Abstract
Background:
Hepatic epithelioid hemangioendothelioma (EHE) is an extremely rare tumor, and no standard therapy has been established yet.
Objectives:
The aim of this study was to investigate the short-term results of combined therapy with sirolimus and interferon-alpha 2b (IFN-a 2b) (SI therapy).
Methods:
From January 2022 to April 2023, 40 patients histologically diagnosed with hepatic EHE and progressive disease received SI therapy. All patients were regularly evaluated for the safety and efficacy of the SI therapy. Patients who received SI therapy for <3 months without a tumor status evaluation after treatment were excluded.
Results:
Twenty-nine patients with hepatic EHE were included in this study. The Eastern Cooperative Oncology Group (ECOG) performance status was 0 in 19 (65.5%) patients and 1 in 10 (34.5%) patients. The median duration of the SI therapy was 8 months (range, 3–15 months). Twenty-three (79.3%) patients showed a decrease in tumor size, including 11 (37.9%) patients who achieved a partial response and one (3.4%) who achieved a complete response; the objective response rate was 41.4%. Stable disease was observed in 13 (44.8%) patients, with a disease control rate of 86.2%. Adverse events (AES) were observed in 18 patients, including leukopenia (31.0%), oral ulcers (13.8%), and liver injury (10.3%). No severe (grade ⩾ 3) AEs were recorded, and SI therapy was not interrupted for any patient due to AEs.
Conclusion:
Sirolimus and IFN-a 2b may have synergistic effects in the treatment of hepatic EHE. SI therapy is a safe and effective treatment for hepatic EHE patients with good ECOG performance status.
Introduction
Epithelioid hemangioendothelioma (EHE) is an extremely rare tumor with an overall incidence of less than one per 1,000,000 person-years. 1 Currently, no standard therapy has been established for hepatic EHE. Favorable long-term results have been reported for hepatic EHE patients who underwent radical surgery.2,3 However, according to our previous studies on the radiological characteristics of hepatic EHE, both multiple intrahepatic lesions and extrahepatic metastases are commonly detected in hepatic EHE patients, which may limit the implementation of surgical resection.4,5 Liver transplantation has also been reported to be an effective treatment for hepatic EHE, but its value has been doubted, considering the risk of post-transplantation recurrence and the indolent nature of the disease.6,7
Sirolimus was reported to be an effective therapy for EHE with a disease control rate (DCR) of more than 80%; however, the objective response rate (ORR) was only about 10%. 8 In our previous study, interferon-alpha 2b (IFN-a 2b) as immunotherapy showed favorable results in hepatic EHE patients with good physical status. 9 Nevertheless, based on our experiences, the period between the start of IFN-a 2b and tumor regression was approximately 1 year in patients with treatment responses. Moreover, the failure of IFN-a 2b monotherapy was commonly observed in patients with large intrahepatic lesions. Therefore, the combined therapy of sirolimus and IFN-a 2b (SI therapy) was conducted, and the successful result of the first hepatic EHE patient who accepted SI therapy has been reported by our group. 10 Since then, SI therapy has been suggested for hepatic EHE patients having tumor progression in our center. We conducted a retrospective study to summarize the short-term outcomes of hepatic EHE patients who underwent SI therapy.
Patients and methods
Since January 2022, SI therapy has been suggested for hepatic EHE patients with tumor progression. IFN-a 2b was administered as we previously reported (3 million units, subcutaneous injection, once every other day). 9 Sirolimus was started at a dosage of 2 mg/day (once daily), and the drug was taken at the same time and in the same conditions (i.e. fasting or after a meal). The plasma level of sirolimus was checked 1 month after the start of treatment and once every 3 months. The dose of sirolimus was adjusted according to patients’ tolerability and the occurrence of adverse events (AEs). Target plasma sirolimus levels were not used. The SI therapy was withheld for Eastern Cooperative Oncology Group (ECOG) performance status ⩾3 and grade 3 AEs, as defined according to the National Cancer Institute Common Toxicity Criteria 3.0. The treatment was restarted after recovery to ECOG ⩽2, and grade ⩽2 AEs, respectively.
Blood counts and biochemistry were evaluated at baseline, at 1 month, and then every 3 months throughout the SI therapy period. Intrahepatic tumor status was assessed at baseline using computed tomography (CT) or magnetic resonance imaging (MRI). The tumor was reevaluated every 3 months by CT or MRI, and the status was assessed according to the Response Evaluation Criteria in Solid Tumors Committee (RECIST) 1.1. 11 DCR was defined as complete response (CR) + partial response (PR) + stable disease (SD) at 6 months, and ORR was defined as RECIST CR + PR. All hepatic EHE patients who received SI therapy were retrospectively investigated. Patients who had received the SI therapy for less than 3 months without a tumor status evaluation after treatment were excluded. Before starting the SI therapy, all patients signed consent forms for treatment and data collection. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental File 1).
Results
Between January 2022 and April 2023, 40 patients histologically diagnosed with hepatic EHE received SI therapy. Six patients with no result of tumor status evaluation were excluded since SI therapy just started and the period was less than 3 months. Three patients had interrupted SI therapy within 3 months due to the deterioration of ECOG performance status, and the treatment has not recovered since then. Two patients chose to discontinue SI therapy within 3 months and were lost to follow-up. Finally, 29 patients were included in this study, and their clinical data were retrospectively analyzed (Figure 1).
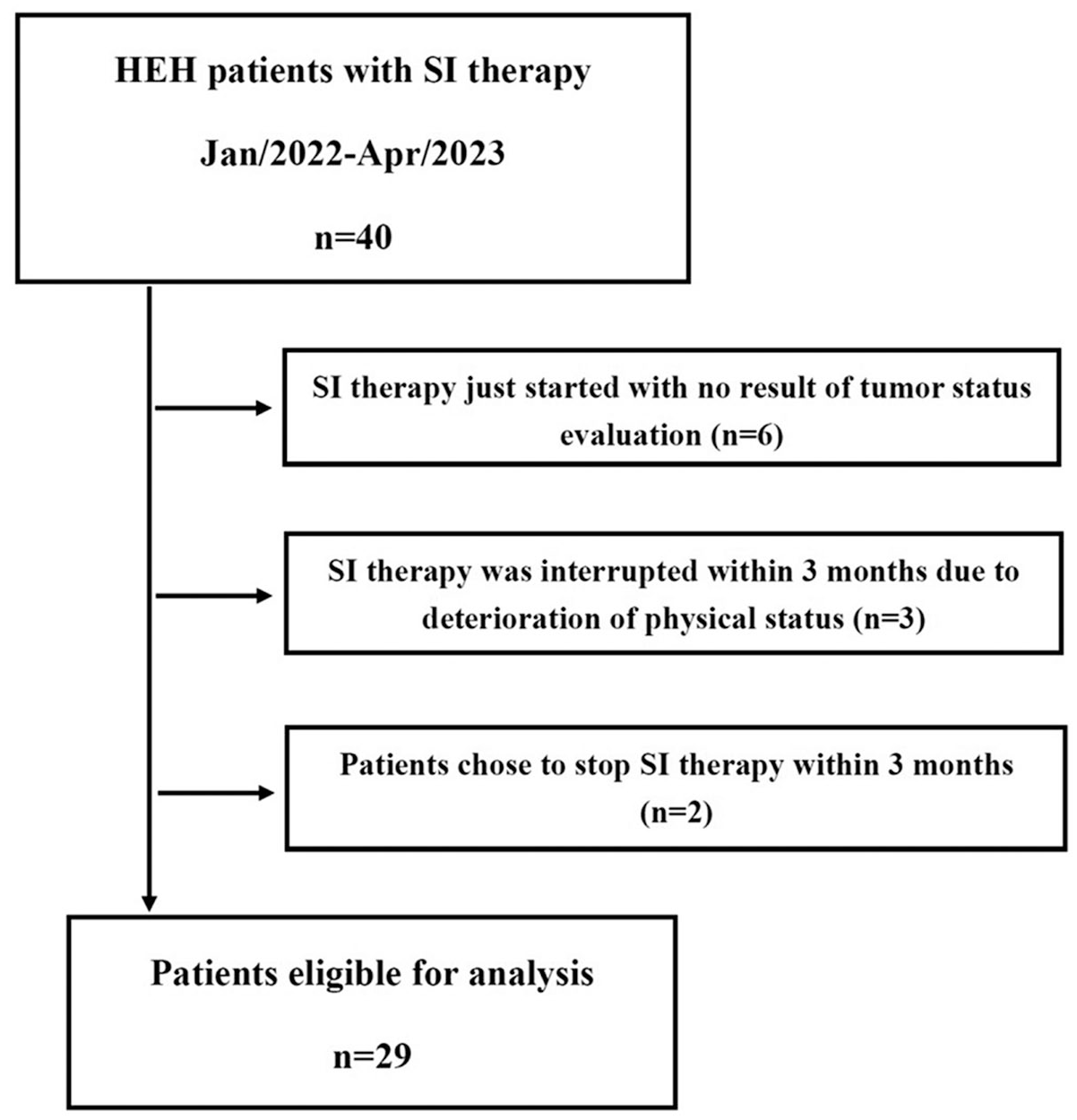
Patient flow chart for inclusion.
Patients
The baseline demographic characteristics of the patients are summarized in Table 1. At baseline, extrahepatic metastases were detected in 14 (48.3%) patients. Eleven patients had metastases at one site (including eight patients in the lungs, one in the soft tissue, one in the spleen, and one in the peritoneum), and three patients had metastases in both the lung and bone. The median time between diagnosis and initiation of SI therapy was 9 months (range, 1–50 months).
Baseline demographic and clinical characteristics of HEH patients with SI therapy (N = 29).

ECOG PS, eastern cooperative oncology group performance status; HEH, hepatic epithelioid hemangioendothelioma; SI, sirolimus and interferon-alpha 2b.
At baseline, 12 patients had progressive disease (PD) with past medical therapies, including IFN-a 2b in seven patients, chemotherapy in three patients, anlotinib in one patient, and sirolimus in one patient. Four patients had intrahepatic recurrences after surgery, including three who underwent radical liver resection and one who underwent liver transplantation. Intrahepatic lesions grew larger, or new intrahepatic lesions occurred in seven patients during observation without any treatment. Six patients with multiple intrahepatic lesions started SI therapy immediately after diagnosis. The detailed demographic characteristics and previous treatments of each patient are presented in Supplemental File 2.
Response
For the 29 patients, the median duration of both SI therapy and follow-up were 8 months (range, 3–15 months). Twenty-three (79.3%) patients showed decrease in tumor size, including 11 (37.9%) patients who achieved PR and one (3.4%) patient who achieved CR, and the ORR was 41.4% (Figure 2). The median time from the start of SI therapy to the observation of tumor regression was 3 months (range, 3–6 months). SD was observed in 13 (44.8%) patients, including 11 with a decrease in tumor size, and the DCR was 86.2%. PD was observed in four patients (13.8%).

A 67-year-old male HEH patient who received SI therapy. (a) CT image at the time of diagnosis (Lesions were marked with black arrows). (b) CT image after 5 months of observation. Tumor progressed during observation (Lesions were marked with white arrows), and then SI therapy was started. (c) CT image after 11 months treatment of SI therapy. All the intrahepatic lesions disappeared, and complete response was achieved. The contour of liver was reshaped due to the disappearance of lesions and regeneration of normal tissue.
Among patients who achieved PR or CR, SI therapy was started in four patients with PD after monotherapy of IFN-a 2b, three patients with tumor progression after observation, one patient with PD after the treatment of sirolimus, one patient with PD after chemotherapy, and one patient with recurrence after surgery (Figure 3). Two patients with PR initiated SI therapy immediately after the histological diagnosis. SI therapy was maintained in all patients with PR and SD. Four patients with PD received chemotherapy (nab-paclitaxel + sirolimus) after termination of SI therapy. All 29 patients survived the study period. Radiological images of all patients at baseline and at the time of best response are provided in Supplemental File 3.

A 46-year-old female HEH patient who received SI therapy. (a) MRI image at the time of diagnosis (Lesions were marked with black arrows). (b) MRI image after 6 months treatment of IFN-a 2b. Tumor progressed (Lesions were marked with white arrows) during IFN-a 2b monotherapy, and then SI therapy was started. (c) MRI image after 13 months treatment of SI therapy. Intrahepatic lesions regressed obviously (marked with yellow arrows), and partial response was achieved.
Safety of SI therapy
Sirolimus was started at a dosage of 2 mg/day for all 29 patients and was decreased in 18 patients due to AEs (eight leukopenias, four oral ulcers, two anemias, one thrombopenia, one diarrhea, one proteinuria, and one liver injury). The maintained dosage of sirolimus was 2 mg/day in 11 patients, 1.5 mg/day in 13, and 1 mg/day in 5. The median plasma level of sirolimus was 7.42 ng/mL (range, 3.25–12.73 ng/mL), and no patient adjusted the dosage of sirolimus due to plasma level. The dosage and frequency of IFN-a 2b was not adjusted for all 29 patients.
AEs were observed in 18 patients, including two in three patients and one in 15 patients. No severe (grade ⩾ 3) AEs were recorded for all 29 patients, and SI therapy was not interrupted for any patient due to AEs. All treatment-related AEs associated with SI therapy are shown in Table 2. Detailed information on drug dosage and AEs for all 29 patients is provided in Supplemental File 4.
Treatment-related adverse events with SI therapy (N = 29).

SI, sirolimus and interferon-alpha 2b.
Discussion
Hepatic EHE is an extremely rare hepatic tumor, and no standard treatment protocol has yet been established. Although liver transplantation has been reported to be effective with favorable long-term survival, the scarcity of organ donation limits the implementation of this procedure for many hepatic EHE patients. 12 Moreover, considering the risk of recurrence and the indolence of the tumor, the value of liver transplantation has been doubted in the treatment of hepatic EHE. 7 Patients with surgical resection have also been reported to achieve satisfying long-term results.13,14 However, according to our studies on the radiological characteristics of hepatic EHE, most patients were not suitable for surgical treatment due to the multiple intrahepatic lesions and extrahepatic metastases, and postoperative recurrence was common.4,5,15 The results of a retrospective study on systemic therapy for EHE showed that chemotherapy such as anthracycline and paclitaxel had limited activity in EHE. 16 Moreover, most of the studies on the systemic therapy of EHE included patients with all primary sites, such as the liver, lung, and soft tissue, instead of focusing on one specific organ.16–18 To the best of our knowledge, the EHE of different organs may have discrepant responses to the same therapy, which is why our research mainly focused on the treatment of hepatic EHE.
Sirolimus, a mammalian target of rapamycin inhibitor, has been reported to inhibit the PI3K/AKT pathway and the expression of endothelial growth factor.19,20 Stacchiotti et al. reported that EHE patients treated with sirolimus achieved favorable DCR, but the ORR was only approximately 10%. 8 Similar results have also been reported in pediatric EHE patients with sirolimus. 21 Although the reported rate of tumor regression was not satisfying, these case-series studies did provide valuable clinical evidence for the treatment choice of hepatic EHE, considering the rarity of the tumor.
Our previous study showed that hepatic EHE patients with monotherapy of IFN-a 2b achieved favorable ORR and long-term survival. 9 Although the mechanism was not clarified, innate and adaptive immune activation were speculated to be relative to the effect of IFN-a 2b treatment.22,23 The microenvironment of the liver may also facilitate the effect of IFN-a 2b, while the EHE of other sites except the liver may not respond to IFN a-2b. In our previous study, for hepatic EHE patients who achieved PR or CR with IFN-a 2b, the median time from the start of IFN-a 2b to tumor regression was 10 months, which indicated the slow reaction of this therapy. 9 Meanwhile, according to our experiences, hepatic EHE patients with large intrahepatic lesions had poor responses to monotherapy of IFN-a 2b, which explained the attempt at SI therapy. We have reported the result of our first hepatic EHE patient with SI therapy as a case report, which is the first hepatic EHE patient ever reported who achieved PR with such a large intrahepatic lesion (>15 cm). 10 Since then, SI therapy has been suggested for hepatic EHE patients who had a recurrence after surgery, tumor progression during observation, or failure of previous treatment in our center.
In this study, 29 hepatic EHE patients who received at least 3 months of SI therapy were included and analyzed. A decrease in tumor size was observed in 23 patients, of whom 12 achieved PR or CR. The ORR of patients who received SI therapy was 41.4%, which is a favorable result. In our previous study on monotherapy of IFN-a 2b for patients with PR or CR, the median period between the start of treatment and observation of tumor regression was 10 months. 10 In this study, the median time between the start of SI therapy and tumor regression was 3 months, which was shorter than IFN-a 2b monotherapy. Seven hepatic EHE patients with tumor progression of IFN-a 2b monotherapy accepted SI therapy, and six of them observed a decrease in tumor size, including four patients with PR. One patient who underwent PD with sirolimus also achieved PR after SI therapy. These results indicated the synergistic effect of sirolimus and IFN-a 2b in the treatment of hepatic EHE. Moreover, this study summarized the short-term outcomes of SI therapy with a median treatment period of only 8 months. With a prolonged period of SI therapy, more PR or CR may be achieved, as a decrease in tumor size was observed in 79.3% of the patients.
The dosage and frequency of IFN-a 2b was the same as we reported in our previous study. 10 However, the dosage of sirolimus was different from that of other studies introduced. Stacchiotti S et al. reported that sirolimus was started at 5 mg, and the dosage was adjusted to reach the target plasma level of 15–20 ng/mL. 24 In this study, sirolimus was started at a dosage of 2 mg, and the dosage was reduced in 18 (62.1%) patients owing to AEs. Leukopenia was the most common AE. No severe (grade ⩾ 3) AEs were observed for all patients, and no patients stopped the treatment due to the AEs of SI therapy. The synergistic effect of sirolimus and IFN-a 2b may be helpful to reduce the dosage of sirolimus and increase the tolerability of the treatment.
This study has two major advantages. First, owing to the rarity of hepatic EHE, most previous studies had to include patients over a long period (usually more than 10 years), when patients may experience a discrepancy in clinical practice. However, all patients in this study were included within 15 months, and consistency in clinical practice was guaranteed. Second, to increase patient inclusion, most clinical studies included EHE patients from all sites. However, EHE originating from different organs may react differently to the same treatment. Several studies have shown that the prognosis differs significantly among EHE patients with different tumor locations.1,25 Therefore, the treatment strategy should be investigated for each tumor location. In this study, we focused on hepatic EHE patients, and the results provided valuable evidence for the administration of hepatic EHE. However, the long-term survival benefits still need more time to be verified.
In conclusion, the results showed that sirolimus and IFN-a 2b may have a synergistic effect in the treatment of hepatic EHE. SI therapy is a safe and effective treatment for hepatic EHE patients with a good ECOG performance status; however, the long-term survival benefits of SI therapy still need more time to be verified.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231220509 – Supplemental material for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma
Supplemental material, sj-docx-1-tam-10.1177_17588359231220509 for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma by Xiaolei Liu, Ruiquan Zhou, Liguo Liu, Shuang Si, Shiwei Yang, Dongdong Han and Haidong Tan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231220509 – Supplemental material for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma
Supplemental material, sj-docx-2-tam-10.1177_17588359231220509 for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma by Xiaolei Liu, Ruiquan Zhou, Liguo Liu, Shuang Si, Shiwei Yang, Dongdong Han and Haidong Tan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359231220509 – Supplemental material for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma
Supplemental material, sj-docx-3-tam-10.1177_17588359231220509 for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma by Xiaolei Liu, Ruiquan Zhou, Liguo Liu, Shuang Si, Shiwei Yang, Dongdong Han and Haidong Tan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359231220509 – Supplemental material for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma
Supplemental material, sj-docx-4-tam-10.1177_17588359231220509 for Short-term outcomes of combined therapy with sirolimus and interferon-alpha 2b for advanced hepatic epithelioid hemangioendothelioma by Xiaolei Liu, Ruiquan Zhou, Liguo Liu, Shuang Si, Shiwei Yang, Dongdong Han and Haidong Tan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
