Abstract
Background:
Patient-reported outcomes (PROs) are increasingly becoming an important part of clinical trials as they are helpful in analyzing the safety and efficacy of treatment in chronic diseases like cancer.
Objectives:
We report PROs and health-related quality of life (HRQoL) with selpercatinib treatment among Chinese patients with rearranged in transfection (RET) fusion-positive non-small-cell lung cancer (NSCLC), RET fusion-positive thyroid cancer (TC), and RET-mutant medullary TC (MTC) as an exploratory analysis of the LIBRETTO-321 trial.
Design:
A total of 77 patients (47 RET fusion-positive NSCLC, 1 RET fusion-positive TC, and 29 RET-mutant MTC) were enrolled. Compliance for European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire-Core 30 (QLQ-C30) was 100% at baseline and >90% at each time point.
Methods:
PROs were assessed using the EORTC QLQ-C30, and a bowel diary assessment for MTC patients with baseline diarrhea using the Systemic Therapy-Induced Diarrhea Assessment Tool. Data were collected at pre-dose; every 8 weeks from cycle 3; and every 12 weeks after cycle 13. A >10-point change from baseline was considered clinically meaningful. PRO changes were summarized through cycle 13.
Results:
Most patients with NSCLC or MTC showed improvement or remained stable on the global health status and functional subscales. For global health status, 47.4% of NSCLC and MTC patients showed definite improvement with only 19.7% showing definite worsening. For functional subscales, less than 30% of the patients showed definite worsening. For symptom subscales, more than 64% of the patients either improved or remained stable for the symptoms. For MTC patients with bowel diary assessment (n = 5), there was no severity or worsening from baseline in the diarrheal episodes observed during treatment with selpercatinib.
Conclusion:
The study demonstrated favorable PROs in Chinese patients with RET fusion-positive NSCLC, TC, and RET-mutant MTC treated with selpercatinib. HRQoL was improved or stable as assessed by EORTC QLQ-30.
Trail registration:
This study was registered at ClinicalTrials.gov (https://clinicaltrials.gov/ct2/show/NCT04280081) ClinicalTrials.gov Identifier: NCT04280081.
Introduction
In China, lung cancer accounts for approximately one-fourth of all cancer deaths with non-small-cell lung cancer (NSCLC) being the most predominant (approximately 85%) form of lung cancer. 1 Thyroid cancer (TC) is the most common endocrine and head-and-neck malignancy globally, and the incidence has been increasing rapidly around the world and in China. 2 The number of newly diagnosed cases in China accounts for 34% of the overall new cases worldwide. 2 Advanced TC has a very poor prognosis. In China, deaths due to TC account for a large percentage of the overall mortality rate globally. In 2020, China reported 9261 deaths out of 43,646 deaths worldwide due to TC. 3 Medullary TC (MTC) is a rare type of TC comprising 3–4% of all TCs with relatively poorer prognosis and higher recurrence and metastasis rates. 4
Rearranged in transfection (RET) is a receptor tyrosine kinase with crucial functions in the normal development, maturation, and maintenance of several adult tissue types, including neural, neuroendocrine, and male germ cell. 5 Point mutations or chromosomal rearrangements of RET are implicated in several human cancers. RET gene fusions have been identified in 1–2% of NSCLCs, and approximately 10–20% of sporadic papillary TCs are associated with adverse prognostic features.6–8 Activating germ line mutations are observed in approximately a quarter of MTC and somatic RET mutations are observed in about 65% of sporadic MTCs. 9
The standard of care for RET-activated NSCLC is platinum-based chemotherapy with or without immunotherapy. For MTC and other cancers with RET alterations, the standard of care is multikinase inhibitors (MKIs), which are only moderately effective and have several other side effects due to their off-target toxicity; therefore, selective inhibition of RET kinase has been shown to be beneficial in such cancers. 10 Selpercatinib (LOXO-292) is a highly selective small molecule inhibitor of RET kinase which is effective against diverse activating RET alterations including RET fusions and RET point mutations.11,12 Selpercatinib has received approval in the United States and China for the treatment of RET-activated locally advanced and metastatic NSCLC, TC, and MTC, and from the European Medicines Agency for the treatment of RET-activated advanced NSCLC, TC, and MTC.12–14 LIBRETTO-001 is a registrational, phase I/II, single-arm, open-label study evaluating the efficacy and safety of selpercatinib in patients with RET-activated solid tumors, including RET fusion-positive NSCLC and TC, RET-mutant MTC, and other tumors with RET activation, and demonstrated durable efficacy with favorable safety profile.11,15 The efficacy and tolerability of selpercatinib were evaluated in Chinese patients with advanced solid tumors, including RET fusion-positive NSCLC and TC, and RET-mutant MTC for the first time in the LIBRETTO-321 trial, which has demonstrated robust and durable antitumor activity with mild and manageable adverse effects regardless of prior treatment line. 16
The prognosis for patients with advanced or metastatic cancer is poor, and some treatments also substantially affect the well-being and functioning of the patient throughout treatment. Therefore, patient-reported outcomes (PROs) are increasingly recognized as an important component of clinical trials to understand the patient experience of symptoms and toxicities not fully captured in physician-assessed outcome measures. Patient-reported health-related quality of life (HRQoL) outcomes are an indication of the patient’s health condition and are evidenced to improve communication, satisfaction, and symptom management.17–20
In this study, we evaluated patient-reported HRQoL outcomes in Chinese patients with RET-activated NSCLC, TC, and MTC treated with selpercatinib.
Materials and methods
Study design and population
LIBRETTO-321 was an open-label, multicenter phase II study in patients with advanced solid tumors, including patients with RET fusion-positive NSCLC, RET fusion-positive TC, and RET-mutant MTC. Eligible patients were aged ⩾18 years with an Eastern Cooperative Oncology Group (ECOG) performance status score of 0, 1, or 2, with no sudden deterioration 2 weeks prior to the first dose of study treatment. Patients with RET alterations in tumor and/or blood were identified at a central laboratory. 16 Patients with prior treatment with selective RET inhibitor(s) (including investigational selective RET inhibitor(s), such as BLU-667, RXDX-105, etc.) were excluded from the study. Patients received 160 mg selpercatinib twice a day from cycle 1 day 1 and cycles were measured in 28-day increments. Individual patients continued daily selpercatinib dosing until progressive disease (PD), unacceptable toxicity, or other reasons for treatment discontinuation. Patients with documented PD were allowed to continue selpercatinib if the patient was tolerating the treatment and was deriving clinical benefit from continuing the study treatment.
Patient-reported outcomes evaluation
Patients with RET fusion-positive NSCLC, RET fusion-positive TC, and RET-mutant MTC enrolled in LIBRETTO-321 were administered the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire-Core 30 (QLQ-C30) version 3.0 at baseline (cycle 1, day 1, prior to study treatment), every 8 weeks from cycle 3, day 1 until cycle 13, and every 12 weeks after cycle 13 until the end of treatment. The QLQ-C30 questionnaire is a well-utilized tool in clinical trial research worldwide to assess the HRQoL in cancer patients and has been validated and translated to more than 100 languages.21,22 It includes global health status and five functional domains (physical, role, cognitive, emotional, and social) and nine symptom domains (fatigue, nausea and vomiting, pain, dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties). All QLQ-C30 subscales were scored from 0 to 100. Higher scores on the global health status/QoL and functional subscales represent better functioning. Lower scores on the symptom subscales represent fewer symptoms.23,24 Patients with RET-mutant MTC having baseline diarrhea were administered a bowel diary questionnaire, a modified version of the Systemic Therapy-Induced Diarrhea Assessment Tool (mSTIDAT), at baseline (cycle 1, day 1, prior to study treatment), weekly during cycle 1, and on day 1 of every cycle until the end of treatment. 25 The mSTIDAT assessed patient’s perception of having diarrhea, daily number of bowel movements, daily number of diarrhea episodes, the presence of urgency, the presence of abdominal discomfort, fecal incontinence, patient’s perception of diarrhea severity, and QoL.
Statistical methods
Patient compliance of QLQ-C30 was calculated as the total number of patients who completed the QLQ-C30 divided by the total number of patients who were on treatment at each visit. Each item on the QLQ-C30 and mSTIDAT was reported descriptively (mean ± SD) at each assessment time point. For QLQ-C30, clinically meaningful differences (CMD) were defined as >10 points difference from baseline on a 0–100 scale. 23 Each patient was categorized as improved or worsened at each follow-up study visit if the CMD was met versus their baseline score, and otherwise defined as stable. Definite improvement or worsening was defined as the first improvement or worsening without any further deterioration or improvement, respectively. The proportion of patients who met the criteria for definite improvement or definite worsening (±10 or more points from baseline without reversal of that change) was reported for each subscale of the QLQ-C30. Time to definite improvement and worsening was defined as duration from first study dose to occurrence of definite improvement or worsening and was analyzed using the Kaplan–Meier method. PRO data were summarized through cycle 13 as only few patients had reached the later time points.
Results
A total of 77 patients [NSCLC: 47, TC (papillary thyroid carcinoma): 1, and MTC: 29] with a mean (SD) age of 50.6 (12.9) years, and 44/77 (57.1%) males were enrolled in the study. The baseline demographics and characteristics are summarized in Table 1. QLQ-C30 completion rates for enrolled patients were 100% at baseline and >90% at each time point during on-treatment visits. The baseline scores for global health status, functional subscales, and symptom subscales of QLQ-C30 among each tumor type are presented in Supplemental Table 1, and their mean changes from baseline are presented in Supplemental Figures 1 and 2. Five MTC patients with baseline diarrhea completed the bowel diary assessment.
Demographics and baseline disease characteristics by tumor type.

BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; MKI, multikinase inhibitor; MTC, medullary thyroid cancer; N, number of subjects in population; n, number of subjects; NSCLC, non-small-cell lung cancer; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; RET, rearranged during transfection; SD, standard deviation; TKI, tyrosine kinase inhibitor.
QLQ-C30 global health status and functional subscales
The change from baseline in the QLQ-C30 scores by study treatment cycle and tumor type for global health status is presented in Figure 1(a). Most of the NSCLC or MTC patients either improved or remained stable during the study treatment with selpercatinib. The proportion of patients experiencing improvement ranged from 33.3% (cycle 3) to 52.4% (cycle 11) for NSCLC, and 45.8% (cycle 3) to 66.7% (cycle 11) for MTC showing an upward trend. Here we are reporting data only until cycle 11, as there were limited number of patients who reached cycle 13 (NSCLC, n = 4; MTC, n = 1). Definite improvement was observed in 40.4% of NSCLC and 60.7% of MTC patients with a median time to definite improvement of 1.87 months; definite worsening was observed in 25.5% of NSCLC and 10.7% of MTC patients (Supplemental Table 2) with a median time to definite worsening of 2.76 months (Supplemental Table 3). No definite change was observed in the rest of the patients. Proportion of patients showing definite improvement or worsening in the enrolled patients (NSCLC, TC, and MTC combined) is presented in Figure 2(a).

Proportion of patients (by tumor type) with changes from baseline on the global health status and functional subscales, by category.

Proportion of patients with definite improvement, definite worsening, or no definite change by global health status and functional subscales.
The change from baseline in the QLQ-C30 scores by study treatment cycle and tumor type for functional subscales is presented in Figure 1(b)–(f). Overall, most of the patients with NSCLC or MTC were either stable or showed improvement on the functional subscales with some patients showing worsening. Except for cycle 13 with limited patients, more than half of the patients with NSCLC or MTC were stable in physical, role, emotional, and cognitive functioning, and the proportion was up to 70–80% during the treatment. For social functioning, most of the MTC patients showed improvement (42–67%), while about half of the NSCLC patients remained stable (43–54%). Improvement was also observed in around 20–30% of patients for almost all the other functional subscales with a relatively lower proportion in cognitive functioning. Limited patients showed function worsening in most subscales and cycles except for cognitive and social functioning evaluations where 20–30% patients reported worsening in some cycles.
For the enrolled population, definitive improvement was observed in 30–40% of patients (NSCLC and MTC) for physical, emotional, and social functioning; and in 26.3% and 22.4% of patients for role and cognitive functioning, respectively. Definite worsening was observed in 17.1% and 11.8% of patients for physical and emotional functioning, respectively, and for other functioning domains definite worsening ranged from 23–29% [Figure 2(b)–(f)]. The rest of the patients showed no definite change. The median time to definite improvement was around 1.8–1.9 months, and median time to definite worsening was relatively longer, from 1.8 to 4.5 months (Supplemental Table 3).
QLQ-C30 symptom subscales
The change from baseline in the QLQ-C30 scores by study treatment cycle and tumor type for symptom subscales is presented in Figure 3. Most of the NSCLC and MTC patients were either stable or showed improvement for the symptom subscales with limited number of patients showing worsening. Around one-third to half of the NSCLC or MTC patients showed improvement in fatigue and pain, and the rest of the patients were stable. For nausea and vomiting, more than 90% of NSCLC patients either improved or remained stable, while all the MTC patients remained stable during the on-treatment evaluation period. For dyspnea, insomnia, appetite loss, and constipation, around 23–36% of NSCLC patients showed improvement, and more than half of the patients remained stable, while more than two-thirds of the MTC patients remained stable with limited patients showing worsening. Diarrhea symptoms were stable in more than half of the NSCLC or MTC patients, and symptom worsening was observed in 19–32% NSCLC patients and 4–11% of MTC patients.

Proportion of patients (by tumor type) with changes from baseline on the symptom subscales, by category.
Definite improvement was observed in around 40% of patients for fatigue, pain, and financial difficulties. More than half of the patients were stable for nausea and vomiting, dyspnea, insomnia, appetite loss, constipation, and diarrhea. Definite worsening was observed in around 20–30% of patients for fatigue, appetite loss, constipation, and diarrhea. The rest of the symptom domains displayed small proportions of worsening (Supplemental Table 2). Figure 4 represents proportion of patients showing definite improvement or worsening in the enrolled patients (NSCLC and MTC combined). The median time to definite improvement was around 1.8–1.9 months, and time to definite worsening was relatively longer, from 1.8 to 4.5 months (Supplemental Table 3).
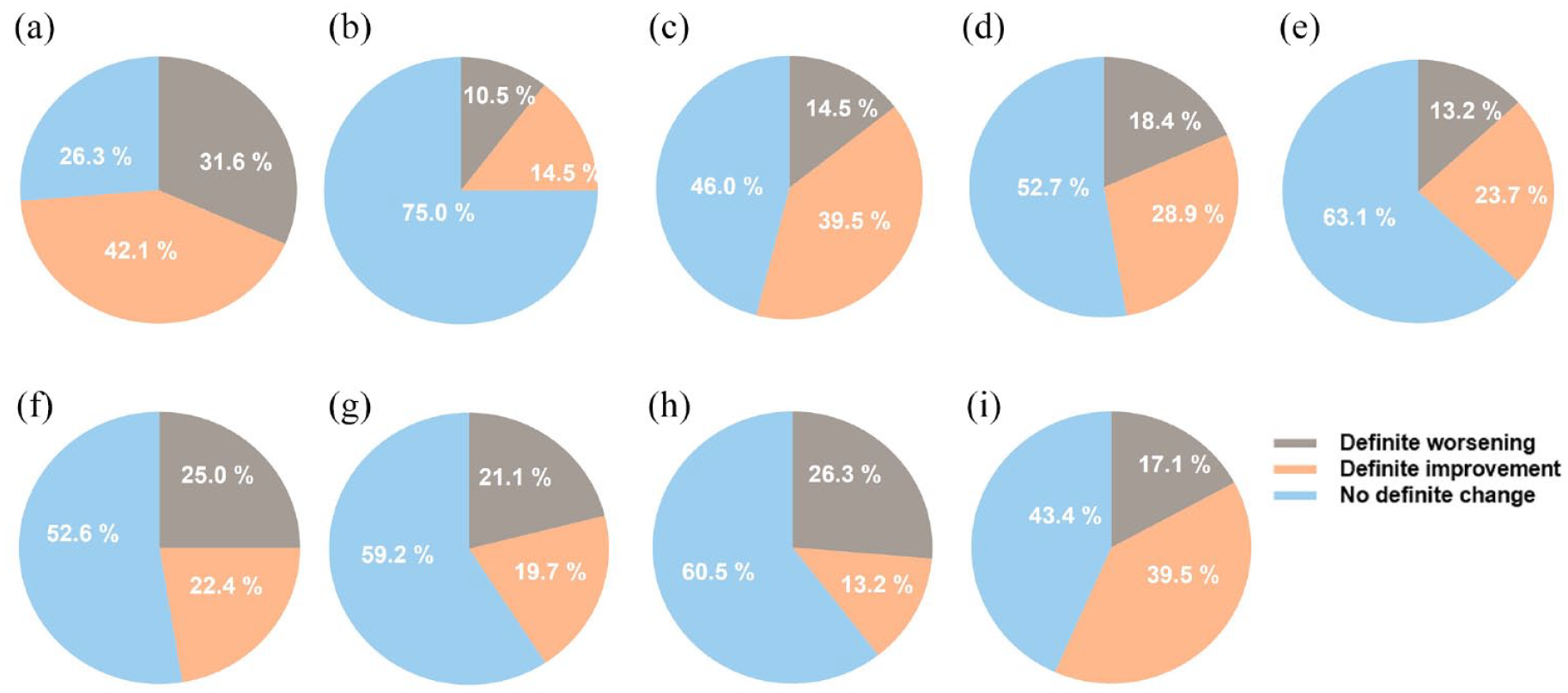
Proportion of patients with definite improvement, definite worsening, or no definite change by symptom subscales.
Bowel diary assessment for MTC patients
Bowel diaries were collected for all the MTC patients with baseline diarrhea (n = 5). The mean (SD) number of diarrheal episodes in MTC patients (n = 5) who completed the mSTIDAT at baseline (cycle 1) was 2.8 (1.1). Patient-reported severity of diarrhea at baseline was minimal among four (80.0%) patients and moderate among one (20.0%) patient. Patient-reported severity of diarrhea by study cycle is summarized in Supplemental Figure 3. From the third week after treatment initiation, only one of the patients still reported minimal diarrhea and the rest of the patients reported no diarrhea along the treatment. There was no severity or worsening in diarrheal episodes as compared to baseline with selpercatinib. The median duration of improvement from baseline on bowel diary was 9.4 months.
Summary of change from baseline bowel diary QoL scores for family life, social life, and overall QoL is depicted in Supplemental Figure 4. Patients reported little impact of diarrhea on any of these aspects of daily living during study treatment with selpercatinib.
Discussion
The overall success of a cancer treatment can be assessed by the prolonged survival, improvement of patients’ HRQoL, intensity alleviation of the symptoms experienced, and thus, the overall physical and mental health of the patient. PROs and HRQoL aid in providing this information and understanding the value of a cancer treatment alongside its efficacy and safety.
Patients with advanced cancer generally report poor general and spiritual well-being and often experience poor HRQoL due to the associated treatment-related side effects and the disease itself. Previous studies have shown that patients undergoing cancer chemotherapy experience significant adverse impact on the HRQoL domains of physical, functional, emotional, and spiritual well-being. 20 Also, the implementation of quality standards for reporting PROs as per the European Society for Medical Oncology–Magnitude of Clinical Benefit Scale (ESMO-MCBS) appears to be important in ensuring the validity and reliability of the PROs. In line with the guiding principles of ESMO-MCBS, the EORTC QLQ-C30 is an important and well-validated tool for PROs and HRQoL assessment and well adopted in our study. 26
The off-target toxicities associated with the nonspecific MKIs have demonstrated moderate efficacy along with tolerability issues which has impacted the HRQoL of cancer patients. 10 As per previous studies, more than 70% of patients with NSCLC and MTC typically experience grade 3 and above adverse events, leading to dose reduction in the majority of trial participants and discontinuation of dose for a few Food and Drug Administration-approved protein tyrosine kinase inhibitors (TKIs) such as cabozantinib, vandetanib, lenvatinib, sorafenib, and sunitinib. The tolerability issues of these TKIs have shown to adversely affect the HRQoL of cancer patients. As an RET-specific TKI, selpercatinib has >20-fold more potent than cabozantinib, vandetanib, or lenvatinib and has demonstrated better tolerability with only 2% of patients undergoing treatment discontinuation due to treatment-related adverse events. 10 In this study, we observed improved outcomes on the various domains of QLQ-C30 in Chinese patients with RET fusion-positive NSCLC, RET fusion-positive TC, and RET-mutant MTC treated with selpercatinib. As per the QLQ-C30 scores for global health status and functional subscales like physical, emotional, cognitive, and role functioning, the majority of the patients with NSCLC and MTC showed improved outcomes or remained stable at each post-baseline assessment during treatment with selpercatinib. These promising results with maintained QLQ-C30 scores in various domains, in addition to the favorable safety profile, explain the good treatment persistence and low discontinuation rate of selpercatinib compared with the other treatment modalities.
Patients with MTC enrolled in this study were generally stable for physical and cognitive functioning of the functional subscales, consistent with a previous study conducted in a global patient population in patients with MTC treated with selpercatinib. 27 Furthermore, patients with MTC were also stable for the symptom subscales, that is, fatigue, nausea and vomiting, dyspnea, insomnia, appetite loss, and constipation; and showed improvement in pain, diarrhea, and financial difficulties as observed by their QLQ-C30 scores. Previous studies have demonstrated the risk of diarrhea associated with the use of MKI inhibitors such as vandetanib leading to dose reduction or discontinuation of treatment. 28 In our study, most of the patients with MTC were stable or showed improvement in diarrhea as observed by the reduction in the severity of diarrhea and overall improvement in the diarrhea-affected family life, social life, and QoL. The results observed were consistent with the global study. 27 The MTC patients with baseline diarrhea (n = 5) also presented high calcitonin levels at baseline; and through treatment with selpercatinib, there was a decrease in the calcitonin levels with each cycle. The bowel diary assessment of these MTC patients treated with selpercatinib demonstrated no worsening in the severity of the experienced diarrheal symptoms.
For patients with NSCLC, improvement was observed with selpercatinib for global health status and social functioning, while patients remained stable for role functioning, cognitive functioning, and emotional functioning. A similar study was conducted with the MKI inhibitor lorlatinib in patients with NSCLC showing improvement in the functional domains such as emotional functioning, role functioning, and symptoms domains such as fatigue and insomnia. 29 For symptom subscales, most of the patients showed improvement for fatigue, pain, dyspnea, insomnia, and appetite loss, with some patients showing worsening of the symptoms for diarrhea. A meta-analysis with checkpoint inhibitors like programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) for advanced cancers demonstrated clinical changes (5- to 10-point difference) for global health status, role and social functioning, fatigue, dyspnea, insomnia, and appetite loss on the QLQ-C30 scales. 30 In our study, we observed improvements on all these functional and symptom domains with a minimum 10-point score difference.
Of note, most of the patients (NSCLC, TC, and MTC combined) either showed definite improvement or were stable for the symptom subscales. A limited number of and very few patients showed definitive worsening on some of the subscales (fatigue, diarrhea, and appetite loss). Moreover, the short time to improvement in all the HRQoL domains is quite in line with the response results of selpercatinib treatment previously reported for the trial. For NSCLC patients, the objective response rate was 87.5% and 61.1% in treatment-naïve and pre-treated patients, respectively, after 9.7 months of median follow-up, and the median time to response was 1.84 months; for MTC patients, it was 58.8% and 55.6% for treatment-naïve and pre-treated patients, respectively, after 8.7 months of median follow-up with a median time to response of 1.87 months.16,31 A post hoc Fisher’s exact test was used to explore the relationship between patients’ clinical response to selpercatinib and patients’ improvement in the EORTC QLQ-C30, indicating a possible correlation between the improvement of PROs and objective response to the treatment (p value = 0.11 for NSCLC patients, p value = 0.28 for MTC patients).
A very high compliance rate of QLQ-C30 questionnaire of >90% is a positive attribute of our study. The lower risk of experiencing key treatment-associated symptoms has demonstrated the relative tolerability of selpercatinib.
Furthermore, previously reported safety data for selpercatinib have demonstrated mild and manageable adverse effects indicating selpercatinib to be a promising treatment option for Chinese patients with RET fusion-positive NSCLC, RET fusion-positive TC, and RET-mutant MTC. The PROs of selpercatinib in the global LIBRETTO-001 trial have also demonstrated favorable outcomes in terms of HRQoL as measured by the QLQ-C30 in most of the patients with RET fusion-positive NSCLC and RET-mutant MTC.27,32 Our results in Chinese patients are consistent with the global study.
There are some limitations in this study. The single-arm nature of the study with open-label design could be a possible source of bias in the results. Only one patient with TC was included in the study, and only five patients were included for the bowel diary assessment in the MTC cohort so could not be representative of the entire disease category. The number of cycles analyzed during this study was limited; a longer follow-up including an additional number of cycles would be of interest. Furthermore, only a small number of patients could reach the end of cycle 13; therefore, results must be interpreted with caution.
Conclusions
PROs including HRQoL provide an extensive insight into the benefits of a treatment in addition to the efficacy and safety outcomes. Our study suggests that the majority of patients treated with selpercatinib either improved or remained stable for HRQoL assessed by the QLQ-C30 on all subscales with limited worsening of symptoms. Overall, our results demonstrated favorable PROs in Chinese patients with RET fusion-positive NSCLC, RET fusion-positive TC, and RET-mutant MTC treated with selpercatinib.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231189429 – Supplemental material for Patient-reported outcomes following selpercatinib treatment in Chinese patients with advanced RET fusion-positive non-small-cell lung cancer and thyroid cancer, and RET-mutant medullary thyroid cancer in the phase II LIBRETTO-321 trial
Supplemental material, sj-docx-1-tam-10.1177_17588359231189429 for Patient-reported outcomes following selpercatinib treatment in Chinese patients with advanced RET fusion-positive non-small-cell lung cancer and thyroid cancer, and RET-mutant medullary thyroid cancer in the phase II LIBRETTO-321 trial by Shun Lu, Xiangqian Zheng, Yuping Sun, Dingzhi Huang, Lin Wu, Qinghai Ji, Chengzhi Zhou, Jianying Zhou, Ye Guo, Minghua Ge, Ding Ding, Jingxin Shao, Wanli Zhang, Ming Gao and Ying Cheng in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This study was sponsored by Eli Lilly and Company. Medical writing services were provided by Deepika Kajarekar and Divya Kamboj from Syneos Health and was funded by Eli Lilly and Company.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
