Abstract
Uterine sarcomas are very infrequent and heterogeneous entities. Due to its rarity, pathological diagnosis, surgical management, and systemic treatment are challenging. Treatment decision process in these tumors should be taken in a multidisciplinary tumor board. Available evidence is low and, in many cases, based on case series or clinical trials in which these tumors have been included with other soft tissue sarcoma. In these guidelines, we have tried to summarize the most relevant evidence in the diagnosis, staging, pathological disparities, surgical management, systemic treatment, and follow-up of uterine sarcomas.
Introduction
Uterine sarcomas are uncommon soft tissue malignancies with an annual incidence of 0.36–0.64 cases per 100.000 women in the United States. 1 Diagnosis and pathological classification of uterine sarcomas is a challenge. The most frequent subtypes are leiomyosarcoma (LMS), followed by endometrial stromal sarcoma (ESS) and undifferentiated uterine sarcoma (UUS). Histological grade deeply impacts on prognosis and clinical behavior. While high-grade ESSs (HG-ESS) are generally highly aggressive, low-grade ESSs (LG-ESSs) have an indolent behavior. LG-ESSs have a 5-year survival rate and a 5-year disease-specific survival of 80–100%. 2 Treatment decision-making in patients with these tumors is complex. The level of evidence is often low and mainly based on retrospective series or case reports. The aim of these guidelines is to summarize evidence available to date and guide decision-making concerning treatment for uterine sarcomas.
Diagnosis and staging of uterine sarcomas
Specific clinical and radiological criteria to differentiate uterine sarcomas from benign uterine leiomyomas are lacking. In both entities, abnormal vaginal bleeding, pelvic pain, and abdominal mass are the most common symptoms. A rapidly growing uterine fibroid in a peri-menopausal or postmenopausal woman should raise suspicion of sarcoma. 3
The first approach to diagnosis is usually a pelvic ultrasound followed by pelvic magnetic resonance imaging (MRI). 4 Algorithms may help to differentiate atypical leiomyomas from uterine sarcomas on MRI, 5 and 18-fluorine-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET-CT) can be useful to distinguish between the two entities. There are no specific serum tumor markers. An endometrial or transvaginal biopsy can be attempted to obtain histological diagnosis but its sensitivity is low. 6 Diagnosis is often reached after analysis of a surgical specimen. Transabdominal biopsy and morcellation are discouraged as they could carry a high risk of peritoneal dissemination [IVA]. 7
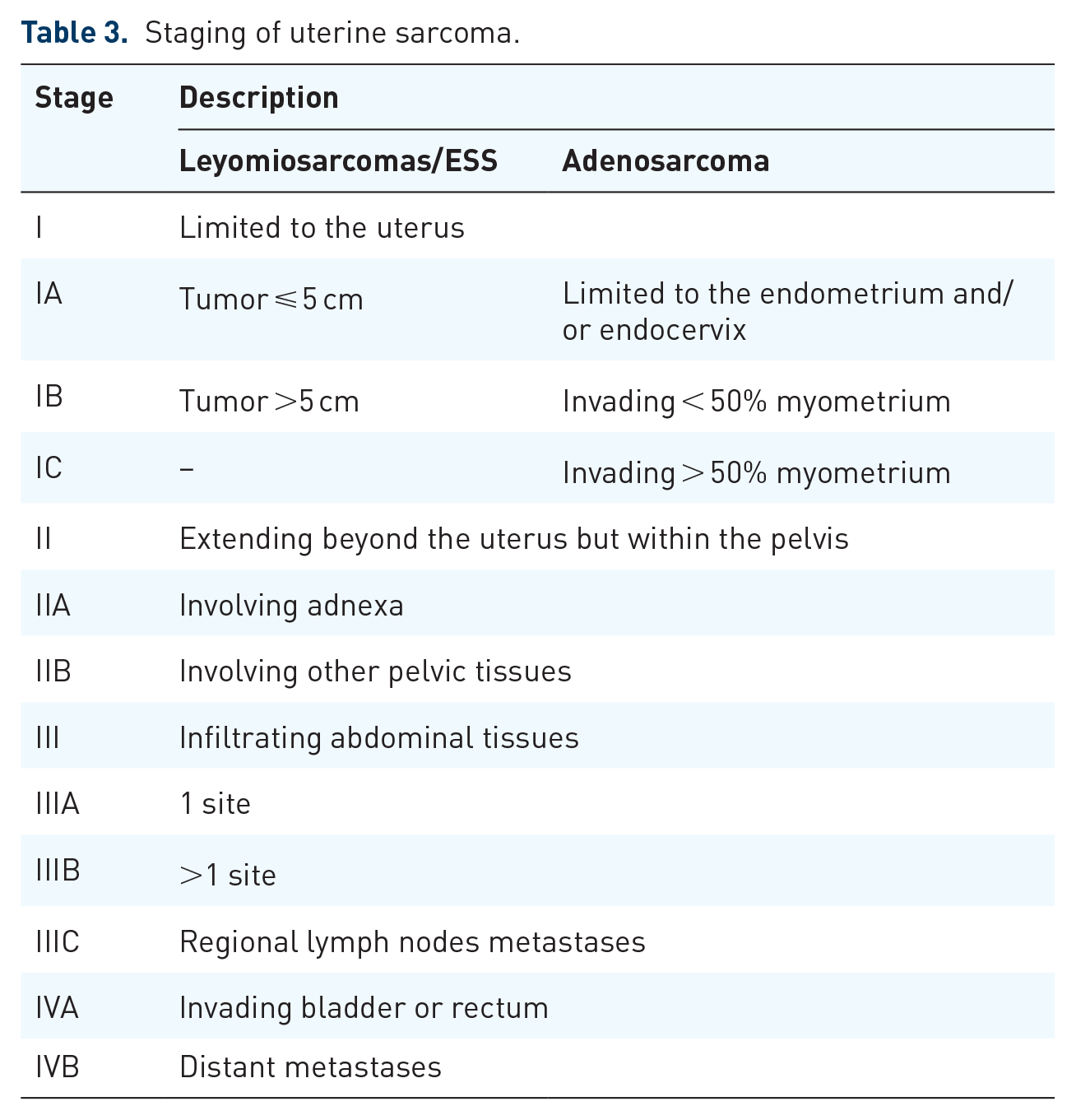
The most widely used staging systems are those of the Federation of Gynecology and Obstetrics (FIGO) and the American Joint Committee on Cancer 8 (Table 1). For distance staging, a CT scan should be performed [IA].
Levels of evidence.

Taken from Lee and Vielemeyer. 9
Pelvic ultrasound and MRI are usually the first approach to diagnosis [IIIA]. A 18F-FDG PET-CT can be useful to distinguish malignancy [VC]. Transabdominal biopsy and morcellation are discouraged due to the risk of dissemination [IVA]. A CT scan is the preferred procedure for staging [IA].
Pathology and molecular diagnosis
Uterine sarcomas a highly heterogeneous group of tumors both clinically and histologically. While some tumors can be classified as simple genomic sarcomas as they harbor specific gene fusions such as endometrial stroma tumors, the most frequent subtype, leiomyosarcomas, has a complex karyotype comprising numerous structural aberrations.
Leiomyosarcomas
Uterine leiomyosarcoma (uLMS) is a malignant mesenchymal tumor originating from myometrial smooth muscle. Three main subtypes have been described: spindle, epithelioid, and myxoid leiomyosarcoma.
Spindle cell (conventional) tumors are typically cellular and composed of spindle-shaped cells with eosinophilic cytoplasm arranged in long, interlacing, often compact, but relatively disorganized fascicles. Nuclear pleomorphism is often striking, but a subset of tumors exhibits uniform cytological features. Diagnosis of spindle leiomyosarcomas requires the presence of two of three features: tumor cell necrosis, marked cytological atypia, and ⩾4 mitoses/mm2 (equating to ⩾10 mitoses/10 HPF of 0.55 mm in diameter and 0.24 mm2 in area). Spindle cell uLMS express estrogen receptors (ERs) and progesterone receptors (PRs) in approximately 40–50% of cases. In addition, p16 and/or p53 overexpression is common.
Epitheloid leiomyosarcomas is a variant composed of more than 50% of cells with epithelioid appearance consisting of polygonal cells with eosinophilic or clear cytoplasm arranged in nested, corded, nodular, or diffuse patterns. Positivity for CD10, EMA (Epithelial membrane antigen), and cytokeratin is common, with EMA and cytokeratin positivity being especially frequent in this subtype. 10
Myxoid tumors are often paucicellular and contain abundant myxoid stroma. They may display vague fascicular or nodular growth. The cells express desmin, h-caldesmon, and smooth muscle actin, but this expression may be focal or weak if the tumor is poorly differentiated or myxoid. 11
High-grade uterine sarcomas
High-grade sarcomas of the endometrium encompass two distinct entities: high-grade ESS (HG-ESS) and UUS.
High-grade ESS
HG-ESS is a malignant endometrial stromal tumor with uniform, high-grade round and/or spindle morphology, sometimes with a low-grade component.
These tumors may show expansive, permeative, or infiltrative growth, and more than one pattern of invasion may be seen. They typically display lymphovascular invasion (LVSI), brisk mitotic activity, and necrosis. FISH HG-ESS can be classified into the following:
1) HG-ESS with YWHAE-NUTM2A/B fusion displays round cells with eosinophilic cytoplasm and high-grade nuclei with irregular nuclear contours, vesicular chromatin, and variably distinct nucleoli. They can also be associated with a fibromyxoid or conventional low-grade endometrial stromal component. 8 The high-grade component is positive for cyclin D1, BCOR, KIT, CD56, and CD99, and negative for DOG1. 12
2) HG-ESS with ZC3H7B-BCOR fusion is typically positive for cyclin D1, whereas only about 50% of cases express BCOR by IHC. This group typically shows diffuse CD10 positivity with variable ER and PR positivity. Also focal expression of SMA and caldesmon can be shown but usually desmin is negative. Tumors may show pan-TRK staining not related to NTRK rearrangement. 13 BCOR internal tandem duplication (ITD) tumors have a different immunoprofile to ZC3H7B-BCOR tumors. They show less positivity for CD10 and are diffusely positive for cyclin D1 and BCOR, but they are negative for ER and PR. Furthermore, they may express desmin but are negative for SMA and caldesmon. 14
3) HG-ESS NOS is a high-grade sarcoma associated with a low-grade endometrial stromal component. 15
Demonstration of gene fusion or BCOR ITD may be helpful for diagnosis. In this context. FISH could be recommended in any case with unusual morphology or any high-grade or recurrent disease.
Undifferentiated uterine sarcoma
UUS is a malignant mesenchymal tumor lacking evidence of specific lines of differentiation. Diagnosis is therefore based on exclusion. These sarcomas show no identifiable line of differentiation and are considered a heterogeneous group. They typically display a destructive pattern of myometrial invasion. In the past, they were classified as uniform or pleomorphic types. 15 In tumors with uniform nuclear features, YWHAE-NUTM2 (FAM22) HG-ESS should be excluded. These tumors may be associated with a low-grade stromal component and express cyclin D1. However, others with pleomorphic histology are also associated with a low-grade component and are designated HG-ESS NOS. UUS typically consists of sheets of uniform or pleomorphic epithelioid and/or spindle cells associated with brisk mitotic activity. Necrosis and LVSI are commonly seen. 16 Tumors are often positive for p53 and p16, and some may show positivity for ER and/or PR, and variable positivity for CD10. 17 P53 mutation is normally seen in fusion negative uterine sarcomas with the exception of a small subset of NTRK1/3-positive sarcomas. 18
YWHAE, JAZF1, and NTRK rearrangements may underpin rare pleomorphic sarcomas that should not be classified in this category. 19
Low-grade uterine sarcomas
LG-ESS is a malignant stromal tumor with cells resembling proliferative-phase endometrial stroma and displaying infiltrative (permeative) growth with or without LVSI.
Microscopically, they show irregular, densely cellular islands of tumor cells with a diffuse growth that permeate the myometrium (tongue-like) and may be associated with LVSI. Cells have uniform, oval-to-fusiform nuclei, with no or minimal atypia, scant cytoplasm, and a delicate arteriolar network. Sometimes, tumor cells whorl around the vessels. The mitotic activity is usually low but can be brisk. 20 Hyaline plaques, foamy histiocytes, cystic change, and necrosis can be seen. 21 Common variant features include smooth muscle differentiation 22 fibromyxoid/fibrous change,23,24 and sex cord–like differentiation. Bizarre nuclei, epithelioid/rhabdoid change, endometrioid glands, and pseudopapillae are uncommon.15,25,26
These tumors usually show diffuse, strong expression of CD10, ER and PR, and focal cyclin D1 positivity. Tumors may be positive for wide spectrum keratins. 27 Desmin and h-caldesmon typically highlight smooth muscle differentiation 28 and are often positive in sex cord–like elements, which also express inhibin, calretinin, melan-A, WT1, and CD99. 29
Demonstration of characteristic gene fusions may be helpful in selected cases. Two-thirds of LG-ESS tumors harbor genetic fusions involving polycomb family genes. 26 The most common of these is JAZF1-SUZ12, followed by JAZF1-PHF1, EPC1-PHF1, and MEAF6-PHF1.30,31 MBTD1-EZHIP (CXorf67), BRD8-PHF1, EPC2-PHF1, and EPC1-SUZ12 have also been documented, but experience is limited (fewer than three cases of each have been reported to date).
BRD8-PHF1 has also been implicated in high-grade tumors. 32
LG-ESS with JAZF1 and PHF1 rearrangements, although rare, can transform into high grade at the time of diagnosis or years later. An increased mitotic index, prominent nucleoli or nuclear enlargement can be seen in this transformation underlying a more aggressive behavior. 33 Table 2 summarizes the most relevant alterations and differences in the diagnosis of high grade and low grade uterine sarcomas.
Main pathological and molecular alterations with diagnostic implications in uterine sarcomas.

ITD, internal tándem duplication.
Surgical management of early stages
Surgery is the standard of care for uterine sarcomas regardless of grade. Complete resection of disease without fragmentation and with negative surgical margins is the gold standard for treatment. 34 Standard procedures include total hysterectomy and bilateral salpingo-oophorectomy. 35 In case of incidental diagnosis, a second surgery should be evaluated to complete surgical treatment.36,37 In case of a casual diagnosis after previous uterine morcellation, a second intervention may be required to complete the surgery, explore the abdominopelvic cavity, and collect peritoneal biopsies. 38
Ovarian-spearing surgery can be performed in leiomyosarcoma in early stage disease and premenopausal women without compromising survival. 39
Systematic pelvic and para-aortic lymphadenectomy is not recommended. Lymph node resection seems to be beneficial only when pre-operative imaging or intra-operative findings suggest lymph node metastasis. 42
In advanced disease, maximal cytoreduction, when feasible, could significantly improve survival outcomes if the resection is R0. Residual disease after surgery is an indicator of poor prognosis.40,41
In LG-ESSs, total hysterectomy and bilateral salpingo-oophorectomy should be the recommended treatment. 43 Ovary-sparing procedures should only be considered in young women, depending on tumor hormonal receptor status, and extensive counselling is advised. 44
Surgery is the standard of care in uterine sarcomas regardless of grade. Complete resection of disease without fragmentation and with negative surgical margins is the gold standard for treatment [IIA]. In premenopausal uLMS patients, ovary-sparing procedures can be performed [IIIA]. In LG-ESS ER+, salpingo-oophorectomy is the recommended treatment [IIIA].
Surgical management of uterine mass (unexpected sarcoma)
The risk of sarcoma is about 1/352 in patients undergoing laparoscopic hysterectomy, but a thorough preoperative examination can help decrease the number of undetected uterine sarcomas. Several reports suggest patients with an unexpected uterine sarcoma who undergo morcellation during surgery have worse prognosis because this procedure may spread disease through the abdominal cavity. 45 It is therefore recommended that (1) morcellation should be avoided if ultrasound shows oval shape, central necrosis, high blood flow, fast growth within 3 months or rapid postmenopausal growth are seen on ultrasound; (2) when planning a power morcellation, a preoperative endometrial biopsy with hysteroscopy is mandatory and a myoma ultrasound-guided Trucut biopsy should be considered; and (3) fibroids from myomectomy should be morcellated in endobag containers.
Adjuvant therapies
Uterine leiomyosarcomas
There is no clear evidence that the administration of adjuvant chemotherapy is associated with improved survival. Two phase III trials were prematurely closed due to low recruitment. In the first one, the SARCGYN phase III trial, the authors compared adjuvant polychemotherapy based on cisplatin, doxorubicin, and ifosfamide followed by radiation versus radiation alone. The study planned to enroll 256 patients but only 81 patients were randomized and 19 of them were patients with carcinosarcomas. A benefit of adjuvant chemotherapy (CT) group was seen on 3-year disease-free survival (DFS) (55% in CT-treated patients versus 41% in the control arm, p = 0.048) was reported but with no impact on overall survival (OS). 46
In the second one, a more recent study, patients with stage I high-grade leiomyosarcoma were randomized to receive four cycles of gemcitabine/docetaxel followed by four cycles of doxorubicin versus observation. The study planned to enroll 216 patients but was prematurely closed after only 38 patients were randomized. No differences were found in OS or recurrence-free survival (RFS). 47 No definitive results can be drawn from these trials. Nevertheless, several phase II studies have suggested that adjuvant chemotherapy provides a benefit in uLMS compared to observation in historical controls. Various chemotherapy schedules have been studied, such as anthracyclines in monotherapy or in combination with other drugs, four cycles of gemcitabine/docetaxel, 48 and four cycles of gemcitabine/docetaxel followed by four cycles of doxorubicin. 49
Several retrospective studies suggest a potential benefit of combining anthracyclines with ifosfamide in uLMS, but it cannot been ruled out that the benefit is mainly due to anthracyclines given the concerns about ifosfamide effectivity in LMS. 50 The combination of anthracyclines with dacarbazine has also shown to be effective in retrospective studies in metastatic disease. 51
Despite the lack of prospective randomized data, the poor prognosis of uLMS has led to a clear increase in the use of adjuvant chemotherapy in localized uLMS over the last decade. 52 Adjuvant chemotherapy options should be individually evaluated by a multidisciplinary tumor board and discussed with the patient. If considered appropriate, antracyclines should be the backbone chemotherapy.
High-grade ESS and UUS
Few prospective data are available regarding the role of adjuvant CT in HG-ESS and UUS. Observation should be the standard of care, but due to the poor prognosis adjuvant CT can be considered in selected cases. 53
Adjuvant CT in high-grade uterine sarcomas is not the standard of care [ID]. In FIGO stage I, adjuvant CT should not be recommended [IB]. Nevertheless, due to the poor prognosis of the disease, adjuvant CT (including antracyclines) could be considered in selected patients with FIGO II-III stage [IVC].
Low-grade uterine sarcoma
To date, adjuvant treatment with endocrine therapy in low-grade endometrial stroma sarcoma (LG-ESS) has only been evaluated in retrospective studies. Beck et al described a recurrence rate of 50% for all patients (38.5% for stage I) without adjuvant treatment. This decreased to 33% (14.3% for stage I) for those treated with adjuvant progestins. 54 Another cohort of LG-ESS found a reduction in the risk of relapse in patients receiving megestrol acetate (31% versus 67%). 55 More recently, Comert et al. described a global recurrence rate of 27%; while 0% in patients who had adjuvant therapy versus 38.5% in those who did not. 56 In addition, Leath et al. showed a trend to a better OS with adjuvant progestin, with a median of 94 months versus 72 months in those who did not receive this treatment. 41
Despite the lack of prospective data, due to the consistent reduction in the recurrence rate observed in retrospective studies, adjuvant hormonal treatment could be recommended in LG-ESS [IVB]. Given the lack of strong evidence, however, no recommendation regarding duration can yet be given. Observation is also an option, particularly for stage I tumors.
Adjuvant radiotherapy
High-grade uterine sarcoma
Adjuvant radiotherapy (RT) has not demonstrated a statistically significant increase in terms of survival versus observation in early-stage high-grade uterine sarcomas. Therefore, its use cannot be considered a standard of care.
In the only phase III randomized study (EORTC 55874) 57 published to date, the rate of pelvic recurrence after adjuvant RT was compared with observation in 224 surgically treated patients with stage I/II uterine sarcoma. Adjuvant pelvic irradiation with 50.4 Gy improved local control in a patient population with various sarcoma entities, but in the subgroup of leiomyosarcomas (n = 99), no effect was found nor in the rate of local recurrence (20% with versus 24% without RT) nor in the OS rate.
Pelvic local recurrence in early stage high-grade uterine sarcomas occurs in 14–34% at 5 years53,58,59 and favorable local control following adjuvant pelvic RT has been reported in retrospective series. 60 In these studies, RT was associated with a lower rate of local relapse, with no clear impact on survival.
A retrospective analysis of 3650 patients of whom the majority were carcinosarcoma, up to 529 patients had uterine leiomyosarcoma. In this cohort, those patients treated with adjuvant RT had a 5-year locoregional failure-free survival rates of 98% (N = 131) versus only 85% in those without adjuvant RT (N = 398) (p < 0.01). 61
In a recent study that used a propensity score approach to reduce comparison biases, adjuvant RT provided a benefit in terms of local control. However, no impact was seen on overall or cancer-specific mortality. 62
A recently published meta-analysis confirmed that adjuvant RT does not reduce the recurrence rate in early-stage uLMS patients. 63
For HG-ESS and UUS patients, retrospective data from the French Sarcoma Group showed that adjuvant RT appears to improve overall survival. Malouf et al. 40 observed that patients who received postoperative RT had better DFS than patients who did not, suggesting that RT could be considered depending on risk factors such as tumor size, involvement of margins, and/or number of positive lymph nodes removed.
Low-grade uterine sarcoma
As far as LG EES patients are concerned, only a few small retrospective studies are available to date, and data mining of large databases has identified a lower rate of local relapse after RT. 64 A retrospective comparative study on the use of pelvic radiation in patients with uterine sarcoma included 361 patients with EES (109 treated with adjuvant RT and 252 patients with no further treatment). In this cohort of EES, adjuvant RT increased 5-year local-regional failure-free survival rates from 93% to 97% (p < 0.05); however, data regarding tumor grade was not available. 61 Because of the evidence based on retrospective designs, unclear data and long-term side effects of adjuvant RT this treatment is generally not indicated.
Adjuvant RT in high-grade uterine sarcomas is not standard of care [ID]. Adjuvant RT can be considered in selected patients with risk factors such as cervical, parametrial, or serosal involvement, or incomplete resection, after discussion with the patient [IVC].
Local management of oligometastatic or locally recurrent disease
High-grade uterine sarcomas
Retrospective studies suggest that complete resection of metastases can increase survival in selected cases of uLMS.65,66 Five-year survival rates of 58% have been reported in isolated lung metastases and rates of 30–50% in isolated liver metastases. In a single-center study in MSKCC with 62 uLMS patients, most of whom had abdomino-pelvic or lung metastases, the most important prognostic factor for survival was complete resection, with no benefit for postoperative chemotherapy. 67
Peritoneal sarcomatosis from uterine sarcoma, specially leiomyosarcoma, could also benefit from complete resection if R0 resection can be achieved. 68
Prognostic factors identified in several studies include histology, number of metastases, a disease-free interval longer than 12–18 months, no extrapulmonary metastases, histological grade, size of metastases, and complete resection. 69
In HG-ESS and US, resection of metastases is not usually considered due to their aggressive clinical course. Stereotactic RT can be proposed in some cases in oligometastic disease, providing disease control with acceptable toxicity. This is of particular interest when surgery is not feasible. 70 Local recurrence should be treated surgically if R0 resection can be achieved, and postoperative RT could be recommended.
Surgical resection of metastatic (particularly lung) or local recurrent disease should be considered by a multidisciplinary tumor board [IVB].
Postoperative RT after complete resection of a local recurrence could be considered. [IVB].
Low-grade uterine sarcoma
Despite its indolent behavior, recurrence occurs in up to one-third of LG-ESS patients. 21 Median time to recurrence in early stages ranges from 5 to 9 years compared to <1 year for stages III or IV. 71 This highlights the need for a long follow-up. Recurrences are generally limited to the pelvis or abdomen but distant metastases are also possible. 72 Locoregional and distant relapses can be managed with local therapies. Locoregional relapses after fertility-sparing are common but can be safely managed through surgery, without affecting survival.44,73,74
Complete surgical removal of metastatic lesions increased survival in several retrospective series – some including other uterine sarcomas.75,76
RT or ablation could also be considered on an individual basis but data supporting these therapies is scarce.
Whenever possible, resection of local or distant recurrences of LG-ESS should be recommended [IVB].
Systemic treatment for advanced or recurrent disease: Chemotherapy
High-grade uterine sarcoma
First-line treatment
Evidence for the use of CT in uterine sarcomas is scarce and mainly extracted from studies including other soft tissue sarcomas (STSs). Nevertheless, in a national database study, first-line CT in uLMS showed a survival benefit of 8 months for patients receiving this treatment. 34 CT achieves a response rate between 17% and 25%. Table 3 summarizes the most relevant trials in first- or second-line settings.
Staging of uterine sarcoma.
Single-agent doxorubicin has been the accepted standard of care for first-line STS and uterine sarcomas. Despite attempts to improve the results with the addition of other drugs, such as ifosfamide, 77 or testing other combinations, such as gemcitabine–docetaxel 78 no benefit of the combination over single-agent doxorubicin was seen.
However, a recent randomized phase III trial compared the combination of doxorubicin plus trabectedin (3-h infusion) followed by maintenance trabectedin for up to 17 cycles versus doxorubicin alone for six cycles in first line in patients with advanced or recurrent LMS. uLMS was a stratification factor. The combination showed a relevant improvement in PFS [13.5 versus 7.3 months; adjusted hazard ratio (HR) = 0.38, 0.27–0.55]. 79 Moreover, the response rate was also superior for the combination (38% versus 13%), with a median follow up of 37 months. OS was 24.1 months in the standard group versus 30.5 m in the experimental arm; HR: 0.73 [95% confidence interval (CI): 0.49–1.12]. Toxicity, particularly hematological and gastrointestinal, was higher in the trabectedin–doxorubicin arm. Based on these results, a combination of trabectedin + doxorubicin should be the first-line regimen of choice for fit patients.
In uLMS patients, the combination of doxorubicin-dacarbazine may also be an option. In a retrospective study, this combination showed a longer OS (median 36.8 months) and a higher response rate (30.6%) than the combination of doxorubicin-ifosfamide or doxorubicine alone. 51 Nevertheless, in prospective trials, anthracycline-based combination chemotherapy has shown to increase toxicity without any benefit on overall survival. 80
Ifosfamide, either as a single agent or combined with another agent, is not commonly recommended in uLMS based on retrospective data. 51 However, a single arm prospective phase II study that examined the role of first-line ifosfamide in CT-naïve ESS patients showed a response rate of 33%. 81
Table 4 summarizes first-line systemic treatment options for high-grade uterine sarcoma.
Most relevant prospective trials with systemic agents including uterine sarcoma patients.
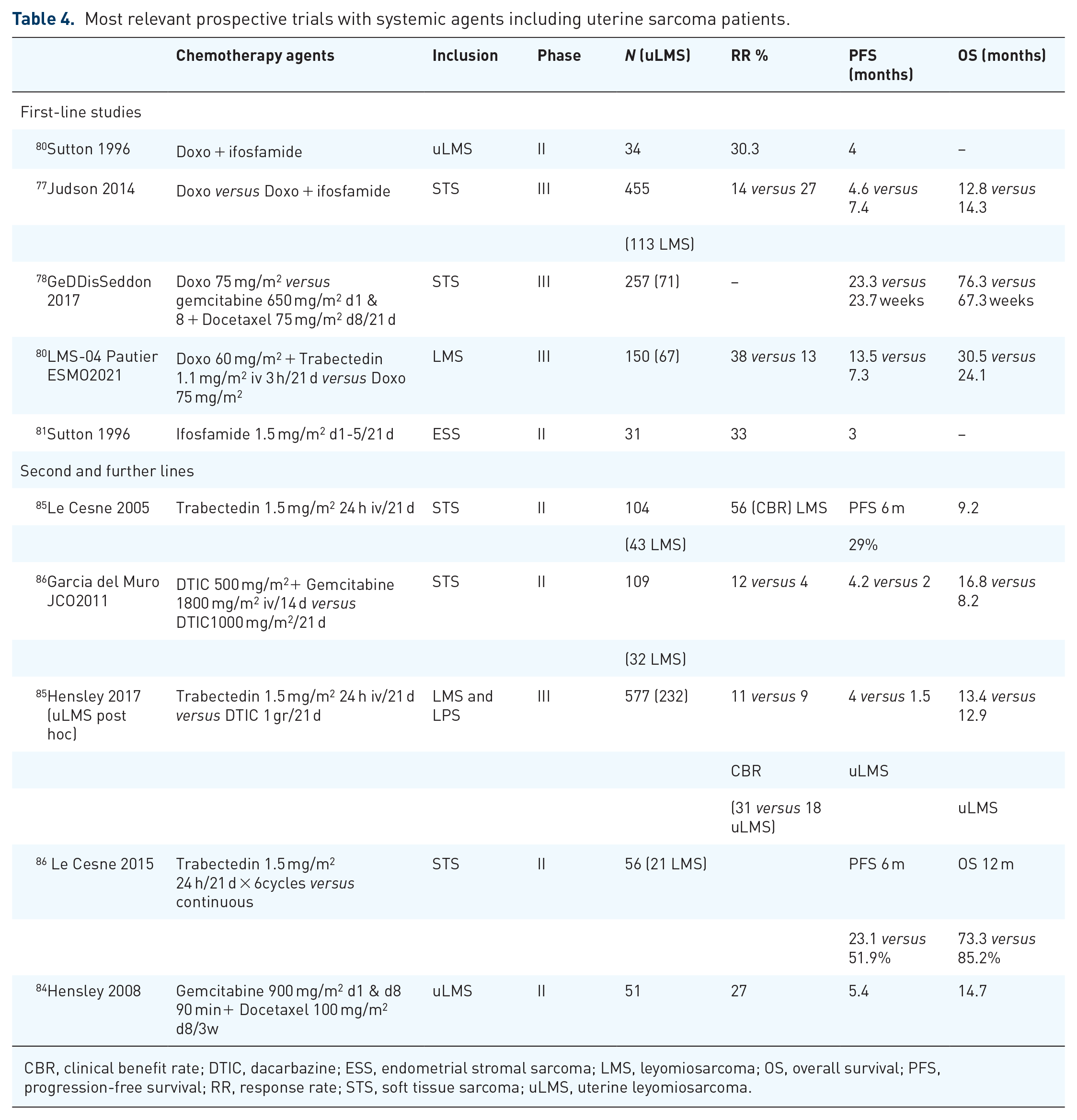
CBR, clinical benefit rate; DTIC, dacarbazine; ESS, endometrial stromal sarcoma; LMS, leyomiosarcoma; OS, overall survival; PFS, progression-free survival; RR, response rate; STS, soft tissue sarcoma; uLMS, uterine leyomiosarcoma.
In fit patients with uLMS the combination of doxorubicin plus trabectedin should be the preferred first-line chemotherapy, particularly when obtaining a response is relevant (IA). Nevertheless, single agent doxorubicin and doxorubicin-dacarbazine are valuable alternatives.
In non-uLMS histologies, single-agent doxorubicin should be considered standard (IB). However, anthracycline-based combinations (with ifosfamide or dacarbazine) are valuable options when obtaining a response is relevant.
Second and later lines
Trabectedin was first approved for STSs in Europe following two phase II trials.82,83 It was recently compared with dacarbazine in a phase III trial in metastatic liposarcoma and leiomyosarcoma patients. In a post-hoc analysis, progression-free survival (PFS) favored trabectedin, especially in uLMS patients, with a clinical benefit rate of 31% versus 18% but no difference in OS. 88 A benefit with the latter strategy was seen in a phase II randomized trial that compared intermittent versus continuous administration of trabectedin in patients achieving disease control. 85
Gemcitabine-based combinations are commonly used. In a phase II trial conducted by the Spanish Sarcoma Group (GEIS) in STS, including uterine sarcomas, the combination of gemcitabine and dacarbazine showed a superior response to dacarbazine alone (49% versus 25%), PFS (4.2 months versus 2 months; 0.58; 95% CI, 0.39–0.86; p < 0.005) and OS (16.8 months versus 8.2 months; HR, 0.56; 95% CI, 0.36–0.90; p < 0.014). Histotype leiomyosarcoma and combination treatment were significantly favorable prognostic factors for PFS. Furthermore, the regimen is well tolerated, and has high compliance despite extensive prior treatment exposure. 86 Combining gemcitabine with docetaxel is also an option and has been evaluated in several phase II studies that included uLMS. 84 Gemcitabine monotherapy could be an option in selected patients.
Pazopanib is an oral antiangiogenic therapy targeting VEGFR. The phase III Palette trial 87 randomized STS patients who had been treated with at least one previous line including anthracyclines to received pazopanib 800 mg once daily or placebo. In all, 372 patients were included, approximately 40% of whom had leiomyosarcoma. With pazopanib, the median PFS increased from 1.6 months (placebo arm) to 4.6 months (pazopanib arm) with an HR = 0.31 (95% CI: 0.24–0.40). A subgroup analysis confirmed that the efficacy of pazopanib in uLMS appears to be similar than in the non-uterine LMS subgroup. 89
Table 2 summarizes second-line systemic treatment options for high-grade uterine sarcoma.
Trabectedin, gemcitabine combinations (preferably with dacarbazine), and pazopanib are second and further-line options depending on the first-line chemotherapy regimen received.
Endocrine therapy
Data supporting endocrine therapy in HG-uterine sarcoma are scarce and restricted to uLMS and aromatase inhibitors (AI). In one retrospective study, 40 patients, 50% of whom were ER+, were treated with AI. A partial response was achieved in 9% of patients (all ER-positive) and 32% achieved stable disease (SD). 90 In another retrospective study including 16 patients who had ER+/PR+ tumors and received AI, the response rate was 12.5%, whereas the clinical benefit reached 62.5% (partial response and SD of at least 6 months). Six of these patients received another AI (exemestane) as second-line treatment, achieving a 1-year progression-free rate of 80%. 91
A phase II non-randomized trial evaluated letrozole in 27 ER+/PR+ patients with unresectable uLMS. The 12-week PFS rate was 50%, and the SD rate was 54%, without any objective response. 92 However, three patients who expressed hormonal receptors in over 90% of tumor cells continued letrozol for at least 24 weeks.
Endocrine therapy with AI may be a reasonable option for some patients with advanced ER/PR-positive uLMS, particularly those who have indolent tumors with a low disease burden and high hormonal receptor positivity [IIIC].
Targeted therapy
NTRK fusions
NTRK rearrangements have also been described in uterine tumors arising mainly in the uterine cervix of premenopausal women.93,94 They exhibit features fibrosarcoma and although few cases have been published to date, they might benefit from targeted therapy. 95
NTRK inhibitors larotrectinib 96 and entrectinib 96 have shown activity in several tumor types harboring NTRK fusions. Larotrectinib achieved an ORR of 75% and entrectinib achieved an ORR of 57%. Both drugs also showed a high proportion of patients maintained prolonged responses. In these trials, only the entrectinib trial enrolled uterine sarcomas, one adenosarcoma and one ESS.
Recently, an NTRK fusion-positive uterine spindle cell sarcoma with features of fibrosarcoma was described. 93 Such tumors harbor NTRK1 and NTRK3 rearrangements, suggesting a potential benefit from NTRK inhibitors.
ALK fusions
Inflammatory myofibroblastic tumors (IMTs) can arise in the uterus. Differential diagnoses must be made with endometrial stromal tumors and also with myxoid variants of uterine smooth muscle tumors. IMTs can exhibit ALK rearrangements, however, as in other STS IMTs, not all are ALK-rearranged. 97 ALK-rearranged locally advanced or metastatic IMTs tumors might be treated with crizotinib. In a single arm phase II study, the objective response rate to crizotinib was 66.7% and median PFS was 18 months for the patients with ALK fusions. 98
NTRK fusion testing could be considered in uterine sarcoma. Patients with NTRK-positive tumors should be offered an NTRK inhibitor [IIIA].
Low-grade uterine sarcoma
LG-ESS is characterized by high expression of ER and PR. Therefore, despite the lack of prospective randomized data, treatment with AI and progestins are commonly used in an advanced or recurrent disease setting. Due to a better toxicity profile, AI is preferred as first-line hormonal therapy. The use of tamoxifen is contraindicated in uterine sarcomas expressing hormonal receptors due to its endometrial proliferative effect. 99
Many retrospective series have reported the effectivity of progestins and AI in the first-line setting. The Royal Marsden study included 13 patients who were treated mainly with AI (11 patients with AI and 2 patients with progestins) and found a response rate of 46.2% and a median PFS of 4 years. 91 A Dutch series with 11 patients treated with AI and megestrol showed a response rate of 82% with a median duration of response of 48 months. 100 A response rate of 77.7% was also reported by Loffe et al. in 18 LG-ESS patients treated with hormonal therapy. 101
Recently, a basket single arm trial of anastrozole in first-line positive ER/PR gynecological cancers enrolled 15 LG-ESS patients. Response rate in the setting of this clinical trial was 26.7% and SD was achieved in 46.7% of the patients. Median PFS was not reached and PFS was 73.3%, 73.3%, and 66% at 6, 12, and 18 months, respectively. 102
In a second-line setting in the Royal Marsden study, patients receiving second-line hormonal therapy had a median PFS of 3 years and a 2-year PFS of almost 90%.
Based on a few individual case reports, other hormonal therapies., include GnRH analogues or fulvestrant. 103 Fulvestrant is currently under evaluation in the FUCHSia phase II trial.
Upon progression to hormonal therapies or in case of no expression of hormonal receptors, chemotherapy regimens used in other STSs might be employed but such practice has only been described in a few case reports.
Treatment with AI or progestins is recommended as first-line treatment. Due to their better toxicity profile, AI are preferred as first- line hormonal therapy (IIIA).
Other infrequent subtypes: Adenosarcoma, PEComa, uterine tumor resembling ovarian sex cord tumor
Uterine adenosarcoma
Uterine adenosarcoma is a rare entity, accounting for approximately 5–9% of uterine sarcomas and only about 0.2% of all uterine neoplasms.104,105 Most patients with adenosarcoma are diagnosed with stage I disease and have a 5-year OS of 60–80%.
Uterine adenosarcoma is considered a biphasic tumor because of the presence of benign epithelial elements combined with a malignant mesenchymal component. Sarcomatous overgrowth is associated with worse prognosis.
The standard treatment is hysterectomy with bilateral salpingo-oophorectomy given the risk of local spread to the adnexa and ovaries. Lymphadenectomy is not recommended as the incidence of nodal metastasis is very low. 106
Adjuvant pelvic RT is not recommended for uterine adenosarcoma, because no survival benefit has been proven. 107 The role of adjuvant CT is uncertain as adenosarcomas have been frequently excluded from clinical trials.
ER/PR positivity could be used as a predictive biomarker for response to hormonal therapy but evidence is limited to case reports or case series in the adjuvant and advanced setting (V, B). Endocrine therapy should be limited to low-grade adenosarcomas.
There is no standard treatment for advanced/recurrent disease. CT or endocrine therapy can be considered (V, A) and in selected cases surgery could be an option (V, A). Regarding CT doxorubicin-based regimens, 108 gemcitabine/docetaxel, 109 and trabectedin 110 have been described in case reports and case series.
A retrospective review showed an increase in PFS in patients treated with doxorubicin and ifosfamide compared to those receiving doxorubicin alone or other regimens. 111
Perivascular epithelioid cell tumors
Perivascular epithelioid cell tumors are ultra-rare vascular neoplasms that can arise in multiple locations including the uterus. The mainstay of treatment consists of hysterectomy plus bilateral oophorectomy. There are no data to support adjuvant treatment either with systemic treatment or RT. In the metastatic setting, due to the mTOR pathway activation in these tumors, mTOR inhibitors such as sirolimus are the preferred treatment choice (III-A). Recently, the phase II single arm AMPEC trial evaluating nab-sirolimus revealed a 39% response rate for the overall population and 89% for the patients with TSC2 mutation. The duration of response also correlated with the mutational status. 112 STS treatment regimens might be an option as second-line and further line therapies but low responses and short PFS are to be expected. 113 (IV-B).
Uterine tumor resembling ovarian sex cord tumors
Uterine tumor resembling ovarian sex cord tumors (UTROSCT) represent fewer than 1% of uterine mesenchymal tumors and their etiology is unknown. These rare tumors affect the uterine corpus (intramural or submucosal), and occasionally the cervix in middle-aged peri-menopausal women. They usually present with abnormal vaginal bleeding or pelvic pain.
UTROSCT are generally benign but sometimes have malignant potential and may recur. Those more aggressive subtypes have been associated with extensive rhabdoid differentiation and GREB1 and ESR1-NCOA2 fusions.114–117
Morphologically, UTROSCT display patterns that resemble those of ovarian sex cord tumors, without a component of recognizable endometrial stroma. 118 UTROSCT are usually well-defined and grow in sheets, cords, nests, insulae, trabeculae, or tubules. Mitotic figures and necrosis are infrequent. Their immunohistochemical profile exhibits variable positivity for sex cord markers (inhibin, calretinin, WT1, CD56, CD99, SF1, FOXL2, and melan-A), epithelial markers (pancytokeratin, EMA), smooth muscle markers (actin, desmin, and h-caldesmon) and miscellaneous markers (ER, PR, CD10).
The staging strategy is similar to that of other uterine sarcomas. Hysterectomy ± bilateral salpingo-oophorectomy is usually the treatment of choice [VA], although fertility-preserving surgery has been successfully reported in younger women. 119
Adjuvant treatment is generally not recommended, and recommended first-line CT is doxorubicin. Other combinations include carboplatin–gemcitabine or bleomycin, etoposide and cisplatin, but evidence to support these alternative regimens is lacking. 120
Follow-up strategy
For high-grade sarcomas, follow-up should include physical examination and imaging every 3–4 months for the first 2–3 years, every 6 months for the following 5 years, and annually thereafter. Imaging should include a CT scan of chest, abdomen and pelvis, with or without pelvic MRI 121 [VA]. A PET-CT can be useful. A chest X-ray could be considered instead of a CT scan after 5 years of surveillance to reduce radiation exposure. 122 Patients should be informed about symptoms that could suggest recurrence and the importance of seeking prompt medical attention.
LGESS is usually a slow-growing malignancy with an indolent course. Several studies suggest that up to 80% of recurrences can be detected by routine radiographic surveillance. 75
Based on expert opinion, a CT scan of chest, abdomen, and pelvis is recommended every 3–6 months for the first 3 years, and then every 6–12 months for the next 2 years. Annual or biannual imaging should be considered thereafter for an additional 5 years. [VB.]. Long-term clinical surveillance is also recommended (VB).
