Abstract
Background:
Advanced triple-negative breast cancer (aTNBC) has a poor prognosis; thus, there is a need to identify novel biomarkers to guide future research and improve clinical outcomes.
Objectives:
We tested the prognostic ability of an emerging, complete blood count (CBC)-based inflammatory biomarker, the pan-immune-inflammation value (PIV), in patients with aTNBC treated with first-line, platinum-based chemotherapy.
Design:
This was a retrospective, monocentric, observational study.
Methods:
We included consecutive aTNBC patients treated with platinum-based, first-line chemotherapy at our Institution, and for whom baseline (C1) CBC data were available. We collected CBC data early on-treatment, when available. PIV was calculated as: (neutrophil count × platelet count × monocyte count)/lymphocyte count. Patients with hormone receptor-positive (HR+), human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer (aBC) were included in a control, non-TNBC cohort.
Results:
A total of 78 aTNBC patients were included. When evaluated as a continuous variable, PIV-C1 was associated with worse overall survival (OS; p < 0.001) and progression-free survival (PFS; p < 0.001). On the other hand, when PIV-C1 was assessed on the basis of its quantile distribution, patients with ‘high PIV-C1’ experienced worse OS [adjusted hazard ratio (HR): 4.46, 95% confidence interval (CI): 2.22–8.99; adjusted p < 0.001] and PFS (adjusted HR: 2.03, 95% CI: 1.08–3.80; adjusted p = 0.027) when compared to patients with ‘low PIV-C1’. Higher PIV-C1 was also associated with primary resistance to chemotherapy. Similarly, a higher PIV calculated from CBC at C2D1 (PIV-C2) was associated with worse survival outcomes. We also created a PIV-based score combining information about both PIV-C1 and PIV-C2 and allowing the stratification of patients at low, intermediate, and high risk of death. No association was observed between PIV-C1 and clinical outcomes of HR+/HER2− aBC patients.
Conclusion:
PIV has a promising prognostic discrimination ability in aTNBC patients treated with first-line, platinum-based chemotherapy. Both baseline and early on-treatment PIV are associated with clinical outcomes and may be exploited for creating PIV-based risk classifiers if further validated.
Keywords
Introduction
Triple-negative breast cancer (TNBC) is the most aggressive and lethal breast cancer (BC) subtype. 1 Chemotherapy still represents the backbone of the first-line treatment for patients with advanced TNBC (aTNBC). In patients with programmed death-ligand 1 (PD-L1)-negative aTNBC, single-agent or combination chemotherapy represents the standard-of-care, first-line therapy. 2 By contrast, in patients with PD-L1-positive aTNBC, which represent about 30–40% of all aTNBC cases, the combination of taxane- or carboplatin–gemcitabine chemotherapy with anti-programmed cell death protein 1/PD-L1 immune checkpoint inhibitors proved to be superior to chemotherapy alone, and it is now considered the first-line standard treatment for most of these patients.3–5
Several cytotoxic agents are effective against aTNBC (both PD-L1 positive and PD-L1 negative), with different activity and safety profiles. Carboplatin, alone or in combination with paclitaxel or gemcitabine, is one of the most effective cytotoxic agents in aTNBC patients;6,7 however, with the exception of germline pathogenic BRCA1/BRCA2 mutations, no other biomarkers associated with benefit (or lack of benefit) from carboplatin chemotherapy (as compared to other cytotoxic agents) have emerged so far.1,8 Despite the most recent therapeutic improvements in aTNBC therapy, including chemoimmunotherapy combinations in patients with PD-L1-positive aTNBC or new antibody–drug conjugates, aTNBC is still characterized by very poor clinical outcomes and long-term tumor control. 1 Thus, novel biomarkers are needed for the prognostic stratification of aTNBC patients and their treatment allocation within available first-line options, and to guide the investigation of novel escalation (or de-escalation) treatment strategies in this specific setting.
Solid translational data indicate that TNBC is the most immune-enriched BC subtype, with a prominent phenomenon of tumor-immune co-evolution, and the prognostic role of intratumor immune contexture (especially in terms of tumor-infiltrating lymphocytes) is largely established. 1 On the contrary, the impact of systemic immune contexture and inflammation on the prognosis of patients with aTNBC is less characterized. Several studies published in the last decade have suggested that various peripheral immune cell-based biomarkers, such as the neutrophil-to-lymphocyte ratio (NLR), the platelet-to-lymphocyte ratio (PLR), and systemic immune-inflammation index (SII), may have a prognostic significance in BC patients.9–11 The main limitation of these immune biomarkers relies on their inability to comprehensively capture the complexity of the immune contexture. Recently, the pan-immune-inflammation value (PIV) has emerged as an easily assessable and comprehensive immuno-inflammatory biomarker encompassing neutrophil, platelet, monocyte, and lymphocyte counts from complete blood count (CBC). 12 PIV was initially developed for the prognostic stratification of patients with advanced colorectal cancer, and further developed as a promising prognostic tool in a variety of solid tumors and in other inflammation- and immune-related diseases.12–19 PIV has also been preliminarily investigated as a biomarker in BC patients18,20; however, no data are available about the potential prognostic significance of PIV in aTNBC patients.
In this study, we conducted a monocentric, retrospective evaluation of the prognostic significance of PIV on the clinical outcomes of women with aTNBC treated with carboplatin-based doublet chemotherapy in the first-line setting.
Patients and methods
Study design and objectives
The primary objective of the study was to evaluate the association between baseline PIV (PIV-C1) and the overall survival (OS) of women with aTNBC treated with first-line platinum-based chemotherapy at our institution. Secondary study objectives were (a) to compare the predictive ability of PIV-C1 (with respect to OS) with the predictive ability of individual CBC parameters and other commonly used, CBC-based inflammatory indexes [i.e. lymphocyte count, neutrophil count, platelet count, monocyte count, NLR, PLR, monocyte-to-lymphocyte ratio (MLR), SII],9,11,12,21 as measured at baseline (i.e. before the initiation of first-line carboplatin-based chemotherapy); (b) to evaluate the association between PIV-C1 and radiological tumor response to the first-line chemotherapy, and to investigate whether PIV is associated with primary tumor resistance and/or overall response; (c) to evaluate the association between PIV-C1 and the clinicopathological patients’ characteristics; and (d) to evaluate the association between early on-treatment modulation of PIV in terms of PIV at C2D1 of chemotherapy (PIV-C2) and delta PIV (both absolute and relative to PIV-C1) with clinical outcomes. To investigate whether PIV has prognostic significance specifically in TNBC patients, we also evaluated the association between PIV-C1 with the clinicopathological characteristics and the clinical outcomes in patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2 (HER2−) advanced BC (aBC; i.e. non-TNBC) treated with chemotherapy in a similar setting (i.e. carboplatin-based chemotherapy as the first-line chemotherapy for advanced disease).
Patient population and enrolment criteria
We retrospectively included consecutive aTNBC patients treated with first-line platinum-based chemotherapy at the ‘Fondazione IRCCS Istituto Nazionale Tumori’ between April 2007 and April 2022, and with available data about baseline (C1D1) CBC (i.e. within 1 week before the initiation of first-line therapy). As a control, non-TNBC cohort, we selected patients with advanced HR+/HER2− aBC treated with carboplatin-based chemotherapy (administered as the first-line chemotherapy for advanced disease) at our institution between September 2008 and February 2020, and with available data about baseline (C1D1) CBC. We also collected CBC data, whenever available, as measured immediately before the administration of the second cycle of chemotherapy (i.e. at the Day 1 of the second treatment cycle, C2D1).
Main enrolment study criteria were as follows: (1) female sex; (2) cytologically or histologically confirmed diagnosis of TNBC (main study population), as defined as estrogen receptor (ER) expression by immunohistochemistry (IHC) in <1% of cells, progesterone receptor (PgR) expression in <1% of cells, and HER2-negative disease, as defined as HER2 IHC score of 0 or 1+, or 2+ with negative in situ hybridization assay; (3) cytologically or histologically confirmed diagnosis of luminal HR+/HER2− aBC (control, non-TNBC cohort), as defined by ER expression ⩾1%, and/or PgR expression at IHC in at least 1% of cells, and HER2-negative disease, as aforementioned; (4) advanced disease, as defined as the presence of unresectable, locally advanced or metastatic measurable disease according to Response Evaluation Criteria in Solid Tumours (RECIST) version 1.1 criteria; (5) having received first-line therapy for aTNBC (main study population) with carboplatin-based chemotherapy doublets (i.e. carboplatin AUC 2, d1,8, q21 plus gemcitabine 800 mg/m2 d1,8, q21; carboplatin AUC 5, d1, q21 plus gemcitabine 800 mg/m2 d1,8, q21; carboplatin AUC 2, d1,8, q21 plus paclitaxel 80 mg/m2 d1,8, q21); (6) having received first-line chemotherapy for the treatment of HR+/HER2− aBC (control, non-TNBC population) with carboplatin-based chemotherapy doublets as aforementioned; and (7) availability of CBC parameters at baseline (i.e. before initiation of first-line carboplatin-based chemotherapy).
Collection of blood parameters
Clinicopathological and biochemical characteristics were extrapolated from institutional electronic records. PIV was calculated for each patient with the formula: [neutrophil count (103/mm2) × platelet count (103/mm2) × monocyte count (103/mm2)]/lymphocyte count (103/mm2), as previously reported. 12 Other CBC-based biomarkers were calculated as reported in the literature: NLR: neutrophil count (103/mmc)/lymphocyte count (103/mmc); PLR: platelet count (103/mmc)/lymphocyte count (103/mmc); MLR: monocyte count (103/mm2)/lymphocyte count (103/mm2); SII: [neutrophil count (103/mmc) × platelet count (103/mmc)/lymphocyte count (103/mmc).9,11,12,21
Statistical analyses
The median and interquartile range (IQR) were reported for continuous variables. Categorical variables were summarized as percentages, and 95% confidence intervals (95% CIs) were provided, when necessary. To evaluate the association between PIV and clinicopathological characteristics or tumor response coded as categorical variables, the Mann–Whitney test was used. We used Fisher’s exact test or chi-square test to study the association between specific cohorts of patients and clinicopathological characteristics or tumor response coded as categorical variables. Wilcoxon signed-rank test was used for matched comparisons of the distribution of PIV-C1 and PIV-C2. Survival curves were extrapolated using the Kaplan–Meier method and compared using the log-rank test. Measures of median survival outcomes (in months) and relative 95% CIs were provided. Follow-up time was estimated using the reverse Kaplan–Meier method. The difference in clinical outcomes between patient groups was tested using Cox’s proportional hazards regression models. In particular, variables significantly associated with survival outcomes at univariable analysis were included in a multivariable Cox regression model to test their independent prognostic significance. Hazard ratios (HRs) and relative 95% CIs were provided. Progression-free survival (PFS) was defined as the time between chemotherapy initiation and clinical/radiological detection of disease progression or death from any cause, whichever occurred first. OS was defined as the time between chemotherapy initiation and patient death from any cause. Radiological tumor response was evaluated as RECIST version 1.1. 22 Primary tumor resistance was defined as the achievement of progressive disease (PD) as the best tumor response according to RECIST v1.1, whereas overall response was defined as the achievement of partial response (PR) or complete response (CR) as the best tumor response according to RECIST v1.1. To compare the discrimination ability of PIV versus other commonly used CBC-based inflammatory biomarkers, a random forest-based approach was used to calculate variable importance (VIMP) and minimal depth. 23 All statistical tests were two-tailed, and a p value of <0.05 was considered statistically significant. Statistical analyses were performed using R software (version 4.1.2) and R Studio (version 2022.07.2) with the following packages: survival, survminer, epitools, stats, easyalluvial, and randomForestSRC.
Results
Study population and clinicopathological characteristics
We screened 436 consecutive patients with aBC treated with carboplatin-based chemotherapy at our institution. Of these patients, 78 had aTNBC, received first-line carboplatin-based doublet chemotherapy and had available baseline CBC data. Supplemental Figure 1 shows the flow diagram of patients’ selection. Median baseline PIV value (PIV-C1) in the whole patient cohort was 228 (IQR: 150–523). As shown in Table 1, PIV-C1 was higher in patients with higher body mass index (BMI; p = 0.021), worse Eastern Cooperative Oncology Group performance status (ECOG PS; p = 0.020), more than one metastatic site (p = 0.002), visceral metastases (p = 0.026), or liver metastases (p = 0.029). We also found a trend, which did not reach statistical significance, between PIV-C1 and the presence of metastases in the lung (p = 0.089) or central nervous system (p = 0.075) (Table 1).
PIV distribution according to clinico-pathological characteristics in aTNBC.

aTNBC, advanced triple-negative breast cancer; BMI, body mass index; CBDCA-GEM, carboplatin-gemcitabine; CBDCA-PTX, carboplatin-paclitaxel; ECOG PS, Eastern Cooperative Oncology Group Performance Status; IQR, interquartile range; PIV, pan-immune-inflammation value.The p values are indicated in bold numbers when statistically significant.
Survival outcomes according to baseline PIV (PIV-C1)
After a median follow-up of 47.4 months (95% CI: 44.2–NA), a total of 63 death events were observed, and median OS was 19.5 months (95% CI: 15.9–27.7) (Supplemental Figure 2, panel A). When PIV-C1 was measured as a continuous variable, we found that higher PIV-C1 was associated with worse OS (HR for 200 units increase in baseline PIV: 1.38; 95% CI: 1.23–1.56; p < 0.001). To gain insight into the power of PIV-C1 in predicting OS with respect to other known CBC-based biomarkers, we used Random Forest models to calculate VIMP and minimal depth of several CBC-based biomarkers (i.e. lymphocyte count, neutrophil count, platelet count, monocyte count, NLR, PLR, MLR, SII, PIV). As shown in Supplemental Figure 3, baseline PIV showed the best prognostic performance, in that it was characterized by the highest VIMP and the lowest minimal depth among all considered CBC-based biomarkers.
Aiming to find a cutoff of PIV-C1 that could be clinically useful in terms of identifying patients at higher versus lower risk of death, we explored the impact of PIV-C1 quintiles on OS. While we found no OS differences among patients in the first four quintiles of PIV-C1 distribution, patients in the last quintile showed a very poor OS (Supplemental Figure 4, panel A). Based on this observation, we divided patients into two cohorts: patients in the first four quintiles of PIV-C1 distribution were classified as ‘low PIV-C1’ patients, whereas patients in the last quintile of PIV-C1 distribution were included in the ‘high PIV-C1’ cohort. Supplemental Table 1 shows the association between PIV-C1 cohort and clinicopathological characteristics. Patients in the ‘high PIV-C1’ cohort experienced a poorer OS (median OS: 7.9 months, 95% CI: 4.7–13.9) when compared to patients in the ‘low PIV-C1’ cohort (median OS: 27.2, 95% CI: 18.2–34.8; HR: 6.48, 95% CI: 3.32–12.64; p < 0.001) (Figure 1(a)). In the multivariable model including all the other variables passing the p value cutoff for association with OS at univariable analysis (i.e. ECOG PS, number of metastatic sites, presence of visceral, lung, and bone metastases), ‘high PIV-C1’ confirmed an independent association with worse OS (adjusted HR: 4.46, 95% CI: 2.22–8.99; adjusted p < 0.001) (Table 2).
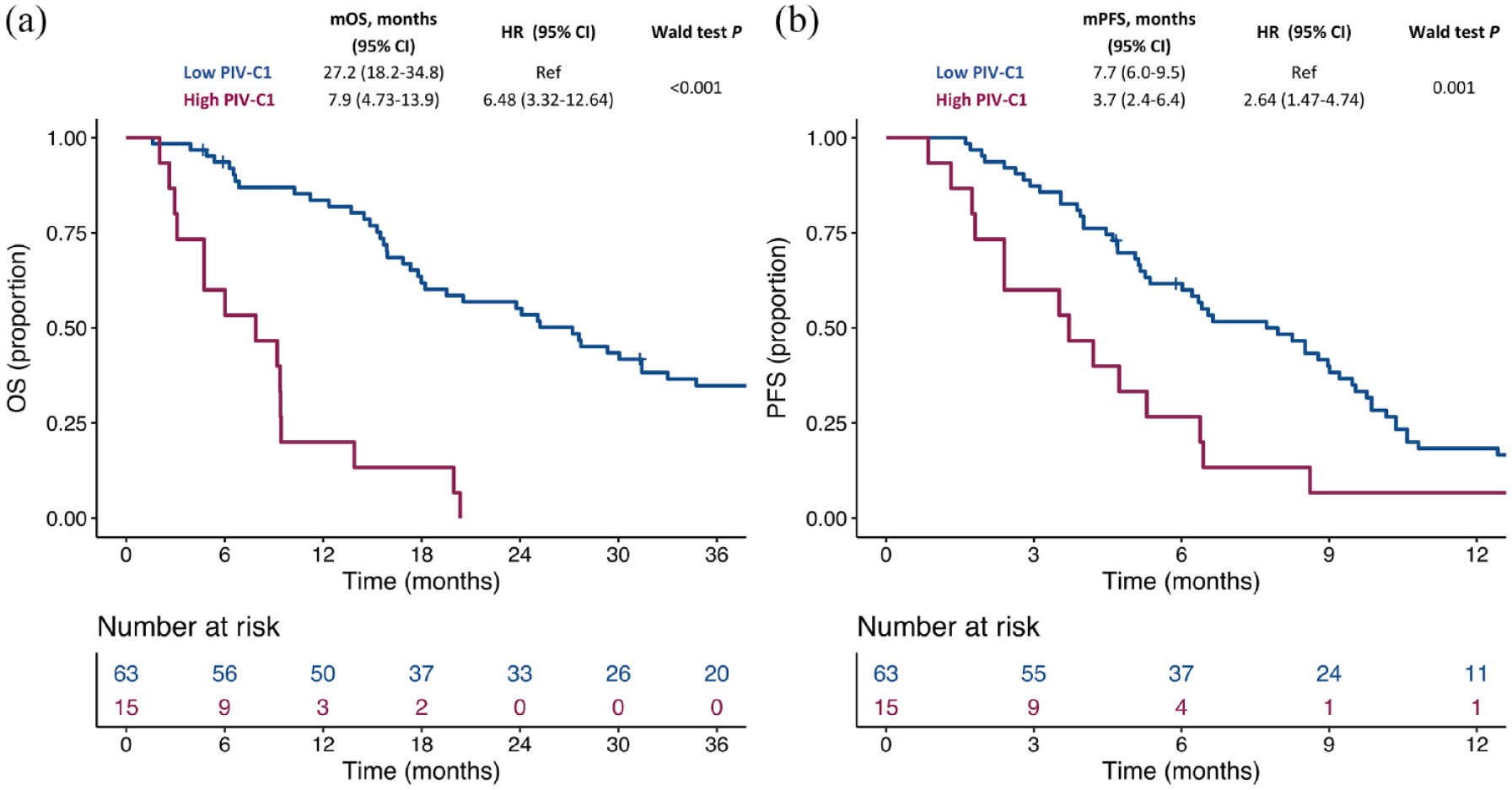
Kaplan–Meier curves for OS (a) and PFS (b) according to the PIV-C1 cohort in aTNBC. Blue lines indicate patients in the ‘low PIV-C1 cohort’, whereas purple lines indicate patients in the ‘high PIV-C1’ cohort.
Cox proportional hazards-regression models for OS and PFS in aTNBC.

aTNBC, advanced triple-negative breast cancer; BMI, body mass index; CBDCA-GEM, carboplatin-gemcitabine; CBDCA-PTX, carboplatin-paclitaxel; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; PFS, progression-free survival; PIV, pan-immune-inflammation value; OS, overall survival.
The p values are indicated in bold numbers when statistically significant.
Then, we explored the association between PIV-C1 and patient PFS. A total of 73 disease progression events were observed during the follow-up period, and median PFS in the entire population was 6.4 months (95% CI: 5.2–8.6) (Supplemental Figure 2, panel B). Consistent with OS data, PIV-C1, as considered as a continuous variable, was associated with PFS outcomes (HR for 200 units increase in PIV-C1: 1.24, 95% CI: 1.12–1.37; p < 0.001). When we analyzed the impact of PIV-C1 quintiles on PFS, we found no PFS differences between patients in the first four quintiles of baseline PIV-C1 distribution, whereas patients in the last quintile showed again a poor PFS (Supplemental Figure 4, panel B). Accordingly, patients in the ‘high PIV-C1’ cohort experienced a worse PFS (median PFS: 3.7 months, 95% CI: 2.4–6.4) than patients in the ‘low PIV-C1’ cohort (median PFS: 7.7 months, 95% CI: 6.0–9.5; HR: 2.64, 95% CI: 1.47–4.74; p = 0.001) (Figure 1(b)). In the multivariable model including all the other variables passing the p value cutoff for association with PFS at univariable analysis (i.e. ECOG PS and the number of metastatic sites), ‘high PIV-C1’ confirmed an independent association with worse PFS (adjusted HR: 2.03, 95% CI: 1.08–3.80; adjusted p = 0.027) (Table 2).
Radiological responses according to baseline PIV (PIV-C1)
We also explored the association between PIV-C1 and tumor response according to RECIST 1.1 criteria. In particular, we studied the association between PIV-C1 and primary tumor resistance (i.e. PD as the best response), as well as between PIV-C1 and overall response rate (ORR, including PR or CR as the best response). When considered as a continuous variable, PIV-C1 was higher in patients with primary tumor resistance (median PIV-C1: 459, 95% CI: 208–723 versus median PIV-C1: 215, 95% CI: 134–454 in patients not experiencing primary resistance; p = 0.016) (Figure 2(a)), whereas we found no PIV-C1 differences according to patient ORR (median PIV-C1 in non-responding patients: 337; 95% CI: 170–532 versus median PIV-C1 in patients with responding tumors: 206; 95% CI: 142–473; p = 0.156) (Figure 2(b)). Accordingly, patients in the ‘high PIV-C1’ cohort experienced a higher rate of primary tumor resistance (46.7%, 95% CI: 21.3–73.4) as compared to patients in the ‘low PIV-C1’ cohort (19.1%, 95% CI: 10.3–30.9; p = 0.025) (Figure 2(c)), whereas no significant difference in terms of ORR was observed in ‘high PIV-C1’ (40%, 95% CI: 16.3–67.7) versus ‘low PIV-C1’ patients (57.1%, 95% CI: 44.1–69.5; p = 0.231) (Figure 2(d)).

Association between PIV-C1 and radiological response in aTNBC. The box plots show the distribution of PIV-C1 according to the achievement of primary resistance (a) or overall response (b). The horizontal line in the box plots indicates the median of the distribution, the whiskers show the IQR, and the dots show the outliers. The bar charts show the primary resistance rate (c) and overall response rate (d) according to the PIV-C1 cohort.
Survival outcomes according to early on-treatment PIV (PIV-C2)
Then, we investigated the prognostic impact of PIV, as calculated from CBC parameters collected before the second cycle of chemotherapy (PIV-C2), as well as of precocious changes in PIV, as evaluated as the absolute or relative difference (delta) between PIV-C2 and PIV-C1. CBC data at C2D1 were available for 77 patients. In this patient sub-cohort, median PIV-C2 was 180 (IQR: 95–309), that is, lower when compared with PIV-C1 (Wilcoxon signed-rank test p = 0.002). Higher PIV-C2 values were associated with a worse OS (HR for 200 units increase in PIV-C2: 1.12; 95% CI: 1.06–1.19; p < 0.001) and PFS (HR for 200 units increase in PIV-C2: 1.09; 95% CI: 1.04–1.14; p < 0.001), whereas we did not observe any statistically significant association between the delta of PIV (C2-C1, both absolute and relative to PIV-C1), as measured as a continuous variable, and survival outcomes (Supplemental Table 2). Then, we explored the association between PIV-C2 quintiles and OS. While we observed no OS differences between patients in the first three quintiles of PIV-C2 distribution, patients in the last two quintiles showed a very poor OS (Supplemental Figure 5). Based on this observation, we divided patients into two cohorts: patients in the first three quintiles of PIV-C2 distribution were included in the ‘low PIV-C2’ cohort, whereas patients in the last two quintiles of PIV-C2 distribution were included in the ‘high PIV-C2’ cohort. The alluvial diagram in Supplemental Figure 6 shows the changes in ‘low’ and ‘high’ cohort compositions when considering PIV-C1 and PIV-C2. Patients in the ‘high PIV-C2’ cohort experienced a poorer OS (median OS: 14.9 months, 95% CI: 9.4–20.5) when compared to patients in the ‘low PIV-C2’ cohort (median OS: 31.4, 95% CI: 23.8–43.9; HR: 2.29, 95% CI: 1.36–3.85; p = 0.002) (Figure 3(a)). Similar results were observed in terms of PFS outcomes: indeed, patients in the ‘high PIV-C2’ cohort experienced a worse PFS (median PFS: 4.7 months, 95% CI: 3.7–7.7) when compared to patients in the ‘low PIV-C2’ cohort (median PFS: 9.0 months, 95% CI: 6.5–10.6; HR: 1.73, 95% CI: 1.07–2.80; p = 0.025) (Figure 3(b)). Then, we created a PIV score system ranging from 0 to 2, assigning 1 risk point for being in the ‘high PIV-C1’ and 1 risk point for being in the ‘high PIV-C2’ cohorts. Interestingly, we found that patients with a PIV score of 0 had the best prognosis both in terms of OS and PFS, and patients with a PIV score of 2 had the worst prognosis, whereas patients with a PIV score of 1 had an intermediate prognosis (Figure 4).

Kaplan–Meier curves for OS (a) and PFS (b) according to the PIV-C2 cohort in aTNBC. Blue lines indicate patients in the ‘low PIV-C2 cohort’, whereas purple lines indicate patients in the ‘high PIV-C2’ cohort.
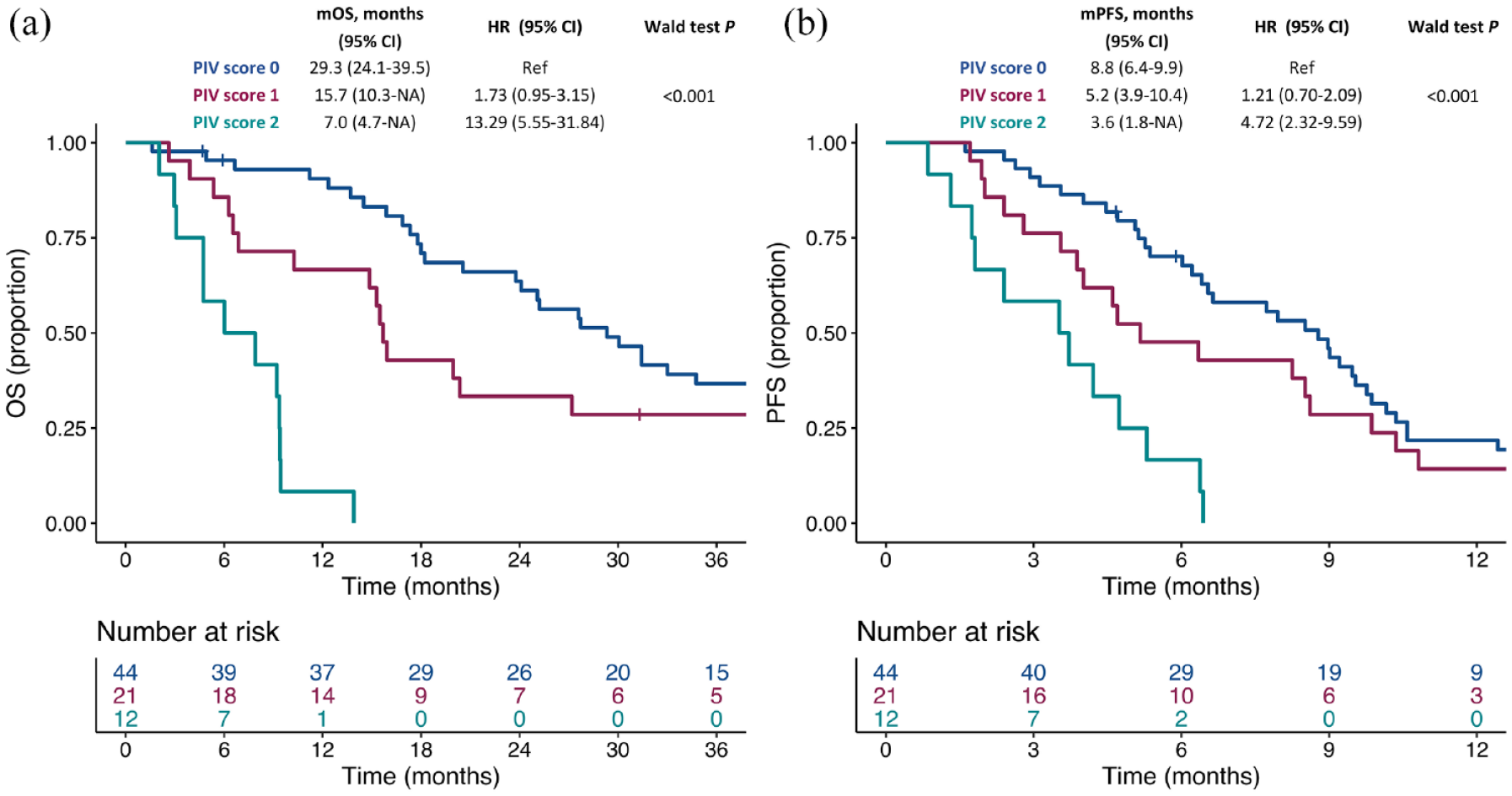
Kaplan–Meier curves for OS (a) and PFS (b) according to the PIV-based score in aTNBC. Blue lines indicate patients with score 0, purple lines indicate patients with score 1, whereas turquoise lines indicate patients with score 2.
Prognostic significance of PIV in the HR+/HER2− aBC cohort
To study whether PIV is specifically prognostic in aTNBC patients, we investigated the association between PIV values and clinical outcomes (OS, PFS) in a control cohort of patients with HR+/HER2− aBC treated with first-line carboplatin-based doublet as the first-line chemotherapy for advanced disease. Of 436 consecutive patients with aBC treated with carboplatin-based chemotherapy, 96 patients had HR+/HER2− aBC and had available baseline CBC data (Supplemental Figure 1). Median PIV-C1 in this non-TNBC cohort was 221 (IQR: 133–339), which was not different from median PIV values in aTNBC patients (p = 0.332). After a median follow-up of 72.3 months (95% CI: 59.8–NA), a total of 78 death events and 93 PFS events were observed, with median OS of 26.7 months (95% CI: 22.5–39.6) and median PFS of 9.7 months (95% CI: 7.4–12.4) (Supplemental Figure 7). PIV-C1 did not differ according to the clinicopathological characteristics, except for the observation of a higher PIV-C1 in patients with lymph nodal metastases (p < 0.001) (Supplemental Table 3).
Of note, PIV-C1 was not associated with survival outcomes, either when considered as a continuous variable (p for OS: 0.733; p for PFS: 0.643) or when the same cutoff values used for TNBC patients, and based on quintile distribution, were used (Supplemental Figure 8). Similarly, PIV-C1 was not associated with radiological responses in non-TNBC patients (Supplemental Figure 9). Taken together, these data indicate that the prognostic impact of PIV in patients with aBC treated with carboplatin-based chemotherapy are specifically observed in patients with aTNBC.
Discussion
In this study, we assessed the prognostic significance of PIV, an emerging and comprehensive, CBC-based inflammatory biomarker in a retrospective cohort of aTNBC patients treated with first-line, platinum-based chemotherapy. We observed that baseline PIV (PIV-C1) was higher in patients with high tumor burden (i.e. presence of visceral and liver metastases, more than one metastatic disease) and worse clinical conditions (in terms of ECOG PS), in line with the well-documented correlation between cancer-related systemic inflammation and tumor metastatic spreading/disease burden. 24 Interestingly, BMI in the range of obesity (i.e. BMI ⩾ 30) was associated with a higher PIV-C1. This observation is consistent with the fact that obesity drives a state of chronic, low-grade inflammation sustained by the secretion of a plethora of adipocyte-derived molecules (adipokines, cytokines, hormones).25,26 As previously reported in the first-line treatment setting in advanced colorectal, melanoma and HER2+ BC patients,12,17,18 we found a strong and independent impact of PIV-C1 on survival outcomes, with high PIV-C1 predicting worse PFS and OS outcomes. Of note, PIV showed the strongest predictive power, in terms of OS, among the most used and established CBC-based biomarkers (i.e. lymphocyte count, neutrophil count, platelet count, monocyte count, NLR, PLR, MLR, SII), as suggested by the lowest minimal depth and the highest VIMP when using a Random Forest approach. Together, our data point to PIV as the optimal candidate biomarker for the inflammatory-based prognostic stratification of aTNBC patients. The better discrimination ability of PIV when compared with other CBC-based inflammatory biomarkers is in line with data previously reported in patients with other solid tumors, and it may be explained by the ability of PIV to comprehensively capture and weigh immune cell types taking part in different steps of inflammatory and immune response. 19 Of note, in our study, we found that the prognostic impact of PIV is limited to aTNBC patients, while it was not observed in patients with HR+/HER2− aBC. These findings, which are consistent with previous data about inflammatory biomarkers in aBC from our group, 9 may be explained by the subtype-specific immune contexture of TNBC as compared to HER2 negative, luminal BC. 27
In aTNBC patients, high PIV-C1 was also predictive of primary resistance to first-line therapy, suggesting that systemic inflammation may also have a role in TNBC resistance to chemotherapy, as previously shown for other solid tumors. 24 In addition, we reported the clinical utility of estimating PIV also early on-treatment, as highlighted by the association of high PIV after one cycle of chemotherapy (PIV-C2) with poor survival outcomes. More interestingly, we observed that a PIV-based score including both PIV-C1 and PIV-C2 can dynamically stratify patients for their survival outcomes early on-treatment.
If validated in future studies, these results suggest the opportunity to design future clinical trials to dynamically tailor treatment-intensification strategies in this specific setting according to the PIV-based score. Although PIV demonstrated a prognostic relevance also when calculated early on-treatment (after one cycle of chemotherapy) and the combination of PIV-C1 and PIV-C2 in a score potentiate the stratification ability of PIV-based prognostication, we did not observe any association between the entity of early on-treatment PIV variation (considered as both absolute difference or relative to baseline) and survival outcomes. This observation is different from what was previously reported in patients with microsatellite instability-high metastatic colorectal cancer treated with immunotherapy, in which early on-treatment PIV increase was associated with worse clinical outcomes. 19 In addition to the tumor-specific inflammatory context, the different prognostic impact of PIV may be explained by the different types of treatment used in these studies. Indeed, chemotherapy may interfere per se with the cancer inflammatory network, 24 for instance by causing hematologic toxicities and affecting PIV values by modifying neutrophil, monocyte, and lymphocyte counts, 28 or through the administration of steroids, which have a remarkable modulatory effect on the dynamics of peripheral blood immune cells and CBC-based biomarkers. 29
We are aware of the limitations of our study. Given the monocentric and retrospective nature of the analysis, the study population may not represent the heterogeneity of aTNBC patients and may suffer from a selection bias. Thus, our results need further external validation. Similarly, the clinical utility and stratification ability of the cutoffs used to define the ‘high PIV’ cohorts (both PIV-C1 and PIV-C2) and based on the quintile distribution of the biomarkers in the study population must be externally validated, since it may be affected by overfitting. Lastly, most of the patients included in this study were treated before the advent of chemo-immunotherapy in aTNBC, and data about PD-L1 expression are not available for most of the patients. Consequently, we do not know whether PIV may have a differential prognostic impact according to the PD-L1 status of the disease, and our results need to be validated in aTNBC patients treated with first-line chemotherapy with a known and negative PD-L1 status (to actualize its prognostic relevance in patients treated with first-line chemotherapy). In parallel, the ability of PIV to predict the clinical outcomes of aTNBC with PD-L1-positive disease and treated with first-line chemo-immunotherapy remains to be elucidated.
Conclusion
Even if further validation is needed in larger patient cohorts, our study suggests that PIV has the potential to become an innovative inflammatory-based biomarker with clinical utility for the prognostic stratification of patients with aTNBC treated with first-line, platinum-based chemotherapy.
Supplemental Material
sj-docx-10-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-docx-10-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-11-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-docx-11-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-12-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-docx-12-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-13-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-docx-13-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-1-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-1-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-2-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-2-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-3-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-3-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-4-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-4-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-5-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-5-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-6-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-6-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-7-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-7-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-8-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-8-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-9-tam-10.1177_17588359231165978 – Supplemental material for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis
Supplemental material, sj-tif-9-tam-10.1177_17588359231165978 for The pan-immune-inflammation value is associated with clinical outcomes in patients with advanced TNBC treated with first-line, platinum-based chemotherapy: an institutional retrospective analysis by Leonardo Provenzano, Riccardo Lobefaro, Francesca Ligorio, Emma Zattarin, Luca Zambelli, Caterina Sposetti, Daniele Presti, Giulia Montelatici, Angela Ficchì, Antonia Martinetti, Alessio Arata, Marta Del Vecchio, Claudia Lauria Pantano, Barbara Formisano, Giulia Valeria Bianchi, Giuseppe Capri, Filippo de Braud, Claudio Vernieri and Giovanni Fucà in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank AIRC, the Associazione Italiana per la Ricerca sul Cancro (MFAG 22977: PI Claudio Vernieri), the Giuliani Foundation – Fondazione Gianmaria e Sabrina Giuliani, Celeste Ungaro and her family, and the Scientific Directorate of Fondazione IRCCS Istituto Nazionale dei Tumori for supporting our research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
