Abstract
Background:
The coronavirus disease 2019 (COVID-19) pandemic was an unprecedented shock to the healthcare systems, and its consequences on managing rare cancers are unknown. We investigated COVID-19’s impact on the activity of sarcoma-labeled networks by comparing key indicators in 2019–2020 (before and during the pandemic, respectively).
Methods:
We compared the incidence of limb and trunk soft tissue sarcomas, surgery rate, surgery center, surgery quality, and surgery delays nationally and in various regions, focusing on the three most severely affected regions.
Findings:
In this study, sarcoma incidence did not decrease, and the tumor and patient characteristics were similar in both years. The number of patients who underwent surgery in the labeled centers increased significantly (63% versus 57%, p = 0.015), the rate of R0 resection increased (55% versus 47%, p = 0.004), and the rate of re-excision decreased (12% versus 21%, p < 0.0001). In the univariate analysis, the time to surgery was similar in both years. Cox regression analysis revealed that the factors associated with a longer time to surgery were age > 70 years (p = 0.003), retroperitoneal location (p > 0.001), tumor size (p < 0.001), deep tumors (p < 0.001), and regions (p < 0.001). However, we have observed an increase in the time before surgery in the regions most stroked by the COVID-19 pandemic.
Interpretation:
The model of the labeled center network for managing rare tumors was resilient. Paradoxically, the quality indicators improved during the pandemic due to the direct referral of patients with sarcomas to the labeled centers.
Summary:
This study shows that a nationwide network organization has made it possible to maintain care for these rare tumors during the pandemic.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic was a major source of stress and shock that deeply impacted the healthcare system. In France, the first COVID-19 outbreak occurred in November 2019. 1 The epidemic spread massively, first affecting the ‘Grand-Est’ and ‘Bourgogne-Franche-Comté’ (around the cluster of Mulhouse), ‘Hauts-de-France’ (around the cluster of Creil-Compiègne), and ‘Ile-de-France’ (Paris) regions. In 2020, to slow down the pandemic, the government implemented campaigns to promote social distancing, self-isolation, and the use of face masks (these became largely available in March and April 2020), and two almost successive national lockdowns (between 17 March and 11 May 2020 and between October 30 and December 15). During the first epidemic wave (January–May 2020), over 98,000 patients with COVID-19 were hospitalized (incidence rate of 146 per 100,000 inhabitants), of whom 20% died. The first vaccination campaign was initiated in January 2021.
In 2020, the healthcare system faced COVID-19 pandemic waves by deprogramming most planned non-urgent care. Access to nonhospital outpatient care was greatly reduced and disorganized. Within hospitals, resources were massively reallocated toward COVID-19 management, and major surgeries requiring critical care were postponed as much as possible. In addition, cancer screening reduced markedly. 2 During that year, shortcomings existed in the supply of anesthetics and muscle relaxants. 3
In an unprecedented crisis, the conditions for caring for patients with cancer, particularly rare cancers, warrant specific studies. Sarcomas require interdisciplinary diagnostic and therapeutic management in compliance with well-established international guidelines. 4 The cornerstone of sarcoma management is specialized and complex surgery. Ideally, this management is supervised by an interdisciplinary tumor board. Since 2010, the French National Cancer Institute has certified a network of 25 reference centers, ensuring that the national network oversees the interdisciplinary coordination of sarcomas. These centers include university hospitals, comprehensive centers, or associations. This network of labeled centers (NETSARC+) prospectively records its activity in a shared database. Thus, we aimed to study the COVID-19 pandemic’s impact on sarcoma management by comparing the sarcoma care pathway in 2019 (before the COVID-19 pandemic) and 2020 (during COVID-19, before access to vaccination). Regarding the massive cancellation of surgical procedures (whatever their indication) observed during the first lockdown, we hypothesized that COVID-19 severely affects sarcoma management and prolongs the delay in sarcoma care.
Patients and methods
Study overview
The study was in two phases: (i) a description of the organization and resources in each labeled center and (ii) an analysis of the sarcoma management pathway documented in the shared database. Regarding the first part, we administered a structured questionnaire to the head of the multidisciplinary board (MDB) at each labeled center. Analyzing this questionnaire allowed us to (i) describe the organizational and resource changes observed in 2020 and (ii) identify each MDB head that appeared critical in sarcoma management. The second part of this study was carried out using a shared network database. Since 2010, all newly diagnosed sarcomas (diagnosis made within or outside the network) or case studies by the MDB have been registered in a curated online national database approved by national health authorities [Commission Nationale Informatique et Liberté (CNIL), no. 910,390] (https://netsarc.sarcomabcb.org/). The database contains 60 items split into four themes: patient and tumor characteristics, diagnosis and second opinion by an expert pathologist, key management steps and follow-up, and successive presentations of the file and decision-making at the MDB discussion. A quality assurance program was established for these databases to ensure the quality of the recorded medical data, and clinical follow-up information was updated at least every 2 years. For this study, 2019 and 2020 data were verified, corrected, and updated for 2021. The cutoff date was 5 December 2022. This was a national multicenter retrospective study of a quasi-exhaustive cohort.
Eligibility criteria and study population definitions
The inclusion criteria were patients aged ⩾18, soft tissue sarcoma of the limbs, girdles, or trunk (external and internal trunk, including retroperitoneum). The exclusion criteria included benign tumors and sarcomas at other primary sites (the primary bone, visceral, head, and neck sarcomas). By definition, the ‘eligible patient population’ included all patients that met all eligibility criteria. More precisely, ‘Eligible patient population assessable for the primary objective’ were those with non-metastatic sarcomas (N0M0), discussed by the MDB, and with available surgical procedure information. In addition, we stratified the population according to French metropolitan regions (https://www.regions-et-departements.fr/regions-francaises).
Objectives, end points, and analysis
The primary end point was the time interval between diagnosis (date of first diagnostic procedure, such as biopsy or surgery) and surgery (date of first curative surgery). This time was estimated for two populations: newly diagnosed (regardless of the diagnosis sample; biopsy or surgical specimen) and those newly diagnosed via a biopsy. The time to surgery was the cumulative incidence using the Aalen–Johansen estimator to consider the competitive risk of death preoperatively. Cumulative incidence curves (2019 versus 2020) were compared using Gray’s test when applicable. Lastly, we used a cause-specific Cox model to identify factors associated with time to surgery. This analysis considered the time interaction identified from Schoenfeld residuals. The time interval between diagnosis and the first MDB discussion (day or post-diagnosis) was calculated in the eligible population using the Kaplan–Meier method and compared using the log-rank test. The median follow-up period was estimated using the reverse Kaplan–Meier method. We have carried out this analysis nationally and in the first most-affected regions (Grand-Est/Bourgogne-Franche-Comté, Ile-de-France, and Hauts-de-France).
Organization and resources declared by the centers were compared between 2019 and 2020 using Wilcoxon matched-pairs signed-rank and McNamar tests.
The demographic and clinical characteristics of the patients were compared by the diagnosis year using chi-squared tests for qualitative variables or the Wilcoxon–Mann–Whitney test for quantitative variables.
All statistical tests were two-sided, with α at 5%.
Statistical analyses were performed using SAS 9.4 (Institute Inc., Cary, NC, USA) software and the ‘survival’ package of R v.4.2.1.
Results
Questionnaires
We administered structured questionnaires to 25 labeled centers. This allowed us to compare the resources available for the diagnosis and surgical treatment of sarcomas in 2019 and 2020 (Supplemental Table 1). The resources needed for diagnostic pathways (imaging and biopathology) remained unchanged during the pandemic. The number of MDB sessions was similar between 2019 and 2020. In contrast, major challenges concerning surgical resources were observed. During the pandemic, there were waves of deprogramming, challenges in supplying propofol and midazolam, and setting up services dedicated to COVID-19 management in most labeled centers. The number of operating rooms available for sarcoma surgery markedly decreased [from a median (interquartile range) of 5 (IQR: 2–6) to 3 (IQR: 2–6)]. The MBDs scored items that seemed critical for sarcoma management. According to the chairs of the MDBs, the critical items most often retained were the number of available operating rooms (68%), deprogramming waves (64%), and the number of expert surgeons (60%). Lastly, most labeled centers were led by comprehensive cancer centers (56%), less involved in the COVID-19 pandemic than university hospitals.
Description of patients managed in 2019 and 2020
Overall, the study population included 2960 patients (Supplemental Figure 1) from 26 centers of the NETSARC network. The newly diagnosed cases were 1486 and 1474 in 2019 and 2020, respectively. Patient and tumor characteristics were similar between 2020 and 2019 (Table 1). Fewer females (52% versus 54%) were diagnosed with sarcoma in 2020 than in 2019, and more were grade 2 tumors (33.8% versus 30.6%). However, this was not significant. The number of cases diagnosed in the Hauts-de-France (99 versus 126) and Grand-Est/Bourgogne-Franche-Comté (151 versus 179) regions – the regions that were first affected by the pandemic – decreased slightly in 2020. Nonetheless, this was not significant (p = 0.0963). A full description of the number and percentages of new cases managed by centers and regions is given in Supplemental Table 2.
Patient and tumor characteristics: in 2019 and 2020.

Missing data: 121 (8.1%) in 2019 and 117 (7.9%) in 2020.
p, p Value of the two-sided Wilcoxon–Mann–Whitney test for quantitative variables and the chi-squared test for qualitative variables at the 5% significance level.
Operated patients
Among the 2436 patients (1225 and 1211 in 2019 and 2020, respectively) eligible and assessable for the primary end point, the proportion of operated patients (80% and 81% in 2019 and 2020, respectively; p = 0.3249) and their demographics and clinical characteristics were similar (Table 2). During the pandemic, the proportion of patients who underwent surgery in referral centers significantly increased (63% versus 57%, p = 0.0147). Simultaneously, surgeries after biopsy (70% versus 66%, p = 0.0446) and preoperative evaluations of surgical quality during MDB (40% versus 35%, p = 0.0138) significantly increased. The most striking observation was a significant increase in the R0 resection rate (56% versus 47%, p = 0.0039) and a decrease in the number of re-excisions (13% versus 21%, p < 0.0001). No changes were observed in the proportion of patients who received neoadjuvant therapy.
Surgical management of sarcoma.
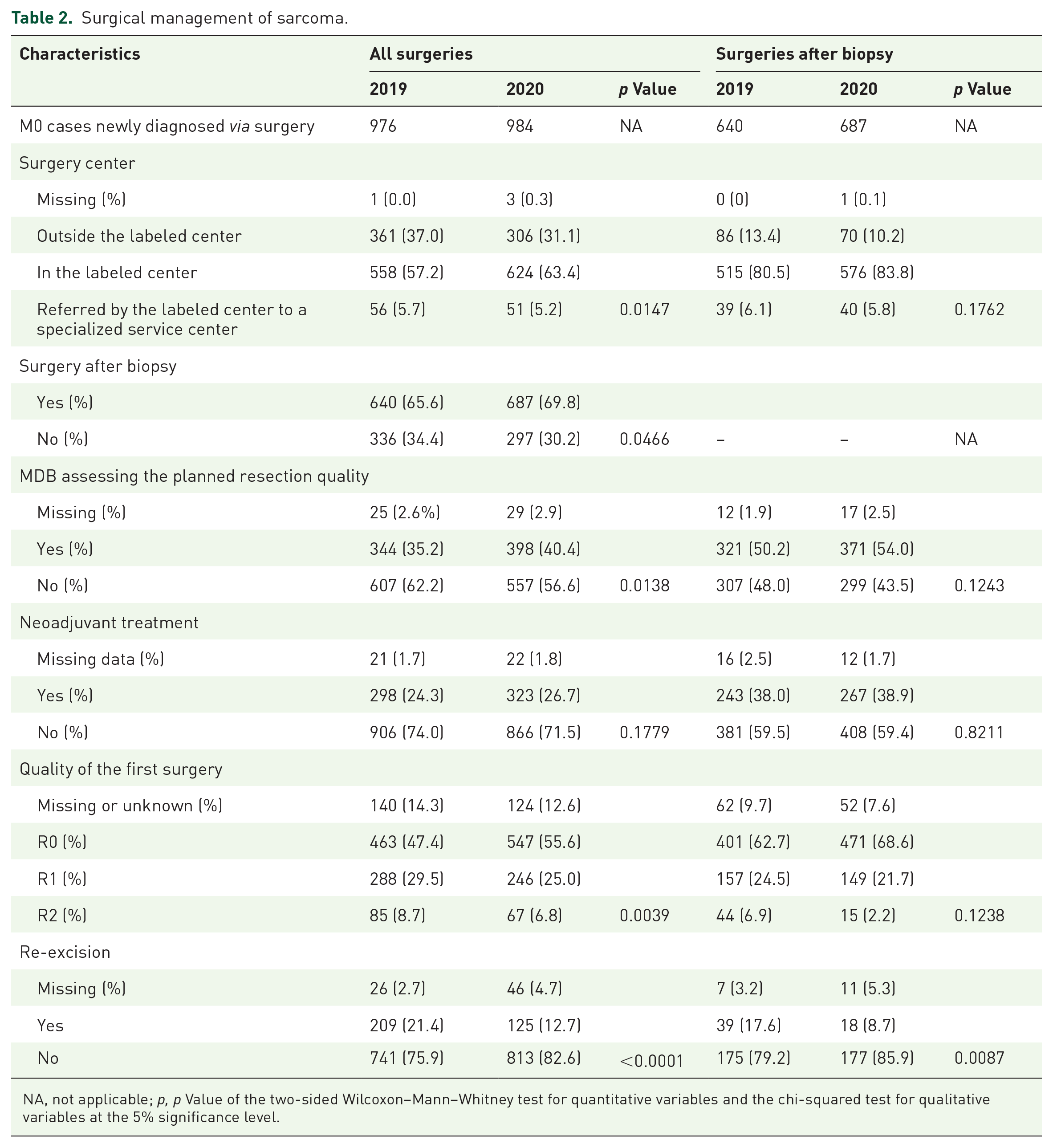
NA, not applicable; p, p Value of the two-sided Wilcoxon–Mann–Whitney test for quantitative variables and the chi-squared test for qualitative variables at the 5% significance level.
Most surgical procedures after biopsy were performed in labeled centers (81% and 84% in 2019 and 2020, respectively; p = 0.1762). There was also a decrease in the number of re-excisions performed in 2020 (8.7% versus 17.6% in 2019; p = 0.0087).
Delays
The median follow-up was 755 days (95% CI: 677–806) in 2019 and 321 days (95% CI: 289–363) in 2020. The median time interval between the diagnostic procedure and the first MDB discussion was 28 days (95% CI: 27–30) in 2019 and 25 days (95% CI: 22–26; p = 0.0038) in 2020. The median time interval between the diagnostic procedure and the first MDB discussion for M0 sarcomas was 29 days (95% CI: 27–32) in 2019 and 25 days (95% CI: 23–27; p = 0.0023) in 2020. The cumulative incidence of operated patients was 53% (95% CI: 50–56) at 3 months in 2019 and 2020, and 75% (95% CI: 72–78) versus 80% (95% CI: 77–82) at 6 months in 2019 and 2020, respectively (Gray’s test was inapplicable) [Figure 1(a)]. Among the operated patients, the median delay between surgery and diagnosis was 46 days (Q1–Q3: 0–132) in 2019 and 53 days (0–128) in 2020 (p = 0.3558). Table 3 summarizes factors associated with the time to surgery according to the cause-specific Cox model. Factors associated with increased time to surgery were diagnosis in 2020 (p < 0.001), age > 70 (p = 0.003), retroperitoneal primary site (p < 0.001), tumor size (p < 0.001), profound tumor (p < 0.001), and the region (p < 0.001).
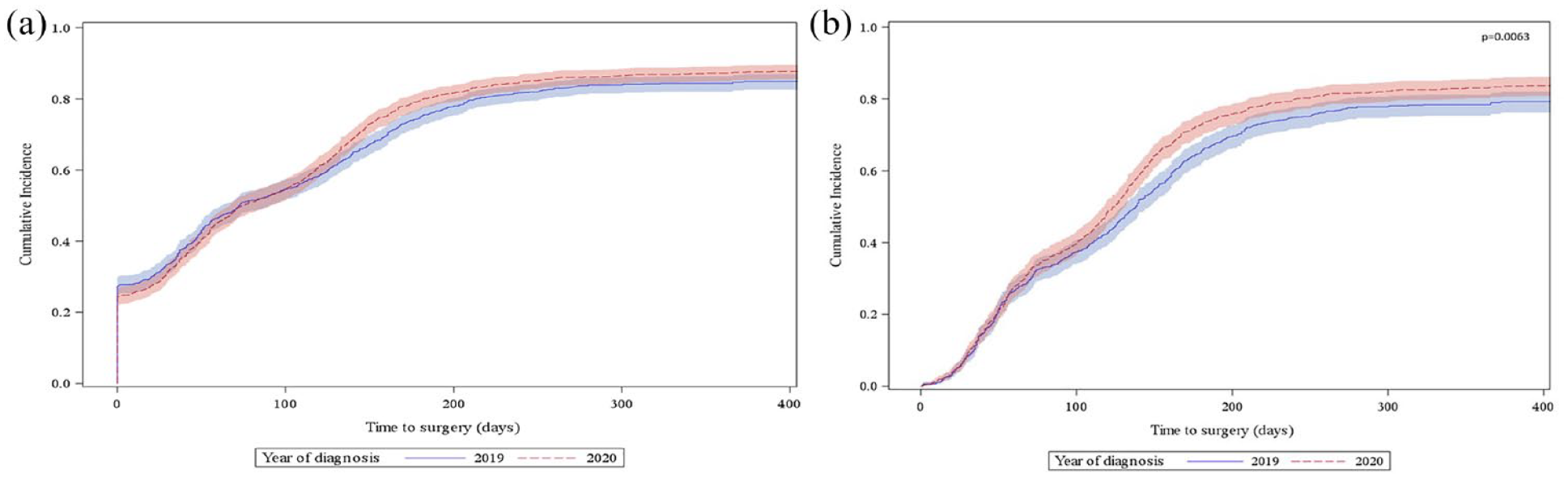
Cumulative incidence functions of surgery (Aalen–Johansen estimator). (a) All surgeries and (b) surgeries after the previous biopsy.
Factors associated with the time to surgery in patients with M0 sarcoma: multivariate cause-specific Cox model (n = 2191).
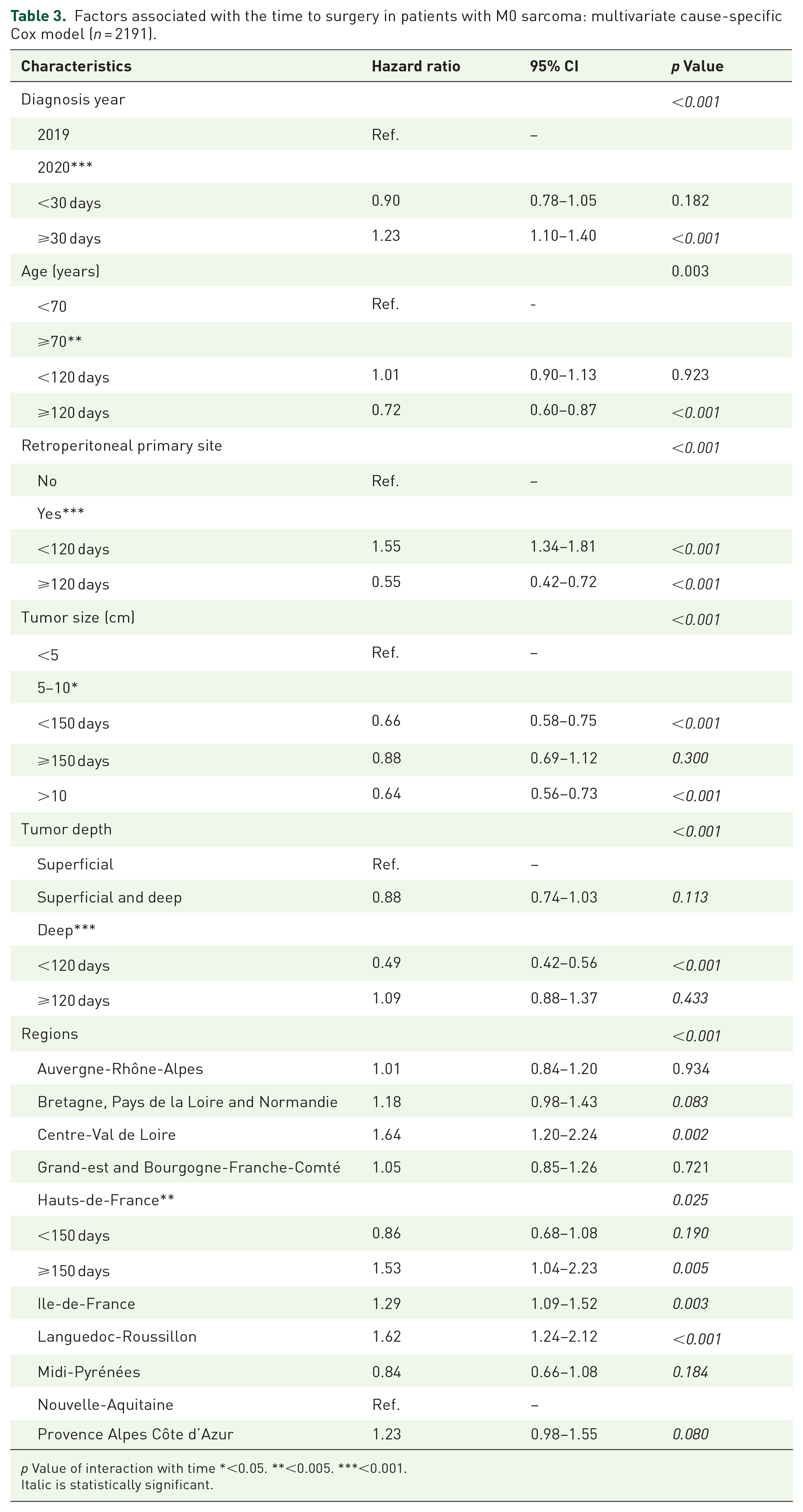
p Value of interaction with time *<0.05. **<0.005. ***<0.001.
Italic is statistically significant.
By stratifying the regions, the cumulative incidence of operated patients at 6 months in 2019 and 2020 were 79% (95% CI: 73–84) versus 86% (95% CI: 80–90; Gray’s test was inapplicable) in Ile-de-France, 67% (95% CI: 58–74) versus 72% (95% CI: 63–80; Gray’s test was inapplicable) in Grand-Est/Bourgogne-Franche-Comté and 72% (95% CI: 62–80) versus 88% (95% CI: 78–94; Gray’s test was inapplicable) in Hauts-de-France.
Among previously biopsied patients with M0 sarcoma discussed by the MDB, the cumulative proportion of operated patients was 35% (95% CI: 32–38) versus 37% (95% CI: 34–41) at 3 months and 66% (95% CI: 62–69) versus 73% (95% CI: 70–76) at 6 months in 2019 and 2020, respectively (Gray’s test p = 0.0063) [Figure 1(b)]. Among the operated patients, the median delay between surgery and diagnosis was 104 days (Q1–Q3: 47–161) in 2019 and 100 days (48–146) in 2020 (p = 0.1967). Table 4 summarizes the factors associated with time to surgery among patients with M0 sarcoma discussed by the MDB. They include age at diagnosis >70 (p = 0.001), retroperitoneal primary site (p < 0.001), tumor size (p = <0.001), profound tumor (p < 0.001), and the region (p < 0.001).
Factors associated with the time to surgery among patients with previously biopsied M0 sarcoma (n = 1632).

p Value of interaction with time <0.001 for all except tumor size (p = 0.062).
Italic is statistically significant.
By stratifying the regions, the cumulative incidence of operated patients at 6 months in 2019 and 2020 were 68% (95% CI: 60–75) versus 80% (95% CI: 73–85; Gray’s test was not applicable) in Ile-de-France; 56% (95% CI: 46–65) versus 65% (95% CI: 54–74; Gray’s test was not applicable) in Grand-Est/Bourgogne-Franche-Comté; and 68% (95% CI: 57–77) versus 86% (95% CI: 75–93; Gray’s test was not applicable) in Hauts-de-France.
Discussion
This study’s results contradict our initial hypotheses. As expected, the analysis of the questionnaire administered to the MBDs revealed profound changes in the organization of the surgical departments; however, the number of sarcomas diagnosed did not decrease. Also, we observed no decrease in the proportion of operated cases, an increase in neoadjuvant treatment use, or an increase in the time to surgery, even in the region most severely affected by the pandemic.
Since 2010, under the auspices of the French National Cancer Institute, the mission of the NETSARC network has been the structuring of sarcoma management on a national scale by setting up MBDs open to external physicians, disseminating guidelines, and providing a free second pathological opinion for pathologists outside the network. First, unlike other models like the Non small cell lung cancer (NSCLC), we did not see the difference in tumor size (p = 0.4454), depth (p = 0.2042) and the presence of metastasis (p = 0.5739) between 2019 and 2020. 5 In 2020, we observed a direct referral of patients for care to labeled centers, with a corollary of improved quality of care indicators. By contrast, the proportion of cases directly managed in labeled centers increased in the same year. This suggests that tertiary hospitals facing the pandemic as the first line immediately referred patients with sarcoma to labeled centers. More precisely (Table 2), the proportion of patients operated on in reference centers increased from 57 to 63%, and that of patients operated on after biopsy increased from 66 to 70%. Files for which the MDB preoperatively estimated the expected resection quality increased from 35 to 40%, the R0 rate increased from 47 to 55%, and the re-excision rate decreased from 21 to 12%. Significant and dramatic changes occurred faster than those observed over the past 10 years. 6 Overall, the cumulative incidence of interventions was similar between 2019 and 2020 (Figure 1). The interaction of some clinical profiles (age > 70 years, retroperitoneal location, large tumor size, and deep tumors) with time must be considered when analyzing the final results (Table 3). These profiles correspond to clinically challenging situations requiring a longer preoperative preparation time (operative prehabilitation) or more morbid procedures requiring the ensured availability of resuscitation beds, which was critical during the pandemic. Finally, the severity of the first COVID-19 wave did not explain the regional disparity in treatment times (Table 3). Indeed, the preoperative delays were longer in Hauts-de-France and Ile-de-France but not in Grand-Est/Bourgogne-Franche-Comté, the first three regions hit by the first wave. Furthermore, the cumulative incidence of surgery at 6 months did not decrease in these three regions in 2020. Overall, these data suggest the strong resilience of the network organization of labeled centers.
We hypothesized that chemotherapy indications might have been slightly modified in patients with operable non-metastatic sarcoma owing to surgery delay. However, we did not observe an overall increase in the time to surgery or neoadjuvant treatment indications.
Comparing this study’s results with data from the literature is challenging and may be inappropriate since healthcare systems differ based on the countries, and their strategies toward the pandemic have been different. The COVID-19 epidemic has profoundly disrupted both cancer care and research, to the point that learned societies have had to guide clinicians to face this unprecedented crisis.4,7 For instance, in an Indian study, our colleagues also observed an increased referral of patients with sarcoma to the expert center (in Mumbai, from 42 to 53%) and decreased time to decision-making (from 36 to 24 days). However, in this study, the number of cases decreased dramatically by approximately 70% during the pandemic. 8 Two other European studies have stressed an increase in the time to diagnosis. In a single-center study of 87 patients, our Polish colleagues revealed an increase in the time from initial symptoms to pathological diagnosis in patients with bone sarcoma (from 7 to 10 months). 9 In a single-center study conducted in Rome, our Italian colleagues revealed, in a cohort of 372 patients, a significant increase in the time of diagnosis (from 90 to 103 days) without a significant increase in the time to treatment (from 144 to 151 days). 10 Lastly, an international multicenter study stressed that stringent lockdown was associated with a major decrease in cancer surgeries such as sarcoma surgery (413 newly diagnosed sarcoma, odds ratio 0.67 (95% CI: .59–0.67) for non-surgical management during stringent lockdown). 11 In France, during the first pandemic year, there were two stringent lockdowns (17 March–11 May 2020 and 30 October–15 December). Their impact on cancer surgery has not been fully analyzed; however, a recent study based on the analysis of a national database of hospital discharge reports suggested that the impact on cancer surgery depends on the primary site. The gap in reported activity in 2020 compared to 2019 was approximately −4.5 to −5.7%, representing 3501–4720 cancer surgical procedures. 12 Our data suggest that this gap was not observed in soft tissue sarcoma surgery with the structured rare cancer network established in 2010. As such, it creates an interesting organizational model for resilience in such disruptive epidemics.
This study had some limitations. We did not study all types of sarcomas because we selected a homogeneous population. Therefore, this study did not consider pediatric cases, bone sarcomas, or all visceral sarcomas. However, more specific studies are required. This study did not consider cases without a pathological diagnosis. Nonetheless, it is unlikely that many cases were missed in 2020, as the incidence was similar to that in 2019. Finally, we do not currently have survival and mortality data that will require follow-up and that will analyze in a future study.
Overall, we observed no deleterious effects of the pandemic on soft tissue sarcoma management in France. The network of labeled centers put in place appears to be particularly resilient.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231192400 – Supplemental material for Impact of the coronavirus disease 2019 pandemic on sarcoma management in France: a 2019 and 2020 comparison
Supplemental material, sj-docx-1-tam-10.1177_17588359231192400 for Impact of the coronavirus disease 2019 pandemic on sarcoma management in France: a 2019 and 2020 comparison by Nicolas Penel, Coralie Cantarel, Claire Chemin-Airiau, Françoise Ducimetiere, François Gouin, François Le Loarer, Maud Toulmonde, Sophie Piperno-Neumann, Carine Bellera, Charles Honore, Jean-Yves Blay and Simone Mathoulin-Pelissier in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to acknowledge the heads of the multidisciplinary sarcoma boards from each labeled center. Loïc Chaigneau (Besançon University Hospital, Besançon); Corinne Delcambre-Lair (Centre François Baclesse, Caen); Pascale Dubray-Lngeras (Centre Jean Perrin, Clermont-Ferrand); Nicolas Isambert (Centre George François Leclerc, Dijon); Fabrice Fiorenza (Limoges University Hospital, Limoges); Florence Duffaud (Marseille University Hospital, Marseille); François Bertucci (Institut Paoli Calmette, Marseille); Didier Cupissol (Institut Régional du Cancer Montpellier); Maria Rios (Centre Alexis Vautrin, Nancy); Emmanuelle Bompas (Institut de Cancérologie de l’Ouest, Nantes); Antoine Thyss (Centre Antoine Lacassagne, Nice); Philippe Anract (Hôpital Cochin, Paris); Céleste Lebbe (Hôpital Saint-Louis, Paris); Jean-Pierre Lotz (Hôpital Tenon, Paris); Jean-Christophe Eymard (Institut Jean Godinot, Reims); Christophe Perrin (Centre Eugène Marquis, Rennes); Cécile Guillemet (Centre Henri Becquerel, Rouen); Jean-Emmanuel Kurtz (Strasbourg University Hospital, Strasbourg); Thibault Valentin (Oncopôle Toulouse); and Philippe Rosset (Hôpital Trousseau, Tours).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
