Abstract
In recent years, major advances have been made toward the individualization of epithelial ovarian cancer care, leading to an overall improvement of patient outcomes. However, real-life data indicate that the oldest populations do not benefit from this, due to aspects related to cancer (more aggressive histopathological features), treatment (i.e. frequently suboptimal), and the host (increased toxicities in patients with lower physiological reserve). A specific risk–benefit perspective should therefore be taken when considering surgery, chemotherapy, and maintenance treatments: the decision for cytoreductive surgery should include geriatric vulnerability and surgical complexity, neo-adjuvant chemotherapy being an option when primary surgery appears at high risk; carboplatin paclitaxel association remains the standard even in vulnerable older patients; and bevacizumab and poly(ADP-ribose) polymerase inhibitors maintenance are interesting options provided they are prescribed according to their indications with a close monitoring of their toxicities. Future studies should aim to individualize care without limiting access of older patients to innovation. A specific focus is needed on age-specific translational analyses (focusing on tumor mutational burden and impaired biological pathways), a better patient stratification according to geriatric parameters, an adaptation of both oncological treatment and geriatric interventions, and treatment adaptations not a priori but according to formal pharmacokinetic data.
Introduction
Epithelial ovarian cancer (EOC) remains the most lethal gynecological malignancy in the Western world despite its decreased incidence over recent decades related to the protective impact of all types of hormonal contraception and the advent of targeted therapies.1,2 Although survival of EOC is increasing, this is more pronounced among younger patients 3 and the prognosis remains markedly poor in older patients. 4 There is therefore a need to draw attention to the inequalities in diagnosis and treatment management in older populations. However, the lack of data specific to older patients, seldom included or highly selected in pivotal trials,5–8 and the fear of excessive toxicity, 9 may explain these inequalities. Furthermore, treatment strategies for older patients are based on subgroup analyses of pivotal randomized trials,10,11 prospective real-life unselected population-based studies,12–14 retrospective studies, 15 and specific clinical trials conducted in older patients,16,17 but these seldom integrate assessment of geriatric covariates. In this context, it appears useful to describe the available evidence on cancer characteristics at diagnosis in older patients, the general impact of age on outcomes, and all successive treatment steps.
Impact of age and geriatric factors on cancer characteristics and treatment outcomes
Where are we now?
Older age has been reported to be a risk factor for excess mortality in several population-based studies.12,18–20 For instance, Pectasides et al. reported that age ⩾70 years is an independent risk factor for premature death, along with FIGO stage III–IV, performance status >1, and residual disease >2 cm 18 ; in a Danish national cohort Jørgensen et al. 12 reported that older age was independently associated with a lower progression-free survival (PFS) and overall survival (OS); in an analysis of the Surveillance, Epidemiology, and End-Result End Results (SEER) American database Wright et al. reported that older age was associated with an increased the risk of premature death both in FIGO stage II, and III–IV cancers at 1 and 5 years; in another analysis of the SEER database Urban et al. found an increased risk of death at 90 days and a decreased OS at 1 year 20 (Table 1).
Impact of age and geriatric factors on cancer characteristics and treatment outcomes.

ADL, Activities of Daily Living; ASA, American Society of Anesthesiologists, d: days; GINECO, Groupe d’Investigateurs Nationaux pour l’Étude des Cancers de l’Ovaire et du sein; GVS, geriatric vulnerability score. (ADL < 6/6, IADL < 25/27, HADS > 14/42, lymphopenia < 1 G/L, albuminemia < 35 g/L); HADS, Hospital Anxiety and Depression Score; HR, hazard ratio; IADL, Instrumental ADL; NACT, neo-adjuvant chemotherapy; OR, odds ratio; OS, Overall survival; PS, performance status; ref., reference; SEER, Surveillance, Epidemiology, and End-Result End Results.
The reasons for such poor outcomes could be cancer-related, treatment-related, and host-related. For instance, age seems to impact cancer characteristics, as reported by Yancik (1993) who found a greater incidence of mixed tumors, high-grade serous sarcomas, and carcinosarcomas 21 ; Petignat et al. (2004) in a hospital database reported a significantly greater proportion of mixed tumors and a lower proportion of differentiated tumors in patients aged ⩾70 years. 22 Unfortunately, descriptive studies on histological characteristics of ovarian cancers according to age are sparse; in particular, the proportion of homologous recombination deficient (HRD) tumors according to age could be of major interest considering the advent of poly(ADP-ribose) polymerase inhibitors (PARPi) and the putative impact of HRD profiling on the risk/benefit ratio of cytoreductive surgery in the oldest old.
Age also impacts cancer treatment as it induces both a priori and a posteriori treatment adaptation. A priori treatment adaptation refers to the classical under treatment observed in oncogeriatrics. Specifically for surgery, procedures are performed less frequently with increasing age, and, when performed, lead less frequently to a complete – or even optimal – cytoreduction; in addition, they are more frequently performed in smaller centers, by non-specialist surgeons, and in emergency contexts. 27 A posteriori treatment adaptation refers to less complex than planned surgical procedures being performed by fear of complications, 9 but also to the high rate of post-operative morbidity, leading frequently to a delay in the initiation of adjuvant chemotherapy, a reduction in its dose-intensity,19,23 and the more frequent use of non-standard chemotherapy regimens (such as monotherapies). 26 Non-standard regimens are also more frequently used upfront a priori, and hematological and non-hematological toxicities lead to more frequent treatment delays, decreased doses, and premature discontinuations.8,10,23,34–38 Moreover, cancer and its treatments increase the onset and severity of comorbidities such as hypertension, congestive heart failure, thrombo-embolic events, infections, and anemia. 39
In parallel, age impacts the general performance of the patients, the presence of comorbidities, and geriatric vulnerability factors, which in turn may impact prognosis either directly or by an increased risk of treatment complications. With regard to geriatric vulnerability factors, it is of note that in 2013 the Groupe d’Investigateurs Nationaux pour l’Étude des Cancers de l’Ovaire et du sein (GINECO, National Investigators’ Group for Studies in Ovarian and Breast Cancer) developed the geriatric vulnerability score (GVS) that includes five vulnerability covariates: activities of daily living (ADL) score < 6/6; instrumental ADL (IADL) score < 25/27, albuminemia < 35 g/L, lymphopenia < 1 G/L and Hospital Anxiety and Depression Score (HADS) score > 14/42; patients being considered as vulnerable if they have at least three of these parameters (GVS ⩾ 3). 40 This score was recently prospectively validated as having a robust prognostic performance for OS regardless of the type of chemotherapy administered, allowing the stratification of populations for clinical research and orientating the geriatric interventions 42 to optimize multidisciplinary care planning. 43
What to do next?
As discussed above, histological data specific to older patients remain sparse. Future subgroup analyses of published or ongoing pivotal studies investigating targeted therapies, including translational analyses, should focus on age-specific analyses of tumor mutational burden and impaired biological pathways. In addition, future trials should gather geriatric covariates (among the subgroup concerned), as it was the case recently in the PAOLA1 trial that included the GVS assessment. However, older-specific prospective trials are also needed to focus on specific challenges related to the geriatric population, such as malnutrition, sarcopenia, polypharmacy, etc., as the more vulnerable patients are usually excluded from randomized studies 44 ; future trials should focus on the adaptation of oncologic treatment strategies according to geriatric assessment, geriatric interventions, and complex interventions and care pathways
Surgery
Where are we now?
The standard of care for ovarian cancer consists of primary cytoreduction followed by platinum-based chemotherapy. Residual tumor after surgery is an independent negative prognostic factor for survival45–47; in the case of the absence of residual disease, the prognosis of older patients is the same as their younger counterparts, but in the case of macroscopic residue the negative effect on survival is greater in older patients. 48 However, complete surgery is less frequently possible in older patients: complete cytoreductive surgery was observed in only 21.7–25% of patients aged ⩾80 years in the SEER database.49,50 Moreover, the rate of completion of a full medical-surgical sequence drops considerably in this population33,51; according to Warren et al. it was only 18.9% in patients aged ⩾75 years. 33
Age is associated with higher rate of medical comorbidities and is an independent risk factor for post-operative morbidity and mortality; advanced ovarian cancer surgery is a complex and heavy procedure that may be challenging to perform in frail patients. 52 The value of surgery depends both on tumor characteristics and on the patient’s health status. For instance, in a cohort of 576 consecutive patients from four centers who had primary cytoreductive surgery for FIGO stage IIIC–IV tumors, a small group of patients (n = 38) aged ⩾75 years with a high tumor dissemination load or FIGO stage IV, and a poor performance status [American Society of Anesthesiologists (ASA) score ⩾3] or low preoperatory albumin level (<3.0 g/dL) had a very poor outcome; their OS reached 17 months while it was 40 months in the total cohort. 53 In selected populations, however, the worse post-operative morbidity profile in older patients was not found as no significant difference in terms morbidity and mortality rate between these and their younger counterparts was found in two large series.25,54 Recently a study reported 70% complete cytoreduction surgery in the old (aged ⩾ 70 years) and oldest old (aged ⩾ 80 years) populations with an acceptable morbidity rate 55 ; postoperative complications and geriatric deconditioning may reduce the dose intensity of further chemotherapy and lead to compromised outcome. 56 Neo-adjuvant chemotherapy (NACT) is an appropriate option in patients with high tumor load, unresectable disease, significant medical co-morbidities, or poor performance status; this also provides time to complete the prehabilitation program.57–59 Tumor load determines the level of surgical complexity and thus is related to post-operative morbidity. The expertise of the team has a major impact on the probability to complete high quality surgery and the ability to diagnose and treat effectively post-operative complications; yet older patients are less likely to undergo surgery in a university hospital, and cancer complications are more frequently managed by non-oncologists and on an emergency basis for occlusion, perforation, or infection). 27 The treatment plan must consider the risk/benefit ratio of cytoreductive surgery, considering an excess in short-term (perioperative) morbidity and an equivalent benefit over the long term in the absence of post-operative residue.12,24 Hence, the multidisciplinary decision for surgery should include tumor burden and surgical complexity, as well as the level of expertise of the surgical and the medical team, comprehensive geriatric and surgical assessment, and the patient’s motivation for surgery. Preoperative assessment should aim to identify patients at higher risk of impaired outcome and qualify the personal involvement of the patients in her treatment plan including nutritional and functional prehabilitation as well as her adhesion to an enhanced recovery after surgery program.
In a recent scoping review on prehabilitation to improve postoperative outcomes in patients undergoing cytoreductive surgery followed by hyperthermic intraperitoneal chemotherapy Strijker et al. provided an overview of modifiable preoperative risk factors for patient outcomes: nutritional status and radiological sarcopenia, performance status [PS, either Eastern Cooperative Oncology Group (ECOG) PS, World Health Organization (WHO) PS, or ASA score], smoking history, health-related quality of life and depression. 60 Nutrition, functionality enhancement, and psychological stress reduction (and for certain authors smoking cessation) are, independently of age, the pillars of prehabilitation 61 and should be proposed to older patients who are expected to obtain the most benefit since post-operative outcomes are poorer and the improvement of physical performance is higher in frailer patients, independently of cytoreductive surgery context. 62 However, a randomized study evaluating the impact of prehabilitation over post-operative rehabilitation only for frail older patients with colon cancer failed to demonstrate any benefit, 63 leading to numerous comments and hypotheses.64–69 Among these, the primary endpoint of the trial, that is, surgical complications according to the Clavien-Dindo classification, 63 should be questioned since older age induces mostly an increase in medical post-operative complications and geriatric events 70 ; this led some authors to consider the National Cancer Institute Common Terminology Criteria for. Adverse Events classification as being better suited for the evaluation of morbidity for such trials 71 or the return to intended oncological therapy 72 , that is frequently postponed after cytoreductive surgery in the older population. In addition, prehabilitation protocols could be adapted to the geriatric population, with the inclusion of specific geriatric interventions such as pharmaceutical optimization, bridging interventions for hospital-to-home transition, and by adapting the physical and nutritional rehabilitation programs to the specificities of the older patient.73–75 Specific attention should be particularly paid to the adherence of the patient to the prehabilitation program, 76 but also of the surgical team to the enhanced recovery after surgery program.
What to do next?
The older population will probably benefit the most from the ongoing international awareness on the need to perform cytoreductive surgery for ovarian cancer in centers specialized in gynecological surgery – with the definition of quotas. Ongoing international, national and regional recommendations will be important relays for such awareness, since older patients are frequently prone to prefer smaller treatment centers. A constant effort must be made to promote prospective older-specific studies investigating surgical strategies and to offer the older population the benefits of innovations, both considering surgical techniques, hospital organization, enhanced recovery after surgery programs, and prehabilitation.
Chemotherapy
Where are we now?
When to treat?
Most patients with EOC, and in particular those aged ⩾70 years, have an advanced stage at diagnosis (FIGO stage III–IV). Advanced disease and comorbidities might often prevent upfront surgery, and NACT is an alternative treatment option (Table 2) that must be considered after assessment of both resectability of tumor and operability of the patient.
Impact of age and geriatric factors on treatment strategies and outcomes.
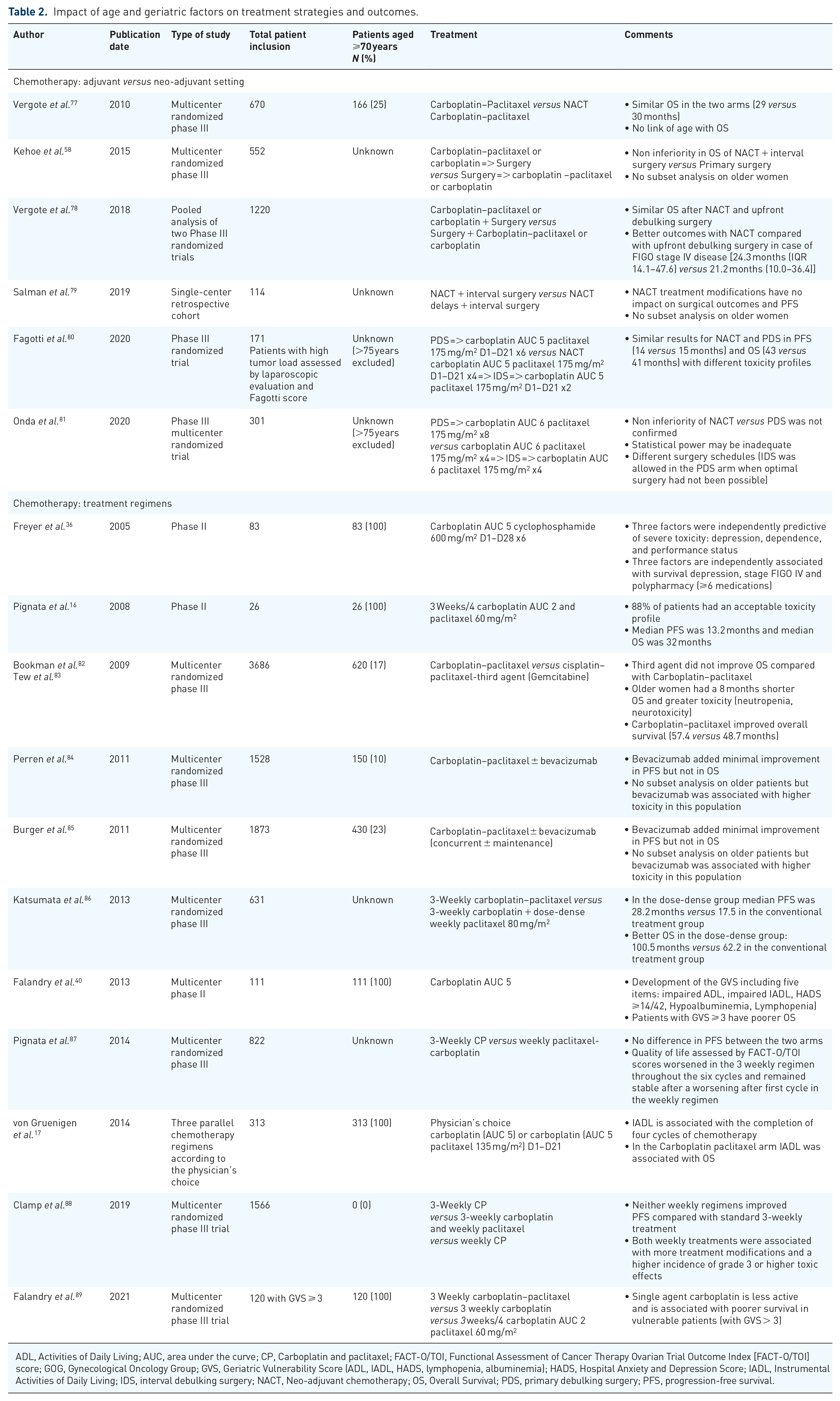
ADL, Activities of Daily Living; AUC, area under the curve; CP, Carboplatin and paclitaxel; FACT-O/TOI, Functional Assessment of Cancer Therapy Ovarian Trial Outcome Index [FACT-O/TOI] score; GOG, Gynecological Oncology Group; GVS, Geriatric Vulnerability Score (ADL, IADL, HADS, lymphopenia, albuminemia); HADS, Hospital Anxiety and Depression Score; IADL, Instrumental Activities of Daily Living; IDS, interval debulking surgery; NACT, Neo-adjuvant chemotherapy; OS, Overall Survival; PDS, primary debulking surgery; PFS, progression-free survival.
In 2015 the non-inferiority of NACT and interval surgery compared to primary surgery was reported 58 ; this can be considered as a safe alternative treatment to achieve complete cytoreductive surgery in unfit patients or those at perioperative risk, or when surgical complexity is deemed at high risk of post-operative deconditioning. Using the SEER database Thrall et al. 90 reported in 2011 a high risk of 30-day mortality in patients aged ⩾75 years with either FIGO stage IV, or stage III and ⩾1 comorbidity, leading to advise avoiding primary surgery in these populations; in this study NACT reduced 30-day mortality by 3-fold in patients aged ⩾65 years. In 2018 the pooled analysis of the two main randomized trials published in 2010 by Vergote et al. 77 and in 2015 by Kehoe et al. 58 confirmed with long-term follow-up that upfront surgery and NACT achieved similar results in terms of OS in women with EOC; 78 it also confirmed that patients with stage IV disease have better OS with NACT. Nevertheless, there is no data concerning older patients in the trial reported by Kehoe et al., 58 and there were only 166 (out of 670 patients) women aged ⩾70 years in the trial reported by Vergote et al. 77 and no link was observed between age and OS. In 2020 Fagotti et al. 80 reported similar achievements in patients with high-load tumor assessed by laparoscopic examination. However, the non-inferiority trial of Onda et al. 81 published in 2020 was negative, a result partly explained by a low statistical power and different surgical procedures. Notably, patients aged >75 years were excluded in those two trials.
How to treat (older patients)?
Carboplatin and paclitaxel (CP) every 3 weeks is the standard chemotherapy regimen in newly diagnosed advanced tubo-ovarian or peritoneal cancer. 91 In older patients, several studies have prospectively evaluated the impact of geriatric parameters on treatment toxicity and efficacy, and aimed to adapt the treatment regimens to geriatric vulnerability. For example, in 2005, Freyer et al. were the first to evaluate prospectively the impact of geriatric parameters on the tolerance of a carboplatin-cyclophosphamide ‘older-specific’ regimen and OS. In this study depression, impaired (⩾2) ECOG performance status and dependence were associated with severe toxicity; FIGO stage IV, depression and polypharmacy (>6 medications) were associated with lower OS. 36 A little later in 2008, Pignata et al. reported that a weekly regimen of paclitaxel and carboplatin every 28 days had, in a small number of patients, acceptable toxicity and efficacy. 16 The EWOT-3 trial of the GINECO evaluated the feasibility of a monotherapy with carboplatin area under the curve (AUC) 5 mg/mL per minute defined as the ability to perform six cycles of treatment without any premature arrest. The feasibility rate reached 72% and the trial led to the development of the aforementioned GVS; a GVS score ⩾3 identified a vulnerable population with significantly worse OS, treatment completion, and increased toxicities. 40 In 2017 the Gynecological Oncology Group (GOG)-273 trial 17 confirmed the link between functional impairment assessed by IADL and the capacity to complete four cycles of chemotherapy in patients aged ⩾70 years; for patients treated with CP the higher the IADL score the longer was the OS. 17 In 2021 the EWOC-1 study found that single-agent carboplatin was less effective with worse survival outcome in vulnerable patients (GVS ⩾ 3) compared to CP associations. 89 Among the two tested CP associations, patients treated with standard 3-weekly CP tended to derive the most benefit, in particular those with a GVS = 3, compared to an adapted 3 weeks/4 carboplatin AUC 2 paclitaxel 60 mg/m2 regimen developed for the MITO5 study. 16 In parallel, another (continuous) weekly carboplatin AUC 2 paclitaxel 60 mg/m2 regimen was found to be comparable to standard CP in the randomized MITO7 trial that was not specifically designed for older patients, and to provide better tolerability and quality of life, 87 leading to consider this regimen as a favorable alternative option, to be evaluated in the future on geriatric patients.
Pharmacokinetic aspects
A challenge of cancer treatment in older patients is the integration of geriatric cofactors that may impact chemotherapy tolerance and prognostic factors. An example of this is the dose optimization of carboplatin in the elderly; 92 this was based initially on the Calvert formula allowing to predict the carboplatin clearance and the choice of target AUC [dose (mg) = target AUC (mg/mL × min) × (GFR mL/min + 25)], where GFR is the glomerular filtration rate. In the older population the main difficulty remains in GFR estimation, despite the improvement of the successive formulas: older creatinine clearance formulas (Cockroft & Gault, Jelliffe), the estimated GFR (eGFR) formulas (MDRD, CKD-Epi, Janowitz), since the measurement of GFR using isotopic methods cannot be performed in routine. The Chatelut et al. 93 and the Thomas et al. 94 formulas rely on the direct calculation of carboplatin clearance, providing a better adjustment in the older, obese and/or sarcopenic populations; both include body weight and age as predicting covariates. The most modern formulas (Thomas, modified Thomas, and CKD-Epi-cysC 95 ) include, in addition, cystatin C, that is filtered but not excreted and with no relationship with muscular mass, contrary to creatinine. Since carboplatin is often prescribed to patients with altered renal function, which may be overestimated in the context of sarcopenia, these dose optimizations including cystatin C seem particularly adapted.
Hematological toxicity is common in older patients but is usually mild; a retrospective study reported by Bruchim et al. found that 75% of all-grade hematological toxicity in patients aged ⩾70 years versus 36.3% in younger patients; p = 0.001; but no significant difference in the frequency of grade 3–4 toxicities; older patients were more likely to have dose reductions and treatment delays compared to the younger patients. For those receiving optimal treatment, age ⩾70 years was not an independent factor for poor prognosis, whereas severe comorbidity was. 23
What to do next?
We do not have evidence-based data concerning patients who are neither fit phase III patients that would have been included in (selective) randomized trials, nor vulnerable patient according to the GVS score. For such patients, pragmatic adaptative approaches could be proposed, using, for example, dose –escalation strategies (‘pre-phase’), as proposed in other tumor models. 96 As raised previously and having demonstrated the need to omit de-escalation strategies in the most vulnerable patients, the field must be open to develop supportive care and geriatric interventions aiming at maintaining or optimize patients’ functional, nutritional and thymic status during chemotherapy. The ongoing EWOC-2/PROADAPT-ovary trial (NCT04284969) addresses specifically the issue of prehabilitation in patients planned for cytoreductive surgery.
Considering pharmacokinetics, results from age-specific subgroup retrospective analyses should be interpreted carefully. Pharmacokinetic analyses should be included in future prospective studies to evaluate the impact of different carboplatin dose calculation formulations on treatment tolerance since a portion of the excess toxicity may be partly attributed to treatment overexposure.
Targeted therapies
Where are we now?
Bevacizumab was the first targeted therapy to improve medical treatment of ovarian cancer (Table 3). Two studies demonstrated a PFS benefit: GOG 218 and ICON7. The GOG 218 study included 1873 patients with stage III (incompletely resectable) or IV ovarian cancer who had a median age of 60 years; bevacizumab was added to standard chemotherapy at 15 mg/kg for 22 cycles. 85 Older patients were included, the oldest was 89 years old, but no subgroup analysis on this specific population was performed. The ICON7 study included 1528 patients with high-risk ovarian cancer (stage I–IV); bevacizumab was added to standard chemotherapy at the dose of 7.5 mg/kg for 17–18 cycles. 84 This study found a benefit in terms of PFS for the whole population and OS in those with poor prognosis. 84 Again, no subgroup analysis was dedicated to the older population. Three studies provide data specific to older patients, the ROSiA and TURBO studies as well as the observational study reported by Beinse et al. 97 The RoSiA single arm phase IIIB study was designed to evaluate the safety and efficacy of bevacizumab extended administration; 1021 patients were included, of whom 121 were aged ⩾70 years 98 and older patients had a higher rate of anemia, diarrhea, grade 3 hypertension and thromboembolic events, but the same PFS. 98 The TURBO case-control study compared the tolerance of bevacizumab in patients aged ⩾65 years with a primary or recurrent ovarian cancer to younger ones; predictive factors of developing severe toxicity were eGFR < 60 mL/min according to the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, and presence of ⩾3 comorbidities. 99 In an observational cohort, Beinse et al. found that baseline hypertension was more prevalent in patients aged ⩾70 years; it was associated with a higher risk of grade 3–4 hypertension under treatment. 97 In addition, the MITO16 study found that bevacizumab rechallenge had a positive effect on PFS (hazard ratio 0.51, [95% CI 0.41; 0.65]; log-rank p < 0.0001); again, the study did not include specific data on older patients. 100 In real-life practice, because bevacizumab use is restricted by the non-reimbursement in many countries and because of the lack of evaluation in the geriatric population, bevacizumab exposure remains limited in the older population. Taken together, due to a higher prevalence of pre-treatment co-morbidities, including hypertension and decreased glomerular filtration rate, particular attention should be paid to the management of bevacizumab in the older population.
Pivotal trials investigating bevacizumab and PARPi.

HR, hazard ratio; HRD, homologous recombination deficient; Mo, Month; OS, overall survival; PARPi, poly(ADP-ribose) polymerase inhibitors; PFS, progression-free survival.
The second revolution in first-line advanced EOC came from PARPi (Table 3). The advent of PARPi as a first-line maintenance treatment, obtained thanks to the SOLO1, PAOLA1, PRIMA and the ongoing ATHENA studies, has led to two major challenges. First, the absolute necessity to obtain HRD status in a timely manner; at the time of writing, the only validated test is commercial (My Choice®; Myriad Genetics, Salt Lake City, UT, USA) but several ongoing studies have the aim to provide prospectively validated academic tests. Second, the respective position of bevacizumab and PARPi in HRP patients is currently unanswered. However, the impact of age on the treatment tolerance and efficacy has been investigated in several subgroup analyses and these did not identify major difficulties in the management of (selected) older patients. 110 In recurrent disease in patients eligible for platinum and no prior PARPi, olaparib, 107 niraparib, 108 and rucaparib 109 demonstrated a benefit in cancer control; and some data specific to older patients have been reported; for olaparib, 111 niraparib, 11 and rucaparib 112 were summarized in a review of the Young Internal Society of Geriatric Oncology in 2019 that highlighted the need to consider pharmaceutical optimization in routine care for older patients, given the high prevalence of polypharmacy in these patients. 113 No difference on toxicity was shown between patients aged < or ⩾65 years for olaparib, 111 for patients aged < or ⩾70 years for niraparib, 11 and in three age subgroups (<65 years, 65–74 years, and ⩾75 years) for rucaparib. 112 Anemia, thrombocytopenia, nausea, and vomiting were the most frequently experienced adverse events, highlighting the need for adequate supportive care and the value of early detection and management, 114 with potentially dose reduction, more frequently observed in elderly versus younger patients for niraparib and rucaparib.11,112 However, a very small proportion of the patients were aged ⩾75 years, and none were aged ⩾85 years11,111,112 which calls into question the applicability of these results among older adults 113 (Table 4).
Impact of age on the safety of PARPi.
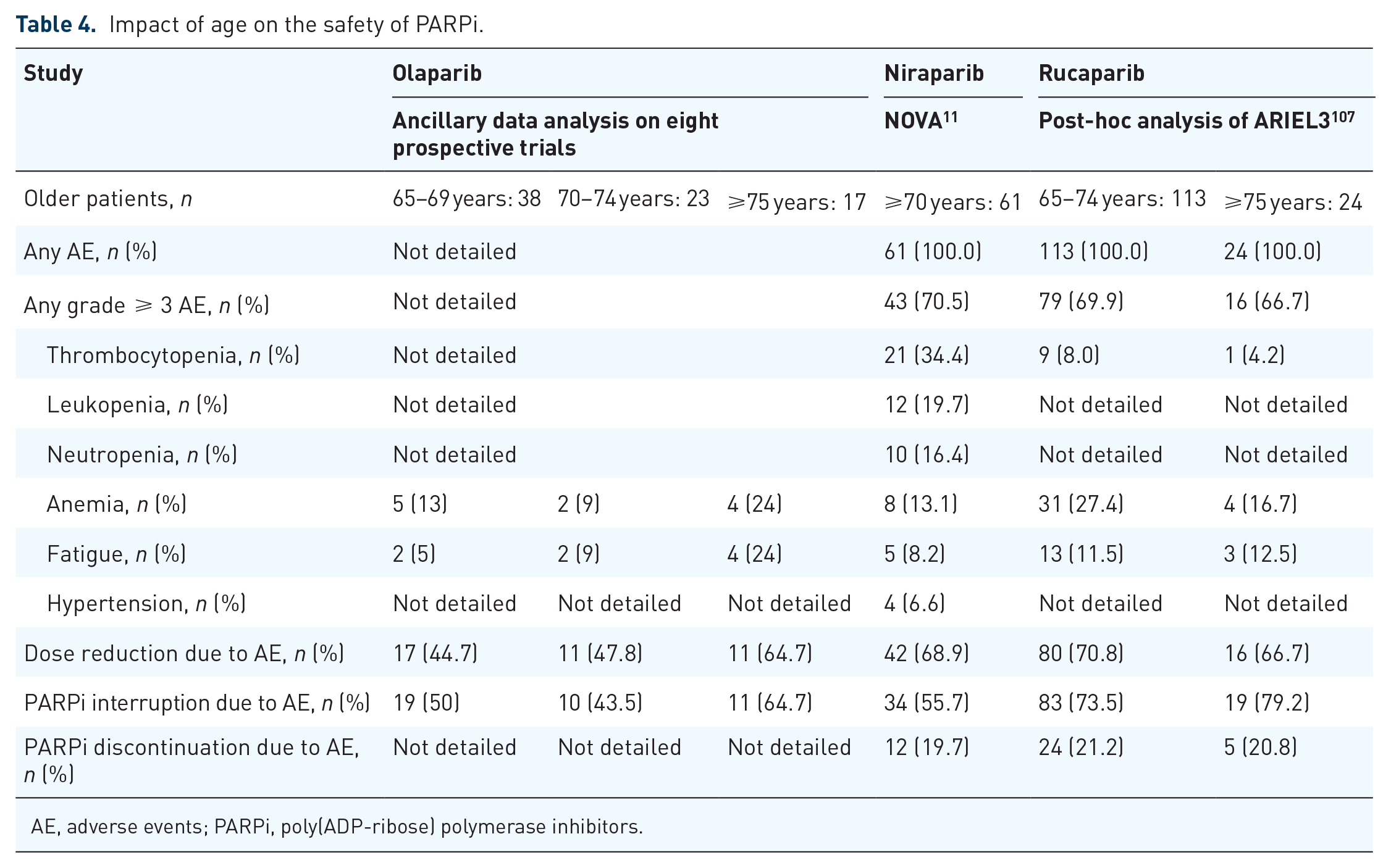
AE, adverse events; PARPi, poly(ADP-ribose) polymerase inhibitors.
In geriatric oncology attention must be paid to both adherence and polypharmacy. Older patients are classically considered as poorly adherent to chronic medications, but they have a better adherence to cancer treatments compared to other medications, and therefore the adherence to both the treatment and supportive medications should be favored.115,116 The frequency of administration differs between the molecules in the class; for example, niraparib should be taken once a day whereas olaparib and rucaparib need to be taken twice a day. A second warning concerns an increased risk of myelodysplastic syndrome. 117 Third, and despite a common mechanism of action, PARPi pharmacokinetic profiles differ substantially and may be a criterion in prescribing PARPi (Table 5). Olaparib and rucaparib are primarily metabolized by the cytochrome P450 enzymatic pathway, which is not the case for niraparib, and which explains the different risk of drug-drug interactions. 118 All concomitant medications and potentially use of complementary medicine should be reviewed before initiating PARPi treatment, especially in case of polypharmacy. No upfront preventive dose adjustment is necessary, as age itself does not seem to significantly increase toxicities in response to PARPi. Whatever the patient’s age, niraparib tolerance was shown to be optimized with an individualized starting dose of 200 instead of 300 mg in case of bodyweight <77 kg. 119 Mild renal impairment, which is a common comorbidity in older patients, requires dose adjustment for olaparib associated to a strong follow-up of adverse drug events in this context. 113
Pharmacokinetics parameters of PARPi.

↑, inductor. ↓, inhibitor. *, in vitro.
PARPi, poly(ADP-ribose) polymerase inhibitors.
What to do next?
The place of targeted therapies in the older population is of utmost importance given both the risk of cumulative toxicities induced by prolonged chemotherapy treatments and the pejorative histopathologic features of their disease. Since bevacizumab has demonstrated benefit in the most to high-risk diseases 123 it is expected that older patients will derive the most benefit; this is supported by the observational study presented at European Society of Gynecological Oncology meeting in 2022 that found a trend toward a positive impact on OS among older patients (⩾70 years) that was not found in younger ones. 124 This indicates that future prospective trials should include geriatric covariates both to further investigate the risk–benefit ratio of bevacizumab addition to chemotherapy, but also to optimize the geriatric and oncological follow-up. Considering PARPi, future trial designs should integrate the possibility of chemotherapy-free regimens, both as first-line treatment in frail older patients ineligible to platins, in platin-eligible relapse, and according to individualized strategies is specific tumor phenotypes (tailored de-escalation strategies). Independently of age, the value of performing iterative biopsies at relapse, to individualize such treatment strategies, remains to be explored. In older populations, there is a specific need to explore added values of therapeutic drug monitoring and a close monitoring of PARPi toxicities, in particular on the hematological system, 125 and more specifically on the myelodysplastic risk. From an ethical perspective, future trials should always consider the patients’ perspective and integrate the specificities of the target population in the choice of study endpoints, with a specific attention to functionality and quality of life preservation. 126
Pharmaceutical optimization
Where are we now?
Polypharmacy (defined as the concurrent use of at least five drugs 127 ) in older adults with cancer is frequent 128 and concerns more than half of older patients with ovarian cancer.129,130 Polypharmacy often includes potentially inappropriate medications (PIMs), which are drugs that lack evidence-based indications, have risks that outweigh therapeutic benefit, or can potentially interact with other drugs. 131 In a recent meta-analysis, the prevalence of PIM, mainly identified using Beers criteria, ranged from 19.0 to 52.0% in older patients with cancer 128 and similar results were found in ovarian cancer patients.36,129 Negative clinical impacts of polypharmacy and PIM have already been reported in cancer patients, contributing to falls, chemotherapy toxicities, postoperative complications, and functional impairment. 128 On 1,213 patients with recurrent ovarian cancer, an increasing amount of medication was associated with overall grade III/IV toxicity (p < 0.001; OR 1.120), and hematological (p < 0.001; OR 1.056) and non-hematological (p < 0.001; OR 1.134) toxicities. 130 Iatrogenic risk of polypharmacy also includes self-medication and use of complementary and alternative medicine. Most of iatrogenic events are evitable. Reducing polypharmacy and PIMs is challenging in cancer patients and requires an interprofessional team with expertise in each assessment domain: oncologists, geriatricians, nurses and pharmacists.132,133
A comprehensive medication review, a systematic process for obtaining and assessing patient-specific information related to all medication therapies, aims to identify and resolve any drug-related problem. Implementing this clinical pharmacy intervention in the multidisciplinary team may have the potential to optimize older cancer patient medication use and health outcomes, as described in several studies (Supplemental Table 1) and recommended by American Society of Clinical Oncology guidelines for geriatric oncology.134,135 Medication reconciliation consisting of obtaining a comprehensive list of all medications taken by a patient and comparing it to the current drug regimen to identify and resolve any discrepancies is of particular interest in older patients with cancer often exposed to multiple care transitions.
In addition, as PARPi are administered orally patient adherence to both the treatment itself and associated supportive drugs has become a major issue. 136 A clear treatment plan needs to be established with the patient and her caregivers with explanations of potential side effects and their prevention, as well as adaptation of the treatment plan to the vulnerabilities identified during the geriatric assessment. 118
What to do next?
To reduce the iatrogenic risk may include integration of hospital pharmaceutical consultations in the interdisciplinary approach and the enhancement of a pharmaceutical community-hospital network. Further studies need to be conducted to investigate risk factors for drug-related problems in patients with ovarian cancers and to develop screening tools for polypharmacy and PIM adapted to elderly cancer patients. Close interdisciplinary collaboration (oncologist, geriatrician, surgeon, pharmacist and nurse) should be promoted, supported by shared electronic medical information, and assessed to optimize healthcare resources and ensure high-quality ovarian cancer care delivery.
Perspectives
In recent years, a major improvement in the outcomes of patients with advanced EOC included in cancer trials, with PFS exceeding 37 months in patients with BRCA1/2 mutated or HRD tumors, treated with surgical standards, adjuvant chemotherapy in combination with bevacizumab and maintenance by the PARPi olaparib. This illustrates the major advent observed in the development of targeted therapies and, in parallel, the individualization of the treatments according to the histopathologic features of the tumors. During the same period and considering real-life data, the outcomes of octogerian and nonagerian patients did not improve, increasing with time the survival gap between younger and older patients. 137 Considering that pejorative outcomes of these patients may be interpreted as cancer-related, treatment-related, and host-related, future work is needed, to improve treatment individualization in the older population, according to their specific histopathologic features, specific safety concerns regarding each treatment procedure (surgery, chemotherapy, targeted treatments. . .) and geriatric covariates and interventions.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231192397 – Supplemental material for Ovarian cancer in the older patient: where are we now? What to do next?
Supplemental material, sj-docx-1-tam-10.1177_17588359231192397 for Ovarian cancer in the older patient: where are we now? What to do next? by Frédérique Rousseau, Florence Ranchon, Christophe Bardin, Naoual Bakrin, Vincent Lavoué, Leila Bengrine-Lefevre and Claire Falandry in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
