Abstract
Background:
The relative lack of specifically targeted agents for HER2-negative metastatic breast cancer (MBC) makes the need for new agents or combination therapies to maximize clinical benefit while reducing toxicity critical.
Objectives:
To retrospectively analyze the efficacy and safety of eribulin combined with antiangiogenic drugs in the treatment of Chinese women with HER2-negative MBC.
Methods:
A total of 85 consecutive MBC patients with HER2-negative who were treated with eribulin + antiangiogenic agents between October 2020 and April 2023 in four institutions were retrospectively included in this study. Patients received eribulin 1.4 mg/m2 (day 1 and 8) plus bevacizumab 7.5 mg/kg (day 1, 64 patients) or anlotinib 10 mg daily (day 1–14, 16 patients) or apatinib 250 mg daily (5 patients) on a 21-day cycle until progression or unacceptable toxicity. The primary end-point was progression-free survival (PFS), according to Response Evaluation Criteria in Solid tumors (RECIST) 1.1. Secondary end-points included toxicities, objective response rate (ORR), disease control rate (DCR), and overall survival (OS). Adverse events (AEs) were graded according to Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.
Results:
The study included 85 HER2-negative MBC patients, with 41 patients (48.2%) in the first to second line group and 44 patients (51.8%) in the greater than or equal to third line group. The median age was 54.0 years. Thirty patients in the first to second line group and 14 patients in the greater than or equal to third line group had triple-negative breast cancer (TNBC). The ORR and DCR were 34.1% (29/85) and 75.3% (64/85). The median PFS (mPFS) of total population was 6.0 months (95% CI: 4.3–7.7), and median OS (mOS) was immature. The mPFS was 7.7 and 4.3 months in the first to second and greater than or equal to third line treatment (p = 0.003), respectively. TNBC patients in first to second line therapy showed a significantly longer PFS (6.5 months versus 2.0 months, p = 0.021) compared to greater than or equal to third line. The incidences of cardiovascular toxicity were 29.4% in grades 1–2 and no grades 3–4. Hematologic toxicity (leukopenia and neutropenia) was the most common grade ⩾3 AEs, and AEs were more common in patients in greater than or equal to third line.
Conclusion:
The results suggest that eribulin combined with antiangiogenic therapy has a meaningful clinical activity and an acceptable safety profile in HER2-negative MBC.
Introduction
Cancer has become one of the major diseases that seriously threaten human health and cause death. According to the GLOBOCAN 2020 released by the International Agency for Research on Cancer, female breast cancer has surpassed lung cancer to become the most commonly diagnosed cancer globally. 1 In January 2023, the American Cancer Society announced that for female cancer patients, the highest prevalence of tumors is still breast cancer, accounting for about 31% of female tumors. 2 Up to now, the cause of breast cancer is still unclear, but various risk factors have been found to increase the burden of breast cancer. 3 In the past decade, from surgery, chemotherapy, endocrine therapy, targeted therapy, and immunotherapy, advanced breast cancer (ABC) treatment methods have been constantly updated – a single treatment strategy transformed into a combination treatment strategy. The survival of patients with ABC has also been significantly improved. 4
In the era of precision medicine for breast cancer, chemotherapy remains the cornerstone. Eribulin, as a new generation of non-taxanes tubulin inhibitor, can not only inhibit the growth of microtubules but also block the formation of the mitotic spindle, causing G2/M phase cell cycle arrest and apoptosis of tumor cells. 5 It can also remodel tumor blood vessels, improve the accumulation of antitumor drugs, and reverse epithelial-mesenchymal transition to delay tumor metastasis. 6 Results of the phase III 305 (EMBRACE) study 7 showed that eribulin significantly prolonged overall survival (OS) compared with the physician’s choice of treatment in patients with metastatic breast cancer (MBC) who had received two to five previous lines of therapy (13.1 m versus 10.6 m). The 1-year OS rate of the two groups was 53.9% versus 43.7%. Similarly, the objective response rate (ORR) was significantly improved with eribulin (12% versus 5%, p = 0.005), and progression-free survival (PFS) tended to be prolonged (3.7 m versus 2.2 m, p = 0.09). Eribulin was first approved in the United States in 2010 for the treatment of patients with MBC. Based on the results of a phase III 304 clinical trial evaluating the efficacy and safety of eribulin and vinorelbine in women with locally recurrent or MBC, eribulin was approved by the National Medical Products Administration of China in 2019. 8 It treats patients with locally recurrent or MBC who have received at least two prior chemotherapy regimens, including anthracyclines and taxanes. In this study, compared with vinorelbine, eribulin was associated with PFS [Hazard Ratio (HR): 0.80, 95% CI: 0.65–0.98, p = 0.036], ORR (30.7% versus 16.9%), clinical benefit rate (CBR, 38.6% versus 23.3%), and disease control rate (DCR, 49.2% versus 33.1%) were significantly improved.
In ABC, the combined treatment mode of ‘chemotherapy plus’ still plays a vital role in treating different molecular types of breast cancer. 9 Angiogenesis plays a crucial role in tumor growth and metastatic spread of breast cancer. Vascular endothelial growth factor (VEGF) is significantly important in angiogenesis, and VEGF receptors are critical regulators of the tyrosine kinase inhibitor (TKI) signaling pathway. Antiangiogenic drugs targeting VEGF therapy include monoclonal antibodies (such as bevacizumab) and small molecule TKIs (such as anlotinib and apatinib). In the E2100, 10 AVADO, 11 RIBBON-1, 12 RIBBON-2, 13 TURANDOT, 14 and ATHENA 15 trials, the addition of bevacizumab to chemotherapy could improve ORR and mPFS but did not significantly improve OS. Due to the lack of an OS benefit, bevacizumab is not FDA approved (approval withdrawn 2011) but is recommended as an option in combination with capecitabine or paclitaxel a first-line chemo option in European Society for Medical Oncology guidelines. A prospective, open-label, single-arm, multicenter phase II study showed a significant PFS/OS benefit with eribulin plus bevacizumab in the first-line treatment of human epidermal growth factor receptor 2 (HER2)-negative MBC, with a 1-year PFS rate was 32% (95% CI: 20–43), ORR was 47% (95% CI: 34–60), a mPFS was 8.3 months (95% CI: 7.0–9.6), the median OS reached 28.3 months (95% CI: 22.8–33.9), and the incidence of grade 3/4 adverse events (AEs) was low. 16
In addition, the phase III clinical trial (ALTER) 17 has confirmed that anlotinib can significantly improve the mOS (9.6 m versus 6.3 m), quality of life, and clinical outcomes of Chinese patients with advanced non-small cell lung cancer as third-line or further treatment. Several basic studies have shown that anlotinib is effective in breast cancer cell lines, but there is a lack of clinical trial results in ABC. The 2022 American Society of Clinical Oncology annual meeting presented that Empathy study involved 56 Chinese patients with ABC or MBC who had received at least two prior chemotherapies and patients were randomized to eribulin alone or eribulin plus anlotinib. It showed that eribulin plus anlotinib significantly improved DCR (100% versus 66.7%, p = 0.007) and PFS (9.7 m versus 3.7 m; HR: 0.20, 95% CI: 0.04–0.91, p = 0.04). Subgroup analysis showed that in the triple-negative breast cancer (TNBC) subgroup, the DCR of eribulin + anlotinib was still as high as 100%, and the mPFS was 3.6 months versus 9.7 months (p = 0.030). 18 Phase II study also reported that camrelizumab combination with eribulin and apatinib had good safety and efficacy (mPFS = 8.1 m) in patients with advanced TNBC. 19 Moreover, a systematic review and meta-analysis from 29 studies released that apatinib monotherapy or combined with other therapies was associated with improved DCR [pooled response rate (RR) = 1.43] and ORR (pooled RR = 1.79) in breast cancer (BC) patients. 20 Therefore, given that eribulin has a unique mechanism of inducing tumor vascular remodeling, improving tumor blood perfusion, and tissue permeability of combined antitumor drugs, we hypothesized that eribulin and antiangiogenic agents might be synergistic and their combination might be an effective regimen for MBC patients.
Thus, we designed this multicenter study to evaluate the efficacy and safety of the combination of eribulin and antiangiogenic agents in the treatment of Chinese HER2-negative MBC patients.
Patients and methods
Patients
Patients were included in the study if they had confirmed MBC and had been treated with eribulin + antiangiogenic therapy between October 2020 and April 2023. The inclusion criteria were as follows: (1) Cytological or histologically confirmed diagnosis of MBC; (2) age ⩾18 years; (3) administration of at least one cycle of eribulin + antiangiogenic therapy until disease progression, unacceptable toxicity, or other factors; (4) availability of clinical-pathological and laboratory parameters before the combination treatment; (5) availability of response evaluation and survival data; (6) availability of previous medication [whether received anthracycline/taxane-based chemotherapy or cyclin-dependent kinase 4/6 inhibitor (CDK4/6i)] in the adjuvant or metastatic setting. MBC was defined as de novo stage IV and recurrent breast cancer confirmed by clinical, imaging, histological, or cytological measures. The exclusion criteria were as follows: (1) Insufficient information on previous clinicopathological features, (2) patients for whom information regarding the first or further cycles of treatment with eribulin + antiangiogenic therapy was not available were excluded from the analysis, (3) male patients, (4) patients with HER2-positive. All data were retrospectively collected from medical records (Figure 1).

Trial profile. Patients were recruited from four hospital sites in China.
Study design and treatment
The purpose of the study was to analyze the efficacy and toxicity of eribulin + antiangiogenic therapy according to different lines of treatment. From October 2020 to April 2023, 117 patients with MBC received eribulin + antiangiogenic therapy as first or further line of treatment across four institutions, including Cancer Hospital Chinese Academy of Medical Science, China-Japan Friendly Hospital, Beijing Cancer Hospital, and Beijing Chaoyang District Sanhuan Cancer Hospital; of these, 85 patients resulted eligible for our analysis. Safety and effectiveness outcomes from patient medical records were collected irrespective of the patient’s age, or length of time of eribulin + antiangiogenic therapy treatment. Data related to treatment patterns were also collected.
Assessment variables included PFS, OS, ORR, DCR, and safety. The primary end point was PFS. Secondary end points included toxicities, ORR, DCR, and OS. AEs were graded according to CTCAE version 5.0 (U.S. Department Of Health And Human Services). PFS was defined as the time from the first eribulin treatment to disease progression or death due to various causes, according to the Response Evaluation Criteria in Solid tumors (RECIST) 1.1. OS was defined as the time from the first eribulin treatment to death from various causes or the last follow-up visit. ORR was defined as the percentage of evaluable patients at baseline who had either complete response (CR) or partial response (PR) as the best objective tumor response. DCR was defined as the percentage of evaluable patients at baseline who had CR, PR, or stable disease (SD) for ⩾24 weeks.
Patients received eribulin 1.4 mg/m2 (days 1 and 8) plus bevacizumab 7.5 mg/kg (day 1) or anlotinib 10 mg daily (days 1–14) or apatinib 250 mg daily on a 21-day cycle. The dosing was adjusted according to dose modification recommended by the FDA prescribing information, 21 AEs, or the physicians’ judgment. Treatment cycles were repeated until progressive disease or unacceptable toxicity or until the patient decided to terminate treatment.
Statistical analysis
Statistical analysis was performed using SPSS 26.0, R Studio (version 4.1.2) and Adobe Illustrator 2020 were used to construct graphics. χ2 test was used to compare the difference in qualitative data between the two groups, and t-test was used to compare the difference in quantitative data between the two groups. The Kaplan–Meier survival curve was used to analyze the mPFS and OS of the patients and the difference in survival rate between the groups. Cox proportional hazards model was used to explore the relationship between the baseline characteristics of the patients and the risk of PFS. Baseline characteristics strongly associated with survival in the clinical setting were included in univariate and multivariate analyses. p Value <0.05 was considered significant.
Results
Patients and treatment
The study included 85 HER2-negative MBC patients at four institutions, with 41 patients (48.2%) in the first to second line group and 44 patients (51.8%) in the greater than or equal to third line group. The characteristics of the 85 evaluable patients are reported in Table 1. Among them, there are 64 patients in the eribulin + bevacizumab group, 21 patients in the eribulin + TKIs (anlotinib: 16 points and apatinib: 5 points) group. The median ages were 52.0 (range, 19–68) and 58.5 (range, 33–73) years, respectively. The Estrogen receptor (ER) - or Progesterone receptor (PgR)-positive rates were 14.6% versus 65.9% (p < 0.001) and 24.4% versus 63.6% (p < 0.001) for the first to second line group and greater than or equal to third line group, respectively. Thirty patients (73.2%) in the first to second line group and 14 patients (31.8%) in the greater than or equal to third line group were TNBC. Fifty patients in the greater than or equal to third line arm had previously received CDK4/6i.
Baseline characteristics of study population.

BRCA, breast cancer susceptibility gene; CDK4/6i, cyclin-dependent kinase 4/6 inhibitor; ECOG, Eastern Cooperative Oncology Group; IDC, Invasive Ductal Carcinoma; ILC, Invasive Lobular Carcinoma.
Fisher’s exact test.
p<0.05 was considered statistically significant.
Safety
Overall, eribulin + antiangiogenic therapy was associated with mild toxicity, with most toxicity being grade 1 or 2 (fatigue, gastrointestinal reaction, and hematologic toxicity). Hematologic toxicity [leukopenia (25.9%) and neutropenia (28.2%)] was the most common grade ⩾3 AEs, and AEs were more common in patients in greater than or equal to third line treatment. But there was no statistically significant difference in hematologic toxicity between first to second line and greater than or equal to third line treatment (Table 2). There was one case of grade 3/4 anemia due to treatment with greater than or equal to third line arm. And there was one case of grade 3/4 thrombocytopenia due to treatment with first to second line arm. Among the grade 1/2 toxicities, asthenia/fatigue was the most common (65.9%), followed by nausea/vomiting, anemia, leukopenia, and neutropenia. In the rate of cardiovascular complications, three cases observed thromboembolic events, seven cases observed hemorrhage, and no cases of grade 3 or higher hypertension. Total incidences of cardiovascular toxicity were 29.4% (25/85) in grades 1/2 and no grades 3/4. Moreover, there was a significant difference in nausea/vomiting (36.6% versus 65.9%, p = 0.007), alanine aminotransferase increased (2.4% versus 25.0%, p = 0.003), and aspartate aminotransferase increased (7.3% versus 29.6%, p = 0.009) between first to second line group and greater than or equal to third line group.
Summary of adverse events.

p<0.05 was considered statistically significant.
Further safety comparison of eribulin combined with bevacizumab and with TKIs group showed that there were statistically significant differences in grade 1/2 AEs (nausea/vomiting, diarrhea, mucositis oral, neutropenia, anemia, alanine aminotransferase increased, aspartate aminotransferase increased, and neurotoxicity). For grade 3/4 AEs, leukopenia was significantly different between the two groups (p = 0.041) (Supplemental Table 1). In addition, a dose reduction of 20–40% and >40% of the total dose was required in 10.6% and 5.9% of patients, respectively. Dose delays were experienced by 15.3% of patients. No treatment-related death was registered.
Efficacy
At the cutoff date (April 2023), 66 (77.6%) patients experienced disease progression and 17 (20.0%) patients had died. Of the total of 85 patients, no patients achieved CR, 29 (34.1%) had PR, 35 (41.2%) had SD, and 21 (24.7%) had PD. The ORR was 34.1% (29/85), while the DCR was 75.3% (64/85) (Table 3) (Supplemental Figure 1). The mPFS of patients treated with eribulin-based treatment was 6.0 months (95% CI: 4.3–7.7). And the OS was immature (Figure 2).
Evaluation of efficacy.

CR, complete response; DCR, disease control rate; ORR, objective response rate; PR, partial response; SD, stable disease.
p<0.05 was considered statistically significant.

Kaplan–Meier plot for PFS and OS in all patients with eribulin + antiangiogenic therapy. (a) Kaplan–Meier plot for PFS. (b) Kaplan–Meier plot for OS.
Univariate analysis (Table 4) indicated that brain metastasis and the number of prior therapies lines were significantly related to PFS in patients treated with eribulin + antiangiogenic therapy. Multivariate analysis (Table 4) demonstrated that brain metastasis and the number of prior therapy lines were independent predictive factors for PFS. Patients without brain metastasis had significantly longer PFS than those with brain metastasis (mPFS: 6.5 m versus 3.0 m, p = 0.020) [Figure 3(a)].
Univariate and multivariate analyses of factors predicting progression-free survival in patients treated with eribulin.
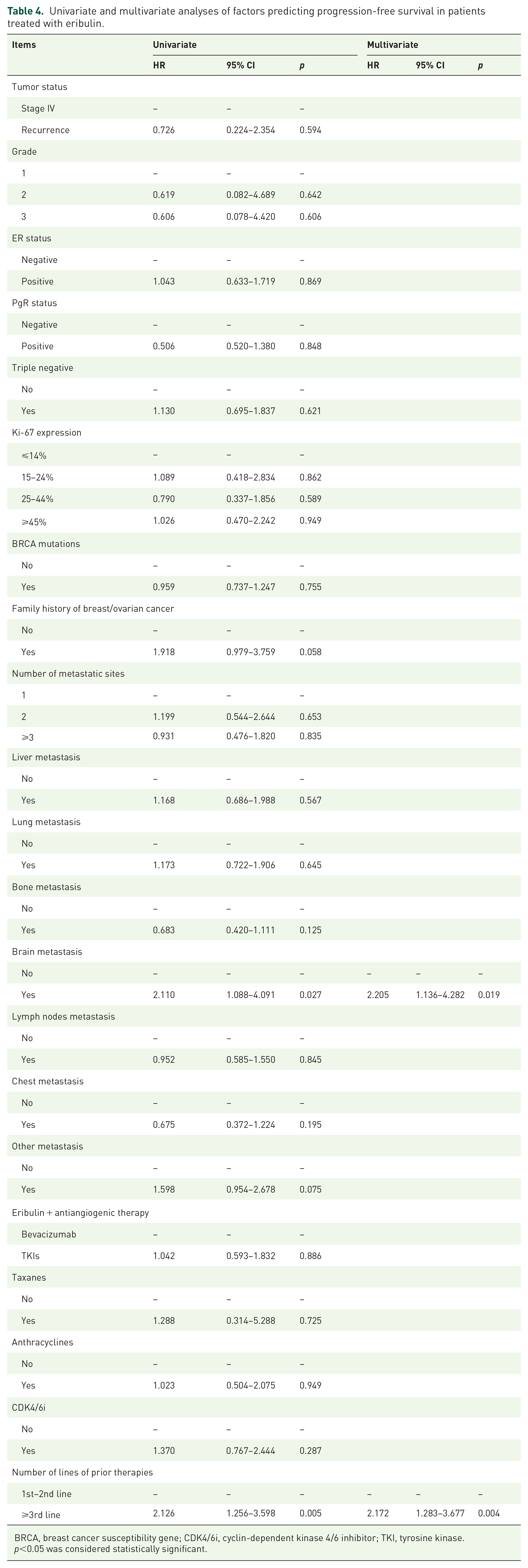
BRCA, breast cancer susceptibility gene; CDK4/6i, cyclin-dependent kinase 4/6 inhibitor; TKI, tyrosine kinase.
p<0.05 was considered statistically significant.

Kaplan–Meier plot for PFS in subgroup. (a) Kaplan–Meier plot for PFS of brain metastatic. (b) Kaplan–Meier plot for PFS of molecular type. (c) Kaplan–Meier plot for PFS of treatment (bevacizumab versus TKIs). (d) Kaplan–Meier plot for PFS of TNBC patients with first to second line and greater than or equal to third line.
The mPFS in the subgroup was 7.7 months and 4.3 months in the first to second line and greater than or equal to third line treatment (P = 0.003), respectively [Figure 4(a)]. The OS in the first to second line and greater than or equal to third line group were immature [Figure 4(b)]. Further analysis, the mPFS was 7.0 months and 5.0 months in HR+/HER2− and TNBC patients (p = 0.612), respectively [Figure 3(b)]. Regarding treatment regimes, most patients (75.3%) used eribulin in combination with bevacizumab. The combination with TKIs presented longer PFS (7.0 m versus 5.0 m, 95% CI: 5.6–8.4 versus 2.8–7.2, p = 0.883), but it didn’t show a significant difference [Figure 3(c)]. A secondary analysis was conducted in TNBC patients. TNBC patients with first to second line therapy showed a significantly longer PFS compared with greater than or equal to third line group (6.5 m versus 2.0 m, 95% CI: 4.4–8.6 versus 0.9–3.1, p = 0.021) [Figure 3(d)].

Kaplan–Meier plot for PFS and OS in first to second line and greater than or equal to third line patients with eribulin + antiangiogenic therapy. (a) Kaplan–Meier plot for PFS. (b) Kaplan–Meier plot for OS.
Discussion
With the advent of the era of precision medicine, numerous targeted or immunotherapy drugs have emerged, further improving the survival benefits of patients with ABC. However, chemotherapy still plays a vital role in the salvage treatment of ABC. Taxanes have become the preferred chemotherapeutic partners among the many combination regimens due to their superior efficacy and relatively low toxicity. 22 In recent years, with the approval of eribulin in China and abroad, the chemotherapy pattern of ABC has changed. Eribulin has become a new choice of chemotherapy for patients with MBC because of its efficacy and safety. 23 A 10-year real-world study in the United States reviewed eribulin’s efficacy in treating MBC. The results showed that the mPFS of eribulin in patients receiving greater than or equal to second-line treatment reached 6.1 months and that in the TNBC subgroup, it was 5.8 months. 24 Real-world studies in Japan25,26 and China 27 have also revealed that eribulin showed excellent antitumor activity in patients with various subtypes of ABC. The frontline application of eribulin can bring long-term survival benefits to patients. Previous studies have shown that eribulin-based combination therapy (targeted therapy, 28 immunotherapy,19,29 etc. 16 ) has better efficacy and safety. In addition, Cortes et al. 6 found that eribulin can increase tumor microvascular density (eliminate hypoxia) and improve tumor vascular abnormalities (alleviate hypoxia, increase tumor perfusion, and reduce tumor interstitial fluid pressure). Meanwhile, many phase III clinical trials have been carried out on the combination therapy of antiangiogenic drugs [bevacizumab (NCT04732598), anlotinib (NCT04405505), and apatinib (NCT04335006)] in the field of locally advanced or MBC. Therefore, this multicenter study aims to investigate further the efficacy and safety of eribulin combined with antiangiogenic drugs in Chinese patients with advanced HER2-negative breast cancer, according to their previous treatment lines.
In previous domestic and international clinical studies, eribulin has shown both efficacy and quality of life benefits. However, eribulin became available in China in 2019. Its clinical application of combination therapy in Chinese patients is still short-term. Therefore, this study analyzed patients receiving bevacizumab, anlotinib, or apatinib in combination with eribulin from four cancer centers in China. From October 2020 to April 2023, a total of 85 women with HER2-negative MBC treated with eribulin combined with antiangiogenetic agents were enrolled. Findings from this study showed the effectiveness of eribulin + antiangiogenic therapy treatment in China women patients with HER2-negative MBC in a real-world clinical setting. Among the 85 patients, 29 patients were PR (34.1%), 35 patients were SD (41.2%), and 21 patients were PD (24.7%). The ORR was 34.1% (29/85), and DCR was 75.3% (64/85). The mPFS of patients treated with first to second line and greater than or equal to third line were 7.7 and 4.3 months, respectively (p = 0.003), and mOS was immature. Concerning safety, eribulin plus antiangiogenic therapy was associated with mild toxic effects, most of grade 1 or 2. Hematologic toxicity [leukopenia (25.9%) and neutropenia (28.2%)] was the most common grade ⩾3 AEs, and AEs were more common in patients in greater than or equal to third line treatment. No statistical difference in hematologic toxicity existed between the first to second line and greater than or equal to third line treatment. Further safety analysis of eribulin combined with bevacizumab and the TKIs group showed many differences in adverse reactions between the two groups. However, the results may be considered a reference due to the significant difference in the patient distribution number of the two groups.
A prospective, open-label, single-arm, multicenter phase II trial (ESMERALDA) showed that eribulin plus bevacizumab was associated with a mPFS of 8.3 months (95% CI: 7.0–9.6) in patients with HER2-negative MBC. The median OS was 28.3 months (95% CI: 22.8–33.9). The incidence of patients who perceived grade 3/4 AEs was low. Brain metastases may cause peri-neoplastic edema. In vivo and in vitro studies have shown that VEGF is the initiating factor of peri-neoplastic edema in intracranial lesions. 30 Bevacizumab can reduce the degree of cerebral edema by reducing vascular permeability and exudate accumulation, thereby reducing symptoms and discomfort in patients. In combination with other chemotherapy drugs, by inhibiting angiogenesis and improving drug delivery, it can also improve the efficacy of chemotherapy drugs in brain metastases and even improve the prognosis. 31 Of the 11 patients with brain metastases in this study, 7 were triple negative. In addition, there were six cases in first to second line group and five cases in greater than or equal to third line group. Ten patients were treated with eribulin plus bevacizumab. The reasons for poor PFS in patients with brain metastases were analyzed comprehensively, including that not all patients received targeted treatment for brain metastases (e.g. brain radiotherapy). Secondly, all 11 patients had ⩾3 metastatic sites, which may indicate that the patient’s own tumor load was also significant. In addition, the phase II Empathy study of eribulin plus anlotinib showed that the DCR of eribulin alone and eribulin plus anlotinib was 66.7% versus 100% (p = 0.007), and the ORR was 37% versus 38.9%, respectively. The mPFS was 3.7 months versus 9.7 months (HR: 0.20, 95% CI: 0.04–0.91, p = 0.04). Regarding safety, the most common grade 3/4 AE in eribulin combination with eribulin + anlotinib group were decreased neutrophil count (25.0% versus 29.2%), increased transaminase (6.3% versus 0%), and reduced platelet count (3.1% versus 0%).
The first to second line mPFS in this multicenter study was comparable with the frontline mPFS reported in previous trials. However, there are apparent differences between patient populations, such as 16% of ESMERALDA patients with TNBC versus 70% of our first to second line patients with TNBC. In addition, the time period of patients receiving this combination in China in our study overlapped with the epidemic time of COVID-19. So, treatment cycles of some patients have been delayed due to the prevention and control policy of the epidemic to a certain extent, which might affect the efficacy. In terms of safety, there appeared to be differences in individual AE items between patients in this study and those in clinical trials. For both study groups, clinically significant safety outcomes, such as discontinuation of treatment due to AEs or AEs requiring hospitalization, appeared to be lower than those reported in clinical trials. This may be due to differences in the frequency of assessments and the retrospective study. Overall, the clinical safety profile of eribulin plus antiangiogenic agents was consistent with the known safety profile,26,32 and didn’t identify new safety concerns.
As most patients with hormone receptor-positive ABC choose first-line endocrine therapy, guidelines recommend salvage chemotherapy as the first choice for patients with visceral metastasis, previous endocrine therapy resistance, or no best endocrine therapy options. Therefore, in this study, most patients in the first to second line group were TNBC patients. Due to the influence of tumor-infiltrating lymphocytes, tumor mutation burden, and Programmed cell death-ligand 1 (PD-L1) expression, immunotherapy has become a hot spot in treating advanced TNBC. Impassion 130 study showed that the PFS of first-line immunotherapy combined with chemotherapy in patients with advanced TNBC was 7.2 months (p = 0.002). The Keynote-355 study indicated that in patients with PD-L1 Combined positive score (CPS) ⩾10, the PFS in the pembrolizumab + chemotherapy group was 9.7 months, the 6-month PFS rate was 65.0%, and the 12-month PFS rate was 46.9% (p = 0.0012). Previous studies have shown that antiangiogenesis therapy can improve the immune microenvironment, so immunotherapy combined with antiangiogenesis treatment has a synergistic effect. 33 A multicenter phase II trial of camrelizumab combined with apatinib and eribulin in patients with advanced TNBC showed a mPFS of 8.1 months (95% CI: 4.6–10.3). Regarding safety, the incidence of grade 3/4 TRAE was 41.3%, mainly including neutropenia and thrombocytopenia. 19 Although this study was more effective, the rate of grade 3/4 AEs was higher. In our study, TNBC patients with first to second line therapy showed a significantly longer PFS than the greater than or equal to third line group (6.5 m versus 2.0 m, 95% CI: 4.4–8.6 versus 0.9–3.1, p = 0.021). Therefore, the combination of eribulin and antiangiogenic drugs might be another option for frontline treatment of TNBC, especially for PD-L1 negative TNBCs with less benefit from immune checkpoint inhibitors.
Despite these positive findings, this study has limitations. A significant limitation is that although it is a multicenter database, the study design is retrospective and observational, and the number of cases is small. Meanwhile, the fact that these are not standard regimens which are supported only by immature phase 2 data. It is necessary to confirm the findings in a large, prospective study. In addition, due to the late introduction of eribulin in China, many patients were in the greater than or equal to third line treatment group (the median number of chemotherapy lines was 4). In addition, some patients have delayed treatment due to the influence of COVID-19 prevention and control treatment. Differences might also exist among physicians selecting patients for eribulin plus antiangiogenic therapy treatment, as uniform assessment criteria were not used to judge the suitability of eribulin. This will more or less affect the results of the study. In comparing AEs of antiangiogenic drugs, the reliability of the results is also affected due to the significant difference in the number of eribulin combined with bevacizumab and with TKIs. Despite these limitations, the current study provides data in a real-world setting and will help future clinical practice.
Conclusion
In conclusion, eribulin combined with antiangiogenic agents has shown promising efficacy and tolerable toxicity in patients with HER2-negative MBC. This combination provided new chemotherapy options for HER2-negative MBC patients, especially in frontline treatments. Further exploration with a larger sample size is warranted in the future.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231204856 – Supplemental material for Eribulin combined with antiangiogenic agents in women with HER2-negative metastatic breast cancer: a retrospective multicenter study
Supplemental material, sj-docx-1-tam-10.1177_17588359231204856 for Eribulin combined with antiangiogenic agents in women with HER2-negative metastatic breast cancer: a retrospective multicenter study by Mingxia Jiang, Bin Shao, Donggui Wan, Jiaxuan Liu, Maiyue He, Yue Chai, Die Sang, Jiayu Wang, Fei Ma, Ying Fan, Peng Yuan, Binghe Xu and Qiao Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-2-tam-10.1177_17588359231204856 – Supplemental material for Eribulin combined with antiangiogenic agents in women with HER2-negative metastatic breast cancer: a retrospective multicenter study
Supplemental material, sj-eps-2-tam-10.1177_17588359231204856 for Eribulin combined with antiangiogenic agents in women with HER2-negative metastatic breast cancer: a retrospective multicenter study by Mingxia Jiang, Bin Shao, Donggui Wan, Jiaxuan Liu, Maiyue He, Yue Chai, Die Sang, Jiayu Wang, Fei Ma, Ying Fan, Peng Yuan, Binghe Xu and Qiao Li in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We deeply appreciate all authors for their contributions, patients who participated in this study, and support from four institutions [National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College; Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Peking University Cancer Hospital and Institute; China-Japan Friendship Hospital; Beijing Chaoyang District Sanhuan Cancer Hospital].
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
