Abstract
Objective:
To investigate the efficacy and safety differences between the cisplatin + paclitaxel (TP) and cisplatin + fluorouracil (PF) regimens in combination with or without immune checkpoint inhibitors (ICIs) in advanced esophageal squamous cell carcinoma (ESCC) first-line treatment and prognostic factors.
Methods:
We selected the medical records of patients with late stage ESCC admitted to the hospital between 2019 and 2021. Based on the first-line treatment regimen, control groups were divided into chemotherapy plus ICIs (n = 243) and non-ICIs (n = 171), 119 (49%) in the TP + ICIs group, 124 (51%) in the PF + ICIs group, 83 (48.5%) in the TP group, and 88 (51.5%) in the PF group in the control group. We analyzed and compared factors related to efficacy, safety, or response to toxicity and prognosis across four subgroups.
Results:
The overall objective response rate (ORR) and disease control rate (DCR) of the TP plus ICIs group were 42.1% (50/119) and 97.5% (116/119), respectively, which were 6.6% and 7.2% higher than those of the PF plus ICIs group. Patients in the TP combined with ICIs group had higher overall survival (OS) and progression-free survival (PFS) than those in the PF combined with ICIs group [hazard ratio (HR) = 1.702, 95% confidence interval (CI): 0.767–1.499, p = 0.0167 and HR = 1.158, 95% CI: 0.828–1.619, p = 0.0055] ORR and DCR were 15.7% (13/83) and 85.5% (71/83) in the TP chemotherapy alone group, significantly higher than the PF group [13.6% (12/88) and 72.2% (64/88)] (p < 0.05), OS and PFS were also better in patients treated with TP regimen chemotherapy than PF (HR = 1.173, 95% CI: 0.748–1.839, p = 0.0014 and HR = 0.1.245, 95% CI: 0.711–2.183, p = 0.0061). Furthermore, following the combination of TP and PF diets with ICIs, the OS of the patients was higher than that of the group treated with chemotherapy alone (HR = 0.526, 95% CI: 0.348–0.796, p = 0.0023 and HR = 0.781, 95% CI: 0.0.491–1.244, p < 0.001). Regression analysis showed that the neutrophil-to-lymphocyte ratio (NLR), the control nuclear status score (CONUT), and the systematic immune inflammation index (SII) were independent prognostic factors for the efficacy of immunotherapy (p < 0.05). The overall incidence of treatment-associated adverse events (TRAEs) was 79.4% (193/243) and 60.8% (104/171) in the experimental and control groups, respectively, and there was no statistically significant difference in TRAEs between TP + ICIs (80.6%) and PF + ICIs (78.2%) (61.4%) and PF groups (60.2%) (p > 0.05). Overall, 21.0% (51/243) of patients in the experimental group experienced immune-related adverse events (irAEs), and all of these adverse effects were tolerated or remitted following drug treatment without affecting follow-up.
Conclusion:
The TP regimen was associated with better PFS and OS with or without ICIs. Furthermore, high CONUT scores, high NLR ratios, and high SII were found to be associated with poor prognosis in combination immunotherapy.
Esophageal carcinoma (EC) is the sixth leading cause of cancer-related deaths worldwide, categorized into esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC). 1 In contrast to the high incidence of EAC in Europe and the United States, ESCC is the dominant histological subtype in Asia, accounting for about 90% of all EC cases.2,3 The mainstay of ESCC treatment continues to be radical surgical treatment supplemented by radiotherapy and chemotherapy. Although this multidisciplinary combination model provides better survival benefit for early stage patients, long-term survival benefit is not yet evident as most patients present with local progression or distant metastatic disease at the time of detection.1,4 Standard first-line regimens for patients with advanced ESCC are predominantly platinum based, when used in combination with chemotherapy with fluorouracil (PF) or paclitaxel (TP), although their clinical benefit is relatively limited, and regimes of paclitaxel plus cisplatin (TP) are common in China, whereas fluorouracil-plus cisplatin (PF) regimens are more popular in other nations.4–7
Targeted molecular drugs have shown promising benefit in a broad range of solid tumors, but their use in ECs is still quite limited, only in EACs with mutations in human epithelial growth factor receptor 2 or vascular endothelial growth factor, and there is no evidence-based targeted therapy that has significant benefit for ESCC.4,8,9 Fortunately, in recent years, immune checkpoint inhibitors (ICIs) based on inhibitors of programmed cell death-1 or programmed cell death-ligand 1 (PD-L1) have progressed in a broad range of solid tumors, including oesophageal cancer, melanoma, colorectal cancer, and some have been approved for first-line therapy. 10 In the CHECKMATE-648 and CHECKMATE-649 studies, nivolumab in combination with chemotherapy was used for advanced first-line ESCC treatment and resulted in longer progression-free survival (PFS) after nivolumab plus chemotherapy compared to the chemotherapy alone group (p = 0.002). 11 The phase III KEYNOTE-590 study (NCT03189719) also found that pembrolizumab in combination with chemotherapy prolonged patients’ overall survival (OS) and PFS by 17.6 months compared with 11 months in the group treated with chemotherapy alone, which is a statistically significant difference.12–14 The guidelines of the Chinese Society of Clinical Oncology state that the main recommendations for advanced ESCC first-line therapy are TP and PF. In addition, patients with advanced ESCC have been shown to have improved disease control rate (DCR) and survival after TP therapy compared with PF.15,16 However, there is a lack of studies and evidence on the association of TP and PF regimen selection with patient efficacy in combination with ICIs for advanced ESCC disease. Accordingly, we performed this retrospective study to explore whether and to what extent the selection of different chemotherapy regimens (TP and PF) had an impact on the survival benefit of patients when combined with ICIs for first-line treatment of advanced ESCC, to compare efficacy and safety differences between TP and PF regimens in combination with ICIs, and to explore the relevant influencers.
Data and methodology
The inclusion and exclusion criteria
Patients with advanced ESCC who were admitted to the Harbin Medical University Cancer hospital between the years 2019 and 2021 were included in the study. Inclusion criteria were as follows: (1) the pathological diagnosis was ESCC, (2) the presence of local progression or distant metastasis, (3) availability of target lesions for assessment of efficacy, and (4) performance status (PS) score ⩽ 2. Exclusion criteria were as follows: (1) pathological diagnosis of EAC, (2) lack of complete case records, (3) combination with other active tumors, and (4) the absence of measurable target lesions.
Therapeutic regime
Upon review of case data from patients included in this study, patients were divided into a combination therapy group and a chemotherapy-alone group based on their treatment regimen during their hospital admission. Experimental group: (1) TP + ICIs, (2) PF + ICIs; control group: (1) TP, (2) PF. Among the ICIs, pembrolizumab, camrelizumab and toripalimab have been used.
Methodology for research
Clinical data
Patient’s name, sex, age, height, weight, smoking and alcohol history, primary site, degree of histologic differentiation, whether or not to add ICIs, different types of ICIs, PS score, history of radical surgery, radiotherapy, distant metastasis and location, hematologic findings (serum albumin, total cholesterol, peripheral blood lymphocyte count, neutrophils, platelet count, leukocyte count, hemoglobin count, and tumor markers), imaging examination and report of gene detection, assessment of tumor efficacy, timing of disease progression, causes of disease progression, adverse events, diagnosis and treatment after hospital discharge, timing and cause of death, and so forth. A waiver of informed consent was granted due to the retrospective retrieval of patient data.
Response evaluation criteria
The effectiveness of the two groups was assessed using the Response Evaluation Criteria In Solid Tumors (RECIST1.1), which may be further divided into complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). If the combined treatment group has a negative progression after evaluation, then evaluated again according to the modified RECIST1.1 (iRECIST1.1). We classified treatment-associated adverse events (TRAEs) based on toxicity criteria version 5.0. The DCR is the proportion of PD cases post-medication. The percentage of patients with CRs and PRs was objective response rates (ORRs).
Follow-up
Follow-up was performed by referral to hospitalizations, readmissions, and telephone contacts, with the last follow-up through June 2022. PFS is defined as the time from the first standard treatment to patient progression or death, and the time between initiation of standard therapy and death (from any cause) is the OS. A total of 383 patients (92.5%) had achieved PFS and 356 patients (86.0%) had achieved OS by the last follow-up date.
Statistical approach
Statistical analysis was carried out using SPSS v26.0 software and was graphed in GraphPad Prism 9.0 and the R software. Metric data were expressed as mean ± standard deviation and paired t tests were used for comparisons between groups. Count data were described by case number (percentage), and comparison between groups was performed using chi-squares test. Survival curves were plotted using the Kaplan–Meier method and survival was compared using log-rank test. Cox hazard regression models were used for the analysis of impact factors, and the hazard ratio (HR) and 95% confidence intervals (CIs) were calculated. A p value of <0.05 was considered to indicate statistical significance (Figure 1).
A retrospective study of the efficacy and safety of a combined immunotherapy arm for advanced squamous cell carcinoma of the esophagus compared to chemotherapy alone.
Results
Analyzed the clinical data from the experimental and control groups
In all, 414 patients with ESCC advanced first-line therapy were included in this analysis. Patients receiving ICIs plus chemotherapy formed the treatment group (n = 243), divided into TP + ICIs and PF + ICIs, 119 and 124, respectively. The control group consisted of those who did not receive additional ICIs (n = 171), of which 83 belonged to the TP group and 88 to the PF group. PS scores ranged from 0 to 1 for all patients included in the study, and both the intervention and control groups were predominantly male in nature. There were 235 male patients (96.7%), only 8 female patients (3.3%), 162 male patients (94.7%), and only 9 female patients (5.3%) in the control group.
The experimental group consisted of 94 patients (38.7%) with organ metastasis, the most common local metastasis was lymph node metastasis in the neck (36.6%), and the most common organ metastasis was metastasis to the lungs (16.5%). In all, 73 cases (42.7%) in the control group had organ metastases. The most common sites of metastasis were the lymph nodes of the neck (28.7%) and liver (20.1%). The results showed that patients with advanced ESCC, PFS, and OS were not statistically different in terms of age, sex, BMI, history of alcohol consumption, location of the primary tumor, site of metastasis, surgery, type of ICIs, squamous cell carcinoma antigen in either the experimental group (ICIs + TP/PF) or the control group (TP/PF) (p > 0.05). There was a correlation with neutrophil-to-lymphocyte ratio (NLR), efficacy evaluation, prior long-term smoking history, combined radiotherapy, control nuclear status score (CONUT) score, systematic immune inflammation index (SII) score, and so on (all p value < 0.05). (Table 1).
Baseline clinical characteristics and PSM analysis of combined immunotherapy group and chemotherapy group in patients with advanced esophageal cancer.

BMI, body mass index; CONUT, control nuclear status score; CR, complete response; MSI, microsatellite instability; MSI-H, microsatellite instability-high; MSI-L, microsatellite instability-low; MSS, microsatellite stability; NLR, neutrophil-to-lymphocyte ratio; PD, progressive disease; PLR, platelet-to-lymphocyte ratio; PR, partial response; PSM, propensity score matching; SCC-Ag, squamous cell carcinoma-antigen; SD, stable disease; SII, systemic immune inflammation index (multiplying the platelet count by the neutrophil count divided by the lymphocyte count).
Efficacy analysis
Four patients (1.6%) achieved a CR in the intervention group, of which three were in the TP + ICIs group (2.5%) and one was in the PF + ICIs group (0.8%). In all, 90 patients (37.0%) with PR were contacted, 47 patients (39.5%) in the TP + ICIs group and 43 patients (30.1%) in the PF + ICIs group. The total number of SD patients was 134 (56.0%), 66 patients (55.5%) in the TP + ICIs group, and 68 patients (54.8%) in the PF + ICIs group. A total of 13 patients (5.4%) had PD, with 3 patients (2.5%) in the TP + ICIs group and 12 patients (9.7%) in the PF + ICIs group. The mean ORR and DCR values for the experimental group were 36.7% (94/243) and 94.6% (230/243), respectively. The ORR and DCR of the patients in the TP + ICIs arm were 42.1% and 97.5%, respectively; and in the PF + ICIs arm were 35.5% and 90.3%, respectively.
Only one individual (1.2%) in the control group achieved a CR, which was in the TP group. A total of 24 patients (14.0%) received PR, including 12 patients in the TP group (14.5%) and 12 in the PF group (13.6%). A total of 110 patients (64.3%) reached SD, including 58 in the TP group (69.9%) and 52 in the PF group (59.1%). A total of 36 patients (21.1%) reached PD, including 12 in the TP group (14.5%) and 24 in the PF group (27.3%). The ORR and DCR of the patients in the control group was 14.6% (25/171)and 78.9% (135/171), respectively, The percentage of ORRs and DCRs was 15.7% (13/83) and 85.5% (71/83) in the TP group and 13.6% (12/88) and 72.7% (64/88) in the PF group, respectively.
Both ORR and DCR were higher in the experimental group than in the control group (36.7% (94/243) versus 14.6% (25/171) and 94.6% (230/243) versus 78.9% (135/171), respectively, at the same time. The ORR and DCR values for the TP + ICIs group were 6.6% and 7.2% greater than those for the PF + ICIs group (35.5% and 90.3%, respectively), and the difference between the two groups was found to be statistically significant. (p < 0.05). In the control group, ORR and DCR were 5.7% (13/83) and 85.5% (71/83), respectively, compared to 13.6% (12/88) and 72.7% (64/88) in the PF group (p < 0.05) (Figure 2).

The ORR and the DCR by the different treatment regimes. (a) ORR by different treatment regimens and (b) DCR by different treatment regimens.
The median (mPFS) and median (mOS) of the patients in the experimental group were 12.42 and 16.65 months, respectively, which were 2.69 and 3.31 months longer than those in the control group (9.73 and 13.34 months), the difference was statistically significant (p < 0.001) (Figure 3). There was a significant prolongation of PFS and OS in the experimental group after ICIs were added to chemotherapy. Patients in the TP + ICIs group had a 4.06-month longer mOS than those treated with the TP regimen alone, respectively (HR = 0.526, 95% CI: 0.348–0.796, p = 0.0023), and patients in the PF + ICIs group also had a 2.81-month longer mOS than those treated with the PF regimen alone (HR = 0.781, 95% CI: 0.0.491–1.244, p < 0.001) (p < 0.05) (Figure 4). PFS and OS were also longer in the TP + ICIs group compared with the PF + ICIs group, based on different chemotherapy regimens combined by ICIs, mPFS, and mOS were 16.63 and 22.87 months in the TP + ICIs group, 3.81 and 6.44 months longer than the PF + ICIs group (HR = 1.702, 95 CI%: 0.767–1.499, p = 0.0167 and HR = 1.158, 95% CI: 0.828–1.619, p = 0.0055) (Figure 5). With respect to the control group, the mPFS and mOS of the TP group patients were 11.37 and 14 months, respectively, who were 3.28 and 3.33 months older than those in the PF arm (HR = 1.173, 95 CI%: 0.748–0.839, p = 0.0014 and HR = 0.1.245, 95% CI: 0.711–2.183, p = 0.0061) (Figure 6).

mPFS and mOS by different treatment regimens. (a) mPFS by different treatment regimens and (b) mOS by different treatment regimens.

Curves of PFS and OS in patients with advanced ESCC regardless of whether they were treated with ICIs. (a) PFS curves of TP and TP + ICIs, (b) OS curves of TP and TP + ICIs, (c) PFS curves of PF and PF + ICIs, and (d) OS curves of PF and PF + ICIs.

PFS and OS curves of patients with advanced ESCC who were treated with ICIs in addition to different chemotherapy regimens. (a) PFS curves of TP + ICIs and PF + ICIs and (b) OS curves of TP + ICIs and PF + ICIs.
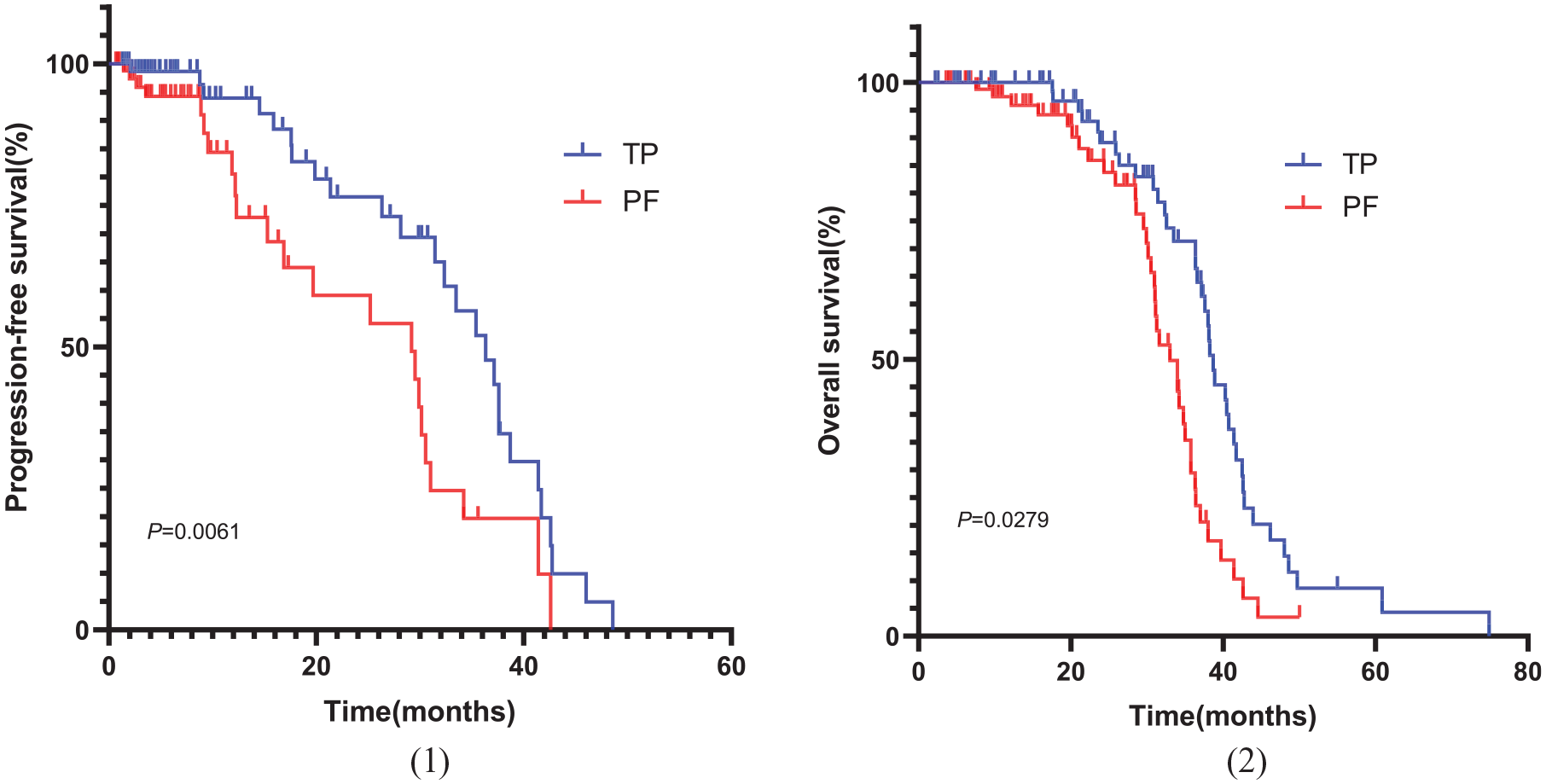
PFS and OS curves of patients with advanced ESCC who were treated with different chemotherapeutic regimens. (a) PFS plots of TP and PF and (b) TP and PF plots of OS.
Analyze the influencers
In conclusion, the results showed that the combination of ICIs had a significant impact on both PFS and OS in patients with advanced ESCC. The PFS was 12.42 months in the experimental group and 9.73 months in the control group, with a difference of 2.69 months; mOS 16.65 and 13.34 months, respectively, with a margin of 3.31 months (p < 0.001). In univariate analysis of patients in the treatment group, factors such as no prior long-term smoking history, combination TP Chemotherapy, improved efficacy assessment, high histologic differentiation, NLR < 3.45, combination radiation therapy, CONUT score < 2 (Table 2), SII < 830 were found to be related to better patient efficacy (all p < 0.05). Cox multivariate regression analysis was performed on the above factors. Results showed that the degree of histologic differentiation, NLR, assessment of efficacy, chemotherapy regimen, combination radiotherapy, SII, CONUT score, history of long-term smoking, and other factors were independent factors affecting the prognosis of the patients in the experimental group (all p value < 0.05) (Table 3). TP was more effective in patients with no smoking history, assessment of CR or PR inefficacy, and chemotherapy regimen; in addition, low NLR ratio, low CONUT score, and low SII value were also associated with better prognosis of patients (Figure 7).
Assessment of nutritional status according to CONUT score.

For the CONUT score, the best-fitting cutoff value is 2, CONUT ⩽ 2 for the low CONUT score group and CONUT > 2 for the high CONUT score group, respectively.
CONUT, control nuclear status score.
Univariate and multivariate regression analyses of survival time of patients with advanced esophageal cancer in combined immunotherapy group.

CEA, carcinoembryonic antigen; CONUT, control nuclear status score; CR, complete response; HR, hazard ratio; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; PR, partial response; SCC-Ag, squamous cell carcinoma-antigen; SII, systemic immune inflammation index; TP, cisplatin + paclitaxel.

PFS and OS curves in esophageal cancer patients treated with ICIs. (a) PFS according to the CONUT score, (b) OS according to the CONUT score, (c) PFS according to the SII value, (d) OS according to the SII value, (e) PFS according to the NLR, and (f) OS according to the NLR.
The results of the receiver operating characteristic (ROC) curve versus time analysis of the patients’ survival (1-year OS) in the experimental group (ICIs + TP/PF) show that the area under the ROC curve (AUC) of the NLR was 0.6422, with 52.17% sensitivity and 72.19%. AUC specificity for CONUT score of 0.6129 was achieved, the sensitivity was 54.17% and the specificity was 64.12%. SII had an AUC value of 0.6971, a sensitivity of 56.3%, and a specificity of 76.61%. We also found that low CONUT scores, as well as low SII and NLR values in the treatment group, were associated with improved PFS and OS in the patients(p < 0.05). Thus, we propose that NLR values, SII, and CONUT scores may be predictive of the efficacy of combination immunotherapy in patients with advanced ESCC (Figure 8).

Time-dependent ROC curve for overall survival at 1 year. (a) ROC according to NLR, (b) ROC according to SII, and (c) ROC according to CONUT.
Adverse events associated with treatment
Overall, 79.4% (193/243) and 60.8% (104/171) TRAEs were observed in patients in the experimental group and those in the control group, respectively. The incidence of immune-related adverse events (irAEs) was 21.0% (51/243) and 3–4 levels of irAEs were 3.7% (9/243) in the experimental group after ICIs were administered in combination; among them, four cases (3.4%) of 3–4 level irAEs in TP + ICIs group were immune capillary hyperplasia (1 case), immune hypothyroidism (1 case), and immune fever (1 case) and five cases (4.0%) were in the PF + ICIs group, which included one case of immune capillary hyperplasia, one case of immune hyperthyroidism, one case of immune pneumonia, and two cases of immune fever. Despite the fact that the experimental group had irAEs that were not found in the control group following the addition of ICIs, this did not affect patients’ final PFS and OS.
In the experimental group, the incidence of TRAEs was 59.7% (145/243) and 19.7% (48/243) for grades 1–2 and 3–4, respectively. In the control group, 47.2% (81/171) and 13.6% (23/171) had 1–2 levels and 3–4 levels of TRAEs, respectively. Overall, 45.7% (111/243) and 52.1% (88/171) of patients in the experimental and control groups experienced hematologic adverse events, respectively, with an incidence of 3–4 levels of hematologic TRAEs of 4.1% (10/243) and 3.5% (6/171), respectively. The incidence of liver and kidney toxicity was 57.4% (140/243) versus 70.2% (120/171) in the two groups, respectively, including an incidence of 3–4 levels of 1.1% (1/83) and 4.5% (4/88).
There were 19.3% and 20.1% (p > 0.05) levels of TRAEs between 3 and 4 in patients with TP + ICIs and PF + ICIs in the experimental arm. The incidence of TRAEs in the control group was found to be 62.7% (52/83) in the TP group and 59.1% (52/88) in the PF group, respectively. The incidence of TRAEs was slightly higher in the TP group as a whole compared to the PF group, but the difference was not statistically significant(p > 0.05). Of the TRAEs in the experimental group, hematologic-related TRAEs (4.1%), hepatic and renal toxicity (4.5%), and gastrointestinal adverse events (5.3%) were the most common 3–4 levels. Hematology-related TRAEs were characterized by decreases in leukocytes, neutrophils, platelets, and hemoglobin, all of which returned to normal after drug treatment and did not appear to be affected by severe hematology-related TRAEs (Table 4).
Comparison of adverse reactions among four groups of patients.

ICIs, immune checkpoint inhibitors; irAEs, immune-related adverse events; PF, cisplatin + fluorouracil; TP, cisplatin + paclitaxel; TRAEs, treatment-related adverse events.
Discussion
In summary, this is a retrospective study of the relationship between the combination of different chemotherapy regimens and the prognosis of patients with advanced ESCC treated with first-line ICIs. Our study showed that compared to ICIs + TP regimen, patients on the ICIs + PF diet had worse PFS and OS, and this did not correlate with patient ICIS types in any obvious way. ORR and DCR in ICIs + TP patients were 6.6% and 7.2% higher, and mPFS and mOS were 3.81 and 6.44 months higher, respectively, than in ICIs + PF. Furthermore, combination ICIs were associated with improved PFS and OS in both the TP and PF diets, consistent with the results of the KEYNOTE-590 study, 13 the JUPITER-06 study, 17 and the ESCORT-1st study (NCT03691090). 13 OS and PFS were significantly longer in the KEYNOTE-590 study in the Pembrolizumab/placebo + TP group compared with the TP chemotherapy-alone group.4,5 However, there was no significant difference between the two groups, with a 2.5-month and 1.6-month difference for OS and PFS, respectively. 13 The JUPITER-06 trial, a phase III clinical study conducted in the country with toripalimab/placebo + TP, also showed significantly longer OS and PFS in the combination group compared to the chemotherapy alone group, at 17.0 versus 11.0 months and at 5.7 versus 5.5 months, and the efficacy and safety of the combination therapy group was confirmed. 17 The results of the ESCORT-1st study showed that the addition of camrelizumab to chemotherapy significantly increased both PFS and OS in patients with advanced ESCC when compared with chemotherapy alone, and mOS by 3.3 months in patients treated with immune combination therapy compared with chemotherapy alone. 18
We also found that in the control group, the TP regimen showed better efficacy than the PF regimen, with mPFS and mOS increased by 3.28 and 3.33 months in the TP group compared to the PF group. A retrospective study conducted by Hsieh JC et al. also showed a DCR of 71.1% after chemotherapy with TP regimen, significantly higher than 51.4% for PF (p = 0.016); therefore, for patients with advanced ESCC, the combination chemotherapy regimen of cisplatin plus paclitaxel may be more suitable. 19 In the univariate regression analysis, we excluded the impact of ICIs types on the effectiveness of patients in the experimental group. Therefore, the difference in PFS and OS of patients in the experimental group is related to the chemotherapy regimen combined with ICIs, ICIs plus TP can benefit patients more than ICIs plus PF regimen. Therefore, we believe that the TP regimen is related to the longer OS and PFS of patients.
In addition, regression analysis revealed that CONUT score, NLR value, and SII were related to patient prognosis. Higher CONUT scores, NLR, and SII values were associated with a worse prognosis. The CONUT score includes total serum cholesterol, serum albumin, and the number of peripheral blood lymphocytes, which reflect the body’s lipid metabolism, protein synthesis capacity, and immune function.20–22 Assessment of nutritional status is important for patients with advanced ESCC, but it is difficult to make a more subjective and comprehensive assessment of nutritional status. CONUT score is a better solution to this problem because it not only provides a simple, objective response to patients’ nutritional status, but also reflects their immune status and inflammatory response status to some degree.22,23 Kheirouri S et al. 24 found that a high preoperative CONUT score was associated with poor survival in patients with all cancer types and that CONUT scores were independent prognostic factors for OS and cancer-specific survival. Similarly, Toyokawa et al. 20 found CONUT score to be an independent predictor of pretreatment OS and relapse-free survival in resectable patients with thoracic ESCC, and showed improved prognostic ability compared to the platelet-to-lymphocyte ratio (PLR), NLR and Glasgow protocol score. However, there have been fewer reports related to CONUT scores in immunotherapies. A recent report on the use of CONUT scores to predict the efficacy of pembrolizumab treatment in non-small-cell lung cancer, and that CONUT scores were independent predictors of OS (p < 0.05).22,25 There are currently no reports of CONUT scores predicting the effectiveness of ICIs in patients with late-stage ESCC. This retrospective study showed that CONUT score could be used to predict effectiveness in patients with advanced ESCC treated with ICI, that a lower CONUT score was associated with a longer OS and PFS and that CONUT score was an independent factor influencing OS and PFS in the patients (p = 0.024). The time-dependent ROC curve for CONUT showed an AUC of 0.6129 with a sensitivity of 54.17% and a specificity of 64.12%.
It has been shown that inhibition of apoptosis, DNA damage, and tumor progression are associated with the systemic inflammatory response, with NLR, lymphocyte-to-monocyte ratio, and PLR being regarded as the most readily available biomarkers of inflammation. 26 Yang et al., 27 Yodying et al., 28 and Zhao et al. 29 explored the relationship between NLR and EC by summarizing studies involving nearly 10,000 patients with esophageal cancer, they found that high NLR was associated with poor survival in ESCC. 30 Similar results were found in our study, with the high NLR group (NLR ⩾ 3.45) demonstrating worse OS and PFS in comparison to the low NLR group (NLR ⩾ 3.45), while NLR was also found to be an independent prognostic factor for OS in patients (p = 0.021). The time-dependent ROC curve for NLR showed an AUC of 0.6422, a sensitivity of 52.17%, and a specificity of 72.19%. In addition, some studies suggest that NLR is the balance between body immunity and tumor inflammation. High NLR reflects the success of high host neutrophil-dependent inflammation, and also signifies the failure of the lymphocyte-mediated antitumor immune response. An imbalance exists between the antibody response of the body and antitumor inflammation, leading to increased tumor biologic activity and disease progression.31,32
The SII is an indicator of inflammation in the body on the basis of platelet, neutrophil, and lymphocyte counts. 33 Some studies have demonstrated that SII can be used as a biomarker to predict survival and quality of life in patients with ESCC and that a higher SII is associated with worse OS and disease-free survival (p < 0.001).34–36 Geng Yiting et al. 35 assessed the prognostic value of SII in 916 ESCC patients and found that SII was an independent risk factor for OS (p = 0.042) and that they considered SII superior to PLR and NLR in evaluating the prognostic value of ESCC patients. These results are consistent with the finding in our retrospective study. Our results also indicate that SII value is related to the prognosis of patients. Patients in the low-SII arm (SII < 830) have longer survival and improved efficacy compared with patients in the high-SII arm (SII⩾830) (p < 0.05). Furthermore, SII is also a prognostic factor independent of both PFS and OS. The time-dependent ROC curve shows an AUC of 0.6971 for SII, a sensitivity of 56.3%, and a specificity of 76.61% (p = 0.033).
Research by Abnet CC et al. 37 revealed that N-nitrosamines (tobacco-specific nitrosamines) and polycyclic aromatic hydrocarbon (PAH) contained in tobacco are the major carcinogenic agents of ECSS, and the risk of oesophageal cancer in smokers is 3–9 times higher than in nonsmokers.38–40 A total of 414 subjects participated in our retrospective study. In our study, long-term smoking history was correlated with poor patient prognosis. Smokers had a reduction in mPFS and mOS of 4.5 and 7.1 months, respectively, compared with nonsmokers (χ2 = 9.243, p = 0.002), and multivariable regression analysis demonstrated that smoking status was an independently influential factor in the survival and prognosis of patients with late-stage ESCC. In addition, Sheikh M et al. 41 found that PAH exposure was associated with risk of ESCC and a poor prognosis.
Radiation therapy is also one of the successful methods of treatment for ESCC. Radiation therapy can significantly inhibit tumor growth and local metastatic lymph node growth, alleviate dysphagia, and rapidly improve patients’ symptoms. 42 In this study, we found that patients in both the experimental and control groups had better outcomes after combined radiotherapy, with a 3.7-month difference in PFS between combined and non-combination radiotherapy (χ2 = 27.693, p < 0.05).
ICIs are immunotherapies that target immune cell surface checkpoints and enhance immunity through the use of antibodies to ‘tumor escape’, ultimately leading to an antitumor response by turning off the immune system’s braking mechanisms.43–47 An increasing number of studies have demonstrated the efficacy and safety of ICIs in patients with ESCC. In a phase III clinical trial of advanced ESCC second-line therapy (RATIONALE-302, NCT03430843), the use of tislelizumab was associated with improved OS and better safety compared to chemotherapy alone. 48 Furthermore, in a phase II clinical trial designed to investigate the efficacy of nivolumab in patients with advanced ESCC who are intolerant or refractory to the drugs fluorouracil, paclitaxel, and cisplatin, it was found that the OS of patients treated with nivolumab was significantly longer than that of untreated patients, which was 10.8 months, indicating that nivolumab is likely to become a treatment option for patients with advanced stage ESCC who are refractory or intolerant to chemotherapy in the future.22,49,50 The increasing stature of immunotherapies has led to the identification and validation of a wide range of therapeutic biomarkers related to the prediction of tumor efficacy, including microsatellite viability and mismatch response, PD-L1 CPS scores, MHC-II molecules, CD73, CD8, and neoantigen.22,51,52 In the present study, however, the association of these biomarkers with ICI treatment has yet to be fully demonstrated and requires further investigation and exploration in the future.
To our knowledge, this retrospective study is the first report of the impact of TP and PF regimen choice on the efficacy of combination ICIs therapy in patients with advanced ESCC. Fortunately, we have a significant result, with the TP option benefitting more than the PF option. However, the study still has some shortcomings; first, there is not enough genetic testing data in the case data to analyze the relationship between genetic test results and effectiveness in patients; second, this was a single-center retrospective study which still lacked sufficient numbers of cases; third, the study did not perform cancer-related genome or exome sequencing to explore possible associations between the efficacy of immunotherapy and the genomes of patients with advanced ESCC to more accurately guide the clinical application of immunotherapy and aid in the effective screening of the target population; fourth, while the study found that factors such as inflammatory and nutritional markers were associated with patient outcomes, since this is a retrospective study, factors influencing inflammation and nutritional status cannot be ruled out as biasing the results, so prospective studies are needed to overcome this limitation.
Overall, in the clinical treatment of advanced ESCC, patients benefit more from a TP combination regimen than a PF regimen based on a combination ICI. Nutritional and inflammatory markers such as CONUT score, SII, and NLR can also be used as biomarkers to predict and evaluate the effectiveness of ICIs, to guide treatment of patients with advanced ESCC and selection of target ICI populations. It should be noted that the number of cases included in the study is still limited due to the short time frame in which ICIs have been used to treat advanced ESCC, but the results can inform practical clinical use and subsequent studies, with the expectation of further documented validation in a larger population in the future.
