Abstract
Ileostomy diverts the flow of feces, which can result in malnutrition in the distal part of the intestine. The diversity of the gut microbiota consequently decreases, ultimately leading to intestinal dysbiosis and dysfunction. This condition can readily result in diversion colitis (DC). Potential treatment strategies include interventions targeting the gut microbiota. In this case study, we effectively treated a patient with severe DC by ileostomy and allogeneic fecal microbiota transplantation (FMT). A 69-year-old man presented with a perforated malignant tumor in the descending colon and an iliac abscess. He underwent laparoscopic radical sigmoid colon tumor resection and prophylactic ileostomy. Follow-up colonoscopy 3 months postoperatively revealed diffuse intestinal mucosal congestion and edema along with granular inflammatory follicular hyperplasia, leading to a diagnosis of severe DC. After two rounds of allogeneic FMT, both the intestinal mucosal bleeding and edema significantly improved, as did the diversity of the gut microbiota. The positive outcome of allogeneic FMT in this case highlights the potential advantages that this procedure can offer patients with DC. However, few studies have focused on allogeneic FMT, and more in-depth research is needed to gain a better understanding.
Keywords
Introduction
Enterostomy is a frequently performed procedure in gastrointestinal surgery. 1 As the incidence of colorectal cancer and other intestinal surgeries has increased, the number of patients requiring permanent or prophylactic colostomy has also risen. Although enterostomy may help patients eat early and recover quickly after surgery, it changes the physiological channel of normal fecal excretion, incorporating a portion of the intestine into an indwelling intestinal tube. Diversion colitis (DC) is a potential complication of this procedure that can be difficult to manage and cannot be ignored. 2 Many diseases, particularly those involving the digestive system, have been shown to be correlated with the gut microbiota. Transplant strategies that utilize the intestinal microbiota have been applied in the treatment of inflammatory bowel diseases such as Crohn’s disease and Clostridium difficile infection with good outcomes.3,4 DC has not been adequately and consistently treated with any effective therapeutic measure to date. We herein describe a patient with severe DC who was successfully treated with ileal stoma placement and allogeneic fecal microbial transplantation (FMT).
Case report
Patient and examinations
A 69-year-old man presented to our hospital in March 2022 with left lower abdominal pain and a high fever that had persisted for 4 consecutive days. He had a history of chronic bronchitis, emphysema, and benign prostatic hyperplasia, but he had experienced no serious symptoms for the past 5 years. On admission, the patient had a body temperature of 39°C. Laboratory findings were as follows: carcinoembryonic antigen, 26.06 ng/mL; neutrophils, 83.2%; fibrinogen, 5.87 g/L; D-dimer, 4.46 μg/mL; fibrinogen degradation products, 12.81 μg/mL; and high-sensitivity C-reactive protein, 107.13 mg/L. Computed tomography examination showed significant localized thickening of the intestinal wall in the descending colon, resulting in narrowing of the intestinal canal. A flaky mixed-density shadow was seen in the left iliac fossa. The patient was suspected to have a malignant tumor of the descending colon and an abscess in the left iliac fossa. In addition, a small hypodense shadow suspected to be an enlarged lymph node was found in the left inguinal region (Figure 1). The patient underwent laparoscopic radical sigmoid colon surgery, preventive terminal ileostomy, and debridement of the iliac fossa abscess. No tumor was observed at the surgical margins, and 12 lymph nodes were excised. The pathological diagnosis of the specimen was differentiated adenocarcinoma, and the tumor had invaded the stromal layer of the intestinal wall (Figure 2). No adverse events occurred intraoperatively or during hospitalization. Two weeks after the operation, the patient began to experience intermittent dull pain in the lower abdomen, anal bleeding, and severe abdominal pain. Approximately 3 months later, colonoscopy showed diffuse congestion, edema, and granular inflammatory follicular hyperplasia of the intestinal mucosa (Figure 3(a–d)). The intestinal mucosal inflammatory damage score was 7 points, indicating severe DC. We did not directly close the stoma because of the risk of hemorrhage and sepsis. However, various local treatments, including short-chain fatty acid (SCFA) enemas, mesalamine, and steroid enemas, were all unsuccessful. Additionally, analysis of the patient’s feces using 16S rDNA sequencing revealed a reduction in the intestinal microbiota diversity and an increase in some conditionally pathogenic genera. Therefore, we considered performing FMT. The patient consented to two rounds of allogeneic FMT from a young, healthy donor who had been recruited according to previously described criteria. 5 A drainage tube was inserted into the ascending colon through the distal part of the bowel stoma (Figure 4). A 50-mL saline-diluted bacterial solution was injected through the tube for 6 consecutive days, and the procedure was repeated 2 weeks later.
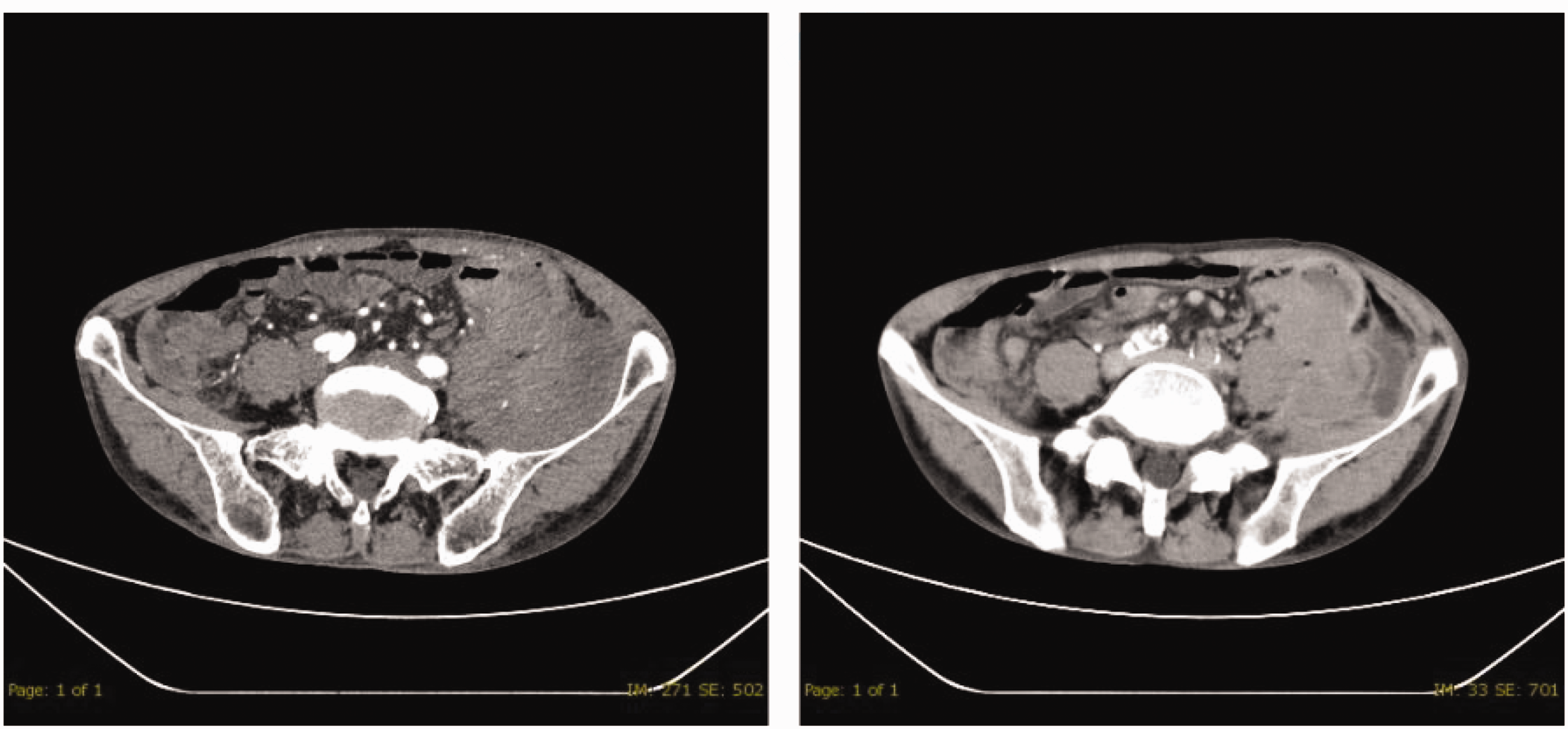
Computed tomography findings in the upper and lower abdomen.

Hematoxylin and eosin staining results: cytokeratin (+) and carcinoembryonic antigen (partly +) (40×).

Enteroscopic observations. (a–d) Before fecal microbiota transplantation. (e–h) After fecal microbiota transplantation. Intestinal mucosal inflammatory damage score: 7 points (mucosal hemorrhage: 3 points, contact hemorrhage: 1 point, edema: 3 points).

Diagram of ileostomy placement.
The procedure was approved by the Ethics Committee of Wuhan Puren Hospital (approval no. 202200101). The patient provided verbal informed consent for publication of his case. The reporting of this study conforms to the CARE guidelines. 6
Outcome and follow-up
After completion of two courses of allogeneic FMT, the edema and diffuse hemorrhage in the intestinal tract disappeared (Figure 3(e–h)). The intestinal mucosal inflammatory damage score was 1 point. Examinations revealed no contraindications for stoma reversal; therefore, the surgery was performed with the consent of the patient and his family. The patient developed no postoperative abdominal pain, diarrhea, bleeding, sepsis, or other symptoms, and his recovery was smooth. The patient was followed up for >6 months after FMT, and no significant adverse reactions were observed. At the time of this writing, the patient was still free of gastrointestinal discomfort and was in good health. The patient and his family were satisfied with the improvement in his clinical condition.
The gut microbiota analysis after FMT showed that the patient’s intestinal microecology had improved. The diversity of gut bacteria had significantly increased, and its composition tended to resemble that of healthy individuals (Figure 5). Before FMT, the patient had exhibited markedly higher abundance of the phylum Actinobacteria and lower abundance of the phyla Proteobacteria and Bacteroidetes compared with health individuals. At the genus level, Corynebacterium, Staphylococcus, and Lactobacillus were significantly elevated; however, the abundance of Bacteroides, Bifidobacterium, Faecalibacterium, and Prevotella was sharply reduced. Notably, the genera Alistipes, Blautia, Parabacteroides, Eubacterium, Streptococcus, and Parasutterella were detected in fecal samples from healthy individuals, but they were not found in samples taken from our patient prior to FMT. Compared with the results before FMT, the abundance of Actinobacteria markedly decreased and that of Proteobacteria markedly increased after FMT. Additionally, Bifidobacterium, Faecalibacterium, and Streptomyces showed a significant increase. Although other previously undetected microbial groups, such as Euryarchaeota, Fusobacteriota, Synergistetes, and Verrucomicrobia, were detected after FMT, Corynebacterium, Lactobacillus, and Staphylococcus were not (Figure 5).

Comparison of gut microbiota in fecal samples before and after fecal microbiota transplantation. (a) Shannon Index. (b) Relative abundance of gut bacteria at phylum level and (c) relative abundance of gut bacteria at genus level.
Discussion
DC is a nonspecific inflammatory reaction of the mucosa that occurs after surgical diversion of fecal flow away from a segment of the colon. It was first discovered by Morson and Dawson 7 and later termed “diversion colitis” by Glotzer et al. 8 The incidence of DC after enterostomy is nearly 100%. Although the majority of patients are asymptomatic, it is important to avoid overlooking the fact that symptomatic patients still account for 30% to 38% of cases. Clinical symptoms of DC include abdominal pain, diarrhea, tenesmus, hematochezia, anorectal pain, and hemorrhagic anal discharge. Colonoscopy can reveal mucosal edema, congestive erythema, erosive ulceration, granular inflammatory follicular hyperplasia, and inflammatory polyps. Additionally, some patients may develop mucosal inflammation and tearing. A small number of patients with severe DC may develop colorectal hemorrhage, sepsis, and other life-threatening symptoms.9–11 Although the use of SCFAs, mesalamine, or corticosteroids has been reported for the treatment of DC, the results vary. 12
The pathogenesis of DC is still unclear and may involve factors such as the intestinal microbiota, SCFAs, and the immune inflammatory response. With the rapid development of multi-omics technology, the significant role of the gut microbiota in intestinal diseases and health is gradually being elucidated. 13 Lloyd-Price et al. 14 performed a systematic study based on multi-omics technology and found that the diversity of the intestinal microbiota in patients with inflammatory bowel disease was low. This was evidenced by an increase in facultative anaerobic bacteria and a decrease in obligate anaerobic bacteria. 14 Watanabe et al. 15 found that fecal stream diversion reduced the diversity of the mucosa-associated microbiota in the dysfunctional ileum. Beamish et al. 16 made a similar observation, suggesting that ileostomy-mediated fecal stream diversion leads to downstream intestinal nutrient deficiencies. This can alter the composition of the intestinal microbiota, thereby affecting intestinal structure and function. 16 Tominaga et al. 12 found that anaerobic bacteria and SCFAs were also significantly reduced in the diverted colon of patients with DC, but immunoglobulin A was significantly elevated. In the present study, changes were mainly observed in the phyla Firmicutes, Bacteroidetes, Proteobacteria, and Actinobacteria. The diversity of the intestinal microbiota decreased, which is consistent with the findings of most other studies. However, the specific changes have not yet been fully integrated.17–19 Although the abundance of Firmicutes, Bacteroidetes, and Proteobacteria in the gut microbiota was low before FMT in our patient, the abundance of Actinobacteria was high. Our patient had an extreme deficiency of Bacteroidetes, which is often associated with inflammatory bowel disease. Many microorganisms in the phylum Bacteroidetes have the potential to produce SCFAs such as propionic acid and acetic acid. For example, Bacteroides fragilis produces propionic acid. As an energy source for colonic epithelial cells, SCFAs play an important role in improving intestinal barrier function, reducing intestinal inflammation, and enhancing host immune function.20,21 Most notably, Corynebacterium (49.51%, phylum Actinobacteria) and Staphylococcus (30.67%, phylum Firmicutes) dominate the microbiota at the genus level. Corynebacterium is often considered an important opportunistic pathogen. Recent findings by Wang et al. 22 suggest a strong relationship between Corynebacterium and the regulation of intestinal defense mechanisms in patients with ulcerative colitis. Similarly, Staphylococcus, an important pathogen associated with antibiotic resistance, has been reported in numerous cases of intestinal disease. After FMT, Bifidobacterium (which has potential anti-tumor effects) can enhance immunity and Faecalibacterium (which can produce butyrate and has anti-inflammatory effects) begins to increase.23,24 Furthermore, we observed a notable rise in the abundance of Proteobacteria and Streptomyces (which were not present in the donor) following FMT in our patient. However, Alistipes, Parabacteroides, Parasutterella, and Prevotella were not successfully colonized in the patient’s gut after FMT. This variation is consistent with the theory proposed by Smillie et al., 25 who suggested that the post-FMT gut microbiota may adopt various colonization patterns, resulting in different outcomes. Although these differences may vary because of individual variances, the shared strains between donors and patients are not coincidental similarities.
Since Gundling et al. 26 reported the first successful treatment of a patient with DC using autologous FMT in 2015, subsequent reports on the potential benefits of autologous FMT in the treatment of DC have been reported by Kalla et al., 27 Tominaga et al., 28 and Donahue et al. 29 Autologous FMT can avoid the influence of potential immune rejection and other factors; however, the gut microbiota of most patients has already been destroyed by the time of their first visit. Direct microbiota transplantation is not conducive to the remodeling of the patient’s intestinal microecology, which undoubtedly places a higher demand on the processing of fecal samples before autologous FMT. Allogeneic FMT can avoid this problem. We have herein presented the first report of successful allogeneic FMT in treating a patient with severe DC. Actually, as early as 2012, Hamilton et al. 30 successfully used FMT to treat Clostridium difficile infection, and FMT and the concept of a “stool bank” thereafter began to attract attention. A stool bank is designed to collect fecal samples from young, healthy individuals for cryopreservation. This allows the use of autologous FMT to improve the health of patients who are aging or have diseases related to microbiota disorders. However, the proportion of individuals with suboptimal health is increasing year by year, making it difficult to obtain “healthy” feces. In addition, the difficulty of preserving samples limits the widespread use of this technology. Therefore, considering both economic cost and technical difficulty, the clinical promotion of allogeneic FMT currently appears to be more advantageous.
Conclusion
Overall, the results of this study suggest that allogeneic FMT may offer advantages in the treatment of DC. However, this study is limited by the analysis of only a single case; therefore, it is important to expand the sample size for future research in clinical practice. Allogeneic FMT is an important technology that will benefit patients in future and should be taken seriously. In addition, continuous and dynamic monitoring of the intestinal microbiota, along with the application of multi-omics techniques, will help us gain a better understanding of the occurrence and development of various diseases.
Footnotes
Acknowledgements
We thank all the doctors and medical staff who were involved in the treatment of this patient.
Author contributions
Cheng CL and Wang XJ performed the surgery on the patient and drafted, edited, and approved the manuscript. Fan LX performed the literature search and review, collected clinical information, and obtained informed consent for publication of the manuscript. Lv YL supervised the study and revised the manuscript. Xiong K and Jiang ZW contributed to the data analysis and presentation. Gan T participated in the care of the patient throughout hospitalization and collected follow-up information. Fu G contributed to the study conceptualization and supervision. All authors read and approved the final manuscript.
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author (Guang Fu).
Declaration of conflicting interest
Yong-Ling Lv, Kai Xiong, and Zhi-Wei Jiang are employees of Maintainbiotech, Ltd. (Wuhan, China). The other authors declare no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
