Abstract
Background:
Modified fluorouracil/leucovorin/irinotecan/oxaliplatin (FOLFIRINOX) regimen (mFOLFIRINOX), comprised of fluorouracil, leucovorin, irinotecan and oxaliplatin, is the first-line standard chemotherapy in patients with advanced pancreatic cancer. The S-1/oxaliplatin/irinotecan (SOXIRI) regimen has also been studied recently under similar conditions. This study compared its efficacy and safety.
Methods:
All cases of locally advanced or metastatic pancreatic cancer treated with the SOXIRI or mFOLFIRINOX regimen in Sun Yat-sen University Cancer Centre from July 2012 to June 2021 were reviewed retrospectively. The data of patients who satisfied the inclusion criteria were compared between two cohorts, including overall survival (OS), progression-free survival (PFS), objective response rate, disease control rate and safety.
Results:
A total of 198 patients were enrolled in the study, including 102 patients treated with SOXIRI and 96 patients treated with mFOLFIRINOX. There was no significant difference in OS [12.1 months versus 11.2 months, hazard ratio (HR) = 1.04, p = 0.81] or PFS (6.5 months versus 6.8 months, HR = 0.99, p = 0.96) between patients treated with SOXIRI and mFOLFIRINOX. In the subgroup analysis, patients with slightly elevated baseline total bilirubin (TBIL) or underweight patients before chemotherapy were more likely to have a longer OS or PFS from SOXIRI than from mFOLFIRINOX. In addition, the carbohydrate antigen (CA)19-9 decline was a good predictor for the efficacy and prognosis of both chemotherapy regimens. All grade adverse events were parallel in all kinds of toxicities except that anaemia was more common in the SOXIRI group than in the mFOLFIRINOX group (41.4% versus 24%, p = 0.03). The occurrence of any grade 3 to 4 toxicity was similar in the two groups.
Conclusions:
For locally advanced or metastatic pancreatic cancer patients, the SOXIRI regimen had similar efficacy and controllable safety compared with the mFOLFIRINOX regimen.
Introduction
Pancreatic cancer is the seventh leading cause of cancer death worldwide. 1 It has a poor prognosis, with a 5-year survival rate of only 9%. Less than 20% of initially diagnosed pancreatic cancers are surgically removed. 2 Most patients are often diagnosed with advanced stages, including locally advanced pancreatic cancer and metastatic pancreatic cancer. Chemotherapy is the main treatment for unresectable pancreatic cancer. Since 1997, gemcitabine has been the standard first-line treatment. 3 However, a large number of prospective studies have demonstrated that combination regimens are superior to monotherapy.4,5
In the MPACT trial, 5 which involved 861 patients with metastatic pancreatic cancer, gemcitabine combined with nab-paclitaxel (Gem/Nab-P) showed a significant overall survival (OS) advantage compared with gemcitabine (8.5 months versus 6.7 months, p < 0.001). Therefore, for patients with advanced pancreatic cancer, Gem/Nab-P is recommended as the preferred first-line chemotherapy. The randomized phase III PRODIGE trial showed that fluorouracil/leucovorin/irinotecan/oxaliplatin (FOLFIRINOX) significantly improved median OS and progression-free survival (PFS) in patients with metastatic pancreatic cancer compared with gemcitabine. 4 However, the incidence of grade 3–4 toxicity of this regimen was too high to be applied to all patients. Thus, several studies have investigated dose modifications of this regimen, modified FOLFIRINOX (mFOLFIRINOX), and verified its similar efficacy and improved safety compared to FOLFIRINOX.6–8 FOLFIRINOX/mFOLFIRINOX has been recommended as the standard first-line chemotherapy regimen for advanced pancreatic cancer with good performance status not only by the National Comprehensive Cancer Network (NCCN) guidelines, but also by numerous European and Asian guidelines.9–13 However, this four-drug regimen containing 5-fluorouracil (5-FU) usually poses some problems for clinical practice. Continuous intravenous administration of fluorouracil within 46 h is now the standard fluorouracil administration protocol.14,15 First, this usually requires the implantation of a central venous access devices, such as implanted venous access ports or peripherally inserted central catheters, which can be associated with complications such as pneumothorax, haemothorax, or catheter rupture. 16 In addition, catheter-related infections 17 or thrombosis 18 can have a serious impact on some patients. Finally, intubation tends to be associated with a greater financial burden. 19 In China and other developing countries, most patients have insufficient awareness of medical safety and self-care, coupled with the unaffordable cost of disconnecting central venous access devices by public health services. Thus, this often requires hospitalization and close monitoring. In summary, FOLFIRINOX or mFOLFIRINOX is often not user friendly enough for patients in China and other developing countries. It is important to develop an alternative that is comparable and more convenient, safe and economical.
S-1 (Taiho Pharmaceutical, Tokyo, Japan) is an oral fluoropyrimidine anti-cancer agent designed to enhance the anti-tumour activity of 5-FU while reducing its toxic effects. 20 S-1 includes tegafur (a prodrug of 5-FU) and two 5-FU modulators: gimeracil, which inhibits degradation of 5-FU, thereby maintaining high 5-FU concentration, and oteracil potassium, which reduces 5-FU levels in the gut, reducing gastrointestinal toxicity.21,22 In the Asian region, S-1 has rapidly become the standard of care for pancreatic cancer, including postoperative adjuvant chemotherapy approved for resectable disease and first-line chemotherapy for metastatic disease.23,24 As a novel fluoropyrimidine derivative, S-1 has several advantages such as sustained activity, low toxicities, favourable efficacy, oral accessibility and cost-effectiveness, thereby rendering it a viable alternative to 5-FU in numerous chemotherapy regimens. The S-1, oxaliplatin and irinotecan (SOXIRI) regimen comprised of S-1, oxaliplatin and irinotecan has been implemented in clinical practice as a replacement for the FOLFIRINOX and mFOLFIRINOX regimens. Employment of this regimen allows for oral administration of S-1, eliminating the need for intravenous administration of 5-FU. Additionally, adoption of an alternate-day dosing strategy for S-1 has been shown to reduce treatment-related adverse events. 25 A multi-centre, randomized, phase II study revealed that in patients with unresectable advanced pancreatic cancer, alternate-Day S-1 dosing was noninferior to daily dosing in terms of OS (9.4 months versus 10.4 months) while significantly reducing the incidence of treatment-related adverse events. 26
Several phase II single-arm trials have preliminarily demonstrated the efficacy and safety of a biweekly regimen of SOXIRI in the first-line treatment of advanced pancreatic cancer.27,28 Nevertheless, head-to-head comparisons between SOXIRI and mFOLFIRINOX are lacking until now. Hence, we conducted this single-centre retrospective analysis to evaluate the efficacy and safety of SOXIRI and mFOLFIRINOX in the first-line treatment of advanced pancreatic cancer.
Methods
Patient selection
This study retrospectively analysed electronic clinical medical records of patients with locally advanced or metastatic pancreatic cancer who received first-line SOXIRI or mFOLFIRINOX at Sun Yat-sen University Cancer Center from July 2012 to June 2021. For locally advanced pancreatic cancer, we first diagnosed according to the criteria of NCCN guidelines. Only when there was a diagnostic dispute did we organize a multidisciplinary team to discuss it and record the final diagnosis in the electronic medical record. Approximately half of the patients in this study had participated in clinical studies led by Sun Yat-sen University Cancer Center. Of these, 61% of the patients in the SOXIRI regimen had participated in a phase II single-centre clinical trial of SOXIRI, whereas 40% of the patients in the mFOLFIRINOX regimen had participated in a phase II multicentre clinical trial of mFOLFIRINOX. 6 Both clinical trials were registered in ClinicalTrials.gov with the numbers ChiCTR1800018233 and NCT02028806. This indicates that the source of our clinical data is reliable and of high quality. The follow-up period was up to 20 February 2022.
Inclusion criteria were as follows: (1) Above the age 18 years, and Eastern Cooperative Oncology Group (ECOG) performance status score of 0 or 1; (2) histologically and pathologically confirmed pancreatic ductal adenocarcinoma; (3) locally advanced or metastatic pancreatic cancer that had not received prior chemotherapy in an unresectable setting or more than 6 months since the last chemotherapy if neoadjuvant/adjuvant chemotherapy was ever given; (4) measurable pancreatic adenocarcinoma and/or metastatic lesions; (5) patients with adequate bone marrow, kidney and liver functions, granulocyte count ⩾1500/mm3; and platelet count ⩾100,000/mm3. Baseline total bilirubin (TBIL) level ⩽1.5 × the upper limit of normal (ULN) or ⩽5 × ULN after percutaneous transhepatic cholangial drainage (PTCD), and slightly elevated TBIL was defined as 1–1.5 × ULN or 1–5 × ULN after biliary drainage; creatinine clearance measured value or estimated value using the Cockcroft-Gault equation, ⩾60 mL/min. Exclusion criteria were endocrine or acinar pancreatic cancer, previous radiation for measurable lesions in 6 months, a history of other major cancers, active infections, chronic diarrhoea, a history of clinically significant heart disease and pregnancy or breastfeeding. The detailed patient selection process is shown in Supplemental Figure S1.
Chemotherapy regimens
The SOXIRI regimen consisted of oxaliplatin (85 mg/m2) for 2 h intravenously and irinotecan (150 mg/m2) for 90 min intravenously. S-1 (80 mg/m2) was orally administered twice daily, every other day for 2 weeks.27,29 The mFOLFIRINOX regimen includes 2 h of intravenous oxaliplatin (65 mg/m2), 2 h of intravenous leucovorin (400 mg/m2), 90 min of irinotecan (150 mg/m2) and 46 h of continuous intravenous fluorouracil (2400 mg/m2). This regimen was repeated every 2 weeks. 6
Treatment evaluation
For patients who responded, up to 12 cycles of treatment were recommended, and then S-1 was first recommended as subsequent treatment or observation for the cases with poor physical status or intolerable adverse effects. Treatment was continued until the disease progressed or an intolerable adverse event occurred. OS and PFS were assessed by the length of time from the initiation of first-line chemotherapy to the date of evaluation. OS was defined as the time from the start of first-line chemotherapy to death or the last follow-up. PFS was defined as the time from the start of first-line chemotherapy to the diagnosis of disease progression, death from any cause or last follow-up. Imaging tests, including computed tomography or magnetic resonance imaging, were performed every 8 weeks to assess treatment response. The judgement was made by the clinician based on RECIST version 1.1. 30 Toxicity was assessed at the beginning of each treatment cycle and graded according to the standard for General Terminology of Adverse Events (CTCAE) version 4.0. 31 For any kind of toxicity, we recorded the highest grade during treatment and compared the difference between the two regimens.
For the baseline evaluation, clinicopathological data and biochemical tumour detection indicators were collected before initial treatment. These included age, sex, ECOG score, clinical stage, pathological grade, metastatic site, tumour site, biliary drainage, hepatitis B, diabetes, height, weight, albumin, bilirubin (TBIL, direct bilirubin and indirect bilirubin), aminotransferase (aspartate aminotransferase and alanine aminotransferase), serum creatinine, and tumour markers [carbohydrate antigen (CA)19-9, CA12-5, CA24-2, CA72-4 and carcino-embryonic antigen]. Height and weight were further analysed by calculating body mass index, which was classified into low weight, normal weight and overweight based on the normal range of 18.5–25.0 kg/m2. The normal ranges of biochemical and tumour markers were based on the standards established by the Clinical Laboratory of Sun Yat-sen University Cancer Center (Supplemental Table S1).
The weight and CA19-9 level of patients before each cycle of chemotherapy were continuously collected for response assessment and survival analysis. Weight loss was defined as two or more consecutive pounds from baseline weight compared to the previous weight without a cut-off threshold. The CA19-9 decline was defined as two or more consecutive decreases of more than 50% from baseline CA19-9 levels compared to the previous CA19-9 level without a cut-off threshold.
Statistical analysis
The end points of this study included OS, PFS, objective response rate (ORR), disease control rate (DCR) and safety. Categorical variables were expressed as counts (percentages) and compared using the chi-square test or Fisher’s exact test. The continuous variables were first tested for normal distribution. Normally distributed continuous variables were expressed as the mean values and compared by Student’s t-test. Non-normally distributed continuous data were represented as medians and compared by a non-parametric (Wilcoxon) test. A Cox proportional risk regression model was used to determine the factors affecting survival. Survival was estimated using the Kaplan–Meier method, and survival data were compared by log-rank test. All of these comparisons were adjusted for stratification. Correlations between reductions in weight and CA19-9 levels and efficacy were calculated using Spearman analysis. All tests were two-sided, and p values less than 0.05 were considered statistically significant. Data were presented as 95% confidence intervals (CIs) and calculated using standard methods based on binomial distributions. All analyses were performed using R software (version 4.0.2; Lucent Technologies Co., Ltd., China).
Results
Patient characteristics
A total of 198 patients with locally advanced or metastatic pancreatic cancer who received SOXIRI (n = 102) or mFOLFIRINOX (n = 96) as first-line chemotherapy were enrolled in this study. Supplemental Table S2 reports the cumulative number of cases per year for the SOXIRI or mFOLFIRINOX regimen from 2012 to 2021. The characteristics of patients in the SOXIRI and mFOLFIRINOX groups are shown in Table 1. There were no significant differences in baseline characteristics between the two groups. The median chemotherapy cycle was five in the SOXIRI group and six in the mFOLFIRINOX group. At the same time, 20.6% (n = 21) of the patients in the SOXIRI group and 16.7% (n = 16) of the patients in the mFOLFIRINOX group did not undergo imaging evaluation, but survival information was still obtained through telephone follow-up. Only two patients in the mFOLFIRINOX group were older than 75 years and both received routine prophylaxis with granulocyte colony-stimulating factor during treatment. No patients in the SOXIRI group were older than 75 years. In the SOXIRI cohort, 30% of patients received second-line chemotherapy, whereas in the mFOLFIRINOX cohort, the corresponding proportion was 40%, with gemcitabine plus nab-paclitaxel being the most commonly administered regimen (Supplemental Table S3).
Baseline characteristics of SOXIRI and mFOLFIRINOX.

Multisite* refers to the presence of a primary tumour in two or more primary sites (head, body or tail) of the pancreas.
BMI, body mass index; CA, carbohydrate antigen; CEA, carcino-embryonic antigen; ECOG, Eastern Cooperative Oncology Group; mFOLFIRINOX, modified fluorouracil/leucovorin/irinotecan/oxaliplatin; PTCD, percutaneous transhepatic cholangial drainage; SOXIRI, S-1/oxaliplatin/irinotecan.
Efficacy
Univariate and multivariate Cox regression analyses were performed for OS and PFS on the basis of included baseline characteristics in all patients with SOXIRI or mFOLFIRINOX (Supplemental Tables S4 and S5). After multivariate Cox regression adjustment, treatment cycle [hazard ratio (HR) = 0.41, p < 0.01], sex (HR = 0.68, p = 0.03) and liver metastasis (HR = 1.59, p = 0.04) were independent prognostic factors for OS, that is, ⩾6 cycles, female sex and no liver metastasis were good prognostic factors. However, only treatment cycle (HR = 0.26, p < 0.01) was an independent prognostic factor for PFS in multivariate Cox regression.
There was no statistically significant difference in median OS and PFS between the two groups. The median OS for SOXIRI and mFOLFIRINOX was 12.1 (95% CI 9.9–15.9) months and 11.2 (95% CI 8.8–14.7) months (HR = 1.04, 95% CI 0.76–1.43, p = 0.81; Figure 1(a)), while the median PFS was 6.5 (95% CI 5.2–8.7) months and 6.8 (95% CI 4.7–8.8) months (HR = 0.99, 95% CI 0.74–1.34, p = 0.96; Figure 1(b)). In locally advanced pancreatic cancer, the median OS for SOXIRI and mFOLFIRINOX was 13.5 (95% CI 11.9–NA) months and 12.0 (95% CI 7.7–NA) months (HR = 0.95, 95% CI 0.46–1.95, p = 0.88; Figure 2(a)), while the median PFS was 8.2 (95% CI 5.2–16.8) months and 7.1 (95% CI 4.7–21.6) months (HR = 0.93, 95% CI 0.48–1.82, p = 0.84; Figure 2(b)). Meanwhile, in metastatic pancreatic cancer, the median OS for SOXIRI and mFOLFIRINOX was 9.9 (95% CI 8.7–15.9) months and 10.7 (95% CI 8.5–14.7) months (HR = 1.02, 95% CI 0.71–1.46, p = 0.91; Figure 2(c)), respectively, while the median PFS was 6.3 (95% CI 4.5–8.7) months and 6.2 (95% CI 4.2–9.0) months (HR = 1.10, 95% CI 0.78–1.54, p = 0.60; Figure 2(d)), respectively. The specific efficacy comparison between the SOXIRI and mFOLFIRINOX is shown in Table 2. The OS rates at 6, 12, 18 and 24 months for SOXIRI and mFOLFIRINOX were 73%, 48%, 32%, 17% and 74%, 46%, 32%, 19%, respectively, and the PFS rates were 51%, 24%, 10% and 50%, 21%, 12%, respectively. The ORRs in the SOXIRI and mFOLFIRINOX groups were 23.5% (95% CI 0.157–0.330) and 22.9% (95% CI 0.150–0.326) (p = 1.00), respectively, and the DCRs were 62.7% (95% CI 0.526–0.721) and 57.3% (95% CI 0.468–0.721) (p = 0.52), respectively. There were 24, 40 and 17 patients with partial response (PR), stable disease (SD) and progressive disease (PD) in the SOXIRI group compared with 22, 33 and 25 patients in the mFOLFIRINOX group, respectively. The waterfall plots showed the distribution of patients with the best response to each regimen (Figure 3). There was no statistically significant difference in the best efficacy evaluation between them (p = 0.43). The two regimens took similar median days to achieve optimal efficacy (76 days versus 69 days, p = 0.28).
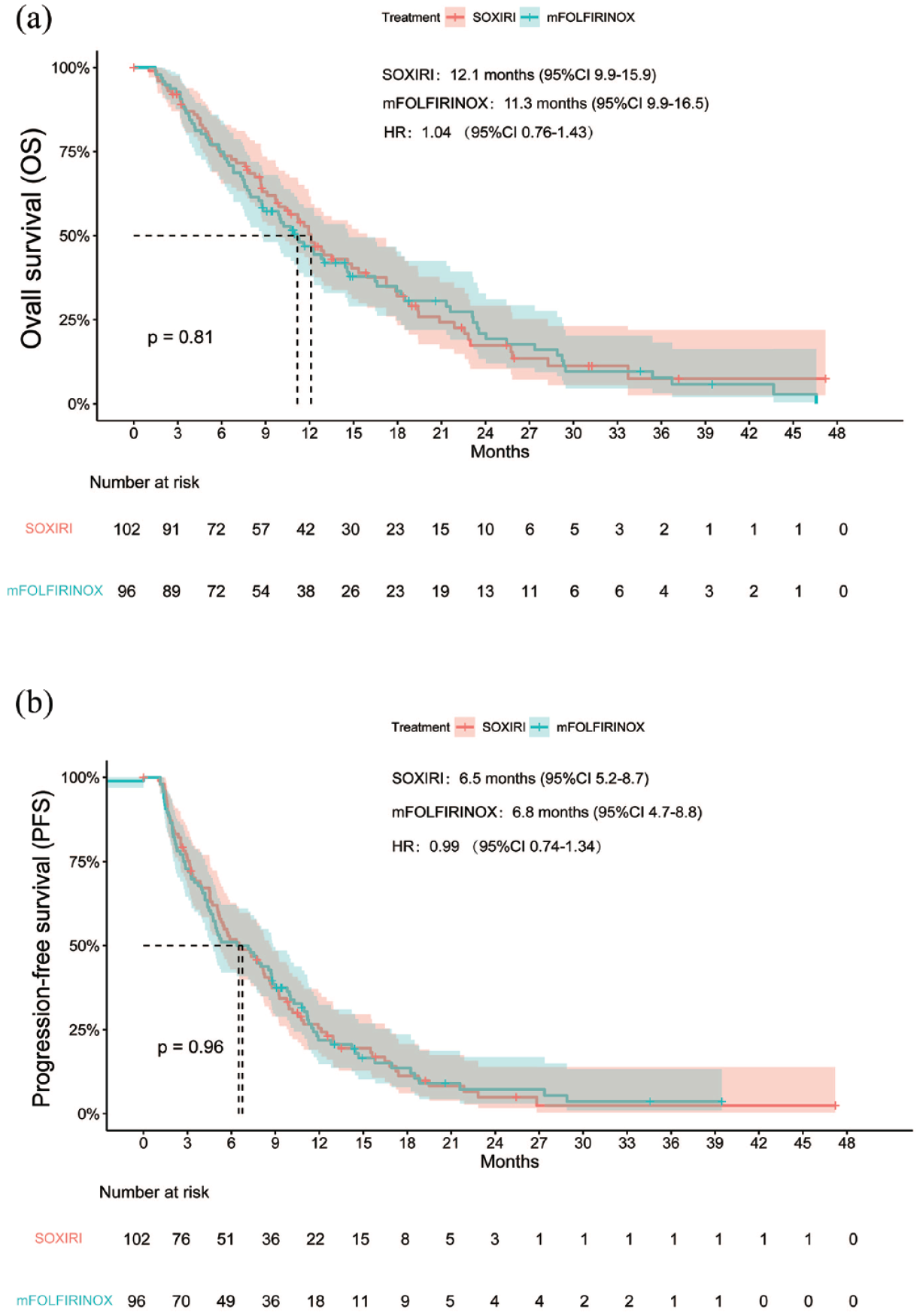
Survival curves of OS (a) and PFS (b) in SOXIRI versus mFOLFIRINOX.

Survival curves of OS and PFS in SOXIRI versus mFOLFIRINOX in stage III (a and b) and IV (c and d) pancreatic cancer.
Efficacy of SOXIRI and mFOLFIRINOX.

CI, confidence interval; mFOLFIRINOX, modified fluorouracil/leucovorin/irinotecan/oxaliplatin; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; SOXIRI, S-1/oxaliplatin/irinotecan.

Waterfall plots of the best efficacy evaluation for SOXIRI (a) and mFOLFIRINOX (b).
Subgroup analysis of OS and PFS by SOXIRI and mFOLFIRINOX was shown through forest plots. SOXIRI may have a better survival benefit than mFOLFIRINOX when patients were underweight (BMI < 18.5 kg/m2; HR = 3.17, 95% CI 1.01–9.95, p = 0.04) or their baseline TBIL was slightly elevated (TBIL was within 1–1.5 × ULN or 1–5 × ULN after biliary drainage; HR = 7.24, 95% CI 1.64–31.96, p < 0.01) (Supplemental Figure S2). Other subgroups showed similar OS benefits between the two groups. The subgroup analysis of PFS found that when the baseline TBIL of patients was slightly elevated (HR = 21.29, 95% CI 2.38–23.92, p < 0.01), the PFS of SOXIRI was superior to that of mFOLFIRINOX (Supplemental Figure S3). Other subgroups showed similar PFS benefits between them.
Comprehensive analysis of weight loss, CA19-9 decline and efficacy
There were no statistically significant differences in baseline weight between PR, SD and PD for the three cohorts, including all groups with SOXIRI plus mFOLFIRINOX cohort, SOXIRI cohort and mFOLFIRINOX cohort (Figure 4(a), (d) and (g)). Meanwhile, the percentage of weight loss between PR, SD and PD showed no statistically significant difference for any cohort (Figure 4(b), (e) and (h); Fisher’s test, p > 0.05). Disease progression was positively associated with only weight loss in all groups (Figure 4(c); R = 0.21, p = 0.025), while neither the SOXIRI (Figure 4(f); R = 0.25, p = 0.06) nor the mFOLFIRINOX cohort (Figure 4(i); R = 0.16, p = 0.23) had found this relationship. In addition, there was statistically significant difference in OS or PFS between patients with or without weight loss for all three cohorts (Supplemental Figure S4A).

Comprehensive analysis of weight loss and efficacy. (a) Baseline weight of PR, SD and PD for all chemotherapy regimens, (b) proportion of patients with or without weight loss in PR, SD and PD for all chemotherapy regimens, (c) Spearman correlation between efficacy and weight loss for all chemotherapy regimens, (d) baseline weight of PR, SD and PD for SOXIRI, (e) proportion of patients with or without weight loss in PR, SD and PD for SOXIRI, (f) Spearman correlation between efficacy and weight loss for SOXIRI, (g) baseline weight of PR, SD and PD for mFOLFIRINOX, (h) proportion of patients with or without weight loss in PR, SD and PD for mFOLFIRINOX and (i) Spearman correlation between efficacy and weight loss for mFOLFIRINOX.
There were no statistically significant differences in baseline CA19-9 levels between PR, SD and PD for the three cohorts (Figure 5(a), (d) and (g)). Interestingly, the percentage of CA19-9 decline between PR, SD and PD showed statistically significant difference for any cohort (Figure 5(b), (e) and (h); Fisher’s test, p < 0.05). In addition, disease progression was inversely associated with CA19-9 decline in all groups (Figure 5(c); R = −0.38, p = 2e-5), SOXIRI (Figure 5(f); R = −0.39, p = 0.0021) and mFOLFIRINOX (Figure 5(i); R = −0.38, p = 0.0042). Despite that there was no statistically significant difference in OS (log-rank test, p = 0.088) between the cases with and without CA19-9 decline for the SOXIRI cohort, both OS of the other two cohorts and PFS of all three cohorts were longer in the patients with CA19-9 decline than in those with no-decline (Supplemental Figure S4B).

Comprehensive analysis of CA19-9 decline and efficacy. (a) Baseline CA19-9 level of PR, SD and PD for all chemotherapy regimens, (b) proportion of patients with or without CA19-9 decline in PR, SD and PD for all chemotherapy regimens, (c) Spearman correlation between efficacy and CA19-9 decline for all chemotherapy regimens, (d) baseline CA19-9 level of PR, SD and PD for SOXIRI, (e) proportion of patients with or without CA19-9 decline in PR, SD and PD for SOXIRI, (f) Spearman correlation between efficacy and CA19-9 decline for SOXIRI, (g) baseline CA19-9 level of PR, SD and PD for mFOLFIRINOX, (h) proportion of patients with or without CA19-9 decline in PR, SD and PD for mFOLFIRINOX and (i) Spearman correlation between efficacy and CA19-9 decline for mFOLFIRINOX.
Safety
Adverse events observed in both groups included haematological toxicity (leukopenia, neutropenia, thrombocytopenia, anaemia), hepatotoxicity (increased bilirubin or transaminase), nephrotoxicity (increased serum creatinine) and other toxicities (fatigue, diarrhoea, vomiting and peripheral sensory neuropathy) (Table 3). All grade adverse events occurred in 83 and 70 patients in the SOXIRI and mFOLFIRINOX groups (81.4% versus 72.9%, p = 0.21), respectively. At the same time, 40 and 23 patients developed grade 3 or 4 adverse events in the SOXIRI and mFOLFIRINOX groups (46.0% versus 30.3%, p = 0.06), respectively. There were no treatment-related deaths in either group. Among all grades of adverse events, only anaemia was more likely to occur in the SOXIRI group than in the mFOLFIRINOX group (41.4% versus 24%, p = 0.03). However, we did not find significant differences in the rates of any grade 3 or 4 adverse events. No patients in the mFOLFIRINOX group experienced thrombocytopenia.
Adverse events of SOXIRI and mFOLFIRINOX.

mFOLFIRINOX, modified fluorouracil/leucovorin/ irinotecan/oxaliplatin; SOXIRI, S-1/oxaliplatin/irinotecan.
Discussion
Previous studies have shown that SOXIRI is a promising and well-tolerated regimen for patients with unresectable pancreatic cancer.27–29,32 However, because all studies were single-arm I/II clinical studies based on SOXIRI regimens, head-to-head studies comparing SOXIRI with standard first-line treatment regimens are lacking. Our study first compared the efficacy and safety of SOXIRI with mFOLFIRINOX, the current standard first-line treatment for advanced pancreatic cancer, using retrospective data analysis from a single centre.
Our study found that SOXIRI and mFOLFIRINOX had comparable efficacy and acceptable safety profiles as first-line treatments for patients with locally advanced or metastatic pancreatic cancer. There was no significant difference in OS (12.1 months versus 11.2 months, HR = 1.04, p = 0.81) or PFS (6.5 months versus 6.8 months, HR = 0.99, p = 0.96) between SOXIRI and mFOLFIRINOX. Interestingly, in the subgroup analysis, patients with baseline slightly elevated TBIL or who were underweight were more likely to benefit more from SOXIRI than mFOLFIRINOX. Meanwhile, only the all groups had a positive correlation between disease progression and weight loss, while in the all groups, the SOXIRI and mFOLFIRINOX groups, a negative correlation was found between disease progression and decreased CA19-9 levels. In addition, although only anaemia of all grade adverse events was more common in the SOXIRI group than in the mFOLFIRINOX group (41.4% versus 24%, p = 0.03), the incidence of grade 3 or 4 adverse events was comparable and well controlled between the two groups.
There are several arguments to support the clinical application of S-1 in place of 5-FU. First, because of the apparent time-dependent nature of the drug, a 46 h continuous intravenous infusion of 5-FU is essential, regardless of whether the FOLFIRINOX regimen is adjusted. However, recent studies have shown that in some regimens, 5-FU continuous infusion chemotherapy can be replaced with oral agents such as S-1 or capecitabine,16,33 without significant changes in efficacy or adverse events. Second, oral S-1 avoids some medical problems. In addition, 5-FU usually requires implantation of a central intravenous infusion devices, which might induce complications related to intubation and catheter care. 16 Meanwhile, the cost of using a central venous infusion is often higher, which imposes a heavier financial burden on patients. 19 Third, S-1 is a combination of tegafur, gimeracil and oteracil potassium that is slowly converted to 5-FU in the body after oral administration, which persists at high concentrations while being able to reduce gastrointestinal toxicity. 21 In contrast, 5-FU has a very short half-life and is rapidly metabolized by the liver upon entry into the body, making it impossible to maintain effective blood levels. 34 Finally, 5-FU requires intravenous administration and regular patient visits to the hospital. As an oral medicine, S-1 has the advantages of simplicity, convenience and economy. 35 In summary, we believe that S-1 is relatively friendly for Chinese patients and even Asian patients, and thus it is very reasonable to replace 5-FU with S-1.
Two single-arm phase II studies reported the efficacy and safety of SOXIRI for advanced pancreatic cancer. A single-arm phase II study conducted by Akahori et al. 27 found that 35 patients with advanced pancreatic cancer treated with SOXIRI had a median OS of 17.7 months, a median PFS of 7.4 months and serious adverse events including neutropenia (54%) and anaemia (17%). Meanwhile, Nie et al. 28 found that 41 patients with advanced pancreatic cancer who used SOXIRI had a median OS of 11.0 months, a median PFS of 4.3 months and serious adverse events including neutropenia (29.27%) and anaemia (12.20%). The median OS and median PFS in the SOXIRI group reported in our study were 12.1 months and 6.5 months, respectively, and the grade 3 or 4 adverse events were neutropenia (27.6%) and anaemia (12.6%). No treatment-related deaths were observed in any of the studies. Our study again confirmed the efficacy and safety of SOXIRI.
Among all grade adverse events, the incidence of anaemia was higher in the SOXIRI group than in the mFOLFIRINOX (41.4% versus 24%, p = 0.03). Meanwhile, although grade 3–4 anaemia was nonsignificant, it was still higher in the SOXIRI group than in the mFOLFIRINOX group (4.6% versus 0%, p = 0.12). Several previous clinical studies have shown that the SOXIRI regimen had a higher probability of patients developing anaemia. A meta-analysis also showed that in advanced gastric cancer, the rate of patients developing grade 3–4 anaemia was higher in the S-1 regimen than in the 5-FU regimen (odds ratio = 1.20, p < 0.001). 33 In Asian populations, anaemia was a unique side effect of S-1, which might be associated with unique anaemia genes, such as HIST1H2BL, C10orf127 and XPXPEP2. 36 In addition, the increase in toxicity was also considered to be related to the difference in oxaliplatin doses between the two regimens, 85 mg/m2 for SOXIRI and 65 mg/m2 for mFOLFIRINOX. Although the dose-limiting toxicity of oxaliplatin was neurotoxicity, 37 the increased dose of oxaliplatin still caused haematologic toxicity, 38 which could also explain the higher anaemia in the SOXIRI regimen. Of note, our research has demonstrated that the common hepatic toxicities observed in both chemotherapy regimens include elevated levels of bilirubin and transaminases. Elevated levels of transaminases may be associated with oxaliplatin-induced sinusoidal obstruction syndrome, while an increase in bilirubin levels was generally unrelated. 39 Furthermore, in the case of colorectal cancer treated using the chemotherapy containing oxaliplatin, there is no observed increase in incidence of elevated bilirubin levels. 40 Therefore, we hypothesized that the rise in bilirubin levels was more likely associated with biliary obstruction caused by pancreatic head primary cancers or metastatic lymph nodes. Although biliary drainage at baseline was not a common occurrence (Table 1), we did not count new biliary drainage cases after chemotherapy. Unfortunately, the present data could not answer the question of whether biliary obstruction could be used to explain the bilirubin elevation instead of chemotoxicity.
Although there was no difference in OS and PFS between the two chemotherapy regimens, we still wanted to determine the population most likely to benefit from each chemotherapy regimen. Subgroup analysis of OS found that the SOXIRI regimen was more effective than mFOLFIRINOX for the underweight (BMI < 18.5 kg/m2) cohort and the slightly elevated baseline TBIL (1–1.5 × ULN or 1–5 × ULN after biliary drainage) cohort. In the subgroup analysis of PFS, the SOXIRI regimen was also superior to mFOLFIRINOX in patients with slightly elevated baseline TBIL. However, two points of concern emerged from the results of these subgroups. First, the population in these subgroups may have potential SOXIRI benefit factors and we did not adjust for some factors within the subgroups that may affect prognosis, which might affect the final results. Moreover, it is worth noting that the calculated HRs for these subgroups were too high and their 95% CIs spanned too wide, which might be related to the small sample size of patients included in these subgroups. In conclusion, the current subgroup results were not very conclusive but indicated a research direction, and a prospective study with a larger sample size is needed to confirm the results.
To further explore the potential predictors of these two regimens, we started with the dynamic changes in weight and CA19-9 level that were common in clinical practice. In our study, we found a positive correlation between weight loss and disease progression in all groups with SOXIRI or mFOLFIRINOX, but weight loss was not a good predictor for efficacy and prognosis when two specific chemotherapy schemes were studied separately. A retrospective study by Wong et al. 41 found that patients with weight loss had a shorter OS than those without weight loss (4.8 months versus 7.1 months, p < 0.01), whereas Hue et al. 42 found that weight loss did not predict poor recurrence-free survival or OS in patients receiving neoadjuvant therapy. Therefore, the prediction of efficacy by weight needs to be explored more comprehensively and carefully, including refining the range of weight changes and excluding the influence of initial weight on poor prognosis. In addition, the CA19-9 decline could well reflect the efficacy and prognosis of these two chemotherapy schemes. In all groups with SOXIRI or mFOLFIRINOX, and either regimen, it was found that CA19-9 decline was negatively correlated with disease progression, and patients with CA19-9 decline generally had better OS and PFS than those without. A retrospective analysis of the PRODIGE4 study 43 showed that patients with CA19-9 decline saw improvements in OS, PFS and ORR when treated with mFOLFIRINOX. The MPACT study 44 also found that in patients with albumin–paclitaxel plus gemcitabine, the CA19-9 decline showed an improvement in OS compared with no decline (11.1 months versus 8.0 months, p = 0.005), with ORRs of 40% and 13%, respectively. Although CA19-9 has been used as a mature diagnostic and predictive biomarker for pancreatic cancer, we think it is also promising to forecast chemotherapy sensitivity in the future and screen out more advantageous benefit groups.
Although the efficacy and safety of SOXIRI and mFOLFIRINOX were comparable in our analysis, the study had several limitations. First, this was a retrospective study conducted at a single institution. Patients received SOXIRI or mFOLFIRINOX according to their physician’s preference, which corresponds to a non-randomized, non-blinded setting with a risk of selection bias. Second, the sample size was not large enough, so a phase III multicentre randomized controlled clinical trial would be necessary to confirm our conclusions. Finally, the conclusion was based only on the Chinese population. With regard to the applicability of this protocol in other countries, some pharmacogenomic differences regarding S-1 metabolism should be noted between ethnic groups.45,46
Conclusions
SOXIRI might be an effective alternative regimen to mFOLFIRINOX as a first-line treatment option for locally advanced or metastatic pancreatic cancer. The safety of this regimen was easily manageable and consistent with previous studies. Patients in the subgroups of slightly elevated TBIL or low BMI at baseline treated with SOXIRI had longer OS than those treated with mFOLFIRINOX in the subgroups of. In addition, CA19-9 decline was a good predictor for the efficacy and prognosis of patients in both the SOXIRI and mFOLFIRINOX groups. Further large-scale prospective trials are valuable to compare the effects of these two regimens.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231186029 – Supplemental material for Efficacy and safety of SOXIRI versus mFOLFIRINOX in advanced pancreatic cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359231186029 for Efficacy and safety of SOXIRI versus mFOLFIRINOX in advanced pancreatic cancer by Xujia Li, Jinsheng Huang, Fenghua Wang, Qi Jiang, Lingli Huang, Shengping Li and Guifang Guo in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
