Abstract
Adamantinoma-like Ewing sarcoma (ALES) of the salivary glands is an exceedingly rare malignancy defined by the t(11,22) EWSR1::FLI1 fusion, with complex epithelial differentiation. To identify features that can allow for better recognition of this disease entity, we reviewed all published reports of molecularly confirmed ALES of the salivary glands and explored epidemiological, clinical, radiological, pathological, and therapeutic characteristics of a population of 21 patients including a single newly reported patient from our group. We searched the English-language literature indexed in PubMed, Medline, Scopus, and Web of Science using the keyword ‘Adamantinoma-like Ewing sarcoma’ published up to June 2022. The median age at diagnosis was 46 years, and a slight female sex predilection was observed. Most tumors originated in the parotid gland (86%) and presented as a painless palpable mass with a median diameter of 3.6 cm. Metastatic dissemination was reported only in one patient (5%), and after a median follow-up of 13 months the 1-year overall survival rate was 92%. Salivary gland ALES were frequently misdiagnosed at presentation (62% of cases) and were pathologically characterized by the presence of highly monomorphic small round blue cells with infiltrative pattern and positive immunostaining for CD99 and high- and low-molecular weight cytokeratins. Epidemiological and clinical features of salivary gland ALES raise questions on the incorporation of this malignancy in the Ewing sarcoma family tumor group.
Keywords
Introduction
Ewing sarcomas comprise a group of neoplasms defined by FET::ETS gene rearrangements. 1 The FET family of transcription factors include EWSR1, FUS, and TAF15, while the ETS family comprises FLI1, ERG, ETV1, E1AF, and FEV. Intraosseous and extraosseous Ewing sarcomas often occur in children and young adults, primarily affecting long bones and pelvis. 2 Adamantinoma-like Ewing sarcoma (ALES) is a rare variant of Ewing sarcoma with complex epithelial differentiation, and is defined by the t(11,22) EWSR1::FLI1 fusion.1,3 Originally described in the long bones and thorax, ALES is now known to primarily affect the head and neck region, with a peculiar predilection for salivary glands, thyroid, and sinonasal tract.4–8
Owing to its rarity, little information is currently available on the clinical course and optimal treatment of ALES. However, outcomes similar to or slightly more favorable than in Ewing sarcoma after treatment with surgery, adjuvant chemotherapy, and radiotherapy have been reported. 5 Since a number of small blue round cell tumors including alveolar rhabdomyosarcoma, olfactory neuroblastoma, NUT midline carcinoma, lymphoma, melanoma, neuroendocrine carcinoma, and desmoplastic small round cell tumors can arise in the head and neck region, the differential diagnosis of ALES can be challenging. Nevertheless, appropriate recognition of this malignancy is key in guiding treatment choices and possibly improving patients’ outcomes.
Here, we report a case of ALES of the parotid gland originally misdiagnosed as poorly differentiated neuroendocrine carcinoma. Moreover, we systematically review the scientific literature concerning salivary gland ALES to provide updated information on the diagnosis and management of this exceedingly rare malignancy.
Case report
A 72-year-old woman presented at our institution with a 5-month history of painless, progressively enlarging right parotid mass. Her family history was unremarkable. She was a nonsmoker and did not consume alcohol. Her past medical history was significant for rheumatic valve disease status post-mechanical aortic and mitral valve substitution, atrial fibrillation, hysteroannexectomy for uterine fibromas, and stage III papillary thyroid cancer status post-radical thyroidectomy and radioiodine treatment. Her physical examination was only remarkable for 2 cm mass in the right preauricular region without skin abnormalities nor enlarged cervical lymph nodes. The results of laboratory tests were unremarkable. Contrast-enhanced computed tomography (CT) of the face and neck revealed an ill-defined mass measuring 19 × 22 × 23 mm in the right parotid gland infiltrating the homolateral masseter muscle (Figure 1(a)). The patient underwent a right parotidectomy with facial nerve dissection, with gross inspection showing a mass of 35 × 25 × 15 mm. Microscopically, the tumor consisted of monotonous small round cells grew in cords and nests containing monomorphic nuclei and infiltrating skeletal muscle structures (Figure 1(b)). Both extra-parotid extension to perisalivary soft tissue and angioinvasion and perineural invasion were detected. Surgical margins were negative. Immunohistochemistry (IHC) showed positivity of tumor cells for CD56 and, occasionally, for synaptophysin, and a diagnosis of poorly differentiated neuroendocrine carcinoma was made. A subsequent pathology revision revealed the positivity of tumor cells for CD99 (Figure 1(c)), CK8/18 and focally for p63 and p40, in the absence of staining for NUT, CK7, TTF1, S100, and androgen receptor. The Ki-67 index was 60%. A fluorescence in situ hybridization (FISH) using the ZytoLight SPEC EWSR1/FLI1 TriCheck Probe showed the presence of the EWSR1 (22q12)-FLI1 (11q12) gene fusion (Figure 1(d) and (e)), and a diagnosis of ALES was made. A post-operatory total-body CT scan demonstrated the absence of disease, while an echocardiography showed a compromised ventricular function, with an ejection fraction of 40%. The patient underwent six cycles of chemotherapy with vincristine, cyclophosphamide, and actinomycin D, reporting G3 thrombocytopenia as main treatment-emergent adverse event. She then received external beam radiotherapy, remaining free of disease at the last follow-up visit after 14 months from surgery.
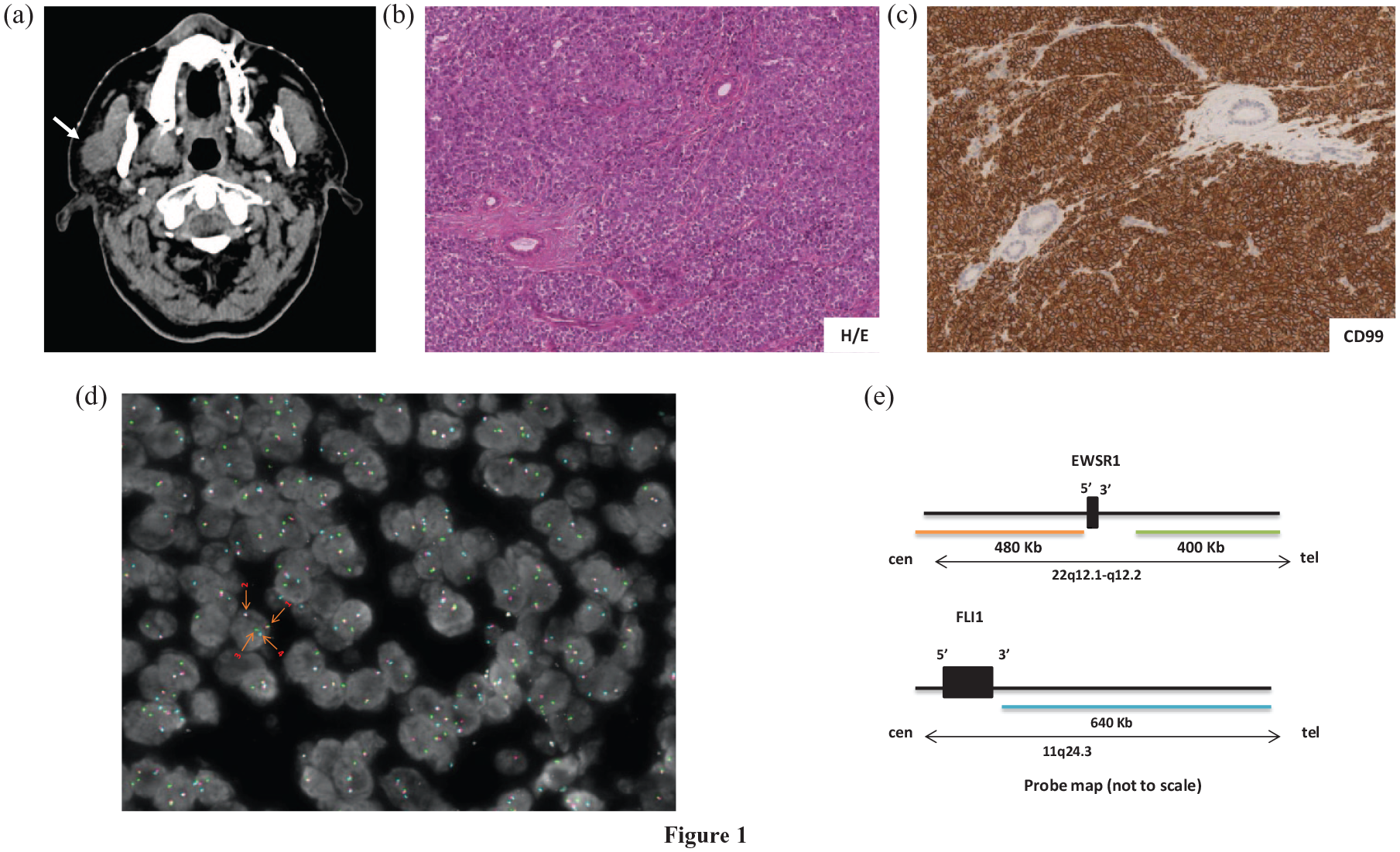
Radiological, pathological, and molecular features of parotid ALES. (a) CT scan showing an ill-defined mass in the right parotid gland. (b) Hematoxylin and eosin staining showing monotonous small round cells grew in cords and nests with monomorphic nuclei (magnification: ×20). (c) IHC demonstrating strong and homogeneous membranous CD99 positivity (magnification: ×20). (d) FISH showing rearrangements of EWSR1 and FLI1. Arrow 1: intact EWSR1 allele; arrow 2: 5′EWSR1 fused 3′FLI1; 3: derivative 3′EWSR1; 4: FLI1 non-translocated allele. (e) Chromosome 22q and 11q map showing the specific binding sequence areas for the 5′EWSR1 probe (spectrum orange), 3′EWSR1 probe (spectrum green) and 3′FLI1 (spectrum aqua).
Literature review and methods of analysis
We searched the English-language literature indexed in PubMed, Medline, Scopus, and Web of Science using the keyword ‘Adamantinoma-like Ewing sarcoma’ published up to June 2022. Only papers reporting individual cases of ALES of the salivary glands were included in this study. Two independent physicians (EL and AM) reviewed the selected papers and completed a detailed analysis of clinical, pathological, molecular, and radiological features as well as treatments and outcomes. Details included age at onset of ALES, sex, race, main symptoms and signs at disease presentation, results of morphological and functional imaging, received treatments, and follow-ups.
Statistical analysis was performed using MedCalc software 12.7 (MedCalc Software bvba, Ostend, Belgium). Overall survival (OS) was determined from diagnosis to death from any cause. Disease-free survival (DFS) was calculated from date of surgery (R0/R1) until evidence of macroscopic recurrence by imaging or death. Survival curves were estimated using the Kaplan–Meier method.
Results
Patient population: We retrieved 11 manuscripts describing patients with ALES arising in the salivary glands (Figure 2). A total of 21 patients matching the inclusion criteria were identified (Table 1). Median age at diagnosis was 46 years (range, 12–79), and the male to female ratio was 1:1.6. Most tumors originated in the parotid gland (18/21; 86%), while the remaining cases (3/21; 24%) arose in the submandibular gland. No cases originating in the sublingual glands nor in minor salivary glands were reported. A history of papillary thyroid carcinoma or ductal breast cancer was described in 3/21 (14%) and 2/21 (10%) cases.

Identification of studies for inclusion in the systematic review as per PRISMA guidelines. 9
Patient demographics and tumor characteristics.

Information available in 19 cases.
ALES, adamantinoma-like Ewing sarcoma.
Disease presentation and imaging features: All patients with ALES of the salivary glands presented with a palpable mass. Such a mass was painless in the majority of cases with available information (14/19; 74%), whereas pain was reported in five cases (26%). By imaging, the median size of the tumor at diagnosis was 3.6 cm (range, 1.5–8.8). The tumor was localized in most of the cases (20/21; 95%), while skeletal and mesenteric metastases were described throughout the clinical course of a single patient (5%). Imaging features were reported only in a minority of cases (4/21), and a contrast-avid, polylobed mass was usually described. Infiltration of the surrounding structures was described in 3/4 cases (75%) with available imaging information.
Pathologic and molecular features: At pathologic evaluation, salivary gland ALES were initially misdiagnosed in 13/21 cases (62%). In all cases, the evaluation was carried out using surgical specimens. A diagnosis of poorly differentiated carcinoma with basaloid features, neuroendocrine cancer, adenocarcinoma, and sialoblastoma was formulated in 5/21 (24%), 4/21 (19%), 1/21 (5%), and 1/21 (5%) cases, respectively. Once correctly diagnosed, salivary gland ALES were microscopically described as nests of highly monomorphic small round blue cells with fine chromatin and prominent nucleoli, often separated by fibrous stroma. An infiltrative pattern was frequently evident, and squamous pearl formation was also reported (3/21; 14%). As summarized in Table 2, IHC uniformly showed positive immunostaining for CD99 (21/21; 100%), frequent expression of p40 (13/13; 100%), CKAE1/AE3 (12/12; 100%), NKX2.2 (9/9; 100%), and synaptophysin (15/18, 83%; focal positivity in five cases). Chromogranin A (4/17, 24%; focal expression in three cases) and S100 (2/13, 15%; focal expression in one case) resulted only occasionally positive. When tested, actin, desmin, and NUT1 stained always negative. In all cases, the EWSR1 gene rearrangements were detected by FISH or next-generation sequencing.
An overview of key pathological characteristics of individual cases of salivary gland ALES.

ALES, adamantinoma-like Ewing sarcoma; F, focal.
Treatments and outcomes: Surgery was performed in all reported cases of salivary gland ALES. In particular, the removal of the tumor was carried out upfront in 20/21 cases and after neoadjuvant chemotherapy in 1/21 patients. Surgery was usually followed by adjuvant chemoradiotherapy (12/21 cases; 57%), with chemotherapy protocols spanning from alternating vincristine/doxorubicin/cyclophosphamide and ifosfamide/etoposide to doxorubicin monotherapy and carboplatin/etoposide combination. The original pathological diagnosis appeared to influence the choice of the chemotherapy regimen. After a median follow-up of 13 months (range, 1–96 months), absence of local or systemic disease was documented in 17/19 cases (89%) with available information. One treatment-related death was reported across the case reports or case series examined. Among 15 patients with available information, the 1-year OS rate was 92% ± 7 (Figure 3(a)). Among 14 patients who underwent R0/R1 resection and with available information, the 1-year DFS rate was 92% ± 8 (Figure 3(b)).

Kaplan–Meier estimate of OS.
Discussion
Salivary gland tumors constitute an uncommon, heterogeneous group of neoplasms that vary considerably in terms of site of origin, histology, and clinical behavior. 17 The parotid gland represents the most frequently involved site of origin, accounting for approximately 85% of the salivary gland neoplasms. Both benign and malignant salivary gland tumors exist and are classified according to the 2022 World Health Organization classification. 18 While approximately three-fourths of the parotid tumors are benign, the majority of the submandibular, sublingual and minor salivary gland neoplasms are malignant. 19 Histologically, the most common type of benign salivary gland neoplasm is pleomorphic adenoma, while the two most frequent malignant tumors arising in the salivary glands are the mucoepidermoid carcinoma and the adenoid cystic carcinoma, comprising one-half of the malignant spectrum of the salivary tract. In addition to primary tumors, up to 10% of major salivary gland masses are constituted by lymph node metastases from malignancies arising in the head and neck region (i.e. lymphoma, melanoma, cutaneous squamous cell carcinoma). 20 In this context, ALES appears as an exceedingly rare form of salivary gland tumor with complex histology, controversial clinical behavior, and debatable incorporation into the Ewing sarcoma group of neoplasms.
Owing to its rarity, little information is currently available on the clinical course of ALES. Table 3 summarizes the key features of ALES at disease presentation as captured by our systematic review of the literature. A slight female predilection (M:F 1:1.6) and peak incidence in the fifth decade can be recorded. This distribution is similar to that of salivary gland neoplasms in general, 21 while contrasting with that of Ewing sarcoma, characterized by slight male predominance and peak incidence between 10 and 15 years of age. 22 The parotid gland represents the site primarily involved by ALES (86%), consistent with the anatomic distribution of salivary gland neoplasms. 18
Key clinical-pathological features of ALES of the salivary glands.

Although the number of cases analyzed in this systematic review does not allow to draw reliable conclusions on factors increasing the risk of developing salivary gland ALES, a personal history of papillary thyroid cancer or breast cancer has been found in 14% and 10% of cases, respectively. Although this association might be coincidental in consideration of the prevalence of both malignancies, administered therapies (i.e. radiotherapy, known to be associated with salivary gland tumors23,24) might contribute to the development of ALES.
Given the absence of pathognomonic characteristics, a wide range of pathologic processes should be considered in the differential diagnosis of a painless salivary gland mass. Such processes should comprise benign and malignant tumors, salivary cysts, cysts of the first branchial cleft, salivary gland stones, Sjogren syndrome, sarcoidosis (Heerfordt syndrome), chronic sclerosing sialadenitis, and lymphoepithelial cysts. A tissue diagnosis is almost always required to distinguish among these pathologic conditions.
Based on our systematic review of the literature, after a median follow-up of 13 months a single patient (5%) with salivary gland ALES developed stage IV disease, with skeletal and mesenteric metastases. Although most of the patients analyzed in this review underwent postoperative chemotherapy, regimens known to be effective in Ewing sarcoma were not necessarily employed. Conversely, locoregional infiltration appears common in salivary gland ALES, with invasion of the surrounding structures detected in the 75% of cases with available imaging. Preoperative neoplastic extent evaluation by CT or magnetic resonance imaging may be thus useful for surgical planning.
The diagnosis of salivary gland ALES is challenging and is based on both pathologic and molecular criteria. According to our systematic review of the literature, almost two-thirds of the cases were initially misdiagnosed, necessitating of re-evaluation in centers with a specific expertise in head and neck pathology. This implies that, as frequently occurs with rare diseases, the frequency of the malignancy is underestimated. Since poorly differentiated carcinomas with basaloid features and neuroendocrine carcinomas of the salivary glands are the two entities most frequently misdiagnosed with salivary gland ALES, revision of such cases in tertiary centers is advisable.
Pathologically, salivary gland ALES is characterized by a heterogeneous morphologic pattern with a mixture of relatively uniform small round blue cells with scant cytoplasm admixed with epithelioid component with peripheral palisading and rare focal squamous differentiation. IHC reveals diffuse and intense immunoreactivity for membranous CD99 and nuclear NKX2.2 combined with diffuse positivity for high and low molecular weight cytokeratins including CKAE1/AE3 and p40. Expression of synaptophysin (either diffuse or focal) is detected in more than 80% of cases, and can orient toward a neuroendocrine differentiation/transdifferentiation. In particular, such an expression appears considerably more frequent in salivary gland ALES rather than in ALES of other head and neck sites (20%). 6 Despite the expression of synaptophysin can be seen as an epiphenomenon of the presumed neuroectodermal origin of Ewing sarcoma, the biologic clues driving its overexpression in salivary gland ALES remain largely elusive. While being detected in all cases of salivary gland ALES, the EWSR1 rearrangements are not pathognomonic of the disease, as myoepithelial carcinomas, 25 hyalinizing clear cell carcinomas, 26 EWSR1-non-ETS sarcomas, 27 desmoplastic small round cell tumors, 28 and the so-called ‘carcinomas of the thyroid with Ewing family of tumors elements’ 29 may also harbor the same gene rearrangements. In this context, as already proposed by Rooper et al., 4 the presence of a EWSR1 rearrangement in a cytokeratin-positive salivary gland neoplasm with low tendency to metastatic dissemination raises questions about whether salivary gland ALES truly belongs to the Ewing sarcoma group or represents a carcinoma with unique morphologic and immunohistochemical features. High-throughput sequencing studies comparing the genomic landscape of salivary gland ALES and Ewing sarcomas might provide an answer to this question, at least partly. Overall, the presence of an identical genetic alteration in ontogenetically different malignancies would not be particularly surprising, as demonstrated for example by the occurrence of recurrent BRAF V600E mutations across multiple solid and hematologic malignancies.30,31
An exact understanding of the ontogenesis of salivary gland ALES is not just a matter of nomenclature. Based on the available evidence, it is indeed currently difficult to conclude whether postoperative chemotherapy or chemoradiotherapy be always required in such a malignancy. As a matter of caution, here we note that the single death recorded in this systematic review was secondary to the postoperative treatment, and not to the disease itself. On the same lines, it is presently impossible to recommend Ewing sarcoma-specific regimens versus therapies for malignant salivary tumors, despite historically the first have been consistently preferred and good outcomes have been recorded.
In conclusion, salivary gland ALES is an overlooked malignancy frequently misdiagnosed with poorly differentiated carcinomas with basaloid features or neuroendocrine carcinomas. Although the detection of EWSR1 rearrangements is crucial in confirming the pathological diagnosis, the presence of such gene fusions does not necessarily collocate this malignancy in the Ewing sarcoma group. Although limited, the evidence emerging from the present systematic review indicate that salivary gland ALES share several epidemiological and clinical characteristics with other salivary gland malignancies, thereby suggesting that there is room for debate about its present classification. Suppression of EWSR1 fusion protein 32 can show activity against the plethora of EWSR1-driven tumors.
Supplemental Material
sj-jpeg-1-tam-10.1177_17588359231165979 – Supplemental material for Adamantinoma-like Ewing sarcoma of the salivary glands: a case report and systematic literature review
Supplemental material, sj-jpeg-1-tam-10.1177_17588359231165979 for Adamantinoma-like Ewing sarcoma of the salivary glands: a case report and systematic literature review by Eleonora Lauricella, Anna Manicone, Federica Cavallo, Gian Paolo Dagrada, Giovanni Centonze, Rossella Bertulli, Pasquale Quattrone, Camillo Porta and Mauro Cives in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
