Abstract
Savolitinib is a highly selective mesenchymal-epithelial transition (MET) tyrosine kinase inhibitor (TKI). Based on its significant efficacy shown in clinical studies, savolitinib was conditionally approved for marketing in China on 22 June 2021, for the treatment of advanced non-small cell lung cancer (NSCLC) with MET 14 exon skipping mutation. Additionally, many studies showed that MET TKIs were equally effective in patients with advanced solid tumors with MET gene amplification or MET protein overexpression. Several relevant registered clinical studies are in progress. The most common adverse reactions (ARs) due to savolitinib administration are nausea, vomiting, peripheral edema, pyrexia, and hepatotoxicity. This consensus was developed through two rounds of extensive national surveys involving multidisciplinary experts in China, aiming to guide clinicians to prevent and treat various ARs scientifically, and improve the efficacy of the drug and the quality of life of patients.
Introduction
Savolitinib is the first approved small molecule inhibitor that specifically targets mesenchymal-epithelial transition (MET) tyrosine kinase in China, which is used in adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with MET 14 exon-skipping mutation who have disease progression after platinum-based chemotherapy or are intolerant to standard platinum-based chemotherapy. 1 Previous studies showed that savolitinib monotherapy for locally advanced or metastatic NSCLC with MET 14 exon skipping mutations resulted in an objective response rate of 49.2%, disease control rate of 93.4%, and median overall survival time of 12.5 months after a median follow-up of 17.6 months, with manageable safety. 2 Several studies are currently exploring the efficacy and safety of savolitinib monotherapy and its combinatory regimens for various solid tumors with MET abnormalities.3–6 With the increasing application of savolitinib, its adverse reactions (ARs) have become a concern to clinicians and patients. The data regarding the safety of savolitinib administration were derived from five clinical studies,2,4,6–8 including a total of 345 oncology patients treated with savolitinib monotherapy. Based on the safety data of savolitinib and the actual needs of clinical physicians in the preliminary survey, this consensus was prepared under the guidance of multidisciplinary experts using evidence-based information and clinical experience to propose standardized management for adverse drug reactions (ADRs), such as nausea, vomiting, peripheral edema, hepatotoxicity, pyrexia, and allergic reactions for clinicians during savolitinib administration.
Nausea and vomiting
In patients treated with ⩾400 mg of savolitinib (n = 338), the incidence of nausea and vomiting was 44.7% and 31.1%, respectively, and the grades ⩾3 were 1.5%. 9
Clinical diagnosis and differential diagnosis
Savolitinib-induced nausea and vomiting should be distinguished from those caused by other factors, including (1) Chemotherapy-related factors: such as high-dose chemotherapy, combination of multiple chemotherapeutic agents, faster intravenous infusion rate, and administration of multiple cycles of chemotherapy; (2) Past medical history, such as anxiety disorder, vestibular dysfunction, history of travel sickness, history of pregnancy sickness, history of alcohol consumption, history of gastropathy, history of constipation, and history of severe nausea and vomiting caused by prior chemotherapy; (3) Comorbidities, such as incomplete or complete intestinal obstruction, electrolyte disturbance, uremia, abnormal liver function, and ongoing use of opioids; (4) Tumor-related factors, such as progression of tumor brain metastasis or radiation-induced brain necrosis. 10
Grading criteria
Nausea is characterized by a queasy sensation and/or the urge to vomit, while vomiting is characterized by the reflexive act of ejecting the contents of the stomach through the mouth. According to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, the grading criteria for nausea and vomiting are shown in Table 1.
Grading criteria for nausea and vomiting.

–, No data available.
Preventive measures
Patients and their families should be informed of possible risks of nausea and vomiting before treatment with savolitinib. Nausea and vomiting should mainly be prevented during treatment by having small but frequent meals, chewing slowly, selecting easily digestible food, avoiding spicy and cold or overheated food, and walking at a moderate pace after meals. In addition to lifestyle interventions, antiemetics may be used for prophylaxis in patients with high-risk factors such as prior history of nausea and vomiting, or medication history of other emetogenic drugs.
Management measures
General principles: Mild nausea and vomiting can be alleviated by eating. Physicians may use antiemetics or traditional Chinese medicine based on clinical judgment.
Dose adjustment of savolitinib: For grades 1–2 nausea and vomiting, savolitinib administration can be continued. For grades 3–4, savolitinib should be interrupted until recovery to grade 1 or below and then resumed at the next lower dosage level, otherwise, permanent discontinuation is necessary (Figure 1).
Interventions: Symptomatic treatment with 5-hydroxytryptamine type 3 (5-HT3) receptor antagonists is recommended for patients who do not use prophylactic drugs. If prophylaxis or treatment with 5-HT3 receptor antagonists fails, the following adjustments can be considered: doublet regimen of 5-HT3 receptor antagonist plus dexamethasone (1–2 weeks appropriately); addition of NK-1 receptor antagonist to the above doublet regimen; triplet regimen containing olanzapine instead of NK-1 receptor antagonist, or triplet regimen containing thalidomide instead of olanzapine; quadruple regimen containing NK-1 receptor antagonist and olanzapine; adding drugs with other mechanisms of action; adjusting dosage/frequency/type of 5-HT3 receptor antagonists; adding anxiolytics or acid suppressants.10,11 The process is shown in Figure 2.

Management process of nausea and vomiting associated with savolitinib.

Drug used for managing nausea and vomiting associated with savolitinib.
Peripheral edema
Savolitinib-induced edema is mainly peripheral edema, characterized by fluid retention in peripheral tissues, such as the face and limbs. In patients treated with ⩾400 mg of savolitinib (n = 338), the incidence of any grade edema was 40.5%, and grades ⩾3 occurred in 3.8%. 9 The median time from medication initiation to the onset of edema was 50 days; grade 3 edema events with an incidence of >1.5% only included peripheral edema (2.4%), and no edema-related deaths were reported. 9
Clinical diagnosis and differential diagnosis
Savolitinib-induced peripheral edema should be considered in patients who develop or experience a significant worsening of peripheral edema after savolitinib treatment. Therefore, the diagnosis should rely on the medication history and clinical symptoms temporally associated with savolitinib. 12 Concomitant conditions that can cause edema should be excluded to determine the etiology.13,14
Peripheral edema is difficult to distinguish in clinical practice due to its complex etiology and various differential diagnoses. 15 Common causes include cardiac, renal, liver, endocrine disorders, and deep vein thrombosis.
Edema caused by cardiac disorders generally spreads from the lower limbs to the entire body and is often accompanied by jugular vein distention, hepatomegaly, ascites, and pulmonary moist rales. Clinical manifestations are palpitation, shortness of breath, inability to lie on the back at night, and paroxysmal dyspnea. Significant increases in brain natriuretic peptide and high-sensitivity troponin are seen on laboratory tests. Additionally, electrocardiography (ECG) and echocardiography are helpful in the differential diagnosis.
Edema caused by renal disorders may be generalized or limited to the eyelids, often accompanied by hematuria, proteinuria, impaired renal function, and hypertension. Nephrotic syndrome and chronic nephritis may cause edema in bilateral lower limbs, eyelids, or even the whole body, with varying severity. Urinalysis, serum albumin determination, and creatinine tests can facilitate the differential diagnosis.
Edema caused by hepatic disorders develops slowly and can affect the whole body, with ascites and edema of lower limb edema being most noticeable. In severe cases, the face may appear yellowish and emaciated, while the abdomen is distended, and lower limbs present swelling, creating a clear contrast. Clinical manifestations may include hepatic impairment, decreased serum albumin, increased globulin, and may be accompanied by jaundice and splenomegaly. Gastroscopy or barium meal examination may reveal esophageal varices.
Endocrine disorders, such as hypothyroidism, may manifest non-pitting edema in lower limbs or the whole body, and may be accompanied by cold intolerance, unresponsiveness, and constipation. Thyroid function tests can facilitate the differential diagnosis. Deep vein thrombosis-related edema mainly affects a unilateral lower limb, manifesting local pain, cyanosis, superficial vein dilatation, and positive Homans sign, which can be differentiated by D-dimer testing, Doppler ultrasound, magnetic resonance imaging, and venography.
Grading criteria
The most common manifestation of peripheral edema is fluid retention in the limbs. According to the CTCAE version 5.0, the grading criteria for peripheral edema are shown in Table 2.
Grading criteria for the severity of peripheral edema.

Activities of daily living using devices: using telephone, shopping, food cooking, housekeeping, and traveling.
Basic activities of daily living of individuals: using the toilet, eating, dressing, making up, bathing, and walking.
Management measures
General principles: Patients and their families should be informed of the possible risk of edema associated with savolitinib. Peripheral edema should be monitored during treatment by measuring body weight and physical examination to facilitate early diagnosis and timely treatment, 16 reduce clinical challenges such as pain, limb swelling, paresthesia, and skin infection. Peripheral edema may be relieved by Lifestyle intervention or physical therapy may relieve peripheral edema before onset or for grades 1–2; for grade 3, pharmacological intervention may be required by the physician judgment (Figure 3).
Dose adjustment of savolitinib: Most patients treated with savolitinib experience mild to moderate peripheral edema and only a small proportion require treatment interruption or discontinuation. For grades 1–2 edema, the drug administration can be continued. For grade 3 edema, savolitinib administration should be interrupted, and when edema recovers to grade 1 or below, the drug should be resumed at the next lower dosage level, otherwise, the drug needs to be permanently discontinued (Figure 1).
Interventions: (1) Lifestyle interventions, such as low-salt and light diet and moderate exercises; (2) Physical therapies, such as elevating affected limb, wearing elastic stockings, lymphatic massage or combinations; (3) Drug intervention: such as diuretics or glucocorticoids according to clinical judgment. Glucocorticoids should be used for ⩽2 weeks. Tolvaptan may be considered for patients with peripheral edema and hyponatremia based on clinical judgment, but evidence-based medical information with a large sample size is still lacking. Additionally, it is important to identify the etiology of edema since savolitinib treatment may be one of several potential causes in patients with malignant tumors.

Management process of peripheral edema associated with savolitinib.
Severe peripheral edema may result in hypovolemia, and caution is necessary when using diuretics to prevent excessive diuresis, worsening renal hypoperfusion, and acute kidney injury. Therefore, it is recommended that daily weight loss should ⩽0.5 kg, especially in elderly patients. Potassium-sparing diuretics and osmotic diuretics should be used with caution in patients with underlying renal insufficiency. When the estimated glomerular filtration rate is <30 mL/min, loop diuretics are recommended. Electrolyte and acid–base balance should be maintained, and the dose adjusted as necessary to avoid ADRs, such as hyponatremia, hypokalemia, and hypotension. Thiazide or loop diuretics should be used with caution in patients with existing hyponatremia, hypokalemia, hypotension, and hyperuricemia. For patients with hypokalemia or a tendency to form hypokalemia, the combination treatment with potassium-sparing diuretics may be used and blood potassium levels should be monitored.
Pyrexia
In patients treated with ⩾400 mg of savolitinib (n = 338), the incidence of any grade of pyrexia was 15.7%, and ⩾grade 3 was 1.2%. 9 Fever-related events were mainly characterized by pyrexia, chills, and flu-like symptoms, with a median time from medication initiation to onset of pyrexia of 20 days, mostly grade 1–2, and no deaths reported. 9 Pyrexia is a precursor event of hepatotoxicity and allergic reactions associated with savolitinib. Although pyrexia does not necessarily indicate hepatotoxicity and allergic reactions, its development should be closely monitored to prevent hepatotoxicity and allergic reactions.
Clinical diagnosis and differential diagnosis
Savolitinib-related pyrexia is associated with the duration of drug use and has clinical characteristics including a body temperature of ⩾37.5°C but generally ⩽38.5°C at least once a day, fever lasting several weeks, negative laboratory tests for infection, no significant antipyretic effects from antibiotics and antipyretics (e.g. aspirin and paracetamol), and sometimes accompanied by decreased appetite, weight loss and significant asthenia.
Most patients treated with savolitinib who develop fever have significant lymphopenia, and C-reactive protein (CRP) level may increase while lymphocyte count decrease on the day of fever. Therefore, changes in CRP level and hematology parameters should be closely monitored during the first 9 weeks of savolitinib treatment, and pyrexia should be considered as an allergic reaction if hematology parameters (especially lymphocyte count) decrease significantly from baseline (close to or ⩾30%) with or without an increase in CRP level.
Fever is a common phenomenon in patients with various types of tumors and has multiple potential causes. Therefore, the differential diagnosis of fever is diversified. 17 Common conditions that need to be distinguished from savolitinib-related pyrexia are as follows:
Pyrexia caused by acute agranulocytosis: Acute agranulocytosis is defined as a decrease in absolute neutrophil count (<0.5 × 109/L) in a short period of time after chemotherapy, radiotherapy, and partial targeted therapy, followed by infection, fever, even hyperthermia, with a body temperature of usually > 38.5°C and accompanied by chills. One week of agranulocytosis (<0.5 × 109/L) means an almost 100% probability of infection. The most common infection is respiratory tract infection (main symptoms: cough and expectoration), followed by urinary system infection (main symptoms: frequent urination and micturition urgency). It may be treated empirically by antibiotics. If the temperature decreases and the accompanying symptoms are relieved after medication, pyrexia-mediated infection is considered.
Simple infectious fever: (1) Obstructive pneumonia may occur in patients with advanced lung cancer due to tumor enlargement that blocks the bronchi; (2) Infection may be caused by more likely invasion of pathogens in the body which retain in the lungs, gastrointestinal tract and other organs due to decreased immunity during chemotherapy. Opportunistic pathogens in the intestine may also cause intestinal infection leading to fever; (3) Indwelling catheter sites are also susceptible to infection. In the event of redness, swelling, fever and pain, the possibility of infection at such sites should be considered. The above infectious fever is not associated with savolitinib administration, so body temperature can decrease rapidly after anti-infective treatment.
Fever caused by brain metastasis: Tumor metastasis to the hypothalamus can cause fever by affecting the thermoregulatory center, resulting in sudden and persistent hyperthermia (body temperature of 40°C–41°C) with dry skin and cold limbs, but no facial flushing. Computer tomography (CT) or magnetic resonance imaging of the head is helpful in the differential diagnosis. This type of fever is rare in the clinical settings.
Tumor fever: The rapid tumor growth, large tumor burden, and endogenous pyrogens produced by tumor cells in advanced patients can trigger immune and inflammatory responses and induce fever symptoms, such as tumor necrosis factor, interleukin (IL)-6, IL-8, IL-10, and ferritin.
Fever caused by other drugs or treatments: If fever is observed in patients treated with savolitinib, concomitant use of other drugs or treatments should also be considered, such as cisplatin, interferon, interleukin, and zoledronic acid. In addition, blood transfusion, albumin infusion, etc. may also cause fever. Detailed medical history and medication history are also helpful in the differential diagnosis.
Grading criteria
According to the CTCAE version 5.0, the grading criteria for fever are shown in Table 3.
Grading criteria for fever.

°F, Degree Fahrenheit.
Management measures
General principles: Patients and their families should be informed of the risk of developing fever before treatment with savolitinib. Body temperature should be monitored regularly during treatment, and blood cell analysis should be performed in time for early diagnosis and timely adoption of appropriate treatment options and strategies. 18
Dose adjustment of savolitinib: For grades 1–2 fever, drug administration can be continued. For grades 3–4, savolitinib should be interrupted and resumed at the next lower dosage level when the fever recovers to grade 1 or below, otherwise the drug needs to be permanently discontinued (Figure 4).
Interventions: The patients should drink more water to promote drug excretion and reduce drug accumulation in the body. If body temperature is not effectively controlled, physical hypothermia or antipyretic analgesics (e.g. paracetamol or ibuprofen) may be used, an appropriate savolitinib dosage reduction should be considered, and antiallergic drugs (e.g. glucocorticoids, antihistamines) may be given prophylactically.

Management process of fever associated with savolitinib.
Hepatotoxicity
Hepatocyte toxicity is mainly indicated by increased levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP), along with hypoalbuminemia, abnormal prothrombin time and activity, and elevated total bilirubin (TBIL). In patients treated with ⩾400 mg of savolitinib (n = 338), the incidence of any grade abnormal hepatic function was 11.8%, mainly grades 1–2, and 5.6% had grades ⩾3; grades ⩾3 adverse events with an incidence of >1.5% include elevated AST (6.2%) and ALT (5.9%). 9 The median time from the drug initiation to the onset of ARs was 31 days (1–420 days). Additionally, among all hepatotoxic events, the incidence of drug-induced liver injuries (DILIs) was 1.2%, and the grades ⩾3 DILIs was 0.9%, one fatal case (0.3%) was reported. 9
Clinical diagnosis and differential diagnosis
The savolitinib-related hepatotoxicity is usually latent and may develop gastrointestinal symptoms, such as asthenia, decreased appetite, oily disgust, distending pain in the liver area, and epigastric discomfort. Significant cholestasis may be accompanied by systemic yellowing of the skin, discolored stools, and pruritus. A few patients may experience allergic manifestations, such as fever, rash, eosinophilia, joint soreness, and symptoms of extrahepatic organ damage.
To detect abnormal liver function early, it is necessary to obtain the chronic liver disease history, including gender, age, medication history (type, dosage, course of treatment, start, and end dates), hepatitis history, previous hepatotoxicity history, reaction to medication, drinking history, and travel history in epidemic areas. Further, testing for autoimmune liver disease-related antibodies and chronic hepatitis, such as hepatitis B five items, and hepatitis C antibody, along with other relevant tests, should be completed.
Preclinical studies found that savolitinib was metabolized by various liver enzymes, mainly cytochrome P450 (CYP) 1A2, CYP3A4, and CYP3A5. 9 Therefore, it is recommended to use caution with inducers or inhibitors of these enzymes during treatment with savolitinib.
Hepatic function should be monitored periodically during treatment with savolitinib. If abnormal liver function or increased levels of liver enzymes are detected compared to pre-treatment level, the following tests should also be performed in addition to the above serological tests for hepatitis virus: blood Immunoglobulin M (IgM) screening for acute hepatitis (e.g. hepatitis A and E); molecular (DNA) testing for hepatotropic viruses [e.g. cytomegalovirus (CMV) and Epstein-Barr virus (EBV)]; liver imaging tests (e.g. CT and MR), and if necessary, a series of tests, such as viral load [hepatitis B virus (HBV) DNA, hepatitis C virus (HCV) RNA] and liver biopsy.
The differential diagnosis of hepatotoxicity includes the following: (1) ARs associated with liver diseases, which can be differentiated by testing for viral loads and antibodies of hepatitis A virus, hepatitis E virus, HBV, CMV, EBV, and human parvovirus B19. (2) Alcoholic liver disease, which can be differentiated by analyzing alcohol consumption, frequency, duration (years), AST/ALT ratio, and glutamyl transferase. (3) Nonalcoholic fatty liver disease, which can be differentiated by performing body mass index, abdominal ultrasound, blood lipid measurement, and liver elastography (if necessary). (4) Autoimmune liver disease, which can be differentiated by testing antinuclear antibodies, antimitochondrial antibodies, smooth muscle antibodies, gamma globulin (especially IgG), IgG4, SP100, and GP120. (5) Cholestatic disease, which can be differentiated by conducting abdominal ultrasound, CT, MRI, and magnetic resonance cholangiopancreatography. (6) Hereditary metabolic liver disease, which can be differentiated by determining ceruloplasmin, alpha 1-antitrypsin, and ferritin. (7) Infection, which can be differentiated by hematology, CRP, erythrocyte sedimentation rate, liver imaging, and signs of systemic infection (sepsis). (8) Hemodynamic abnormalities, which can be differentiated by signs of cardiac insufficiency, blood pressure measurements, and signs of shock. (9) Vascular occlusive disease, which can be differentiated by thrombosis or phlebitis caused by various non-drug factors, compression, or invasion of extra venous lesions such as tumors.
Grading and classification criteria
According to the CTCAE version 5.0, the grading criteria for AST/ALT increases are shown in Table 4.
Grading criteria for AST/ALT Increases.

–, no data available; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ULN, upper limit of normal.
For patients with DILI, the guidelines for the diagnosis and treatment of DILIs 19 are often recommended to assess the severity of the liver injury as shown in Table 5 (Grading Criteria for DILIs), with grades 1–5 representing mild, moderate, severe, liver failures, and fatal liver injuries, respectively.
Grading criteria for DILIs.

ALP, alkaline phosphatase; ALT, alanine aminotransferase; DILI, drug-induced liver injury; INR, international normalized ratio; PTA, prothrombin time activity; ULN, upper limit of normal; TBIL, total bilirubin.
Treatment options may vary among patients with different types of DILI. The hepatic injuries can be classified according to the guidelines for the diagnosis and treatment of DILIs 19 : (1) Hepatocellular injury: ALT of ⩾3 × ULN, and R of ⩾5; (2) Cholestatic injury: ALP of ⩾2 × ULN and R of ⩽2; (3) Mixed type: ALT of ⩾3 × ULN, ALP of ⩾2 × ULN, and 2 < R < 5. 19 R is calculated as (Measured ALT/ALT ULN)/(Measured ALP/ALP ULN). Some studies have proposed a new R-value that takes the high value of ALT or AST for calculation. If ALT and ALP do not meet the above criteria, they are referred to having an abnormal liver biochemical profile.
Management measures
General principles: Liver injuries caused by savolitinib can be prevented by the following tasks: educating patients and their families about the risk of hepatic injuries caused by savolitinib, improving the management of informed consent for medication; using caution when taking drugs that may interact with savolitinib (refer to package insert in marketed countries); strengthening baseline assessment and regularly testing liver function (once a week for the first 6 weeks of treatment with savolitinib and every 3 weeks thereafter); and adhering to package inserts and guidelines.
Dose adjustment of savolitinib: Most patients improve after hepatoprotective therapy, but attention should be paid to the dose adjustment of savolitinib with reference to Table 6. 9
Interventions: Most AST and ALT increases caused by savolitinib are mild to moderate, therefore, the dose adjustment according to the package insert is enough, and no additional medication is required. For definitive DILI, the treatment principles include (1) discontinuing savolitinib promptly and avoiding the re-use of similar drugs; (2) carefully weighing the risk of primary disease progression caused by discontinuation against the risk of worsening liver injury caused by continuation; (3) selecting appropriate drug therapy based on the clinical type of DILI; (4) providing treatment under the guidance of hepatologists for severe cases such as acute/subacute liver failures (ALF/SALF), and considering additional treatments like artificial liver if necessary.
Treatment modification measures for hepatotoxicity associated with savolitinib.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; INR, international normalized ratio; TBIL, total bilirubin; ULN, upper limit of normal.
For adults with ALF/SALF, it is recommended to administer N-acetylcysteine early as early as possible. Magnesium isoglycyrrhizinate is recommended for acute hepatocellular injury or mixed DILI with markedly increased ALT. For mild to moderate hepatocellular injury and mixed DILI, bicyclol and glycyrrhizic acid preparations (diammonium glycyrrhizinate enteric-coated capsules or compound glycyrrhizin) may serve as a treatment option for patients with severe inflammation, while silymarin may be a treatment option for those with mild inflammation. Ursodeoxycholic acid or ademetionine can be selected for patients with cholestatic DILI, but high-level evidence-based medical information is needs to support this. It is not recommended to use ⩾2 hepatoprotective and anti-inflammatory drugs in combination nor prophylactic medication to reduce the incidence of DILI. 19
Hepatoprotective drugs are available in various categories based on their mechanisms of action, including anti-inflammatory and hepatoprotective drugs, cell repair drugs, detoxification hepatoprotective drugs, antioxidant drugs, and cholagogue drugs. Anti-inflammatory and hepatoprotective drugs include magnesium isoglycyrrhizinate, diammonium glycyrrhizinate, and compound ammonium glycyrrhizinate. Glycyrrhizic acid preparations are recommended for patients who have mild to moderate hepatocellular injury or mixed DILI with severe inflammation. Magnesium isoglycyrrhizinate can be used to treat patients with acute hepatocellular injury or mixed DILI with marked ALT increase. Diammonium glycyrrhizinate injection can be administered to patients who are unable to use oral glycyrrhizin preparations or those with liver failure. The representative cell repair drug is polyene phosphatidylcholine, and its injections are limited in the event of liver failure or rescue treatment but are contraindicated in children. The representative of detoxification and hepatoprotective drugs include glutathione, N-acetylcysteine, and tiopronin, which protect hepatocytes in many ways. N-acetylcysteine is recommended for adult patients with early drug-induced acute or subacute liver failure. The representative antioxidant drugs are silymarin and bicyclol, which are recommended for patients with increased ALT and AST, especially those with mild to moderate hepatocellular injury or mixed liver injury with increased ALT. Silymarin is indicated for patients with mild inflammation; bicyclol is indicated for those with more severe inflammation. The representative of choleretic drugs are ademetionine and ursodeoxycholic acid, which are recommended for cholestatic liver injury. Injections of ademetionine should not be mixed with alkaline or calcium-containing solutions, and its enteric-coated tablets must be swallowed whole and not chewed.
Allergic reactions
In patients treated with ⩾400 mg of savolitinib (n = 338), the incidence of any grade allergic reactions was 1.5% and the grades ⩾3 serious allergic reactions (including serious immediate hypersensitivity reactions) was 1.2%. 9 The median time from the medication initiation to the onset of a general allergic reaction was 15 days (10–90 days), and no relevant deaths were reported. 9 Severe immediate hypersensitivity reactions often occur within a very short time after medication administration, and may be life-threatening and must be treated immediately.
Clinical diagnosis and differential diagnosis
General allergic reactions: The diagnosis of general allergic reactions associated with savolitinib is the identification of attack forms. Common symptoms include drug-related skin allergic reactions, fever, and elevated liver enzymes. Most reactions occur within 6 weeks of medication. When a patient experiences a fever or increased liver enzymes while taking savolitinib, it is important to differentiate between other causes and manifestations of savolitinib-related allergic reactions. The rash of general allergic reactions is mainly manifested as maculopapular rash, erythema multiforme, and skin exfoliation. Clinical studies have reported that some patients who initially experienced general allergic reactions may develop severe immediate hypersensitivity reactions when resuming savolitinib after short-term discontinuation.
Antibiotics and alcohol consumption can also cause similar manifestations of allergic reactions. Differential diagnosis relies on medical history, including details of drug exposure, experiences before the onset of symptoms (e.g. exercise, eating seafood, or drinking alcohol), insect bites, and acute infectious diseases. Allergen skin test and/or determination of serum allergen-specific Immunoglobulin E (IgE) levels for suspected allergens confirms the diagnosis. Serum protease testing can distinguish between allergic reactions caused by drug injections and insect bites. 20
Severe immediate hypersensitivity reactions: Severe immediate hypersensitivity reactions are predominant type I hypersensitivity reactions, usually occurring within minutes to 1 h after the drug administration and are mainly caused by IgE-mediated release of intracellular active mediators (e.g. histamine and 5-hydroxytryptamine). Typical manifestations include attacks of bronchial asthma, laryngeal edema, anaphylactic shock, and acute urticaria. Asthma attacks are mainly characterized by expiratory dyspnea with wheezing, accompanied by polypnea, chest tightness, and cough. Laryngeal edema is a critical emergency that can cause significant hoarseness, dyspnea, and pharyngolaryngeal pain. Anaphylactic shock is the most serious ARs, with early-stage symptoms including dysphoria, pallor, cold extremities, increased respiratory rate and heart rate, and reduced pulse pressure difference. With the progression of the disease, patients may appear indifferent, unresponsive or even coma, lip cyanosis, cold sweat, increased heart rate, and significantly decreased blood pressure. In severe cases, cyanosis may present throughout the body, with undetectable blood pressure levels, and may be accompanied by metabolic acidosis and disseminated intravascular coagulation. Acute urticaria, a common accompanying symptom of anaphylactic reactions, is an edematous mass of varying size that is localized to the surface of the skin and mucosa, accompanied by pruritus and angioedema.
Management measures
General principles
Allergic reactions can be managed by educating patients and their families about allergic reactions, obtaining informed consent for medication, understanding the patient’s allergic history before administration, reminding patients to avoid allergens during the drug administration, and seeking medical attention immediately in the event of suspected allergic reactions.
Dose adjustment of savolitinib
For acute immediate hypersensitivity reaction, the medical intervention must be performed immediately and savolitinib should be permanently discontinued. When other general allergic reactions occur, intervention, dose adjustment or discontinuation may be adopted depending on the type and severity of symptoms. Specific medication modification regimens are shown in Table 7. 9
Treatment modification measures for allergic reactions associated with savolitinib.

Intervention
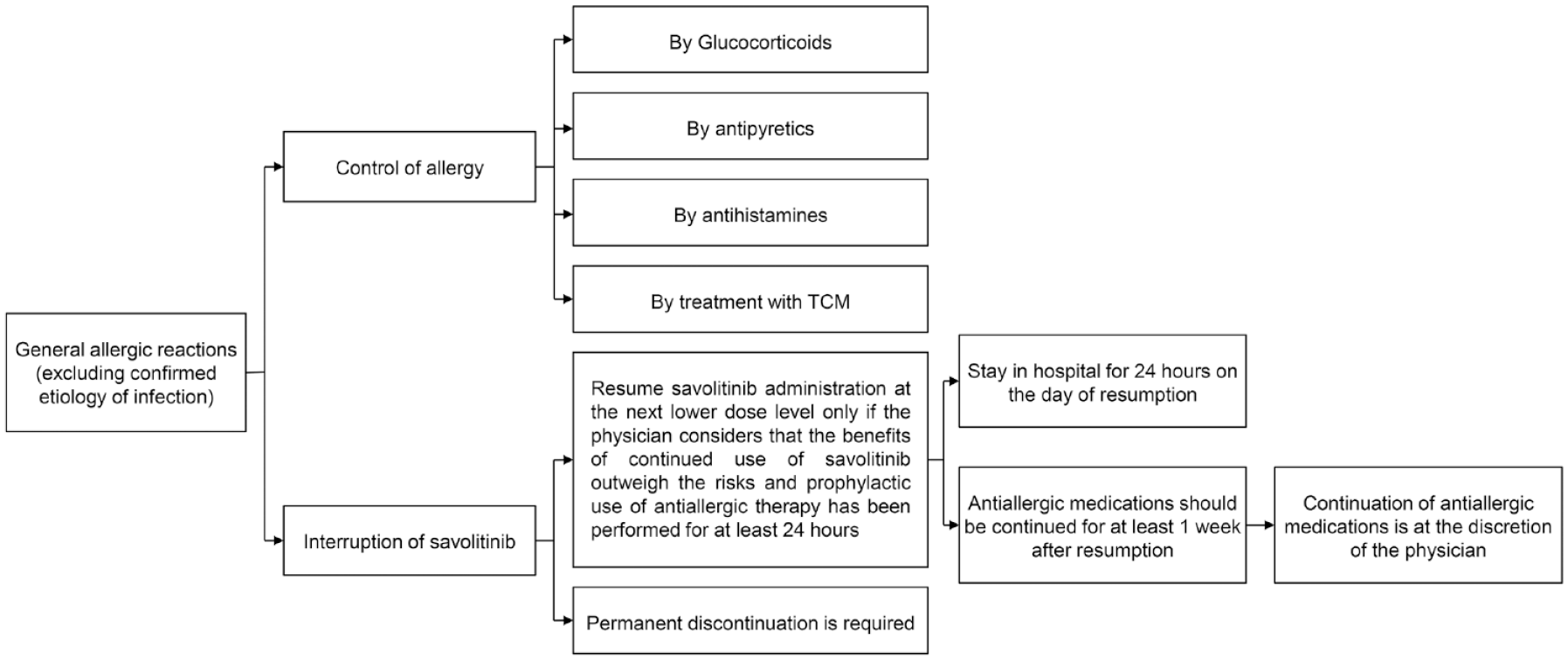
(1) General allergic reactions: For suspected general allergic reactions (excluding confirmed infection), patients should receive appropriate treatment with antihistamines, glucocorticoids, and/or antipyretics, and discontinue savolitinib. After symptom recovery, dose reduction should only be considered if the physician believes that the benefits of continued use outweigh the risks. Antiallergic drugs (e.g. glucocorticoids and antihistamines) should be initiated prophylactically and concurrently at least 24 h before resuming savolitinib. Patients must be monitored in the hospital for 24 h on the day of resuming savolitinib. Antiallergic drugs should be continued for at least 1 week after resuming savolitinib, and the physician should determine whether to continue their use. Antihistamines are the most frequently used antiallergic drugs in the clinical setting, including chlorpheniramine, diphenhydramine, promethazine, loratadine, cetirizine, and desloratadine. In cases of acute fever and multisystem involvement or associated exfoliative dermatitis, intensive treatment with glucocorticoids such as prednisone should be performed, which usually lasts for 1–3 months. The management process is shown in Figure 5.
(2) Severe immediate hypersensitivity reactions: For severe immediate hypersensitivity reactions, permanent discontinuation and timely intervention are required. Hematology, blood chemistry, blood gas analysis, coagulation, and urinalysis should be completed. Interventions include monitoring vital signs, establishing venous access, antiallergic therapy, and, if necessary, life-supporting and symptomatic treatment. Antiallergic drugs include antihistamines (e.g. diphenhydramine, loratadine) and glucocorticoids. Short-acting β2-agonists (salbutamol) can be added to patients with bronchial asthma episodes to relieve symptoms. For patients with laryngeal edema, the primary objective is to maintain airway patency and provide oxygen inhalation and mechanical ventilation, with cricothyrotomy or tracheostomy performed if necessary. Epinephrine is the drug of choice for anaphylactic shock. Rapid volume expansion, fluid replacement, maintaining water and electrolyte balance, oxygen inhalation, and ECG monitoring are also required. Second-line drugs are mainly glucocorticoids, preferably hydrocortisone succinate or methylprednisolone. Additionally, antihistamines and short-acting β2-agonists can be added depending on whether the patient experience rash, dyspnea, or asthmatic attack. Patients with severe hemolytic anemia can be transfused with washed red blood cells, combined with antihistamines, calcium supplements, and vitamin C to improve vascular permeability. The management process is shown in Figure 6.
Management process of general allergic reactions associated with savolitinib.

Management process of severe immediate hypersensitivity reactions associated with savolitinib.
Conclusion
Savolitinib is a potential treatment choice for various solid tumors with MET abnormalities. Most ADRs associated with savolitinib are grades 1–2 and can be well managed by dosage adjustment and symptomatic treatment, making its safety and tolerability acceptable. Unlike other MET inhibitors (crizotinib and tepotinib, whose main ADR leading to discontinuation is edema), the main ADRs leading to savolitinib discontinuation are hepatotoxicity and severe allergic reactions. Therefore, it is important to monitor hepatic function and identify allergic symptoms when administering savolitinib in clinical setting. Additionally, improving patient education, promoting cooperation between doctors and patients’ families, and observing patients’ signs and symptoms at any time can help identify and manage ARs, especially serious ones, as early as possible. Oncologists and physicians from other departments should also cooperate to standardize the management of possible ARs that may occur during savolitinib treatment using comprehensive prevention and treatment measures, lifestyle intervention, and drug intervention to avoid unnecessary dosage reduction or premature discontinuation, which can improve the therapeutic effect and treatment compliance and the quality of life for patients.
Members of the consensus expert group (in alphabetical order by surname)
Huang Zijing (Department of Oncology, the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine), Li Wei (Department of Dermatology, West China Hospital of Sichuan University), Li Yue (Department of Gastroenterology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences), Lin Lizhu (Department of Oncology, the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine), Lu Shun (Department of Oncology, Shanghai Chest Hospital, Shanghai Jiaotong University School of Medicine), Miao Kang (Department of Respiratory Medicine, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences), Qin Yan (Department of Nephrology, Peking Union Medical College Hospital, Chinese Academy of Chinese Academy of Medical Sciences), Si Xiaoyan(Department of Respiratory, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences), Tan Bei (Department of Gastroenterology, Peking Union Medical College Hospital, Chinese Academy of Chinese Academy of Medical Sciences), Tian Guoqing (Department of Traditional Chinese Medicine, Peking Union Medical College Hospital, Chinese Academy of Chinese Academy of Medical Sciences), Wang Yongsheng (Department of Oncology, West China Hospital of Sichuan University), Xie Yan (Department of Gastroenterology, West China Hospital of Sichuan University), Yu Yongfeng (Department of Oncology, Shanghai Chest Hospital, Shanghai Jiaotong University School of Medicine), Zhang Li (Department of Respiratory, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences), and Zhong Hui (Department of Nephrology, West China Hospital of Sichuan University).
Footnotes
Acknowledgements
This article was translated in its entirety by several coauthors, including Li Zhang (Department of Respiratory, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Beijing, China), Yongshen Wang (Department of Oncology, West China Hospital, Sichuan University, Chengdu, China), Lizhu Lin (Department of Oncology, the First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, China), Yongfeng Yu (Department Oncology, Shanghai Chest Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China) and Shun Lu (Department Oncology, Shanghai Chest Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China).
Disclaimer
This article is based on a consensus first published in the Chinese Journal of Oncology (Chinese multidisciplinary expert consensus on the management of adverse drug reactions associated with savolitinib. 2023, 45[4]: 298-312; DOI: 10.3760/cma.j.cn112152-20221226-00853). The article is translated and reprinted by permission of Chinese Medical Association. Minor revisions to the article have been made as a result of the peer-review process conducted by Therapeutic Advances in Medical Oncology.
