Abstract
In recent years, endocrine therapy (ET), an effective systemic treatment for the management of estrogen receptor (ER)-positive breast cancers, has regained interest as a neoadjuvant therapy based on evidence that ET can fulfill the aim of neoadjuvant systemic treatment for tumor shrinkage as well as elucidate important clinical information on endocrine sensitivity that enables the prognostication of patients. Moreover, neoadjuvant endocrine therapy (NET) potentially provides an opportunity for early assessment of the clinical efficacy of novel agents. Furthermore, recently reported trials have generated evidence for a more tailored approach for perioperative management of ER-positive breast cancer using clinical and molecular biomarkers, and this has provided a rationale that enables the broadening of clinical indications for NET. This review discusses the current evidence for NET, the evolution of NET trials, clinical indications, and NET-based treatment strategies.
Keywords
Introduction
Neoadjuvant systemic treatment constitutes an important therapeutic strategy in patients with non-metastatic breast cancer. It is aimed at achieving tumor shrinkage and, possibly, conversion to breast-conserving surgery besides enabling early response evaluation for systemic treatment. Estrogen receptor-positive (ER+) breast cancer accounts for the majority of all breast cancer subtypes. 1 ER+ breast cancer, despite the lower response to chemotherapy, has a relatively favorable outcome, with pathologic complete response (pCR) rates of <10%, especially in low-grade tumors. Unlike in other breast cancer subtypes, the correlation between survival outcome and pCR is weaker.2,3 Given the low response to chemotherapy in these patients, there exists a significant body of research on the feasibility of neoadjuvant endocrine treatment (NET) and its comparable efficacy with neoadjuvant chemotherapy (NCT), with significantly lower toxicity. 4
Furthermore, although NET was an underutilized tool until recently, current international guidelines endorse NET as a reasonable alternative for NCT, 5 and novel NET regimens and clinical trial designs that enable appropriate patient selection for NET have been suggested in the last few years. NET has increasingly garnered clinical interest during the COVID-19 pandemic because of the medical resource limitations. 6 This review presents an assimilation of the current evidence for NET, the evolution of NET trials, and the clinical indications and treatment strategies for NET.
Efficacy of NET compared with NCT
A limited number of trials have prospectively compared the efficacy of NET versus NCT and these have reported similar results (Table 1). A phase II trial conducted by Semiglazov et al. randomized 239 postmenopausal patients with ER+ breast cancer to either NET with an aromatase inhibitor (AI) (anastrozole or letrozole) or NCT (doxorubicin and paclitaxel) for 3 months and reported similar clinical response rates based on palpation [according to the World Health Organization (WHO) criteria, 64% in both arms]. The pCR rates were low in both arms (3% versus 6% in the NET and NCT arms, respectively). 7 The GEICAM 2006-03 trial enrolled patients with ER+, progesterone receptor-positive (PR+), human epidermal growth factor receptor 2-negative (HER2−) breast cancer, regardless of the menopausal status. Accordingly, a total of 95 participants, of whom 51 were premenopausal, were randomized to NET with exemestane [plus ovarian function suppression (OFS) if premenopausal] versus NCT with epirubicin plus cyclophosphamide followed by docetaxel for 6 months. In the overall study population, differences in the clinical response rates were nonsignificant [66% (NCT) versus 48% (NET)]. Although the pCR rates were low in both groups (none in the NET versus one patient in the NCT arm), subgroup analyses showed lower response rates to NET in premenopausal patients [75% (NCT) versus 44% (NET)] and patients with high baseline Ki-67 levels [>10%; 67% (NCT) and 42% (NET)]. 8 Similarly, the NEOCENT study, which randomized 44 postmenopausal patients with ER+ breast cancer, reported comparable radiologic response rates for the NET and NCT groups (59% versus 55%). 9
Randomized clinical trials that compared NET and NCT.

NET versus NCT, unless otherwise indicated.
ALN, axillary lymph node; BCS, breast-conserving surgery; ER, estrogen receptor; FEC, 5-fluorouracil, epirubicin, and cyclophosphamide; HER2, human epidermal growth factor 2; MMG, mammography; MRI, magnetic resonance imaging; NCT, neoadjuvant chemotherapy; NET, neoadjuvant endocrine therapy; NR, not reported; NS, not significant; OFS, ovarian function suppression; ORR, overall response rate; pCR, pathologic complete response; PR, progesterone receptor; RECIST, Response Evaluation Criteria in Solid Tumors; US, ultrasonography; WHO, World Health Organization response criteria.
While the majority of clinical trials that evaluated the efficacy of NET enrolled postmenopausal patients, Kim et al. randomized premenopausal patients with ER+ HER2− breast cancer to either NET with tamoxifen with OFS or NCT with doxorubicin/cyclophosphamide followed by docetaxel for 6 months and obtained similar results as in the GEICAM 2006-03 premenopausal patient subgroups: inferior radiologic response rates in NET groups compared with NCT groups [53% (NET) versus 84% (NCT) by magnetic resonance imaging (MRI), p < 0.001]. However, comparable BCS conversion rates among patients who were initially identified as candidates for mastectomy were observed (12% versus 14%). 10
These clinical trials suggested the feasibility of 3–6 months of endocrine treatment as neoadjuvant therapy in postmenopausal patients with ER+ breast cancer, although pCR rates were low in both the NET and NCT arms. Despite these results, NET implementation in clinical practice has been limited as these trials used different inclusion criteria, treatment regimens and durations, and outcome measurements, as well as the lack of prognostic and predictive biomarkers for appropriate patient selection.
Treatment regimens
Choice of endocrine agents
Large-sized randomized trials (P024, IMPACT, PROACT, etc.) that compared tamoxifen and AI11–13 as well as a meta-analysis that included 20 prospective NET trials 4 suggested similar conclusions with regard to the choice of endocrine agents for NET (Table 2).
Response rates by endocrine regimens.

Results are sequentially ordered according to the study arms presented in the ‘intervention’ column, unless otherwise indicated.
MRI, magnetic resonance imaging; N, number; OFS, ovarian function suppression; ORR, overall response rate; Pre, premenopausal; Post, postmenopausal; RECIST, Response Evaluation Criteria in Solid Tumors; US, ultrasonography; WHO, World Health Organization response criteria.
In postmenopausal patients treated with NET, compared with tamoxifen, AI induced higher response rates as well as breast-conserving surgery rates; a meta-analysis showed that the odds ratios (ORs) for the clinical response and BCS rates for AI, compared with tamoxifen, was 1.69 and 1.62, respectively. 4 Nonetheless, the results of the IMPACT trial showed that tamoxifen–AI combination therapy did not generate higher response rates. 12 Although limited data are available, similar results were observed in premenopausal patients. In the STAGE trial, the clinical response rates were higher with 6 months of neoadjuvant AI (anastrozole), compared with tamoxifen, both with OFS, in premenopausal ER+ patients (70% versus 51%, p = 0.004). However, bone mineral density (BMD) reduction was greater in the anastrozole group (mean BMD at L-spines, −5.8% in the anastrozole group versus −2.9% in the tamoxifen group, p < 0.0001). 19
Among AIs, steroidal AI showed comparable efficacy with non-steroidal AI in terms of clinical response rates in the Z1031 trial that compared neoadjuvant letrozole, anastrozole, and exemestane (clinical response rate by WHO criteria, 75% versus 69% versus 63%, respectively). Moreover, the proportion of patients who achieved endocrine response, represented by a Preoperative Endocrine Prognostic Index (PEPI) 0 (discussed further in the ‘Response evaluation and endpoints for NET’ section of this review), did not differ among the treatment groups (16% versus 17% versus 16% for letrozole, anastrozole, and exemestane, respectively). 15
Fulvestrant, a selective estrogen receptor degrader (SERD), showed similar clinical response rates compared with AI in postmenopausal patients in two prospective phase II trials.17,18 Furthermore, pooled analysis of these two trials showed comparable relapse-free and overall survivals at 5 years. 20 In addition, the ALTERNATE trial, which included 1299 postmenopausal ER+ patients who were randomized to receive anastrozole, fulvestrant, or both (anastrozole–fulvestrant), showed similar endocrine-sensitive disease rates [ESDRs: defined as the proportion of patients achieving modified PEPI (mPEPI) 0 or pCR] for fulvestrant or anastrozole–fulvestrant, as compared with anastrozole monotherapy (23% versus 21% versus 19%, respectively). 21
Duration of treatment
There is currently no clear consensus on the treatment duration for NET. Although many prospective clinical trials implemented 3–6 months of NET, higher response rates were reported with longer-duration endocrine therapy. An exploratory phase IIb/III trial observed clinical response amenable for BCS at 4 months of NET in more than half of the originally BCS-ineligible patients; however, the authors observed further response at 8 months in patients who did not show a clinical response at 4 months, suggesting the clinical benefit of extended duration of NET. 22 Similarly, a phase II trial revealed a median time to maximal response of 4.2 months with NET, although 37% achieved maximal response in 6–12 months. 23 The phase II TEAM IIA trial, which compared 3 months versus 6 months of neoadjuvant exemestane, reported higher clinical response rates in the 6-month treatment group. 16 In addition, higher pCR rates of 18% have been reported with 1-year endocrine treatment. 24 Although these data suggest that extended-duration NET is likely to induce further tumor response, the optimal NET duration remains to be determined because the risk for clinical progression during longer treatment and the optimal timing for surgery needs to be balanced.
Novel neoadjuvant endocrine regimens: Combination with CDK4/6 inhibitors
The efficacy of three different cyclin-dependent kinase 4/6 (CDK4/6) inhibitors (palbociclib, abemaciclib, and ribociclib) has been explored in multiple phase II trials, which showed similar results. In the phase II PALLET trial, 307 postmenopausal patients with ER+ breast cancer measuring >2 cm were randomized to four arms with different induction schedules (14 weeks of letrozole monotherapy, 2 weeks of induction therapy with either palbociclib or letrozole followed by palbociclib–letrozole combination therapy for 14 weeks, or palbociclib–letrozole combination therapy for 14 weeks). A greater decrease in Ki-67 was observed in palbociclib–letrozole combination therapy arms, compared with letrozole monotherapy. The rates of complete cell-cycle arrest (CCCA), defined as posttreatment Ki-67 ⩽2.7%, were higher in the combination arm (90% versus 59%, p < 0.001). However, clinical response rates or breast-conserving surgery rates did not significantly differ between the palbociclib–letrozole combination therapy arms versus letrozole monotherapy arm (54% versus 49%). 25 Similarly, the neoMONARCH trial compared 2 weeks of lead-in treatment with anastrozole, abemaciclib, or a combination of both, followed by 14 weeks of combination treatment with anastrozole and abemaciclib for all groups, and identified a greater decrease in the Ki-67 at week 2 with higher CCCA rates in the abemaciclib combination arms (58–68% versus 14%). 26 The FELINE trial compared letrozole, with or without ribociclib, with two dosing schedules (intermittent or continuous). The CCCA rate at 2 weeks was higher in the letrozole–ribociclib combination arms (92% versus 52%); however, the CCCA rate at surgery did not differ between arms (71% versus 63%), with the majority of patients showing a significant Ki-67 increase from 2 weeks of treatment to surgery in the letrozole–ribociclib combination arms. The PEPI-0 rates did not differ between arms (25% in both arms) nor did the clinical response rates. 27
The CORALLEEN 28 and NeoPAL29,30 trials compared NET in combination with CDK4/6 inhibitors versus NCT. The CORALLEEN trial included patients with luminal B intrinsic subtype only, and compared ribociclib plus letrozole versus NCT. The primary endpoint comprised the rates of low PAM50 risk-of-relapse (ROR) disease at surgery, which was comparable between the study arms (47% and 46% in the NET and NCT arms, respectively). The result of the CORALLEEN trial suggests that NET with CDK4/6 inhibitors can induce ‘molecular downstaging’, which might enable safe avoidance of chemotherapy in patients with luminal B breast cancers. Molecular downstaging was more rapidly achieved in the NET arm, as the rate of conversion to the luminal A subtype at day 15 was 96% in the NET arm and 37% in the NCT arm. However, pCR rates were low in both NET and NCT arms (0% versus 6%). Although limited by the lack of long-term follow-up survival data, the authors suggested that NET with CDK4/6 inhibitors can substantially induce early molecular downstaging in patients with luminal B breast cancers assessed by the standard PAM50 (Prosigna®) assay. 28
The NeoPAL trial compared NET with letrozole plus palbociclib for 19 weeks versus NCT with FEC regimen followed by docetaxel for a total of six cycles. Pathologic response rates, defined as the residual cancer burden (RCB) 0/1 at surgery, were numerically higher in the NCT arms (8% in the NET arm versus 16% in the NCT arm), although low in both arms. Conversely, PEPI-0 was achieved in 18% versus 8% of NET and NCT arms, respectively. However, follow-up survival data did not show significant differences in progression-free survival (PFS) or invasive disease-free survival (iDFS) between the arms.29,30
To summarize, NET in combination with CDK4/6 inhibitors can induce early cell-cycle arrest, which results in an earlier, potent reduction of Ki-67. However, evidence is currently lacking on whether these treatments can improve clinical response rates or survival outcomes, and pCR rates to NET remain low even with the addition of CDK4/6 inhibitors or the exclusive selection of patients with luminal B breast cancers (Table 3). Moreover, consensus on which endpoint is optimal for these patients has not been reached. The clinical meaning of the early cell-cycle arrest that was achieved with the addition of CDK4/6 inhibitors is unknown. Therefore, without long-term outcomes, NET with CDK4/6 inhibitors currently remains an investigational treatment.
Neoadjuvant trials with CDK4/6 inhibitors (N ⩾ 50).
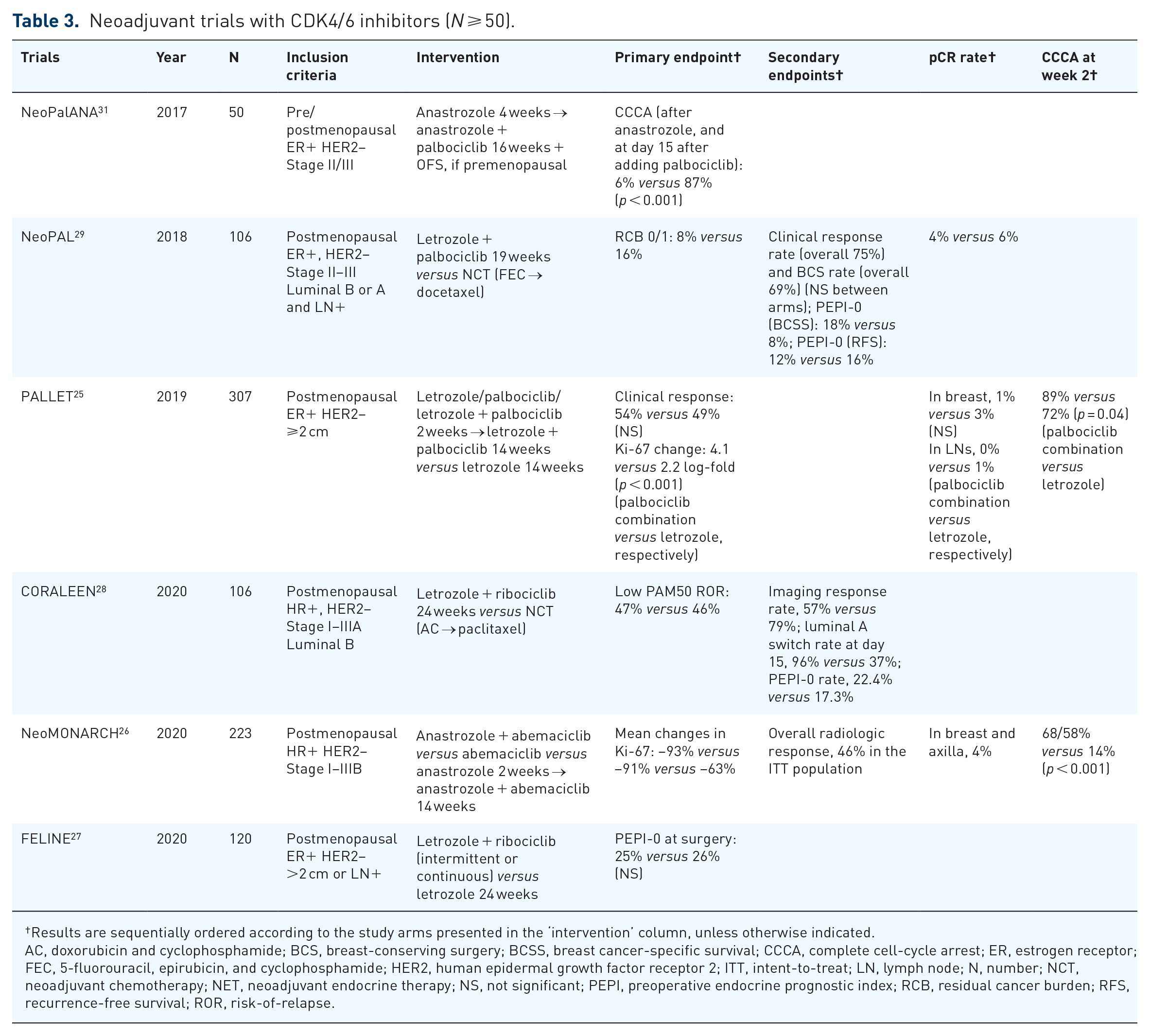
Results are sequentially ordered according to the study arms presented in the ‘intervention’ column, unless otherwise indicated.
AC, doxorubicin and cyclophosphamide; BCS, breast-conserving surgery; BCSS, breast cancer-specific survival; CCCA, complete cell-cycle arrest; ER, estrogen receptor; FEC, 5-fluorouracil, epirubicin, and cyclophosphamide; HER2, human epidermal growth factor receptor 2; ITT, intent-to-treat; LN, lymph node; N, number; NCT, neoadjuvant chemotherapy; NET, neoadjuvant endocrine therapy; NS, not significant; PEPI, preoperative endocrine prognostic index; RCB, residual cancer burden; RFS, recurrence-free survival; ROR, risk-of-relapse.
Novel neoadjuvant endocrine regimens: Other targeted therapies and novel endocrine agents
Few trials have investigated the role of phosphoinositide 3-kinase inhibitors in combination with NET. In the LORELEI trial, 334 postmenopausal ER+ patients were randomized to letrozole with or without taselisib. A marginal increase in the radiologic response rates by MRI at 16 weeks was observed in the taselisib combination arm (50% versus 39%, OR: 1.55, p = 0.049). However, pathologic response rates were low in both groups (2% versus 1%), and both groups had similar Ki-67 change after 3 weeks. 32 The NEO-ORB trial randomly assigned 257 postmenopausal patients with HR+ breast cancer to NET with letrozole with or without alpelisib for 24 weeks. As a result, the addition of alpelisib to letrozole did not result in improved radiologic response rates or pCR rates compared with letrozole monotherapy, regardless of the presence of PIK3CA mutation (response rates to alpelisib combination versus letrozole monotherapy, 43% versus 45% for mutants, 63% versus 61% for wild type subgroups). The decrease in Ki-67 was similar across arms. Of note, only 52% of the cohort completed 24 weeks of alpelisib treatment. 33
As for novel agents, the phase II coopERA trial compared 2 weeks of window-of-opportunity phase with oral giredestrant, a novel oral SERD, versus anastrozole (n = 221). Both groups were then followed by 16 weeks of combination treatment with palbociclib administered with each agent. Ki-67 suppression at 2 weeks and the rates of CCCA were greater in the giredestrant arm compared with those in the anastrozole arm. However, the objective response rate did not differ between the treatment arms. 34 Additionally, a small phase II window-of-opportunity trial compared 2 weeks of preoperative AZD9496, an oral SERD, with fulvestrant before curative surgery, but failed to demonstrate the superiority of AZD9496 with regard to the ER H-score reduction and decrease in Ki-67 levels. 35
Lastly, with the growing interest in the HER2-directed antibody-drug conjugates in advanced ‘HER2-low’ cancers – a population that was previously considered HER2-negative – a neoadjuvant trial that compares trastuzumab deruxtecan (T-DXd), with or without AI, in patients with ER+, HER2-low breast cancer is currently underway. In the phase II TRIOS-US B-12 TALENT trial, HR+ HER2-low pre- and postmenopausal patients were randomized to receive 6–8 cycles of T-DXd, with or without anastrozole (plus OFS if premenopausal). 36 The interim results, which were reported in the 2022 San Antonio Breast Cancer Symposium, showed that the addition of endocrine therapy (ET) to T-DXd did not increase treatment efficacy, given that the primary endpoint – the pCR rate – was 68% in the T-DXd alone arm versus 58% in the T-DXd with anastrozole arm. 37
Response evaluation and endpoints for NET
Pathologic response
In luminal A breast cancer treated with NAC, the pCR rates are known to be low and are not well-correlated with survival outcomes. 3 pCR rates are low with NET, as discussed earlier, but the clinical outcome of these patients is favorable without achieving pCR. Similarly, the prognostic implication of residual disease on pathology specimens after NET is unclear. In the NeoPAL trial comparing NET with letrozole plus palbociclib versus NCT, rates of RCB 0/1 were low in both arms (8% versus 16%), and were not associated with PFS or iDFS.29,30 In the exploratory pooled analyses of CAMINA02 and HORGEN trials, 20 both of which compared neoadjuvant anastrozole and fulvestrant in postmenopausal women with HR+ HER2− breast cancers, the pathological response assessed by the Sataloff classification was also not associated with recurrence-free survival (RFS).
Clinical and radiologic response
Previous large-sized NET trials reported discrepancies in the clinical response rates, ascertained by palpation, and the radiologic response rates,11,12,13,19 as all of these methods have some limitations for response assessment. For example, clinical response rates by palpation can be inaccurate for reasons, such as treatment-related changes in the tumor, and although MRI is known as the most accurate imaging method, the extent of residual disease might not be accurately delineated because of reasons including fibrotic change and non-mass enhancement. Furthermore, response evaluation in the axilla by imaging is a challenging issue.38,39 Although tumor shrinkage with neoadjuvant systemic treatment allows a reduction in the surgical extent and possibly facilitates conversion to breast-conserving therapy, it is unclear whether the clinical tumor response assessment can be a surrogate marker for survival in patients with breast cancer treated with neoadjuvant treatment, especially in the context of NET.
Preoperative Endocrine Prognostic Index
In the P024 trial, the baseline Ki-67 was not associated with RFS, whereas the post-NET Ki-67 was significantly associated with RFS. As such, a post-NET prognostic index called PEPI was suggested that incorporated other prognostic factors, including ER status, pathological tumor size, and nodal status. A low (0), intermediate (1–3), and high (⩾4) PEPI was significantly associated with relapse rates in NET-treated patients in the P024 trial data and was validated in the IMPACT trial dataset. 40 Moreover, the prognostic significance of the PEPI was validated in the Z1031 trial which compared neoadjuvant exemestane, letrozole, and anastrozole. The recurrence rates of patients with PEPI 0 were low without chemotherapy (after 5.5 years of follow-up, 4% versus 14% recurred among patients with PEPI 0 and >0, respectively), suggesting that these patients were likely to have favorable survival outcomes with endocrine treatment alone. 41 Based on these data, multiple recent clinical trials now include PEPI as the efficacy endpoint for NET. Considering the low pCR rates in patients with ER+ HER2− breast cancers and the significant differences in RFS according to the posttreatment PEPI, PEPI is now widely used as a marker to assess the NET response. 42
The ALTERNATE trial used a mPEPI, which excluded the ER status from the original PEPI. It was proposed as the study intervention included fulvestrant, a SERD, 21 and retrospective analyses of the P024/POL trial data suggested the prognostic value of mPEPI. 43 The primary endpoint, the ESDR (defined as the proportion of patients who acquired mPEPI-0 or pCR) did not differ between the three treatment arms (anastrozole, fulvestrant, and combination of both for NET) and long-term survival data is awaited.
Short-term endocrine response assessment with Ki-67
As the PEPI is available after 3–4 months or more of NET followed by surgery, the role of the early-treatment Ki-67 response was explored as a marker for an early endocrine response. The POL trial and IMPACT trial data showed that the Ki-67 after 2–4 weeks of NET were associated with PEPI, and a nonresponse indicated with a Ki-67 >10% was associated with a higher PEPI and poorer RFS.14,43,44 Accordingly, prospective clinical trial designs using Ki-67 after a brief course of NET to select patients with endocrine resistance and to guide subsequent neoadjuvant treatment strategies in ER+ BC patients were proposed. In the Z1031B trial, patients with Ki-67 >10% at 2–4 weeks post-NET were switched to NCT, which comprised about 20% of the patient population. 41 In the ALTERNATE trial where patients were randomized to the three NET arms (anastrozole, fulvestrant, or a combination of both), the Ki-67 was assessed at baseline, at 4- and 12-weeks post-NET, and at surgery. Patients with Ki-67 >10% at weeks 4 or 12 were recommended to switch to NCT 45 ; similar to the results of the Z1031B trial, approximately 22% of patients were included in this category. 46
The POETIC trial implemented a ‘window-of-opportunity’ design using a short course of preoperative NET with AI in patients with operable ER+ breast cancer. A total of 4480 patients were randomized (2:1) to 2 weeks preoperative AI followed by surgery or upfront surgery. The authors showed that paired baseline and 2-week post-AI Ki-67 is prognostic for survival (6Y recurrence rates by Ki-67 at baseline and Ki-67 at weeks, low-low: 4.3%; high-low: 8.4%; high-high: 21.5%). 47
The WSG-ADAPT HR+ HER2− endocrine trial combined the Oncotype DX® recurrence score (RS), clinical staging, and Ki-67 response after a short course of NET to ‘adapt’ perioperative treatment strategy in HR+ breast cancer patients. The authors suggested that patients who can forego chemotherapy can be successfully identified without a significant impact on the survival outcomes, as patients whose RS were intermediate (12–25) and were endocrine responders (Ki-67post <10% at 3 weeks) showed comparable survival outcomes as those of patients with low RS (<12) among patients with clinical N0–1 disease. 48 As discussed above, recent trials using novel agents such as CDK4/6 inhibitors also incorporate brief lead-in phases of 2–4 weeks of ET and analyze Ki-67post as a marker of early endocrine response to the study treatments.25–27,31
To summarize, Ki-67 assessment at 2–4 weeks post-ET allows early assessment of endocrine response and helps to tailor further neoadjuvant/adjuvant treatment strategy in ER+ breast cancer, while serving as a prognostic biomarker. Furthermore, continuing efforts have been made to standardize and automatize the analysis to improve the analytical validity of Ki-67. 49
Outcome of endocrine-resistant patients switched to NCT
Although the reported clinical progression rates during NET are not high (0–9%),7,8,25,28 prospective trials showed that approximately 20% of patients treated with NET were endocrine nonresponders, with Ki-67 >10% after 2–4 weeks of NET.21,41,48 These endocrine nonresponders remain a challenging subgroup. Adaptive clinical trial designs where these patients were switched to NAC yielded only unsatisfactory outcomes. The pCR rates after switching to NAC remain low (5–6%) in these patients and, despite additional chemotherapy, survival outcomes are inferior compared with that of endocrine responders (Table 4).
Outcome of nonresponders to ET after switching to chemotherapy.

dDFS, distant disease-free survival; ET, endocrine therapy; iDFS, invasive disease-free survival; NAC, neoadjuvant chemotherapy; NCT, neoadjuvant chemotherapy; NET, neoadjuvant endocrine therapy; NS, not significant; OS, overall survival; pCR, pathologic complete response; RFS, recurrence-free survival; RS, Oncotype DX® recurrence score.
Changes in the gene expression profiling
In the CORALEEN trial that compared 24 weeks of NET with ribociclib and letrozole versus NCT in 106 postmenopausal patients with luminal B breast cancer, genomic profiling by Prosigna® PAM50 ROR score was undertaken at baseline, day 15, and at surgery. NET resulted in comparable rates of ‘molecular downstaging’, showing a similar proportion of patients converted to low ROR at surgery (primary endpoint, 46% versus 47% with NET and NCT), suggesting the feasibility of ‘molecular downstaging’ as potential endpoints for neoadjuvant trials, although the long-term implications are undetermined. 28
Serum thymidine kinase 1 activity
An elevated level of serum thymidine kinase 1 (TK1) activity, a cell-cycle-regulated enzyme that is involved in DNA precursor synthesis and a marker of cell proliferation, 50 is known to be a poor prognostic factor in advanced breast cancer, 51 and its role as a predictor for endocrine resistance has been suggested. 52 In the NeoPalAna trial, patients with ER+ HER2− breast cancer received 4 weeks of anastrozole followed by 16 weeks of anastrozole and palbociclib. A significant reduction in the TK1 activity was observed on day 15 after palbociclib plus anastrozole, whereas no statistically significant differences in TK1 activity were observed after the anastrozole monotherapy treatment period. In addition, a high level of concordance was observed between Ki-67 level and serum TK1 activity (concordance level, 89.8%), suggesting the role of TK1 as a predictor for response to CDK4/6 inhibitors in the neoadjuvant setting. 53
Markers for patient selection for NET
Menopausal status
Most NET trials were conducted in postmenopausal patients and, although NET with AI showed comparable clinical response with NCT in postmenopausal patients, 4 the limited evidence from previous data indicated inferior response rates to NET compared with NCT in premenopausal patients.8,10 However, more recent trials showed clinically meaningful results, which suggest that the efficacy of NET in premenopausal can be improved with an appropriate choice of agents in combination with OFS and that the endocrine responsiveness, after a short course of preoperative endocrine treatment, can help patient selection among premenopausal patients (Table 5).
NET outcome in premenopausal patients.

AC, adjuvant chemotherapy; BCS, breast-conserving surgery; ER, estrogen receptor; DFS, disease-free survival; GnRHa, gonadotrophin-releasing hormone agonist; LN, lymph node; MRI, magnetic resonance imaging; N, number; NCT, neoadjuvant chemotherapy; NET, neoadjuvant endocrine therapy; NR, not reported; NS, not significant; OFS, ovarian function suppression; ORR, overall response rate; pCR, pathologic complete response; PEPI, Preoperative Endocrine Prognostic Index; RECIST, Response Evaluation Criteria in Solid Tumors; US, ultrasonography; WHO, World Health Organization response criteria.
The STAGE trial showed superior efficacy with anastrozole compared with tamoxifen (both with OFS) in premenopausal patients, similar to the postmenopausal patients (clinical response rates, 70% versus 51%, p = 0.004). 19 Dellapasqua et al. explored the role of degarelix, a GnRH antagonist, in comparison with triptorelin, a GnRH agonist for OFS. A shorter time to adequate OFS in the degarelix group was observed, although this did not result in clinically meaningful improvement of tumor response rates or Ki-67 reduction rates. 56
Most recently, pooled analysis of WSG-ADAPT and ADAPTcycle trial suggested that the endocrine response rates (defined as posttreatment Ki-67 ⩽10%) of premenopausal patients treated with OFS was similar to that of AI-treated postmenopausal patients, thus highlighting the need for OFS and the feasibility of NET in appropriately selected premenopausal patients. 57 Moreover, 5-year follow-up of the WSG-ADAPT trial data 48 suggested that, among the subgroup of patients ⩽50 years of age, patients who were RS16–25 endocrine responders showed comparable survival outcomes to that of patients with RS < 11 or 12–25 endocrine responders without chemotherapy – a subset of patients who often receive adjuvant chemotherapy in current clinical practice based on the results of TAILORx data. 58 Currently, a phase II trial exploring the role of palbociclib and letrozole combination with OFS in premenopausal patients, stratified by Oncotype Dx® RS, is ongoing (NCT03628066).
Taken together, NET had not been routinely recommended as a preferred neoadjuvant approach in premenopausal patients outside a clinical trial owing to insufficient data supporting its efficacy compared to NCT. Recent data indicate the possibility for improved efficacy of NET in these patients with appropriately selected regimens, along with OFS. In addition, assessment of early endocrine response using a short-course preoperative endocrine treatment, in combination with genomic assays, allows the identification of patients who would have favorable outcomes without chemotherapy. Although NET should be implemented cautiously in premenopausal patients, these strategies can facilitate patient selection and may broaden the indications for perioperative ET in this population.
ER expression level
In the P024 trial, a linear relationship between the ER expression, assessed by the Allred score, with response rates to NET (letrozole or tamoxifen) was observed. 11 Similarly, the IMPACT trial showed that a higher ER expression, which was assessed by the H-score, was associated with higher response rates to NET. 12 Based on these results, later trials such as Z1031 or ALTERNATE incorporated an Allred score ⩾6 in the inclusion criteria.15,45
Functional imaging
In a small substudy of the NEOCENT trial, the utility of 16α-18F-fluoro-17β-estradiol (18F-FES) positron emission tomography, a functional imaging method that allows visualization of ER expression status, for prediction of pathologic response to NCT has been suggested. In this feasibility analysis, none of the patients with baseline low FES uptake (non-FES avid tumors) responded to NET (0/5), whereas they responded to NCT (5/7). 59
Ki-67
Exploratory analysis of the GEICAM 2006-03 trial showed higher clinical response rates to NCT compared with NET in patients with higher baseline Ki-67 (>10%; 67% for NCT versus 42% for NET, p = 0.075), whereas patients with low baseline Ki-67 showed comparable response rates (63% for NCT versus 58% for NET, p = 0.74). 8 However, P024 trial data showed that baseline Ki-67 was not associated with RFS, whereas posttreatment Ki-67 showed significant association with RFS, and was therefore incorporated into a post-NET prognostic model, ‘PEPI’. 40
Compared with the limited use of baseline Ki-67 for endocrine response prediction, multiple trials now use the posttreatment Ki-67 level after 2–4 weeks of ET as a marker of early endocrine response, as discussed earlier. This early endocrine response evaluation using Ki-67 supports the clinical decision for subsequent perioperative systemic treatment strategy (e.g. whether to switch to chemotherapy),41,45,48 allows prognostication of patients with a short-term preoperative treatment,47,48,57 and serves as an earlier marker for the efficacy of investigational neoadjuvant regimens.25–27,32,31
Intrinsic subtypes
The intrinsic subtypes of breast cancer were initially suggested using microarray-based analyses in 2000. 60 The Prosigna gene signature assay was later developed by analyzing the differential expression of 50 genes (PAM50 gene signature) and is now used to differentiate intrinsic subtypes of breast cancers in practice, as well as to provide risk-of-recurrence (ROR) scores for predicting the probability of distant recurrence.61,62 Among patients with ER+ breast cancers, luminal subtypes are known to respond better to NET compared with non-luminal subtypes. In the Z1031 trial, all patients with basal-like and HER2-enriched subtypes demonstrated endocrine resistance with high posttreatment Ki-67 values after NET. 15 In the phase II LETLOB trial that compared the efficacy of letrozole with or without lapatinib in postmenopausal patients with ER+, HER2− breast cancers, non-luminal breast cancers showed significantly lower response rates to NET compared with luminal breast cancers. 63
Z1031 investigators also showed that, compared with those who had luminal B subtypes, a higher proportion of patients with luminal A breast cancer achieved endocrine response (PEPI 0) to neoadjuvant AI (27% versus 11%). However, the clinical response rate or BCS rate did not differ between the two groups. 15 In the CORALEEN trial, patients with luminal B subtypes showed substantial rates of ‘molecular downstaging’ with NET with ribociclib and letrozole, which was comparable to NCT (conversion rates to the low ROR group at surgery, 47% versus 46% with NET and NCT, respectively). However, the pCR rates in the breast and axilla were low in both arms (0% and 6% in the NET and NCT groups, respectively). 28 Similarly, the NeoPAL trial which compared NET with letrozole plus palbociclib for 19 weeks versus NCT in luminal B or node-positive luminal A breast cancers (approximately 89% of the study population had luminal B subtypes), reported low rates of pCR rates in both NET and NCT arms (4% versus 6%). In addition, survival outcomes (PFS or iDFS) did not differ between the two arms. 29
These data suggest that intrinsic subtypes assessed by gene expression profiles are associated with the effectiveness of NET, as non-luminal subtypes respond significantly less to NET compared with luminal A/B subtypes. Although response rates to NET were higher in luminal A subtypes, NET is a useful treatment strategy both in luminal A and B subtypes. In addition, recent data suggest that changes in the gene expression profiles, that is, ‘molecular downstaging’ can also be used as a marker for response to neoadjuvant systemic treatment.
Oncotype Dx® RS
The Oncotype Dx® RS, is a 21-gene-based assay originally developed for prognostication of tamoxifen-treated, node-negative ER+ patients. 64 It is now widely used for risk prediction for recurrence and guiding adjuvant chemotherapy decisions in ER+, lymph node-negative, or up to three lymph node-positive breast cancers.58,65 In the neoadjuvant setting, few studies suggested the association between baseline RS and NET response.
Ueno et al. 66 observed that a low RS (<18) at baseline was associated with higher clinical response rates to NET (59% versus 20%) as well as the BCS rates (91% versus 47%), compared with patients with a high RS (⩾31). In addition, the authors suggested that the combined pre- and posttreatment RS was associated with DFS, independent of other prognostic factors. 67 Similarly, in the transNEOS study, a translational analysis of the phase III NEOS trial in which postmenopausal ER+ breast cancer patients were treated with neoadjuvant letrozole for 6 months with or without adjuvant chemotherapy, participants with low baseline RS (<18) achieved higher radiologic response rates compared with patients with high RS (⩾31; 55% versus 22%). 68
A small pilot study by Bear et al. used the baseline RS score for neoadjuvant treatment selection, and patients with RS11–25 were randomized to either NET or NCT, whereas patients with RS < 11 and >25 received NET and NCT, respectively. Although limited by the small sample size, patients with low RS showed high response rates to NET (83%), whereas patients with intermediate RS had lower clinical response rates with NET than with NCT (50% versus 73%). 69
Finally, the WSG-ADAPT HR+ HER2− endocrine trial combined RS with clinical stage and endocrine response assessed by Ki-67 after 3 weeks of induction ET (Ki-67post) to guide systemic treatment decisions in pre- and postmenopausal ER+ HER2− breast cancer patients. 48 Among patients with N0–1 diseases, patients were treated with ET alone if their RS was low (<12; control arm) or intermediate (RS12–25) and responsive to ET (Ki-67post ⩽10%; experimental arm), whereas patients who were N2–3, N0–1 with high RS (>25), or N0–1 with intermediate RS(12–25) nonresponsive to ET (Ki-67post >10%) received chemotherapy. With 5 years of follow-up, comparable survival outcomes were observed between the control and experimental arm, without significant difference in the 5Y invasive DFS rates (94% versus 93%) and 5Y distant DFS rates (96% versus 96%), and this provides clinical evidence for perioperative strategy with ET alone without chemotherapy in selected patients with intermediate RS. As discussed earlier, the result was consistent in the subgroup of patients aged ⩽50 years. The subsequent trial, WSG-ADAPTcycle, aims to further broaden the indication for perioperative ET without chemotherapy in ER+ breast cancers by comparing the ribociclib–ET combination versus chemotherapy in ‘intermediate-risk’ ER+ HER2 breast cancers. These ‘intermediate-risk’ patients are those at higher risk compared with the ‘experimental arm’ of the previous WSG-ADAPT trial, such as patients with RS ⩽25 endocrine nonresponders, RS >25 endocrine responders with N0–1 disease, and RS ⩽25 endocrine responders with N2–3 disease. 70
Current use of NET in clinical practice and limitations
NET was significantly underutilized in clinical practice, and it was primarily considered for older patients with comorbidities. A National Cancer Database (NCDB) analysis showed that only a small proportion (1.2%) of patients with non-metastatic breast cancer received NET. 71 Although several clinical trials have generated promising efficacy data for NET in the past two decades, only a small increase in NET use was observed during the period. 72 These findings are attributed to the limitations of NET. First, NET requires longer time to achieve tumor response compared with NCT. In addition, in many NET trials, the primary endpoint was the clinical response rate, which is not an optimal surrogate marker for long-term survival. Furthermore, the decision for adjuvant chemotherapy after NET remains uncertain. Limited evidence is available for the clinical interpretation of genomic testing results from either surgical specimens or diagnostic biopsy specimens in the context of NET. However, more recent data provide insights for the appropriate adjuvant treatment decision by utilizing endocrine sensitivity and genomic biomarkers. Survival analyses of the Z1031 trial showed that although most patients who achieved PEPI 0 did not receive chemotherapy, these individuals still demonstrated excellent survival outcomes. 41 Importantly, the long-term survival data of the ALTERNATE trial, which omitted adjuvant chemotherapy for patients who achieved mPEPI 0 with NET, are eagerly awaited as these data will provide valuable information that could guide post-NET adjuvant treatment decisions. Furthermore, novel trial designs that incorporate brief course preoperative ET showed that early endocrine response assessment in combination with genomic assays may help adjuvant treatment decisions.47,57
NET during COVID-19 pandemic
Due to the scarcity of medical resources during the COVID-19 pandemic, guidelines recommended delaying of elective surgeries and to consider initiating NET if necessary. 73 A survey conducted among US physicians reported that approximately 91% of the respondents answered that their practices changed during the pandemic to consider delaying of surgery. 6 In alignment with these findings, a separate study observed a 4.4-fold increase in the utilization of preoperative systemic therapy in the initial phases of the pandemic, with a significant proportion of this change attributed to the use of NET. 74 Multiple evidence supported this approach by showing that delaying surgery with NET was feasible in the pandemic setting. For example, a multicenter cohort study in European countries showed that, with a median period of 53 days of preoperative ET, only 2% of patients required expedited surgery. 75 In addition, a matched historical cohort study showed that the outcome of early breast cancer patients treated with NET (median time to surgery, 68 days) was comparable to that of historical control who underwent upfront surgery (median time to surgery, 26.5 days). 76 Furthermore, a prospective observational study showed that patients with clinical node-negative breast cancer who received NET with multidisciplinary decision had similar rates for sentinel node positivity with historical control in the pre-COVID-19 era. 77
Summary and future directions
A growing body of evidentiary data supports the use of NET as an effective, safe alternative treatment option for ER+, HER2− breast cancer. Moreover, NET trials can facilitate the early evaluation of the clinical efficacy of newer agents, and novel NET regimens in combination with CDK4/6 inhibitors and other targeted agents are currently actively being explored (Table 6). The use of genomic assays and early evaluation of endocrine sensitivity based on the Ki-67 response in the preoperative setting helps us to select patients who may safely forgo chemotherapy without compromising clinical outcomes. These adaptive trial designs that incorporate the clinical and the molecular characteristics of each patient allow a more tailored perioperative treatment strategy in ER+ breast cancer. Trials now in progress, as well as further research, will help broaden the indications for NET.
Ongoing neoadjuvant ET trials.
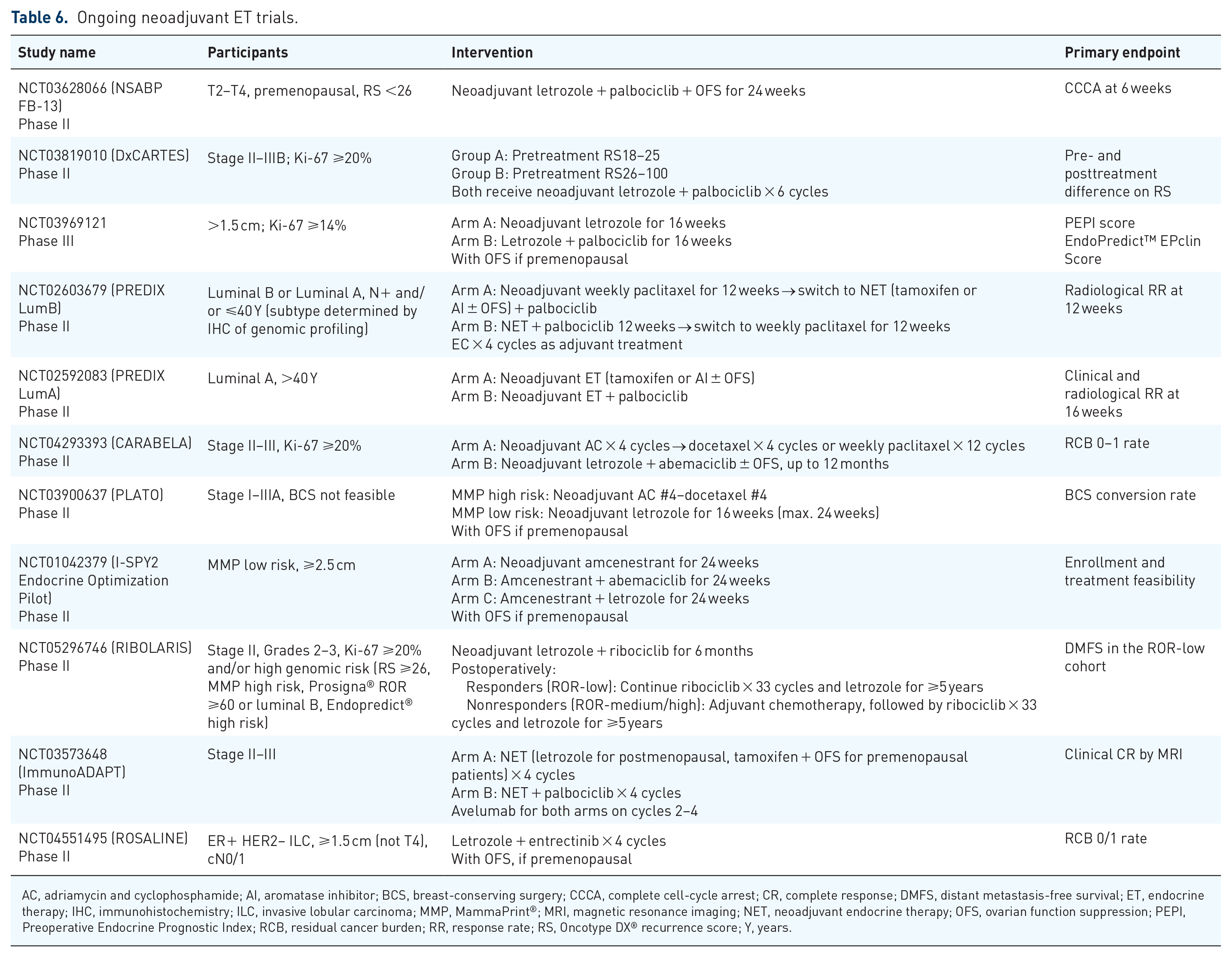
AC, adriamycin and cyclophosphamide; AI, aromatase inhibitor; BCS, breast-conserving surgery; CCCA, complete cell-cycle arrest; CR, complete response; DMFS, distant metastasis-free survival; ET, endocrine therapy; IHC, immunohistochemistry; ILC, invasive lobular carcinoma; MMP, MammaPrint®; MRI, magnetic resonance imaging; NET, neoadjuvant endocrine therapy; OFS, ovarian function suppression; PEPI, Preoperative Endocrine Prognostic Index; RCB, residual cancer burden; RR, response rate; RS, Oncotype DX® recurrence score; Y, years.
