Abstract
Background:
Selpercatinib, a highly selective and potent REarranged during Transfection (RET) kinase inhibitor, is effective in advanced RET-altered thyroid cancer (TC). However, the efficacy and safety in Chinese patients are unknown.
Patients and methods:
In the open-label, multi-center phase II LIBRETTO-321 (NCT04280081) study, Chinese patients with advanced solid tumors harboring RET alterations received selpercatinib 160 mg twice daily. The primary endpoint was objective response rate (ORR; RECIST v1.1) by independent review committee (IRC). Secondary endpoints included duration of response (DoR) and safety. Efficacy was assessed in the primary analysis set [PAS; treated patients with RET fusion-positive TC or RET-mutant medullary TC (MTC) confirmed by central laboratory] and all enrolled patients with MTC.
Results:
Of 77 enrolled patients, 29 had RET-mutant MTC and one had RET fusion-positive TC. In the PAS (n = 26), the ORR by IRC was 57.7% [95% confidence interval (CI), 36.9–76.6]. Median DoR was not reached and 93.3% of responses were ongoing at a median follow-up of 8.7 months. In all enrolled MTC patients (n = 29), the ORR by IRC was 58.6% (95% CI, 38.9–76.5). One RET fusion-positive TC patient treated for 23.4 weeks achieved a partial response at week 8 that was ongoing at cutoff. In the safety population (n = 77), 59.7% experienced grade ⩾3 treatment-emergent adverse events (TEAEs). TEAEs led to dose reductions in 32.5% (n = 25) and discontinuations in 5.2% [n = 4; 3.9% (n = 3) considered treatment related] of patients.
Conclusions:
Selpercatinib showed robust antitumor activity and was well tolerated in Chinese patients with advanced RET-altered TC, consistent with global data from LIBRETTO-001 (NCT04280081).
ClinicalTrials.gov Identifier:
NCT04280081 (first posted Feb 21, 2020)
Keywords
Introduction
Medullary thyroid cancer (MTC) is a rare endocrine tumor with a global prevalence of approximately 5–10% of all thyroid malignancies. 1 However, data suggest that MTC accounts for 2% or less of thyroid cancer (TC) cases in China. 2 This may be due to more cases of papillary thyroid microcarcinoma (tumors less than 1.0 cm) being diagnosed in China, thereby reducing the proportion of MTC cases. 2 The REarranged during Transfection (RET) gene is a primary oncogene in multiple subtypes of TC. 3 In MTC, RET mutations have a particularly high incidence and are present in over 90% of hereditary cases and over 40%–50% of sporadic cases.4,5 RET fusions also occur in other types of TC, such as papillary TC, but with a lower frequency (<10%). 6 Furthermore, the presence of RET alterations is associated with more aggressive TC phenotypes. 4 For patients with advanced or metastatic RET-altered thyroid carcinomas, a new generation of highly selective and potent RET inhibitors (selpercatinib and pralsetinib7,8) have recently been approved and are recommended by current treatment guidelines. 9 These drugs exert antitumor activity through RET targeting with lower toxicity compared to older agents such as cabozantinib and vandetanib that target RET as well as multiple other kinases.10 –12 Selpercatinib, a first-in-class highly selective and potent RET kinase inhibitor with central nervous system (CNS) activity, is approved in multiple countries for the treatment of advanced/metastatic RET-altered non-small-cell lung cancer (NSCLC) or TCs.6,13 –15 In the phase I/II global LIBRETTO-001 trial, selpercatinib showed robust antitumor activity in patients with advanced RET-mutant MTC. 6 In 55 patients with RET-mutant MTC who had previously received vandetanib, cabozantinib, or both, the objective response rate (ORR) was 69% [95% confidence interval (CI), 55–81] with 82% (95% CI, 69–90) of patients remaining progression-free at 1 year. Furthermore, among 88 patients who had not received previous treatment with a kinase inhibitor, the ORR was 73% (95% CI, 62–82) with 92% (95% CI, 82–97) progression-free at 1 year. The updated safety analysis of LIBRETTO-001 which included 746 patients showed that most adverse events (AEs) were low grade and only 2% of patients had discontinued treatment due to treatment-related AEs (TRAEs).6,16
Calcitonin and carcinoembryonic antigen (CEA) are well-known diagnostic and prognostic biomarkers in MTC. 17 Data also suggest that calcitonin and CEA levels may represent markers of treatment response in MTC. For example, the global LIBRETTO-001 trial showed a high biochemical response rate for calcitonin and CEA in patients with MTC who received selpercatinib. 6
Based on the findings from LIBRETTO-001, the United States Food and Drug Administration granted selpercatinib accelerated approval in May 2020 for advanced or metastatic RET fusion-positive TC and RET-mutant MTC. 15 However, to date, there are no data on selpercatinib in Chinese patients and, at the time of this analysis, there were no approved RET-targeted therapies for patients with RET-altered TC in China. We report the efficacy and safety of selpercatinib in patients with RET-mutant MTC and RET fusion-positive TC included in the phase II LIBRETTO-321 trial, which was conducted to evaluate the efficacy and safety of selpercatinib in Chinese patients with solid tumors harboring an activating RET alteration.
Materials and methods
Study design and treatment
LIBRETTO-321 was an open-label, multi-center phase II trial conducted across 15 centers in China to explore the antitumor activity of selpercatinib in Chinese patients with advanced solid tumors with RET activation, including RET-mutant MTC and RET fusion-positive solid tumors (thyroid and NSCLC) (Supplemental Figure 1). The study was conducted in-line with Good Clinical Practice guidelines, the principles outlined in the Declaration of Helsinki, and all applicable country and local regulations. The protocol was approved by the institutional review boards at each study site (Supplemental Table 1) and all patients provided written informed consent before inclusion. The study protocol was prospectively registered on ClinicalTrials.gov (NCT04280081, first posted 21 February 2020).
Eligible patients received selpercatinib 160 mg orally twice a day continuously in a 28-day cycle. Treatment was continued until disease progression, unacceptable toxicity, or discontinuation at the patient or investigator’s discretion. Patients with clinical benefit who tolerated treatment could continue selpercatinib beyond progression at the investigator’s discretion.
Patients
Adult Chinese patients (⩾ 18 years of age) with an advanced tumor harboring an activating RET alteration or a prospectively identified RET alteration (fusion or mutation) confirmed by a certified laboratory were eligible for this study. Patients were categorized into three cohorts. Cohort 1 included patients with advanced RET fusion-positive solid tumors who had progressed or were intolerant to one or more prior standard therapies and who had declined, or were considered unsuitable by the investigator for, standard first-line therapy. Cohort 2 included patients with advanced RET-mutant MTC regardless of prior systemic therapy (at the time of initiation of this study, there was no approved standard of care for patients with RET-mutant MTC in China). Patients in Cohorts 1 and 2 were also required to have measurable disease as determined by the investigator, evidence of RET alteration in the tumor (except in germline DNA for patients with MTC in Cohort 2) and have RET status confirmed by central laboratory. Cohort 3 included patients with advanced RET-altered solid tumors and patients who did not fulfil the requirements for Cohorts 1 or 2. Other inclusion criteria included an Eastern Cooperative Oncology Group performance status of 0–2, adequate organ function, and a life expectancy of > 3 months. Key exclusion criteria included prior therapy with a selective RET inhibitor including investigational agents, presence of an additional validated oncogenic driver that could cause resistance to selpercatinib (such as targetable BRAF mutations for patients with TC, targetable re-arrangements of ALK in patients with MTC, or activating RAS mutations in patients with TC or MTC) in Cohorts 1 and 2, and symptomatic primary CNS tumors or metastasis. More details can be found in the protocol.
Endpoints and assessments
The primary endpoint was the proportion of patients in the primary analysis set (PAS) with a confirmed ORR [complete response (CR) or partial response (PR)] as determined using RECIST version 1.1. by an independent review committee (IRC) of expert radiologists. Secondary endpoints included investigator-assessed ORR (RECIST 1.1), clinical benefit rate [the proportion of patients with a best overall response (BOR) of CR, PR, or stable disease (SD) lasting ⩾16 weeks], duration of response (DoR), time to response (TTR), and time to best response (TTBR) assessed by IRC and investigator, progression-free survival (PFS) by IRC and investigator, overall survival (OS), and safety. An exploratory endpoint was biochemical response, assessed by changes in calcitonin and CEA levels. A complete biochemical response was defined as normalization of serum levels following treatment and confirmed ⩾ 4 weeks later; partial response (PR), ⩾ 50% decrease from baseline levels maintained for ⩾ 4 weeks; SD, between +50% and −50% change from baseline levels maintained for ⩾ 4 weeks; and progressive disease, ⩾ 50% increase from baseline maintained ⩾ 4 weeks. The objective biochemical response rate was defined as the proportion of patients achieving a complete or partial biochemical response.
Tumors were assessed by chest, abdomen, and pelvis computed tomography or magnetic resonance imaging at baseline, week 4 (± 7 days, optional), week 8 (± 7 days), and then every 8 weeks (± 7 days) until week 48 following the first dose and every 12 weeks (± 7 days) thereafter. AEs were assessed at each treatment cycle and were categorized and graded according to the National Cancer Institute-Common Terminology Criteria for Adverse Events (CTCAE) v5.0.
Statistical analyses
Efficacy was primarily assessed in the PAS, which comprised all treated patients in Cohorts 1 and 2 who had RET fusion-positive TC or RET-mutant MTC with status confirmed by central laboratory using the KingMed next-generation sequencing (NGS) 529 plus kit. Efficacy was also evaluated in all enrolled patients with MTC. The biochemical response evaluable population included RET-mutant MTC patients in the PAS with abnormal baseline levels of calcitonin or CEA. The safety population consisted of all enrolled patients who received at least one dose of selpercatinib. The planned sample size for Cohorts 1 and 2 was at least 20 patients per cohort, to provide a preliminary assessment of the antitumor activity of selpercatinib. If the observed ORR was high (⩾ 45%) within a cohort of 20 patients, the corresponding lower limit of a two-sided exact 95% CI would exclude true response rates considered marginal (< 40%). With an overall sample size of 75 patients, the probability of observing one or more instances of a specific AE with a true incidence rate of 2% and 5% was approximately 80% and 98%, respectively. DoR was defined as the time from first achieving a PR or CR to disease progression or death. PFS and OS were defined as the time from initiation of selpercatinib to disease progression or death, or death from any cause, respectively. All survival data were summarized descriptively using the Kaplan–Meier method.
Results
Patient characteristics and disposition
A total of 77 patients were enrolled between 16 March 2020 and data cutoff on 25 March 2021 (Supplemental Figure 1), including 29 patients with RET-mutant MTC and one with RET fusion-positive TC. At the time of data cutoff, 27 patients with RET-mutant MTC and one patient with RET fusion-positive TC were still receiving treatment.
The PAS MTC comprised 26 treated patients with MTC and RET-mutant status confirmed by central laboratory. All enrolled patients with MTC (n = 29) were also evaluated. One treatment-naïve patient with TC was included in the TC PAS. This was a 19-year-old female patient diagnosed with a papillary thyroid carcinoma at 12 years old whose tumor NGS testing result showed CCDC6-RET. The patient had previously received multiple surgeries and radiotherapy but no systemic therapy and had lung and lymph node metastasis.
Baseline characteristics for all patients in the PAS MTC and PAS TC are summarized in Table 1. The RET M918T mutation was the most common RET alteration in patients with MTC. Among all 29 patients with MTC, the most common sites of metastasis (present in > 2 patients) were lymph node (86.2%; n = 25), lung (65.5%; n = 19), liver (51.7%; n = 15), bone (31.0%; n = 9), adrenal gland (10.3%; n = 3), and soft tissue (10.3%; n = 3). No patients were diagnosed with CNS metastases at baseline. Among the 26 patients with RET-mutant MTC, 17 (65.4%) were treatment-naïve and nine had previously received therapy, including multikinase inhibitors (n = 4), other systemic therapies (n = 7: anlotinib, n = 4; apatinib, n = 1; etoposide, n = 1; and gimeracil/oteracil/tegafur, n = 1) and platinum-based chemotherapy (n = 1). Baseline characteristics for all enrolled patients with RET-mutant MTC were consistent with the PAS.
Patient demographics and baseline characteristics.

Patients with RET-mutant MTC and RET fusion-positive cancer whose RET status was confirmed by central laboratory, RET alterations in the tumor were detected using the KingMed NGS 529 plus kit.
All enrolled patients with MTC.
Extracellular cysteine mutations included C634R, C634W, and C630Y.
Other mutations included E632-L633DEL and V899-E902DEL in one patient each.
ECOG PS, Eastern Cooperative Oncology Group performance status; Inv, investigator; MTC, medullary thyroid cancer; PAS, primary analysis set; RET, REarranged during Transfection; TC, thyroid cancer.
Antitumor activity
RET-mutant MTC
After a median follow-up of 8.7 months (range, 3–11), the ORR by IRC for the PAS MTC was 57.7% (95% CI, 36.9–76.6; 2 CR; 13 PR; 3 with a PR pending confirmation) and the clinical benefit rate was 61.5% (Table 2). The ORR by IRC was similar for pre-treated (55.6%) and treatment-naïve (58.8%) patients in the PAS. Almost all patients had reductions in tumor size during treatment (Figure 1). The median TTR and TTBR in patients in the PAS with RET-mutant MTC who had CR or PR confirmed by IRC (n = 15) was 1.87 months (min/max: 0.89–5.52) and 1.94 months (min/max: 0.89−5.52), respectively. The median DoR was not reached and 93.3% of responses were ongoing after a median follow-up of 8.7 months (Figure 2(a)). All responses for the pre-treated patients in the PAS MTC (n = 5) and 90% of responses for treatment-naïve patients (n = 10) were ongoing at data cutoff. Median PFS and OS data are not mature.
Antitumor activity of selpercatinib by IRC in patients with RET-mutant MTC.

Patients with RET-mutant MTC whose RET status was confirmed by central laboratory.
All enrolled patients with RET-mutant MTC.
Not including three patients who achieved PR pending confirmation.
CIs estimated using the Clopper–Pearson method.
CI, confidence interval; CR, complete response; IRC, independent review committee, MTC, medullary thyroid cancer; ORR, objective response rate; PAS, primary analysis set; PR, partial response; RET, REarranged during Transfection; SD, stable disease.
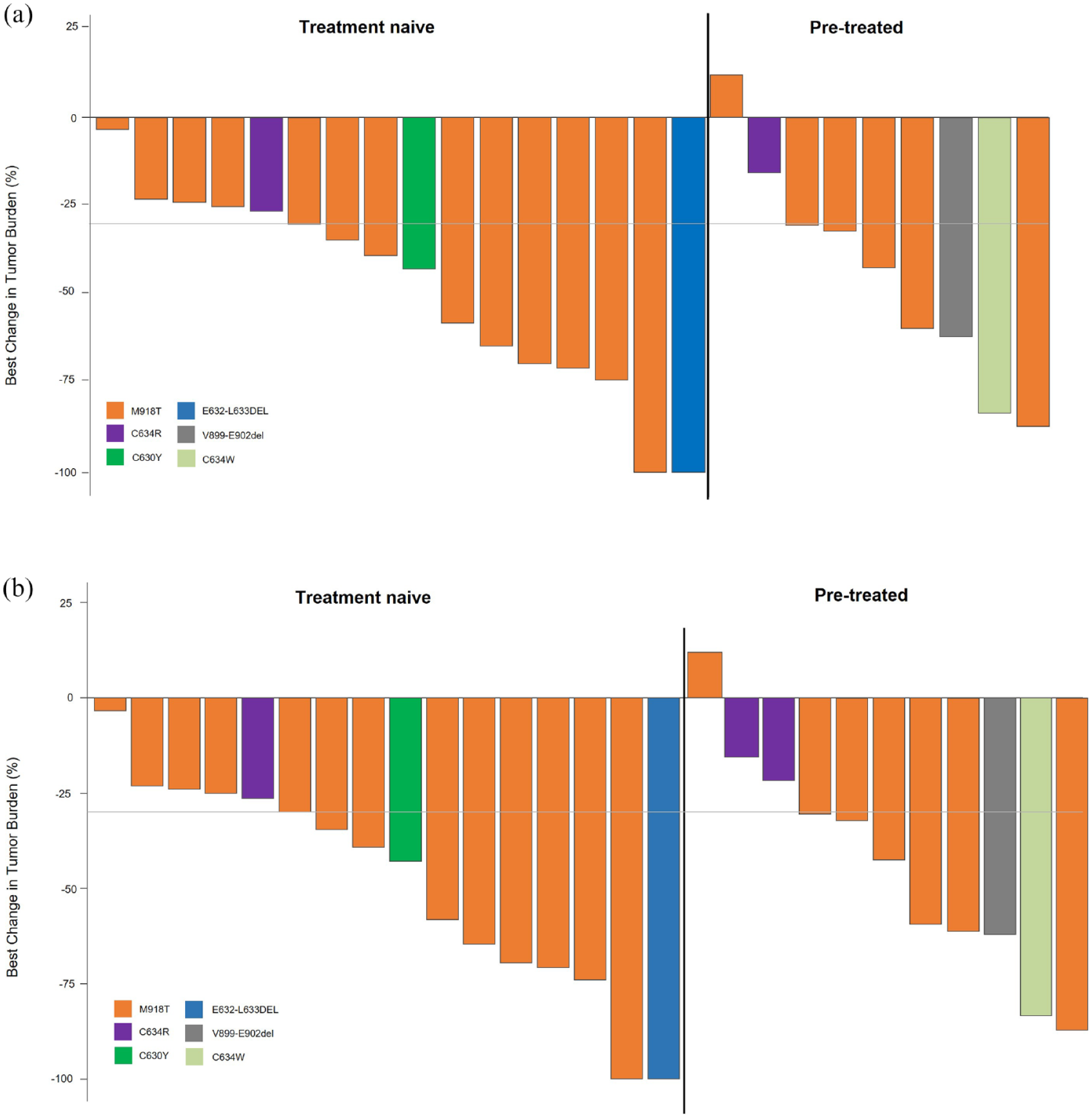
Antitumor activity of selpercatinib in patients with RET-mutant MTC. Waterfall plots of the best change in tumor size for (a) the PAS assessed by the IRC and (b) all enrolled patients with MTC. Each bar represents an individual patient.

DoR in patients with RET-mutant MTC. Kaplan–Meier estimates of DoR in (a) the PAS and (b) all patients with MTC who had CR or PR confirmed by IRC.
Among all enrolled patients with MTC, the ORR by IRC was 58.6% (95% CI, 38.9–76.5) and the clinical benefit rate was 65.5% (Table 2). The ORRs by IRC for pre-treated and treatment-naïve patients were 58.3% and 58.8%, respectively. Among all patients with MTC who had CR or PR confirmed by IRC (n = 17), the median TTR and TTBR was 1.87 (min/max: 0.89−5.52) and 1.94 (min/max: 0.89−5.52), the median DoR was not reached and 94.1% of responses were ongoing after a median follow-up of 9.0 months (Figure 2(b)).
The three patients in Cohort 3 (all of whom had pre-treated MTC) achieved a BOR of CR with duration of 7.2 months, SD with duration of 7.4 months and a PR with duration of 7.4 months. Treatment was ongoing for all three of these patients at data cutoff.
In the PAS (n = 26), one treatment-naïve patient with RET-mutant MTC did not have an evaluable response. Among all enrolled patients with MTC (n = 29), one patient did not have an evaluable response and one had only non-target lesions.
In the biochemical response evaluable population, the percentage of patients with a biochemical response was 92.0% (23/25; 95% CI, 74.0–99.0) for calcitonin and 87.0% (20/23; 95% CI, 66.4–97.2) for CEA (Figure 3).

Biochemical responses in patients with RET-mutant MTC included in the PAS: (a) calcitonin and (b) CEA.
RET fusion-positive TC
The treatment-naïve patient with RET fusion-positive TC was treated for 23.4 weeks and achieved a confirmed PR at week 8 (Supplemental Figure 2). A maximum tumor burden shrinkage of 43% was determined by the IRC and the response was ongoing at cutoff.
Safety
In the safety population (n = 77), the median duration of treatment with selpercatinib was 40.29 weeks (range, 2.29–51.29) and the median number of treatment cycles was 10. Overall, 75 (97.4%) patients experienced at least one treatment-emergent adverse event (TEAE), of which 46 (59.7%) were grade ⩾3 (Table 3). A total of 40 patients (51.9%) experienced grade ⩾ 3 TRAEs and the most common was hypertension (19.5%). Serious TRAEs occurred in 13 (16.9%) patients. In all, 25 (32.5%) patients required a dose reduction and 41 (53.2%) had ⩾1 dose omission (39 due to AEs, 4 due to scheduling conflict, and 1 due to treatment availability). TEAEs leading to dose reductions were hypersensitivity (7; 9.1%), aspartate aminotransferase (AST) increased (n = 6; 7.8%), alanine aminotransferase (ALT) increased (n = 5; 6.5%), platelet count decreased (n = 4; 5.2%), abnormal hepatic function (n = 3; 3.9%), electrocardiogram QT interval prolonged (n = 2; 2.6%), pyrexia (n = 2; 2.6%), blood creatinine increased (n = 1; 1.3%), liver injury (n = 1; 1.3%), and anemia (n = 1; 1.3%). TEAEs leading to dose interruptions occurred in 39 (50.6%) patients and included AST increased and hypersensitivity (both 7.8%), hypertension, ALT increased, and blood creatinine increased (all, 5.2%). TEAEs leading to selpercatinib discontinuation occurred in four (5.2%) patients, of which 3.9% (n = 3) were considered treatment related by the investigator: hypersensitivity, platelet count decreased, and abnormal liver function in one patient each. There was one (1.3%) grade 5 TEAE considered unrelated to selpercatinib; acute pancreatitis in a 46-year-old male patient with RET-mutant MTC.
AEs in all selpercatinib-treated patients (n = 77).
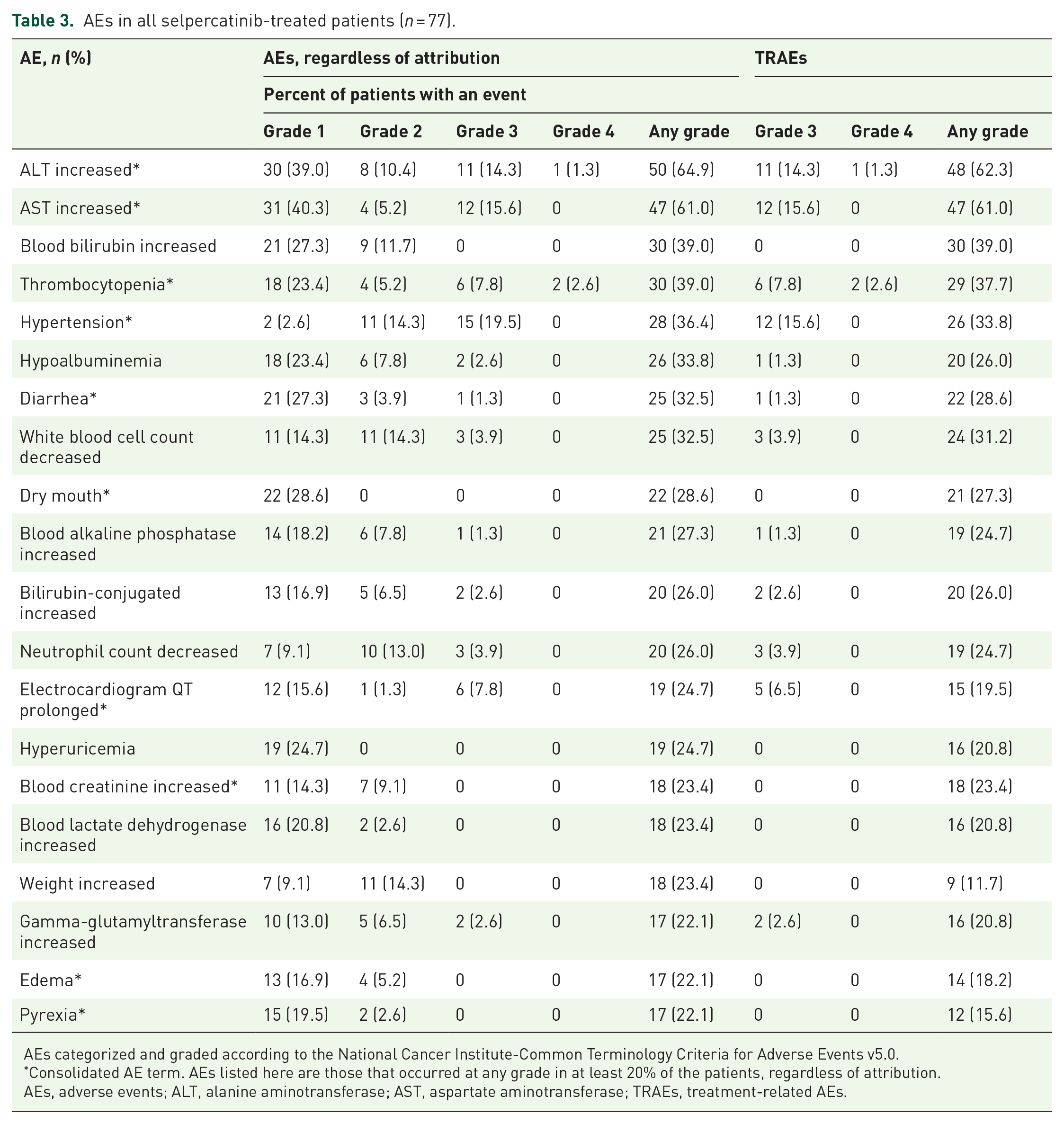
AEs categorized and graded according to the National Cancer Institute-Common Terminology Criteria for Adverse Events v5.0.
Consolidated AE term. AEs listed here are those that occurred at any grade in at least 20% of the patients, regardless of attribution.
AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TRAEs, treatment-related AEs.
The one patient with RET fusion-positive TC experienced TRAEs including increased ALT/AST levels, diarrhea, electrocardiogram QT prolonged, hypocalcemia, decreased neutrophil and white blood cell counts, purpura and increased weight. No grade ⩾ 3 TRAEs occurred. The AEs that occurred in all RET-mutant MTC patients (n = 29) are summarized in Supplemental Table 2.
Discussion
The results of this phase II study show that selpercatinib has robust and rapid antitumor activity in Chinese patients with advanced RET-altered TC; the median time to CR or PR among all patients with MTC was 1.87 months. Selpercatinib also showed an acceptable safety profile. Continued follow-up of all patients in this trial will provide further information on the durability of response to selpercatinib.
The results of the present study are broadly consistent with data from the global phase I-II LIBRETTO-001 trial. 6 In LIBRETTO-001, the ORR in pre-treated and treatment-naïve patients with RET-mutant MTC was 69% (95% CI, 55–81) and 73% (95% CI, 62–82), respectively, compared with 56% (95% CI, 21–86) and 59% (95% CI, 33–82) in the present study. However, it should be noted that LIBRETTO-001 had a longer median follow-up time and a larger number of patients. In addition, responses for the patients in the present study were still ongoing at cutoff, with three patients with an unconfirmed PR and multiple patients with SD that may be reclassified as a PR after longer follow-up. These factors are reflected in the lower 95% CI lower bounds as well as lower point estimates for ORR from the present study, and these would be expected to increase with further follow-up as the data mature. The antitumor activity of selpercatinib in our study also compares favorably with previous reports of responses in patients with MTC treated with vandetanib (45%) and cabozantinib (28%).11,18 In December 2020, pralsetinib was approved for the treatment of patients with advanced RET-altered TC following positive ORR results from the phase I/II ARROW study. 19 Together, these findings suggest that selective RET inhibitors may provide greater therapeutic benefit for patients with RET-mutant MTC and RET fusion-positive TC compared to multi-kinase inhibitors. Results from the first global head-to-head comparative phase III trial of selpercatinib versus physician’s choice of cabozantinib or vandetanib for the treatment of multi-kinase inhibitor-naïve progressive advanced or metastatic RET-mutant MTC (LIBRETTO-531, NCT04211337) will provide further evidence on the benefits of highly selective RET inhibitors as targeted therapy for RET-mutant MTC.
Selpercatinib treatment was associated with grade ⩾ 3 TRAEs such as hypertension and increased ALT/AST levels, which were manageable and reversible with dose interruptions, dose reductions, or addition of concomitant medications. These observations in Chinese patients are generally consistent with results from the global LIBRETTO-001 population, with a similar proportion of patients experiencing at least one TEAE of any grade (100% and 97.4%) and a similar profile of AEs. 6 In addition, a similar proportion of patients in the LIBRETTO-001 trial and the present study experienced TEAEs leading to dose reductions (30.0% and 32.5%) and selpercatinib discontinuation due to TRAEs (2.0% and 3.9%). 6 However, most AEs in the present study were low grade and manageable, suggesting that the safety profile of selpercatinib is acceptable in this patient population, possibly due to the high selectivity of selpercatinib for RET.
One of the limitations of this study was its single arm, open-label design, which could have introduced a source of bias. However, the efficacy results were based on independent review of tumor assessment, which may have reduced this potential bias. This study also had a relatively small sample size, especially for subgroup analyses, and the results should be interpreted cautiously. The efficacy and safety of selpercatinib will be investigated in a larger population in the global phase III study, which includes Chinese study sites.
Conclusions
Selpercatinib showed robust and durable antitumor activity in Chinese patients with advanced RET-altered TC and was well tolerated, consistent with previously reported data from the global LIBRETTO-001 trial. These findings suggest that selpercatinib represents a valuable treatment option for Chinese patients with advanced RET-altered TC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221119318 – Supplemental material for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study
Supplemental material, sj-docx-1-tam-10.1177_17588359221119318 for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study by Xiangqian Zheng, Qinghai Ji, Yuping Sun, Minghua Ge, Bin Zhang, Ying Cheng, Shangtong Lei, Feng Shi, Ye Guo, Linfa Li, Lu Chen, Jingxin Shao, Wanli Zhang and Ming Gao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359221119318 – Supplemental material for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study
Supplemental material, sj-jpg-2-tam-10.1177_17588359221119318 for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study by Xiangqian Zheng, Qinghai Ji, Yuping Sun, Minghua Ge, Bin Zhang, Ying Cheng, Shangtong Lei, Feng Shi, Ye Guo, Linfa Li, Lu Chen, Jingxin Shao, Wanli Zhang and Ming Gao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359221119318 – Supplemental material for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study
Supplemental material, sj-jpg-3-tam-10.1177_17588359221119318 for Efficacy and safety of selpercatinib in Chinese patients with advanced RET-altered thyroid cancers: results from the phase II LIBRETTO-321 study by Xiangqian Zheng, Qinghai Ji, Yuping Sun, Minghua Ge, Bin Zhang, Ying Cheng, Shangtong Lei, Feng Shi, Ye Guo, Linfa Li, Lu Chen, Jingxin Shao, Wanli Zhang and Ming Gao in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Editorial support and submission support for this manuscript was provided by Jake Burrell PhD (Rude Health Consulting Limited, 2 Pulteney Road, Bath, BA2 4HD, +44 07708935518) and paid for by Eli Lilly and Company.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
