Abstract
In this article, we describe the case of a 34-year-old woman presenting a multifocal and metastatic epithelioid hemangioendothelioma (HEHE) of the liver. Under classical chemotherapy using cyclophosphamide, there was a fast tumor progression in liver and extra-hepatic metastatic sites (lungs and mediastinal lymph node). Taking into account the patient’s age and the natural history of the HEHE, our goal was to try to bring her to liver transplantation (LT) and lenvatinib was an acceptable candidate for this reason. Shortly after the initiation of lenvatinib before LT and surgery, we observed the enlargement of large devascularized necrotic areas in most of the liver HEHE masses, suggesting a good response. The patient was finally transplanted 6 months after initiation of lenvatinib treatment. Eight months after LT, progression occurred (ascites, peritoneal recurrence, and mediastinal lymph node). After restarting lenvatinib, ascites disappeared and the lymph node decreased in size, suggesting a good response, more than 1 year after her transplantation. This is the first case report to our knowledge that illustrates the benefit of lenvatinib as a neoadjuvant bridge until LT for a multifocal and metastatic HEHE. In addition, this drug has also shown a benefit in term of disease control after a late recurrence of the tumor. We suggest that lenvatinib should be proposed as a bridge to the LT for nonresectable HEHE. Moreover, this drug was also beneficial in the treatment of late recurrence after LT. The absence of pharmacologic interactions between classical immunosuppressive drugs and lenvatinib may allow its use as an early adjuvant approach when the risk of recurrence is high. The strength of our case consists in the long follow-up and the innovative message allowing changing palliative strategies into curative ones in case of advanced HEHE.
We report here on the case of a 34-year-old woman with no particular medical history. In July 2018, she presented with severe asthenia and nonspecific abdominal pain. A computed tomography (CT) scan revealed multiple liver nodules, a large anterior mediastinal lymph node, and several infra-centimetric pulmonary nodules. Liver tests and standard digestive tumor markers (i.e. CEA, AFP, and CA 19-9) were within normal range. In the absence of any known, underlying chronic liver disease and the lack of specificity of imaging, a guided biopsy of the liver nodules was performed. It showed typical histological features of hepatic epithelioid hemangioendothelioma (HEHE) with bulky and fusiform tumor cells that strongly expressed eryhroblast transformation-specific related gene (ERG), cluster of differenciation 31 (CD31), and calmodulin-binding transcription actIvator 1 (CAMTA1) (not shown). Of note was the fact that the tumor cells were negative for programmed death-ligand 1 (PD-L1).
Liver transplantation (LT) and surgery were not proposed at that time due to the large size and multiplicity of the liver masses and the synchronous lung and mediastinal lymph node metastases.1–4 Despite very few data in the literature,5–8 an initial therapeutic approach combining systemic anti-angiogenic treatment with bevacizumab and radio-embolization targeting almost all the liver nodules was proposed, but was finally refused by the patient. In April 2019, tumor progression justified systemic chemotherapy with cyclophosphamide that was well tolerated. However, after 4 months under chemotherapy, imaging revealed rapid growth of all liver nodules and of the metastases in the lung and anterior mediastinal lymph node (Figure 1(a1)).
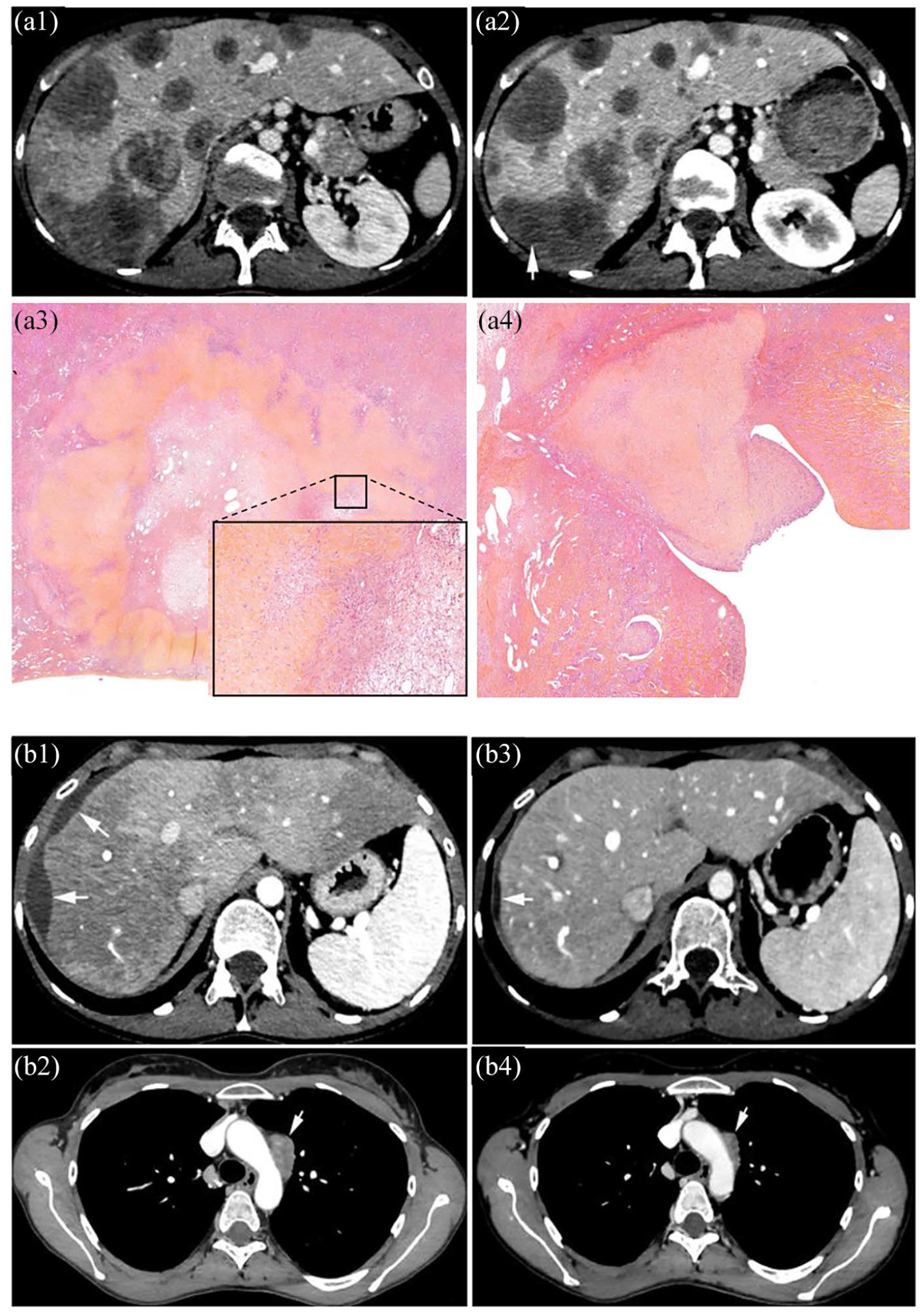
HEHE in a 34-year-old woman. HEHE after the first line of chemotherapy with cyclophosphamide. (a1) Contrast-enhanced CT shows multiple liver lesions that coalesce in the right liver. Stable disease obtained after the second line of chemotherapy using lenvatinib. (a2) Contrast-enhanced CT shows liver lesions of the same size, with some intra-tumor necrotic areas (arrow). (a3) Histological analysis of native liver revealed necrotic changes to a subcapsular nodule – HES original magnification ×10 and (a4) tumor thromboses of spigelian veins protruding into the lumen of the vena cava – Hematoxyline and eosine (HES) original magnification ×17. (b1) Contrast-enhanced CT performed 8 months after LT shows tumor scalloping of the liver surface (arrows) with ascites and (b2) an increase in size of the anterior mediastinal lymph node (arrow). (b3) More than 1 year after LT and 4 months under lenvatinib, contrast-enhanced CT shows slight perihepatic ascites and (b4) a reduction in size of the mediastinal lymph node (arrow).
In view of this progression, another systemic anti-angiogenic treatment with lenvatinib was started in September 2019, the aim being to achieve a significant tumor response or at least disease control, so that, the patient could be advanced to LT. 9 For this purpose, lenvatinib was initiated at 8 mg per day, which was increased to 12 mg per day after 2 weeks and finally to 16 mg per day as a function of tolerance, which remained excellent throughout follow-up. 9 At that time, immunotherapy was not a therapeutic option due to the absence of PD-L1 expression (not shown).10,11
After 3 weeks of this treatment, the patient presented with severe abdominal pain in the right hypochondrium. Liver tests displayed only slight changes (i.e. GGT = 196 IU/l, AST = 46 IU/l, ALT = 47 IU/l, and bilirubin = 10 μmol/l). An abdominal CT scan revealed stability in terms of the number and size of liver nodules, but a significant increase in hypodense and hypovascular areas within these nodules which was suggestive of early necrotic changes (Figure 1(a2)). On the lung CT scan, infra-centimetric pulmonary nodules were stable in number and size, without new lesions. In addition, 18 F-fluorodeoxyglucose (F-FDG) uptake was evidenced in the mediastinal lymph node (SUVmax: 5.8) and different liver lesions (SUVmax: 5.3) but not in the lung nodules at that time. Lenvatinib was not discontinued because abdominal pain rapidly improved under a simple analgesic treatment without any complication and because of the tumor response.
Given the unusual natural history of this tumor and its prolonged stability under lenvatinib after months of progression under other chemotherapy, the patient was then screened for LT. The French Biomedicines Agency (ABM) finally accepted her prioritization at 6 months, dependent on tumor control under treatment until LT. The rare safety information available in the setting of surgery recommended discontinuing lenvatinib for at least 6 days prior to scheduled surgery, which corresponds to five times the half-life of the drug (i.e. 28 hours). To reduce the risk of complications attached to the anti-angiogenic treatment in terms of healing9,12 and digestive fistula after surgery 13 and at the same time to maintain tumor control until LT, we withdrew lenvatinib 2 weeks before LT was scheduled.13,14 Lenvatinib was therefore stopped on 1 January and the patient was successfully transplanted on 17 January. LT was made from a deceased donor. Neoadjuvant treatment with lenvatinib was performed for overall 4 months before LT. No immediate or late surgical complications were observed after LT and the patient left the hospital 15 days after the procedure.
Histology of the native liver revealed a multitude of confluent nodules of 0.6–9.5 cm in diameter, with large necrotic and fibro-edematous changes affecting approximately 40% of total tumor volume (Figure 1(a3)), as well as numerous macro/microvascular and lymphatic embolisms (Figure 1(a4)). The histological analysis revealed metastatic lymph node involvement by ERG and CAMTA1 expressing epithelioid cells. However, there was no suggestive image of hilar lymph node involvement before LT.
In the absence of tumor progression at the other sites of HEHE (i.e. lung and lymph nodes), lenvatinib was not restarted after LT despite the pejorative aspect of the histological lesions evidenced on the explanted liver (see above).
During initial follow-up, clinical examinations and imaging were performed at 1, 3, and 5 months post-LT. The CT scan did not reveal any liver recurrence and a showed complete stability in terms of the size and number of pulmonary and anterior mediastinal lymph node metastases. The positron emission tomography (PET) scan performed in May 2020 (i.e. 5 months after LT) confirmed the absence of liver recurrence and the stability of the mediastinal lymph node with stable 18F-FDG uptake. In addition, the infra-centimetric pulmonary nodules were not hypermetabolic. After discussions during multidisciplinary meetings, lenvatinib was not restarted in the absence of liver recurrence or progression of the extra-hepatic tumors.
However, contrary to this initial favorable development, imaging performed 8 months after LT (i.e. August 2020) once again revealed significant tumor progression. A CT scan showed the occurrence of localized perihepatic ascites with a liver imprint strongly suggestive of peritoneal recurrence (Figure 1(b1)), with an increase in the size of the anterior mediastinal lymph node (i.e. 19 versus 14 mm) (Figure 1(b2)). The PET-CT scan demonstrated metabolic activity of the anterior mediastinal lymph node (SUVmax: 4.8). After multidisciplinary discussions, lenvatinib was restarted at that time (8 mg per day) and immunosuppression (IS) was adjusted, with mycophenolate being withdrawn and a switch toward everolimus plus tacrolimus.
After 2 months under lenvatinib (October 2020), imaging revealed complete stability in terms of the size and number of the infra-centimetric pulmonary metastases and mediastinal lymph node that remained hypermetabolic but with a significant decrease in the FDG uptake (SUVmax: 2.7 versus 4.8 in August 2020). More importantly, on a CT scan, perihepatic ascites and the liver imprint had almost completely disappeared, suggesting an early and partial tumor response under treatment (Figure 1(b3)). After 4 months of lenvatinib therapy (December 2020), a PET scan showed stable FDG uptake in the anterior mediastinal lymph node (SUVmax: 2.6 versus 2.7 in October 2020) and a CT scan revealed a significant reduction in size of this nodule (9 versus 19 mm in August 2020) (Figure 1(b4)). Liver magnetic resonance imaging (MRI) confirmed the complete disappearance of perihepatic ascites and of the peritoneal recurrence, thus demonstrating complete disease control 6 months after the resumption of lenvatinib and more than 1 year after LT (February 2021).
Discussion
To our knowledge, this is the first case report to have illustrated the benefits of lenvatinib as a neoadjuvant bridge until LT in the context of multifocal and metastatic HEHE. In addition, this drug also showed its benefits in terms of disease control following a late recurrence of the tumor.
Hepatic HEHE is considered as a low-grade vascular sarcoma originating from endothelial cells and associated with variable malignant potential. It is a very rare tumor, with an incidence of 1/1,000,000. 15 It mainly affects patients between the ages of 30 and 40 years.16,17 The molecular mechanisms underlying its development remain largely unknown. HEHE cells strongly express endothelial markers, such as CD31, CD34, and factor VIII. The expression of nuclear CAMTA1 in approximately 85%–90% of cases is currently the main immunohistochemical diagnostic test for HEHE, which may be localized or multifocal and metastatic.1,4,5,10
Liver resection is the first choice among curative treatments for HEHE.5,15,18 However, in a majority of patients, an oncologic resection is not possible due to the multicentricity of the lesions. It should be noted that unresectable HEHE, with or without a few extra-hepatic metastases, does not represent a systematic contraindication to LT. The HEHE recurrence rate is about 30% after hepatectomy and about 40% after LT. LT may therefore be considered at an early stage in these patients. Long-term survival is good and is much better than for hepatocellular carcinoma (HCC); indeed, overall survival may reach 92% at 5 years after LT.19,20 An analysis of data on 149 transplant recipients with HEHE in the European Liver Transplant Registry showed that the presence of macrovascular invasion, a pre-LT waiting time of 120 days or less, and the presence of hepatic hilar lymph nodes (but not of pre-LT extra-hepatic disease) were significant risk factors for HEHE recurrence after LT.18,21 These findings were confirmed by a propensity score analysis and enabled the development of a HEHE-LT prognostic score. Patients with a score of 2 or lower had much better 5-year disease-free survival than those with a score of 6 or higher.18,21–23
Unexpectedly, some cases of multifocal HEHE display an indolent course with stable disease for years; 7 they may benefit from a watch-and-wait attitude or active systemic therapy, when possible, in clinical trials. There is some evidence in the literature to support the benefits of some chemotherapies, such as paclitaxel, and of anti-angiogenesis targeted therapies such as bevacizumab or oral tyrosine kinase inhibitors (TKI) (sorafenib, sunitinib, pazopanib).6,24–26 However, no systemic chemotherapy has yet been definitively validated for HEHE in first-line use or beyond.27,28 Sorafenib and sirolimus have been assessed in a few patients, with marginal responses.29,30 Bevacizumab has been shown to be an effective and well-tolerated treatment for some metastatic or locally advanced angiosarcomas and has been proposed for epithelioid hemangioendotheliomas.2,6 However, no treatment (including bevacizumab) has been validated as a neoadjuvant for HEHE before surgery or LT, in view of the high risk of recurrence. In addition, surgical complications, such as fistulae, gastrointestinal perforations, and delayed surgical healing have been reported with bevacizumab, suggesting that it is not the best candidate for this purpose.31–33
Lenvatinib is an oral TKI that targets vascular endothelial growth factor (VEGFR), fibroblast growth factor receptor (FGFR), and platelet-derived growth factor receptor (PDGFR), as well as the receptor tyrosine kinase (RET) and receptor tyrosine kinase (KIT) pathways.34–36 Lenvatinib displays clinical activity and acceptable toxicity profiles in patients with advanced thyroid and colorectal cancers or those with advanced HCC. 37 Early dose modification was necessary at 8–12 mg per day in patients with a lower body weight during a phase II trial for HCC, but the safety and tolerability profiles of lenvatinib were confirmed in a phase III trial. 38
To our knowledge, lenvatinib has never previously been reported as being beneficial in HEHE and even less as a bridge to LT. Before LT, we had no macrovascular invasion and no hepatic hilar lymph nodes, so that, the HEHE–LT score was 3. After histological examination we noticed that the lymph nodes removed during LT were metastatic, so that, the HEHE–LT score after LT was 5 (HEHE score = 5 × pathological macrovascular invasion + 3 × waiting time until LT ⩽ 120 days + 2 × pathological invasion of hilar lymph nodes). Based on these criteria, no surgery or LT could be initially considered. Under classic chemotherapy using cyclophosphamide, there was rapid tumor progression in and liver and at extra-hepatic metastatic sites. In view of the patient’s age and the natural history of the HEHE, our goal was to try to bring the patient to LT for a definitively nonresectable disease using a treatment with good efficacy and tolerance in this setting and a reduced risk of complications after surgery. Lenvatinib was therefore an acceptable candidate. Bevacizumab could not be a therapeutic alternative given the impossibility of scheduling the date of LT and the high risk of severe AEs (i.e. hypertension, bleeding), including during the postoperative period. 39 The other question concerned the timing of the withdrawal of lenvatinib treatment before LT. 6 In light of the pharmacokinetics of lenvatinib, we proposed stopping lenvatinib 15 days before the scheduled time for LT after prioritization by the French Biomedicines Agency, and this approach was safe. 9
Shortly after the initiation of lenvatinib before LT, we observed an enlargement of large devascularized necrotic areas in most of the liver HEHE masses. These results suggested that modified RECIST (mRECIST) or the European Association for the Study of the Liver (EASL) criteria based on the concept of viable tumor might be of value to assessing the response of HEHE to lenvatinib. 40
Another point concerns the use of lenvatinib as an adjuvant treatment after LT in the context of a very high risk of recurrence based on the history of the disease and/or histological findings. In the present case, we only restarted lenvatinib when there were imaging features of tumor recurrence and progression at the different sites (ascites, peritoneal recurrence, and mediastinal lymph node) that occurred 8 months after LT. Should we have reintroduced it sooner after LT as an adjuvant treatment? The answer is probably yes, in order to maintain disease control alongside IS. Case reports concerning other very rare liver tumors such as fibrolamellar HCC have suggested starting adjuvant chemotherapy as soon as possible after surgery.41–43 Of course, no such recommendation has been yet validated for lenvatinib. In terms of safety, no serious adverse events have occurred under lenvatinib combined with everolimus (no hand–foot syndrome, hypertension, or edema), and notably, no interactions have been reported between lenvatinib, tacrolimus, and other IS drugs.19,44–46 Furthermore, lenvatinib levels remained within the therapeutic range (20.6 ng/ml) after LT. 9
In conclusion, our case report showed that lenvatinib should be proposed as a bridge to LT in the case of nonresectable HEHE. Moreover, this drug was also seen to be beneficial in the treatment of a late recurrence after LT. The absence of pharmacologic interactions between classic immunosuppressive drugs and lenvatinib could also permit its use under an early adjuvant approach when the risk of recurrence is high. Finally, we propose a simple decision-making algorithm for the management of unresectable HEHE based on this original report and recent data in the literature concerning LT and HEHE (Figure 2).

Proposed decisional algorithm for the management of HEHE based on this case report.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient’s consent
The patient gave permission for the publication of the manuscript. The revised form of the manuscript was shown to the patient before publication.
