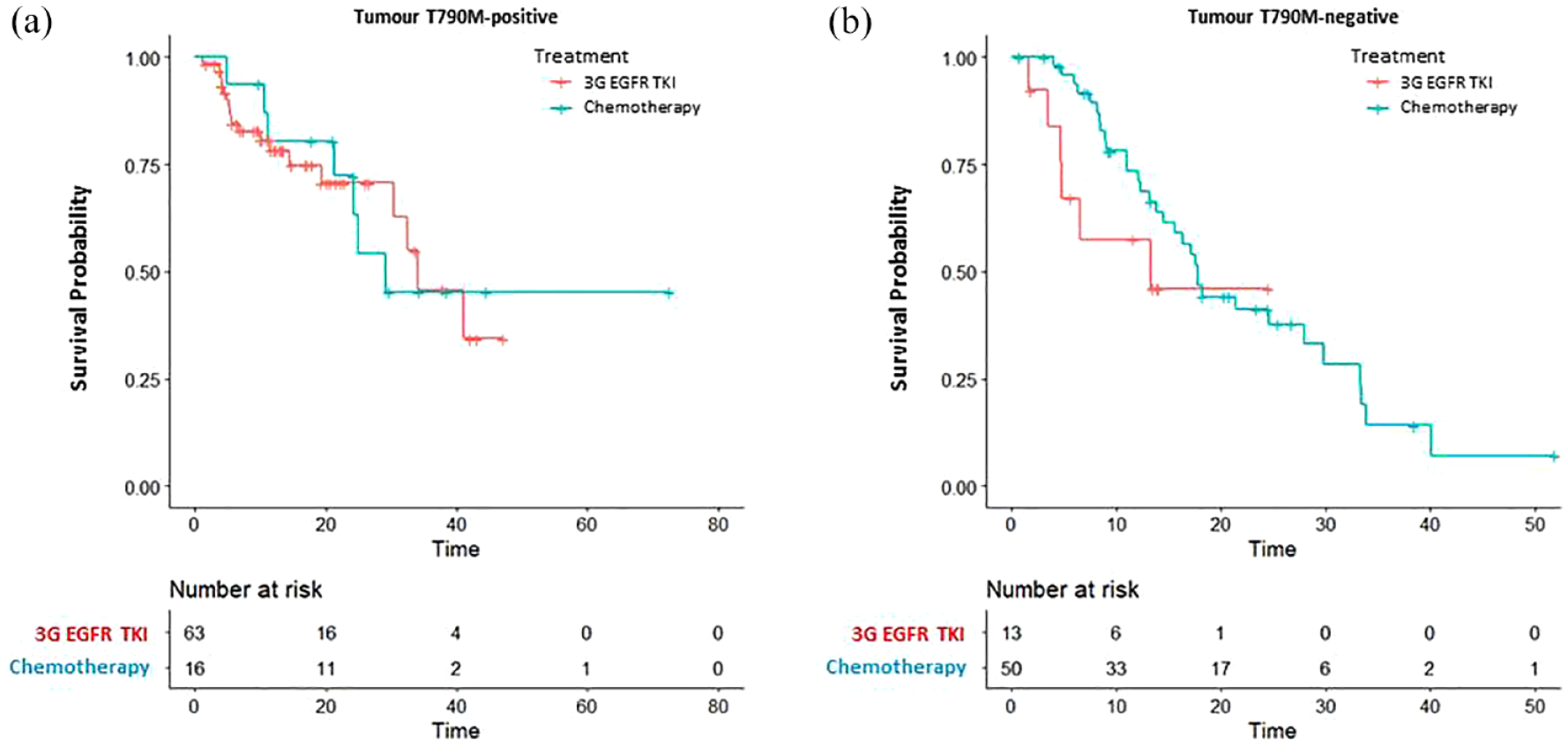
Post-progression OS for osimertinib treatment and chemotherapy in (a) tumor T790M-positive patients and (b) tumor T790M-negative patients.
Post-progression OS of third-generation EGFR-TKI treatment or chemotherapy stratified by plasma T790M status
Of the 53 patients with plasma T790M-positive status, 37 (69.8%) patients underwent treatment with a third-generation EGFR-TKI, and 10 (18.9%) patients received chemotherapy. Interestingly, patients who received chemotherapy exhibited a trend toward longer post-progression OS (not reached versus 15.1 months; log-rank test p = 0.100) and a higher reduction in the risk of death [HR 0.36 (95% CI, 0.10–1.28); p = 0.116] compared to patients who received a third-generation EGFR-TKI [Figure 4(a)]. Of the 37 patients with plasma T790M-negative status, 11 (29.7%) patients received chemotherapy and 14 (37.8%) patients were treated with a third-generation EGFR-TKI. Neither of the groups reached the median post-progression OS [Figure 4(b)] and the chemotherapy group demonstrated a similar reduction in the risk of death as the third-generation EGFR-TKI group [HR 0.36 (95% CI, 0.07–1.80); p = 0.214].
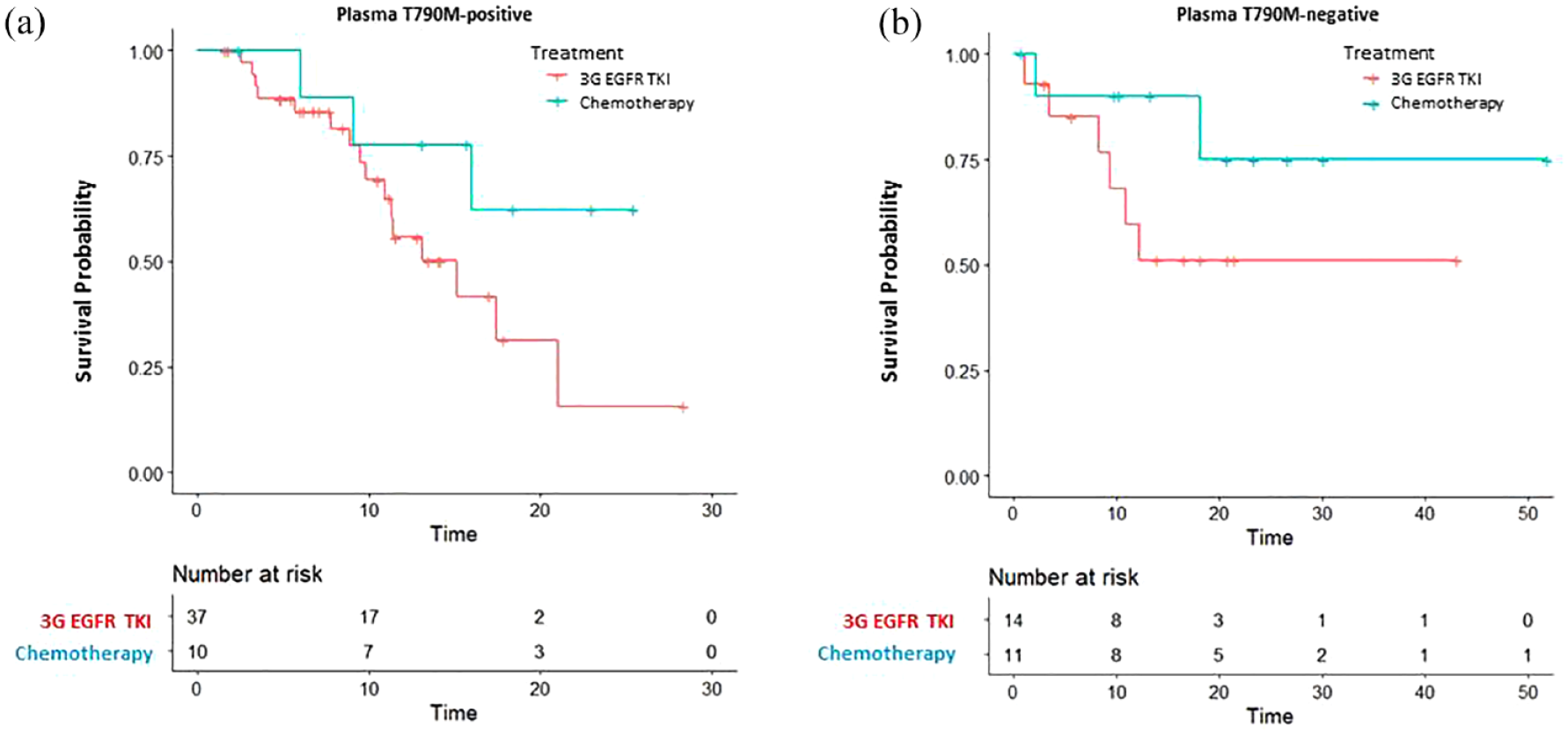
Post-progression OS for osimertinib treatment and chemotherapy in (a) plasma T790M-positive patients and (b) plasma T790M-negative patients.
Discussion
This analysis provides real-world evidence of the prognostic value of tumor and plasma T790M mutation status in a cohort of patients with advanced EGFR-mutant NSCLC who progressed from first-line treatment with gefitinib/erlotinib or afatinib. This study demonstrates that patients with T790M-positive mutation status achieved better post-progression survival, which was mainly due to the better outcomes of patients with T790M tumor-positive status. Osimertinib and chemotherapy led to similar post-progression survival in tumor T790M-positive patients. However, chemotherapy was associated with a trend toward longer post-progression survival in plasma T790M-positive patients compared to osimertinib.
In this real-world analysis, we observed that patients with unknown T790M status had the poorest post-progression survival compared to patients with either positive or negative T790M status. This result reflects the finding that the group of patients with unknown T790M status were more likely to have poor performance status and thereby more likely to be ineligible for tumor re-biopsy. Another real-world study made a similar observation that T790M testing might lead to the selection of a patient population with a better prognosis. 20 Other reasons for the unknown T790M status in this analysis included procedural failure during re-biopsy and acquisition of invalid tissue samples or insufficient sample/nucleic acid for molecular testing; these factors have been previously documented.21,22 Although liquid biopsies can be an alternative to tumor biopsy, the application of liquid biopsies varies in different countries for multiple reasons, including the additional cost 23 and the obligatory requirement of a tissue-based T790M assay to obtain reimbursement for osimertinib treatment in Taiwan.
Compared to previous studies, we observed some similarities and differences in the predictive value of tumor and plasma T790M for survival outcomes. Both the AURA I trial and our study demonstrated tumor T790M has a consistent predictive effect, with tumor T790M-positive patients having a significantly reduced risk of disease progression and a significant reduction in the risk of death compared to tumor T790M-negative patients. 16 The impact of plasma T790M status on survival was also comparable in the AURA I trial and this analysis, in that the risk of disease progression and risk of death were similar for plasma T790M-positive patients and plasma T790M-negative patients. 16 This finding is likely related to the biphasic association of prognosis in the plasma T790M-negative cohort, as patients who had T790M-positive tumors but T790M-negative plasma, that is, non-shedders, had a more favorable prognosis than patients with T790M-negative tumors. On the other hand, both tumor and plasma T790M positivity predicted a similar response to osimertinib in the AURA I study, with an objective response rate and PFS of approximately 62% and 9.7 months in both groups. By contrast, the prognosis of tumor and plasma T790M-positive patients was different in this analysis, as tumor T790M-positive patients achieved significantly longer post-progression survival.
Multiple factors may underlie the differences between our analysis and previous studies. First, in the AURA I trial, the patients with T790M-positive plasma who were evaluable for the response to osimertinib mainly had T790M-positive tumors; the tumor T790M-positive, -negative, and unknown rates in this subgroup were approximately 70%, 10%, and 20%, respectively. Therefore, one could rationally conclude that the predictive value of osimertinib defined by plasma T790M-positivity would be approximately the same as the predictive value defined by tumor T790M-positivity. In contrast to the AURA I trial, the patients who underwent tissue and liquid biopsies in this analysis were essentially different cohorts, with only 20 patients undergoing both types of biopsy. Second, the methods used to capture plasma T790M may also have an impact. The AURA I study used the Cobas EGFR mutation assay, whereas we used a high-sensitivity digital PCR assay to assess plasma T790M. 24 Hence, the cohort of plasma T790M-positive patients in our study may include a higher proportion of patients whose tumors contain minor T790M clones and thus contain patients whose tumor biopsies were more likely to be T790M negative as a result of tumor heterogeneity.
This analysis further provides practice-based evidence that may help to inform treatment decision-making relative to T790M status. Previously, the AURA III trial investigated osimertinib or platinum-doublet chemotherapy as therapeutic strategies in patients with T790M-positive tumors who progressed after treatment with an EGFR TKI. Although osimertinib led to significantly better PFS, OS was similar between the two treatment groups – mainly due to a high cross-over rate (73%) to osimertinib treatment from the chemotherapy group.12,25 In this real-world analysis, as expected, the cross-over rate to osimertinib from the chemotherapy group was somewhat lower at 50%, and the cross-over rate to platinum-doublet chemotherapy from the osimertinib group was similar to AURA III at 38%. Thus, the pattern of OS observed in this study is generally similar to the AURA III study. On the other hand, the patients with plasma T790M positivity in this analysis tended to obtain better outcomes after chemotherapy than osimertinib. Several factors may be associated with this finding. First, the digital PCR-based high-sensitivity platform used in this study was more likely to capture patients with minor T790M clones, in whom the efficacy of osimertinib treatment is less favorable. Second, the impact of patient selection associated with different treatments may also play a role in clinical practice-based settings.
PD-L1 status was determined in 54% of patients in this analysis. However, we did not identify an association between the expression of PD-L1 and post-progression OS. This finding echoes our earlier finding that the response rate and OS after front-line EGFR-TKI treatment were similar across the groups of patients with strong, weak, and negative PD-L1 expression. 26 Currently, the relationship between PD-L1 expression and the treatment outcomes of EGFR-TKIs remains controversial.27–29 In addition, although treatment of EGFR-mutant NSCLC using an immune checkpoint inhibitor had higher efficacy in patients with T790M-negative tumors than T790M-positive tumors, this treatment only demonstrated limited antitumor activity and combination with chemotherapy was usually required to achieve tumor control.30,31 Therefore, previous studies explained the finding that very few patients in this cohort received an immune checkpoint inhibitor, either as a stand-alone treatment or a part of subsequent treatment after tumor progression after front-line treatment with an EGFR-TKI.
One of the limitations of this study is its retrospective nature and thus the risk of inherent bias. In addition, we only performed T790M testing and did not conduct molecular analysis of other mechanisms of molecular resistance to front-line EGFR-TKIs, which limits further clarification of our findings.
Conclusion
The presence of T790M mutations after disease progression from first-line EGFR-TKIs is a positive predictor of post-progression survival; tumor T790M positivity has a stronger prognostic value than plasma T790M positivity. In patients with T790M-positive tumors who progress after a front-line EGFR-TKI, subsequent treatment with osimertinib or chemotherapy provides similar survival benefits.
