Abstract
Background:
The study on skip-N2 metastasis in small-cell lung cancer (SCLC) is lacking. Therefore, this study aimed to explore the prognostic significance of skip-N2 metastasis based on a multicenter cohort.
Methods:
We collected 176 SCLC patients with pathological categories T1-4N1-2M0 from four hospitals in China. Survival curves were drawn through the Kaplan–Meier method and compared by the log-rank test. The Cox regression method was used to calculate the hazard ratio (HR) and 95% confidence interval of the characteristics for cancer-specific survival (CSS). Two propensity-score methods were used to reduce the bias, including the inverse probability of treatment weighting (IPTW) and propensity-score matching (PSM).
Results:
This multicenter database included 64 pN1 patients, 63 non-skip-N2 cases, and 49 skip-N2 cases. Skip-N2 and the non-skip-N2 patients had gap CSS rates (skip-N2 no versus yes: 41.0% versus 62.0% for 1-year CSS, 32.0% versus 46.0% for 2-year CSS, and 20.0% versus 32.0% for 3-year CSS). After PSM, there were 32 pairs of patients to compare survival differences between N2 and skip-N2 diseases, and 34 pairs of patients to compare prognostic gaps between N1 and skip-N2 diseases, respectively. The results of IPTW and PSM both suggested that skip-N2 cases had better survival outcomes than the non-skip-N2 cases (IPTW-adjusted HR = 0.578; PSM-adjusted HR = 0.510; all log-rank p < 0.05). Besides, the above two analytic methods showed no difference in prognoses between pN1 and skip-N2 diseases (all log-rank p > 0.05).
Conclusions:
Skip-N2 patients were confirmed to have a better prognosis than non-skip-N2 patients. Besides, there was no survival difference between pN1 and skip-N2 cases. Therefore, we propose that the next tumor-node-metastasis staging system needs to consider the situation of skip metastasis with lymph nodes in SCLC.
Keywords
Introduction
Lung cancer is still one of the most common malignancies worldwide, which mainly includes two histological types: non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC).1,2 The prognosis for SCLC is unfavorable because this histological type of lung cancer has metastatic diseases at the time of initial diagnosis. 3 SCLC accounts for 10–15% of all lung cancers, and its 5-year survival rate is only about 6%.3,4 The characteristics affecting patient prognoses of SCLC include treatment approaches, tumor-node-metastasis (TNM) descriptor, and peripheral blood markers.5,6 However, the effect of lymph node-related indicators on the postoperative survival of SCLC patients is still unclear. The previous study reported that lymph node ratio might be a more accurate prognostic factor than N classification based on the analysis for the Surveillance, Epidemiology, and End Results database. 7 The database has some unavoidable limitations, such as lacking the exact number of lymph nodes in the N1 stations, although its sample size is large. Therefore, using this database to analyze the phenomenon of lymph node skip metastasis is full of limitations.
The skip-N2 metastasis occurs during the pathological N2 involvement without the involvement of the N1 station; non-skip-N2 metastasis has pathological N2 and N1 involvement. 8 Many previous studies explored the significance of skip-N2 metastasis in resected NSCLCs.9–11 One of those reports confirmed that the prognosis of skip-N2 metastasis was better than non-skip-N2 metastasis and even was similar to the pathological N1 (pN1) classification in NSCLC patients. 9 However, the study on the significance of skip-N2 metastasis in SCLC patients is lacking. Thus, we performed a multicenter study to explore the effect of skip-N2 metastasis on the postoperative prognosis of SCLC patients.
Material and methods
Patient selection
We collected the data from four hospitals in China. A total of 176 SCLC patients with pathological categories T1-4N1-2M0 were included in this study. Those patients received endobronchial ultrasound-guided transbronchial needle aspiration, positron emission tomography-computed tomography, or computed tomography to confirm the N0 disease before surgery. Those eligible patients underwent surgical resection between 2004 and 2018. All patients for main analyses met the following standards: (1) age was over 17 years old; (2) pathologically diagnosed as T1-4N1-2M0 SCLC; (3) underwent lobectomy or pneumonectomy; and (4) the number of examined lymph nodes was over zero. Patients were excluded if they (1) died within 1 month after surgery and (2) received neoadjuvant therapy. Detailed information about patient selection standards is presented in Figure 1. The work has been reported in line with the STROCSS criteria. 12
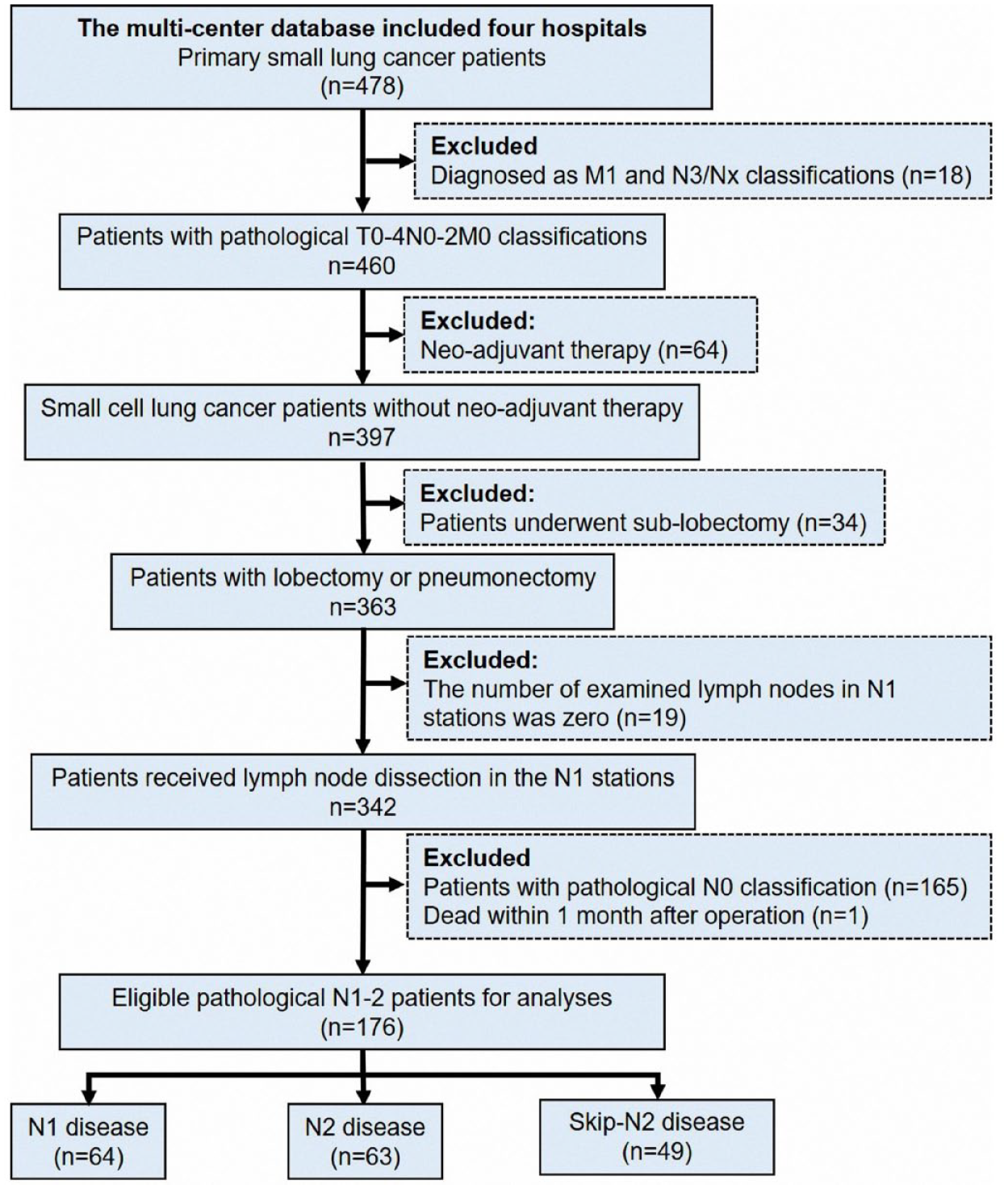
The standards of case selection in this study.
Follow-up information
The follow-up information was updated in March 2022. The median follow-up interval was 35.0 months (from 1 to 118 months). Patients after the operation visited the outpatient clinic at 3- or 6-month intervals for the first 3 years and 12-month intervals after that. We used telephone and outpatient visit records for follow-up updates. The time interval between the diagnosis of the SCLC and the cancer-caused mortality was defined as cancer-specific survival (CSS). Cases were censored at the end of the follow-up. CSS was considered best concerning clinical relevance.
Statistical analyses
The chi-square test and Fisher’s exact test were used to compare differences among groups through the software SPSS 25.0 (IBM SPSS, Inc., Armonk, IL, USA). Survival curves were drawn through the Kaplan–Meier method using ‘survival’ and ‘survminer’ packages in the R 4.1.2 software (https://www.r-project.org/) and compared by the log-rank test. Univariable and multivariable Cox proportional hazards regression models were conducted in the ‘survival’ package of R software to calculate the hazard ratio (HR) and 95% confidence interval (CI) of the characteristics for cancer-specific mortality. A two-sided p < 0.05 was defined as statistically significant.
We used two propensity-score methods to reduce the bias: inverse probability of treatment weighting (IPTW) and propensity-score matching (PSM).13,14 PSM was conducted to reduce potential selection bias. However, the sample size was reduced after PSM. Thus, we added another propensity-score-adjusted analysis, IPTM, to reserve the sample size and further confirm results from PSM. IPTW was performed by ‘RISCA’, ‘tableone’, ‘survey’, ‘MatchIt’, ‘foreign’, and ‘reportReg’ packages using R 4.1.2 software. We defined pN classification as the treatment variable. The treatment probabilities were calculated from a multivariable logistic regression using a set of covariates deemed to have affected baseline differences, including sex, age, combined SCLC, pT classification, smoking history, location, surgical approach, grade, adjuvant chemotherapy, adjuvant radiotherapy, and lymph nodes in N1 stations. A one-to-one PSM with a caliper width of 0.01 was applied to balance between two groups using software SPSS 25.0. The baseline characteristics for PSM included sex, combined SCLC, pT classification, surgical approach, adjuvant chemotherapy, adjuvant radiotherapy, and lymph nodes in N1 stations.
Results
Patient characteristics
The patient’s baseline characteristics are presented in Table 1. In this multicenter database, 64 patients were identified with pN1 disease (36.4%) and other patients with pN2 diseases, including 63 non-skip N2 cases (35.8%) and 49 skip N2 cases (27.8%). Men outnumbered females, constituting 86.9% of the patients. 100 (56.8%) patients were 60 and over, whereas 76 (43.2%) were below 60 years old. The median lymph nodes in N1 stations were 5. Thus, 56.8% and 43.2% of patients were categorized into the lymph nodes in N1 stations ⩽5 and lymph nodes in N1 stations >5, respectively. Over 80% of patients underwent lobectomy, and about 30% of patients did not receive adjuvant chemotherapy.
The baseline characteristics of SCLC patients.

Fisher’s exact test calculated the p values of those variables, and others were calculated by the chi-square test.
CSCLC, combined small cell lung cancer; pN, pathological nodal; pT, pathological tumor; SCLC, small-cell lung cancer.
Survival analyses for pN2 patients
The median survival time was 21.5 months, ranging from 1.0 to 108.0 months. The 1-year, 2-year, and 3-year CSS rates were 56.0%, 47.0%, and 31.0% in this cohort, respectively. We performed Kaplan–Meier methods and Cox regression to compare the prognostic differences between skip-N2 and non-skip-N2 patients. Skip-N2 and the non-skip-N2 patients had gap CSS rates in 1 year, 2 years, and 3 years (skip-N2 no versus yes: 41.0% versus 62.0% for 1-year CSS, 32.0% versus 46.0% for 2-year CSS, and 20.0% versus 32.0% for 3-year CSS). However, the survival curves of the above two categories of patients did not significantly differ (Figure 2(a), log-rank p = 0.26). The univariable Cox regression showed similar results (Table 2, skip-N2 no versus yes: unadjusted HR = 0.754, 95% CI: 0.459–1.239, p = 0.265). Interestingly, after adjusting for other confounders, patients with skip-N2 disease had a better survival benefit than those with the non-skip-N2 disease in the analysis of the multivariable Cox regression (Table 2, skip-N2 no versus yes: adjusted HR = 0.491, 95% CI: 0.275–0.877, p = 0.02). Therefore, two methods of propensity-score analysis were performed. Detailed information about baseline before and after PSM is presented in Table 3. After PSM, there were 32 pairs of patients to compare survival differences between N2 and skip-N2 diseases. The results of IPTW and PSM both suggested that skip-N2 cases had a better trend of survival outcomes than the non-skip-N2 cases (Figure 2(b)–(c), IPTW-adjusted HR = 0.578; PSM-adjusted HR = 0.510; all log-rank p < 0.05). Skip-N2 patients had a more significant improvement than the non-skip-N2 patients in 1-year, 2-year, and 3-year CSS rates after PSM (skip-N2 no versus yes: 42.0% versus 74.0% for 1-year CSS, 30.0% versus 60.0% for 2-year CSS, and 12.0% versus 41.0% for 3-year CSS).

The survival curves of pN2 patients before propensity-score analysis. (a) After the IPTW adjusting. (b) and after PSM (c).
Cox regression analyses for cancer-specific mortality of SCLC patients with pN2 or skip-N2.

CI, confidence interval; HR, hazard ratio; SCLC, small-cell lung cancer.
The baseline characteristics of SCLC patients with PSM.

The p values of those variables were calculated by the Fisher’s exact test, and others were calculated by the chi-square test.
CSCLC, combined small-cell lung cancer; N, nodal; PSM, propensity-score matching; pT, pathological tumor; SCLC, small-cell lung cancer.
Prognostic analyses for pN1-2 cases
In the Kaplan–Meier analysis, the pN1 patients showed more satisfactory outcomes over the pN2 patients (Figure 3(a), log-rank p = 0.01). Moreover, we compared patient prognoses among patients with pN1, non-skip-N2, and skip-N2 diseases, and we found no statistical difference between pN1 and skip-N2 patients (Figure 3(b), log-rank p = 0.13). The univariable Cox regression showed similar results: the unadjusted HR was 1.509 with 95% CI: 0.879–2.592 (Table 3, p = 0.14). However, the multivariable analysis presented that skip-N2 disease had a worse survival than pN1 disease (Table 4, adjusted HR = 1.983, 95% CI: 1.043–3.767, p = 0.04). IPTW and PSM analyses were used to reduce potential bias to confirm those findings further. Detailed information about baseline before and after PSM is presented in Table 3. After PSM, there were 34 pairs of patients to compare survival differences between N1 and skip-N2 diseases. The above two analytic methods showed similar results: there was no difference in prognoses between pN1 and skip-N2 diseases (Figure 3(c)–(d), all log-rank p > 0.05).

The survival curves of pN1-2 patients (a and b). The survival curves between pN1 and skip-N2 patients after the IPTW adjusting (c) and PSM (d).
Cox regression analyses for cancer-specific mortality of SCLC patients with pN1 or Skip-N2.

CI, confidence interval; HR, hazard ratio; SCLC, small-cell lung cancer.
Discussion
To our knowledge, the number of studies on skip metastasis of mediastinal lymph nodes in SCLC patients is lacking. Previous studies mainly collected data from NSCLC patients to explore the significance of skip-N2 metastasis in prognosis.8,15 The proportion of SCLC cases in all lung cancers is only 10.0–15.0%, and amount of SCLC patients who received surgical resection is small.2,16 Therefore, the data to study the significance of skip-N2 metastasis in SCLC patients is valuable. Thus, we performed the multicenter study and collected 176 eligible SCLC patients. After multivariable regression, IPTW, and PSM analyses, skip-N2 patients were confirmed to have better survival than non-skip-N2 patients with SCLC in this study. Besides, pN1 patients were more prone to acquire survival benefits than skip-N2 patients in the multivariable Cox regression analysis. However, there was no prognostic difference between pN1 patients and skip-N2 patients after propensity-score analyses. Those findings may improve the accuracy of the pN category in SCLC patients after surgery.
As in the results obtained in NSCLC, resected SCLCs also have skip metastases of the mediastinal lymph nodes. In the previous studies on NSCLC, some researchers had found that skip-N2 patients had better survival than non-skip-N2 patients.8,10,17 Therefore, it is essential to subdivide the pN2 diseases to improve the accuracy of pN classification. In this study, there was a gap difference between skip-N2 and non-skip-N2 SCLC patients for CSS rates. The possible reason for this phenomenon is that the existing pN classification may not be sufficient to distinguish the prognosis of lung cancer patients. Patients with non-skip N2 are considered to have a higher tumor burden and, therefore it has a worse prognosis. 18 Thus, this situation encountered in clinical practice suggests that further refinement of pN classification is inevitable in the next version of the TNM staging system with lung cancer.
The prognosis of skip-N2 patients is likely to be similar to pN1 patients in SCLC. The study by Wang et al. 9 confirmed that skip-N2 disease had a similar prognostic advantage as pN1 in patients with adenocarcinoma. The abovementioned research, in fact, provided proof that the prognosis of skip-N2 cases was better than it was in non-skip-N2 patients in another view. Although our study explored that the pN1 descriptor was more prone to be a protective prognosticator than skip-N2 classification in the multivariable Cox regression, pN1 cases did not show better survival than skip-N2 patients in other statistical analyses, especially after propensity-score analyses. Accordingly, we propose that pN1 patients with SCLC have similar prognoses to skip-N2 patients, as viewed in NSCLC. Regrettably, as discussed by Li et al., the reason skip-N2 metastasis has a survival similar to that of pN1 disease still needs to be explored. 19
A previous study found that the skip-N2 metastasis might be related to the anatomical location of lymph nodes. It showed that supra-tracheobronchial lymph nodes (level 4) and subcarinal lymph nodes (level 7) were prone to occur the phenomenon of skip metastasis. 19 Some basic studies explored the relationship between tumor microenvironment and lymph node metastasis. For example, the high expression of synaptonemal complex protein 3, long non-coding RNA HOTAIR, and circular RNA 0008594 in primary tumor tissues were confirmed to promote tumor cells to metastasize to lymph nodes.20–22 Another profound study mainly revealed that tumor cells needed to undergo a metabolic shift toward fatty acid oxidation before lymph node metastasis. 23 Several bioactive bile acids accumulated in lymph nodes and could activate the yes-associated protein pathway to regulate lymph node metastasis. 23 Therefore, skip metastasis of lymph nodes may depend on the relation among subtypes of tumor cells, tumor microenvironment, and lymph node microenvironment. Overall, the actual mechanism of skip-N2 metastasis needs to be explored.
This study still has some limitations. First, this study was a retrospective study; although two propensity-score analysis methods were used in the study, selection bias is still inevitable. Second, the study included data from four hospitals; however, the sample size was relatively small. Therefore, we still need to collect more cases of SCLC. Third, we successfully compared prognoses among pN1, skip-N2, and non-skip-N2 patients; however, we could not explore the significance of adjuvant radiotherapy in different pN classifications because of lacking cases. Finally, the skip-N2 category was confirmed to have a better performance in survival than the non-skip-N2 classification, but this does not mean that skip-N2 SCLC patients could avoid adjuvant radiotherapy. We need more studies to confirm our findings.
Conclusions
In SCLC, skip-N2 patients were confirmed to have a better prognosis than non-skip-N2 patients. Besides, there was no survival difference between pN1 and skip-N2 cases. Therefore, we propose that the next TNM staging system needs to consider the situation of skip metastasis with lymph nodes in SCLC.
Footnotes
Acknowledgements
L. L. W. sincerely thanks Prof. T. H. R. for instructing clinical knowledge, surgery, and research in thoracic oncology. L. L. W. also sincerely thanks the international star Mr. Leslie Cheung, whose songs brought him much encouragement to persevere on his research path.
