Abstract
Background:
Grading system for resected invasive pulmonary adenocarcinoma proposed by the International Association for the Study of Lung Cancer (IASLC) was validated as a strong prognostic indicator. Nonetheless, the efficacy of utilizing such grading system in prognostic assessment of patients receiving neoadjuvant therapy still needs elucidating.
Methods:
A retrospective study was conducted including patients with resected adenocarcinoma following neoadjuvant chemotherapy or targeted therapy from August 2012 to December 2020 in Shanghai Pulmonary Hospital. All the surgical specimens were re-evaluated and graded. The prognostic value of the grading system was further validated.
Results:
Ultimately, a total of 198 patients were enrolled in this study, and subdivided into three cohorts according to the grading system. There were 13 (6.6%), 37 (18.7%), and 148 (74.7%) patients belonging to Grades 1, 2, and 3, respectively. IASLC grading system demonstrated significant power in prognosis differentiation of the entire cohort [recurrence-free survival (RFS), p < 0.001; overall survival (OS), p < 0.001] and the neoadjuvant chemotherapy and targeted therapy cohorts separately, and was further verified as a significant prognostic indicator for RFS and OS in multivariable Cox analysis. Since the majority of the patients (84.8%) did not achieve major pathologic response (MPR), representing a wide spectrum of survival, the prognostic value of grading system in non-MPR cohort was further evaluated. Similar results were also obtained that IASLC grading system was assessed significant in univariable analysis of RFS (p < 0.001) and univariable analysis of OS (p = 0.001).
Conclusions:
The prognostic efficacy of pathological evaluation of the residual proportion of pulmonary adenocarcinoma post-neoadjuvant therapy using IASLC grading system was preliminarily verified. Such grading system might assist prognostic evaluation of neoadjuvant cohort other than traditional pathological parameters.
Introduction
Recent years have witnessed the emergence and wide application of neoadjuvant therapy in non-small-cell lung cancer (NSCLC). 1 Simultaneously, treatment regimens have been enormously enriched, from the initial chemotherapy2,3 to systematic modalities including targeted and immunotherapy.4–7 Since the inflexibility and time-consuming nature of survival outcomes, other effective parameters of evaluating neoadjuvant therapy efficacy need exploring. The percentage of residual viable tumor cells in the tumor bed no more than 10% was brought up as a significant indicator for survival improvement for lung cancer patients receiving neoadjuvant chemotherapy in 2012, 8 and was subsequently specified as major pathologic response (MPR) and proposed as a surrogate for survival of neoadjuvant therapy-treated cohort. 9 Despite the strong association between MPR and survival benefits, those who did not achieve MPR still occupied approximately 60–80% of the entire cohort,10–13 which represented a wide spectrum of prognosis. Hence, other clinicopathological parameters assisting treatment response status in prognostic assessment are still warranted.
The previous subclassification criteria of invasive pulmonary adenocarcinoma (IPA) were primarily based upon architectural proportions, specifically, five predominant histomorphologic patterns, 14 demonstrating distinct survival outcomes.15,16 Nevertheless, other pathological features, for instance, complex glandular patterns, were also found to confer poor prognosis,17,18 but were not defined as independent subcategories. A recent proposal to renew the grading system of IPA put forward by the International Association for the Study of Lung Cancer (IASLC) pathology committee has taken the concept of high-grade patterns (including solid, micro papillary, and complex glandular patterns) into account with a 20% threshold, and subdivided IPA into three grades. 19 Such grading system was subsequently validated in large cohort studies of Asian patients and verified as a robust prognostic indicator,20–22 which harbored promising potential in prognosis evaluation. Our previous study further stratified the prognosis of completely resected stage I IPA utilizing IASLC grading system and verified its clinical significance. 23 Nevertheless, the feasibility of using such grading system in prognosis differentiation after neoadjuvant therapy still needs exploring.
Hence, on the basis of pathological evaluation of the residual proportion post-neoadjuvant therapy in accordance with the IASLC proposed IPA grading system, we would like to further elucidate its value in prognostic stratification. Such grading system might assist or even substitute for pathological response assessment under certain circumstances.
Methods
Patient cohort
We retrospectively enrolled lung adenocarcinoma patients who received neoadjuvant chemotherapy (platinum-based doublet therapy, mostly carboplatin combined with pemetrexed or paclitaxel) or targeted therapy [epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors, started from March 2016] between August 2012 and December 2020 in Shanghai Pulmonary Hospital. The main inclusion criterion was pathologically confirmed IPA patients undertaking neoadjuvant therapy prior to curative resection. Patients with distant metastasis, pathologically proven invasive mucinous adenocarcinoma, or other variants of invasive adenocarcinoma, participating in any other clinical trial were excluded from the study, while those who achieved pathologic complete response (pCR, defined as no residual viable tumor in primary tumor bed) were also excluded. The neoadjuvant regimen was decided and administered after a multiple disciplinary team consultation, which comprised pulmonologists, oncologists, radiologists, and thoracic surgeons. Besides, postoperative consolidation therapy was administered based on the clinical response status of the neoadjuvant therapy. This study was conducted in accordance with Declaration of Helsinki (as revised in 2013), while informed consent was waived by the Institutional Review Board of Shanghai Pulmonary Hospital (IRB ID: K22-294) due to its retrospective nature.
Clinical and pathological evaluation
All the enrolled patients received computed tomography (CT) evaluation both prior to and after neoadjuvant therapy, and hence the treatment response was initially evaluated radiologically. Clinical responses were separately defined as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) according to RECIST Version 1.1 criteria. 24 Besides, clinical staging based on the baseline CT scan was conducted conforming to the UICC/AJCC TNM classification system (TNM stage classification, 8th edition). 25 After the neoadjuvant therapy, all the patients were reevaluated for the eligibility for surgery and type of resection. After the surgery, primary tumor and lymph node specimens were sectioned and restaged according to the IASLC recommendations for assessing specimens after neoadjuvant therapy (ypTNM stage), 26 according to which ypT stage was specifically estimated by multiplying the total size of the tumor bed times the percentage of viable tumor. Pathological response was also evaluated in accordance with such recommendations, as the percentages of viable tumor cells, necrosis, and stroma were respectively determined. MPR was defined as no more than 10% viable tumor in the primary tumor bed while the circumstance where no viable tumor was observed was defined as pCR. Furthermore, several prominent pathological patterns including tumor spread through air spaces (STAS) as well as pleural invasion of the specimens were also appraised.
Histological grading
The IASLC issued grading system for IPA comprised three levels of invasion descriptions based on the histological subtype of tumor, which were well-differentiated adenocarcinomas (Grade 1), moderately differentiated adenocarcinomas (Grade 2), and poorly differentiated adenocarcinomas (Grade 3). 19 The typical pathological proportions are illustrated in Figure 1. In accordance with the IASLC system, all the surgical specimens were separately graded by two pathologists (Dr. Likun Hou and Dr. Shaoling Li). When disparities were encountered, consensus was reached through discussion.
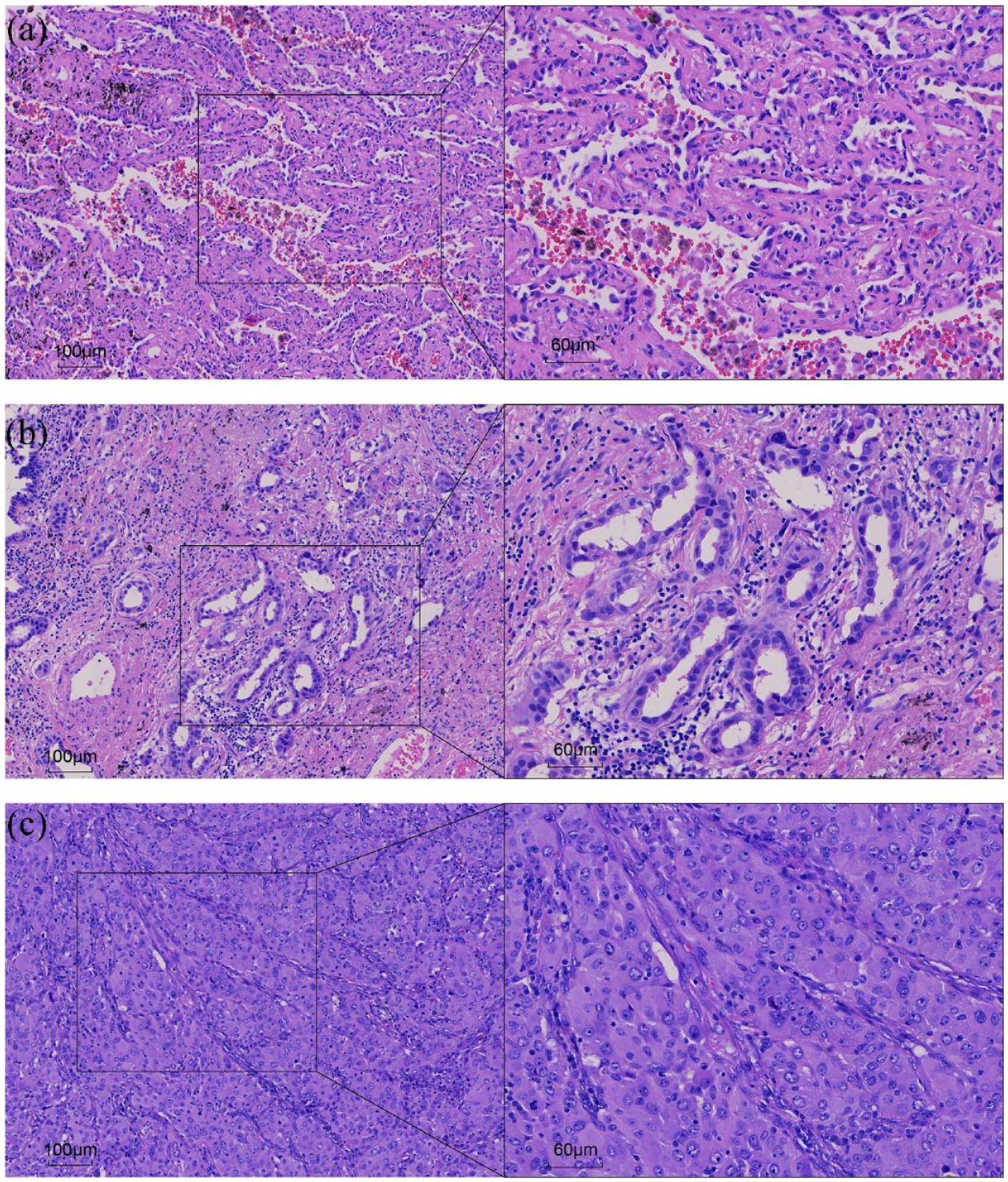
Typical histological patterns of the surgically resected IPA specimens. Lepidic, acinar, and solid patterns are separately illustrated in a, b, and c. The first case (a) was subsequently evaluated as Grade 1 as the residual proportion contained a 60% of lepidic component and 40% of acinar component. The second case (b) was evaluated as Grade 2 as the proportion contained a 90% of acinar component and 10% of micro papillary component. The third case (c), on the contrary, contained a 30% of acinar component and 70% of high-grade patterns (20% of solid pattern, 10% of micro papillary component, and 40% of complex glandular pattern) and was consequently evaluated as Grade 3.
Follow-up strategy
During the first 2 years post-surgery, physical examination, blood tests, and chest radiography were conducted every 3 months, along with chest CT scans every 6 months. Afterwards, chest radiography was performed every 6 months and chest CT scanning annually. For patients with high-risk factors for recurrence or exhibiting any symptom of recurrence, a whole-body bone scan, ultrasound of the abdomen/neck/supraclavicular area, and brain magnetic resonance imaging was performed annually. The follow-up information was collected through outpatient visits or by telephone calls. Recurrence-free survival (RFS) was defined as the time from the operation to disease progression, relapse or death, whichever event came first, while overall survival (OS) was defined as the time from the operation to any cause of death. All the data censored at the last time of follow-up in October, 2021.
Statistical analysis
Differences concerning nominal and ordinal variables of three grades were evaluated using Pearson’s chi-squared test or Fisher’s exact test, while Kruskal–Wallis one-way ANOVA was performed when comparing interval variables. Kaplan–Meier estimates were utilized to analyze the survival outcomes concerning different grade groups, and a log-rank test was subsequently conducted to test the significance. Univariable and multivariable Cox proportional hazard analyses were conducted to identify the prognostic predictors. All the comparisons between the clinical and pathological information and Cox proportional hazard analyses were performed using SPSS (version 26.0; IBM Corp, Armonk, NY, USA), and all the violin plots and Kaplan–Meier estimates were conducted using the ‘ggplot2’, ‘survival’, and ‘survminer’ packages in R software (version 4.1.1, https://cran.r-project.org). All the statistical tests were two-sided with a significant level when p < 0.05.
Results
Clinical and pathological characteristics
In total, 198 patients were enrolled in this study (Supplemental Figure S1), and patients’ demographic, clinical, and pathological characteristics are demonstrated in Table 1. There were 13 (6.6%), 37 (18.7%), and 148 (74.7%) patients finally evaluated belonging to Grades 1, 2, and 3, respectively, with an interobserver agreement k value of 0.913 (Supplemental Table S1). The median age of the entire cohort was 60 years (interquartile range: 53–65 years), with a predominant sex of male (117/198, 59.1%). Most of the patients (146/198, 73.7%) were diagnosed as clinically stage III, and 76 (38.4%), 117 (59.1%), and 5 patients (2.5%) were observed as PR, SD, and PD after neoadjuvant therapy, respectively. Most of the patients undertook neoadjuvant chemotherapy (128/198, 64.6%), while patients diagnosed with Grade 3 tended to receive chemotherapy more often (p < 0.001), and the specific treatment modalities of the entire cohort are presented in Supplemental Table S2. In regard to the pathological findings, 38.9% (77/198) and 18.2% (36/198) of the specimens were evaluated positive for STAS and pleural invasion, respectively. Pathological evaluation also showed that half of the patients were diagnosed as ypN2 (99/198, 50.0%). Besides, the status of major driver oncogene alterations, including EGFR, K-Ras, B-Raf, PIK3CA, ALK, and ROS1 gene mutations, was also evaluated, and the specific information was presented in Supplemental Table S3. Moreover, MPR was observed in 30 patients (15.2%), and the MPR rate dropped significantly in Grade 3, compared with its counterparts (p < 0.001). Pathological response rate was also compared among three grades, while Grade 1 exhibited a significant high proportion of response (Figure 2(a), p < 0.001). Furthermore, 51 patients of the entire cohort (25.8%) relapsed within 1-year post-surgery.
Demographic, clinical, and pathological characteristics of the entire cohort.

cTNM stage and ypTNM stage was classified according to TNM classification eighth edition, clinical response was evaluated in accordance with RECIST V1.1.
Comparison of p values between three cohorts.
IQR, interquartile range; MPR, major pathologic response; PD, progressive disease; PR, partial response; SD, stable disease; STAS, tumor spread through air spaces. Significant p values are in bold form.

Pathological response status of different grades concerning the entire cohort (a) and the non-MPR cohort (b). The central dots represented the median pathological response rates of different grades, and the box plots demonstrated the IQRs and ranges of the data. The density curves of the data are also shown through violin plots.
IASLC grading system served as a strong indicator for RFS and OS
Based on the follow-up information of the entire cohort, IASLC grading system harbored significant power in differentiating the RFS (Figure 3(a); p < 0.001) and OS (Figure 3(b); p < 0.001) of the entire cohort. The univariable predictors reaching a significance of p < 0.05 comprised the grading system (p < 0.001), neoadjuvant (p < 0.001) and adjuvant regimens (p = 0.003), presence of STAS (p < 0.001), ypN stage (p < 0.001) and MPR status (p = 0.001), and after adjustment, grading system, ypN stage, neoadjuvant regimen, STAS, and MPR status were subsequently included into the multivariable model (Table 2) (adjuvant regimen excluded due to its close relation with neoadjuvant therapy). IASLC grading system was determined as a strong indicator for RFS [Grade 2 versus Grade 1, hazard ratio (HR): 4.243, 95% confidence interval (CI): 0.870–20.689, p = 0.074; Grade 3 versus Grade 1, HR: 10.215, 95% CI: 2.312–45.134, p = 0.002] along with ypN stage (ypN1 versus ypN0, HR: 1.000, 95% CI: 0.488–2.051, p = 0.999; ypN2 versus ypN0, HR: 2.047, 95% CI: 1.257–3.335, p = 0.004) in the multivariable analysis. Similar results were also obtained concerning grading system in predicting the OS status of the entire cohort (Table 2, Grade 2 versus Grade 1, HR: 2.075, 95% CI: 0.378–11.387, p = 0.401; Grade 3 versus Grade 1, HR: 7.038, 95% CI: 1.510–32.811, p = 0.013) after multivariable adjustment including grading system, ypN stage, neoadjuvant regimen, STAS, and MPR status into analysis. Furthermore, to better illustrate the prognostic significance of IASLC grading system concerning patients receiving different treatment modalities, we extracted the patients from the neoadjuvant chemotherapy and targeted therapy cohorts separately, and discovered that grading system was verified as a robust prognosticator for both cohorts (Supplemental Figure S2, neoadjuvant chemotherapy cohort: RFS, p = 0.001, OS, p = 0.002; Supplemental Figure S3, neoadjuvant targeted therapy cohort: RFS, p < 0.001, OS, p = 0.001).

Survival curves of the entire cohort (198 patients). Kaplan–Meier estimate curves of RFS (a) and OS (b) curves of the included patients with invasive pulmonary adenocarcinomas receiving neoadjuvant therapy. p Value listed demonstrated the significance of the log-rank test between three grade groups.
Univariable and multivariable Cox regression analyses of RFS and OS concerning the entire cohort (N = 198).

chemo, neoadjuvant chemotherapy; CI, confidence interval; HR, hazard ratio; IASLC, International Association for the Study of Lung Cancer; MPR, major pathologic response; OS, overall survival; RFS, recurrence-free survival; STAS, tumor spread through air spaces. Significant p values are in bold form.
Prognostic evaluation of IASLC grading system in Non-MPR cohort
Since there was only a minority of patients (15.2%) achieving MPR, non-MPR cohort still represented for a wide spectrum of patients with distinct clinical prognosis. We then evaluated whether such cohort could be further stratified according to IASLC grading system. Pathological response rate was first compared among three grades, and phenomenon similar to the entire cohort was obtained (Figure 2(b), p = 0.004). Kaplan–Meier curves also demonstrated the strong prognosis differentiation power of IASLC grading system of the non-MPR patients (Figure 4). Adjusted multivariable analysis including grading system, ypN stage, neoadjuvant regimen, and STAS status found that neoadjuvant regimen (Table 3, targeted versus chemotherapy, HR: 0.468, 95% CI: 0.257–0.852, p = 0.013) along with ypN stage (ypN1 versus ypN0, HR: 1.034, 95% CI: 0.473–2.260, p = 0.933; ypN2 versus ypN0, HR: 1.967, 95% CI: 1.165–3.321, p = 0.011) were significantly correlated with RFS. The grading system was further validated as a strong factor in the univariable analysis of OS (p = 0.001) in the non-MPR cohort (Table 3), but not in the adjusted multivariable analysis (p = 0.079).
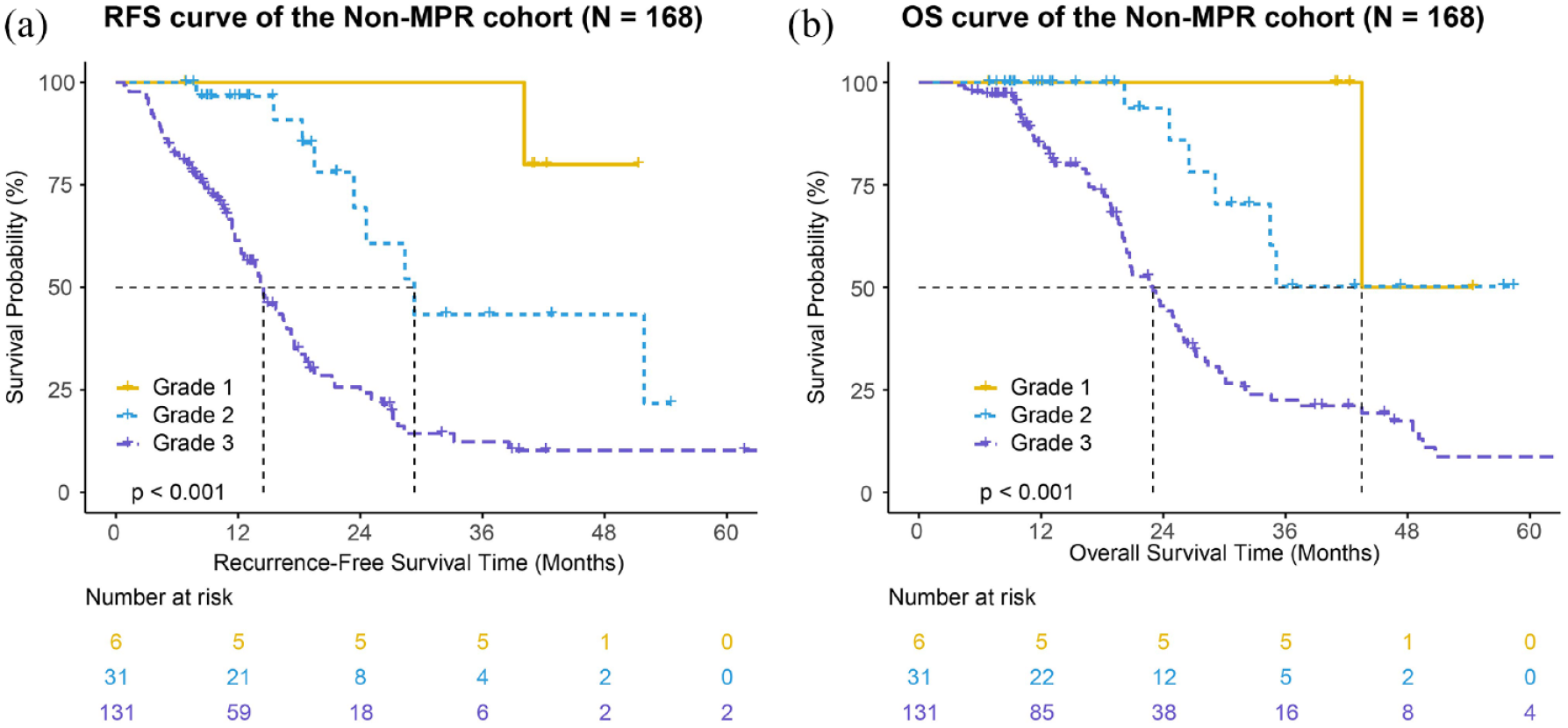
Survival curves of the non-MPR cohort (168 patients). Kaplan–Meier estimate curves of RFS (a) and OS (b) curves of the patients not achieving major pathologic response. p Value listed demonstrated the significance of the log-rank test between three grade groups.
Univariable and multivariable Cox regression analyses of RFS and OS concerning the non-MPR patients (N = 168).

chemo, neoadjuvant chemotherapy; CI, confidence interval; HR, hazard ratio; IASLC, International Association for the Study of Lung Cancer; OS, overall survival; RFS, recurrence-free survival; STAS, tumor spread through air spaces. Significant p values are in bold form.
Prognostic comparison between Grade 1 and pCR cohorts
To further elucidate whether grade 1 IPA correlated with fairly better prognosis than grades 2 and 3 in the entire cohort, such group of patients was separately extracted and compared with patients pathologically evaluated achieving pCR between August 2012 and December 2020. The external pCR cohort comprised of 10 patients and the clinicopathological characteristics are summarized in Supplemental Table S4. We further compared the prognoses between Grade 1 and external pCR cohorts, and Kaplan–Meier curves demonstrated that there was no significant difference between these two groups of patients in regard to RFS and OS status (Supplemental Figure S4).
Discussion
The prognostic value of IASLC proposed grading system for IPA was preliminarily validated by this retrospective single-center study in Chinese cohort receiving neoadjuvant therapy. IASLC grading system was a significant classifier for RFS and OS status of the neoadjuvant therapy-treated IPA patients, and compensated as an important indicator for pathological response and ypN stage in regard to subgroup analysis. Specifically, patients with IPA classified as grade 1 was depicted as one cohort with satisfactory prognosis, approximate to those who achieved complete pathologic response, while conversely, those with grade 3 IPA tended to encounter disease progression or relapse more frequently. Such grading system harbors the potential in survival differentiation of the IPA cohorts receiving neoadjuvant therapy, specifically for those not achieving MPR.
The subclassification benchmark of IPA was initially based upon the 2011 international multidisciplinary recommendation, introducing the concept of predominant architectural patterns, 14 which was afterwards adopted by the 2015 WHO classification system. 27 Such architectural grading system was reportedly well correlated with contrasting prognosis15,16 and was proven predictive of response to adjuvant chemotherapy.28,29 Albeit the high efficacy in survival stratification, inaccuracy still existed for certain group of patients, specifically for acinar-predominant subtype which reportedly represented a wide variety of prognosis.17,18 To further differentiate IPA patients, IASLC pathology committee consequently initiated a novel grading system exploiting predominant subtype in combination with proportional distribution of high-grade patterns. 19 Subsequently, Weng et al. investigated the value of new grading system in survival outcome estimation of advanced stage lung adenocarcinoma and proved a close interaction between poorly differentiated arm with poor prognosis. 30 Deng et al. further validated the survival discrimination efficacy of new grading system in a large Chinese cohort and illustrated a pathologic-genetic subclassification modality in survival prediction. 20 Subsequently, the prognostic significance of the system was verified by Rokutan-Kurata et al. in a large Japanese cohort as well. 21 Fujikawa et al. further demonstrated the distinct clinicopathologic traits and genotypic features of different grades. 22 Another recent study conducted by our group explored the clinical significance of the novel system in evaluation of survival status of completely resected stage I IPA and direction of adjuvant therapy through external institutions integrated validation. 23 Despite the retrospective nature of these studies, the efficacy of IASLC grading system in survival differentiation was preliminarily confirmed.
Since the advent of including neoadjuvant therapy into the systematic regimen of locally advanced NSCLC, it served as one of the passionately debated issues. Neoadjuvant chemotherapy was initially evaluated beneficial for survival improvement through large cohort of clinical trials2,3 and recommended as one effective downstaging treatment modality. 31 Hereafter, numerous clinical trials evaluating the efficacy of neoadjuvant targeted and immunotherapy were conducted worldwide, continually extending the treatment regimens.4,5,7,12,13,32,33 Parameters other than survival outcomes were consequently needed to evaluate the trials in a more effective way. Pathological response was evaluated as a strong predictor for survival benefits in numerous solid tumors. 34 Dislike other tumors including breast and bladder cancers, where pathological complete response was more frequently encountered after neoadjuvant therapy and was thus selected as a prominent parameter for pathological response assessment,35,36 the frequency of pCR in NSCLC post-therapy was scarcely low. 34 Hence, Junker et al. initially exploited <10% viable tumor cells of the tumor tissue as a critical prognosticator for preferable survival outcome of lung cancer patients, 37 which was subsequently defined as MPR by Hellmann et al. and recognized as substitute endpoint for survival outcomes regarding neoadjuvant trials. 9 Specific recommendations for pathologic assessment of the residual proportion of neoadjuvant therapy-treated lung cancer lesions were proposed later in 2020. 26 In our study, MPR was evaluated significant for survival outcomes in the univariable analysis concerning the entire cohort, while it was insignificant in the multivariable analysis when included along with IASLC grading system, ypN stage, and other critical clinicopathological parameters. Such phenomenon might be attributed to the prognostic prediction efficacy of pathological response status lower than traditionally robust ypN stage and aforementioned grading system. Nonetheless, such speculation still needed future studies’ confirmation.
Another controversy over pathological response evaluation of IPA existed concerning the threshold of residual viable tumor cells in defining MPR. Qu et al. reported a distinct optimal cutoff percentage of viable tumor between lung adenocarcinoma and squamous cell carcinoma, where 65% of viable tumor in the tumor bed harbored seemingly high efficacy in survival prediction. 38 Such phenomenon was subsequently confirmed by Liu et al. with a calculated optimal cutoff of 58% of residual viable tumor, close to the 65% threshold. 39 Despite these studies’ results, IASLC still recommended a 10% of viable tumor as a threshold for defining MPR when assessing of resected specimens. 26 Since such uncertainty still existed, IASLC grading system might serve as a valuable substitute for MPR, especially IPA, which customarily represented a large variety of non-MPR patients under a cutoff value of 10%.
Limitations still exist concerning our study. First, due to the retrospective nature of the study and a limited enrolled sample size, patient selection bias was inevitably encountered and hence the efficacy of the study was relatively restricted. Further large-scale studies even with a prospective design are still warranted. Besides, the follow-up period of this study was short due to the fact that most patients enrolled were treated after 2018; hence, future studies with long-term surveillance are needed to comprehensively evaluate patients’ prognosis. Third, since numerous trials already exploiting immunotherapy as neoadjuvant regimen, while patients receiving immunotherapy were all excluded in this study, whether new grading system is applicable to neoadjuvant immunotherapy-treated cohort is in continuing need of further evaluation.
In conclusion, our study preliminarily verified the efficacy of pathological assessment of the residual IPA proportions post-neoadjuvant therapy using IASLC grading system in prognostic stratification. Such grading system could assist or even substitute for pathological response evaluation in survival outcome prediction and clinical management.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221148028 – Supplemental material for The IASLC grading system for invasive pulmonary adenocarcinoma: a potential prognosticator for patients receiving neoadjuvant therapy
Supplemental material, sj-docx-1-tam-10.1177_17588359221148028 for The IASLC grading system for invasive pulmonary adenocarcinoma: a potential prognosticator for patients receiving neoadjuvant therapy by Haoran E, Junqi Wu, Yijiu Ren, Lang Xia, Long Xu, Shaoling Li, Yue Zhao, Chongwu Li, Yunlang She, Chunxia Su, Chunyan Wu, Likun Hou, Deping Zhao and Chang Chen in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank the biostatistician, Prof. Aihong Zhang (Department of Medical Statistics, School of Medicine, Tongji University, Shanghai, 200092, China), for the design and guidance of statistical analysis in this research. Dr Haoran E would particularly like to thank Drs Yifan Zhong and Hai Tang for their consistent encouragements and supports during the past years.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
