Abstract
Background:
Oncotype DX (ODX) is a validated assay for the prediction of risk of recurrence and benefit of chemotherapy (CT) in both node negative (N0) and 1–3 positive nodes (N1), hormone receptor positive (HR+), human epidermal growth factor receptor 2-negative (HER2−) early breast cancer (eBC). Due to limited access to genomic assays in Brazil, treatment decisions remain largely driven by traditional clinicopathologic risk factors. ODX has been reported to be cost-effective in different health system, but limited data are available considering the reality of middle-income countries such as Brazil. We aim to evaluate the cost-effectiveness of ODX across strata of clinical risk groups using data from a dataset of patients from Brazilian institutions.
Methods:
Clinicopathologic and ODX information were analyzed for patients with T1–T3, N0–N1, HR+/HER2− eBC who had an ODX performed between 2005 and 2020. Projections of CT indication by clinicopathologic criteria were based on binary clinical risk categorization based on the Adjuvant! Algorithm. The ODX score was correlated with the indication of CT according to TAILORx and RxPONDER data. Two decision-tree models were developed. In the first model, low and high clinical risk patients were included while in the second, only high clinical risk patients were included. The cost for ODX and CT was based on the Brazilian private medicine perspective.
Results:
In all, 645 patients were analyzed; 411 patients (63.7%) had low clinical risk and 234 patients (36.3%) had high clinical risk disease. The ODX indicated low (<11), intermediate (11–25), and high (>25) risk in 119 (18.4%), 415 (64.3%), and 111 (17.2%) patients, respectively. Among 645 patients analyzed in the first model, ODX was effective (5.6% reduction in CT indication) though with an incremental cost of United States Dollar (US$) 2288.87 per patient. Among 234 patients analyzed in the second model (high clinical risk only), ODX led to a 57.7% reduction in CT indication and reduced costs by US$ 4350.66 per patient.
Conclusions:
Our study suggests that ODX is cost-saving for patients with high clinical risk HR+/HER2− eBC and cost-attractive for the overall population in the Brazilian private medicine perspective. Its incorporation into routine practice should be strongly considered by healthcare providers.
Keywords
Introduction
Breast cancer (BC) is now the most common type of cancer among women worldwide and also the leading cause of cancer-related mortality – accounting for an estimated 2261.419 new cases and 684,996 deaths in 2020, respectively. 1 In Brazil, 66,000 new cases of female BC were estimated in 2021, representing almost 30% of all neoplasms. 2 Hormone receptor positive, human epidermal growth factor receptor 2-negative (HR+/HER2–) subtype accounts for 65% and 75% of cases of BC among younger and older women, respectively, of which 70% are expected to be of stage I–II. 3 Therefore, a precise assessment of the risk of recurrence for individual patients with early-stage HR+/HER2– BC is paramount to avoiding both undertreatment and overtreatment.
The Early Breast Cancer Trialists’ Collaborative Group overview showed that, despite approximately 60% of patients having received adjuvant chemotherapy (ACT), only a minority derived meaningful benefit from this treatment.4,5 In addition, traditional clinicopathological features such as age, tumor size, node status, histological grade, ER and PR expression, and HER2 status have not been consistently predictive of benefit from ACT. 6
Studies based on molecular signaling pathways and gene expression signatures (GES) rely on methodologies to determine recurrence risk and have provided prognostic and predictive information beyond that provided by standard clinicopathological features.7,8 The main GES available for prognostication of patients with HR+/HER2– early breast cancer (eBC) are Oncotype DX® (ODX) [Exact Sciences, Madison, Wisconsin, US], MammaPrint® (MMP) [Agendia, Amsterdam, Netherlands], Prosigna® [NanoString Technologies, Seattle, Washington, US], Breast Cancer Index® [Biotheranostics, San Diego, California, US], and EndoPredict® [Myriad Genetics, Salt Lake city, Utah, US]. Only ODX and MMP though have been considered level of evidence I.9,10 ODX is a 21-gene assay that is currently validated for the prediction of risk of recurrence and benefit from ACT in both node negative (N0)1–3 and node-positive (N1) disease. 10
In times of rapidly increasing costs and growing awareness about toxicity and quality of life issues, GES are becoming valuable tools in driving treatment de-escalation strategies. 11 Decision-making impact studies of ODX Recurrence Score (ODXRS) have demonstrated the clinical utility of this test especially in terms of its ability of decreasing the proportion of patients who are recommended ACT by ~20–40%.12–15 However, both decision-making impact and cost-effectiveness studies can be profoundly influenced by local realities. Reassuringly, the clinical utility of ODX has been replicated in our reality by a prospective study conducted in two Brazilian public hospitals, with a reported 63% reduction in ACT indication after ODX test results. 16
Many studies have specifically addressed the cost-effectiveness of these assays, having largely shown that both MMP and ODX are indeed cost-effective. Importantly, these studies were conducted across a wide range of healthcare systems and regions, such as Europe, North America, Latin America, and Asia.17–25
In Brazil, despite the growing evidence supporting the clinical utility of GES in HR+/HER2– eBC, treatment decisions remain largely driven by traditional clinicopathological features. The high cost of these tests remains a significant barrier, and limited data on their cost-effectiveness are currently available. Our aim in this study is to provide further insight into the cost-effectiveness of ODX in the current reality of the Brazilian private health system.
Methods
Objectives of the study
The primary objective of this study was to estimate, retrospectively, the rate of change in the indication of ACT and the cost-effectiveness of ODX testing in patients with HR+/HER2–eBC from the Brazilian private health system. We evaluated the cost-effectiveness of ODX across strata of clinical risk groups using data from a large dataset of patients from various Brazilian institutions.
Study design and population
This is a multicenter, retrospective study developed and conducted by GBECAM (Grupo Brasileiro de Estudos em Cancer de Mama) involving seven cancer centers in Brazil (Hospital Sírio-Libanês in São Paulo and Brasília, Hospital Israelita Albert Einstein in São Paulo, A. C. Camargo Cancer Center in São Paulo, Rede D’Or São Luiz in São Paulo and Rio de Janeiro, Grupo CAM in Salvador, Hospital Beneficiência Portuguesa in São Paulo, and Hospital Moinho de Ventos in Porto Alegre).
Clinicopathological and ODX information were retrospectively collected from a large cohort of patients with T1–T3, N0–N1, HR+/HER2− eBC who underwent an ODX between 2005 and 2020. Cases were retrieved from medical records and the review was undertaken by the co-authors from the participating institutions.
Patient data are presented in an aggregated or non-identifiable manner and no informed consent was deemed necessary by the institutional ethics committee board. The Ethics Committee provided approval.
Model structure
The population was divided into the three genomic risk groups by ODX, as reported in the TAILORx trial: low (<11), intermediate (11–25), and high (>25) recurrence score (RS). 26 Clinical risk of recurrence was defined as low or high based on binary clinical risk categorization based on the Adjuvant! Algorithm as used in Microarray in Node-Negative Disease May Avoid Chemotherapy trial. Clinical risk was defined as low if the tumor was 3 cm in diameter or smaller and had a low histologic grade, 2 cm or smaller and had an intermediate grade, or 1 cm or smaller and had a high grade for node negative patients; and if the tumor was 2 cm in diameter or smaller and had a low histologic grade for node-positive patients. The clinical risk was defined as high if the low-risk criteria were not met.27,28 Patients with micrometastatic nodal involvement were considered as node positive for clinical risk classification as in MIDACT trial, 29 whereas these patients were not included in TAILORx trial and were excluded after protocol amendment in RxPONDER trial.
Two decision-tree models were developed using Microsoft Excel®. In the first model, the overall population (containing low and high clinical risk patients) were entered into the model. In the second model, only high clinical risk patients were included.
In the first model, all patients would undergo ODX, and ACT would be given according to TAILORx26,30 and RxPONDER 31 data as well as ASCO 10 and NCCN 9 guideline recommendations (because menopausal status was not always clearly documented in the charts, we used age ⩽/>50 as a surrogate for pre-/postmenopausal status): patients >50 years with pN0-1, pT1-3, and RS > 25; patients ⩽50 years with pN0, pT1b-2 and either RS 16–20 with high clinical risk or RS > 20; patients ⩽50 years with N1 and any RS. In this model, the scenario considered as default (i.e. usual care comparator) was all women with high clinical risk would be offered ACT.
In the second model, only women with high clinical risk would undergo ODX, and ACT would be given according to the aforementioned criteria.26,30,31 In this model, the scenario considered as default was all women with high clinical risk (100% in this model) would be offered ACT.
Costs
Treatment and the cost of the assay were estimated in January 2022 and were converted from Brazilian Reais (R$) to United States Dollars (US$), based on an exchange rate of R$5.00 = US$1.00. The Brazilian private health costs are variable because they are based on multiple factors such as type of health insurance, geographic region, and the profile of the care provider. All costs plotted in this model derived from Planserv (Assistência a saúde dos servidores públicos estaduais, Bahia, Brazil) and the pricing establishment and regulation agency, CMED (Camara de regulação do mercado de medicamentos) listings which are available for public consultation in Brazil.
The cost of the ODX assay was informed by the Brazilian representative for GenomicHealth (now ExactSciences) US$ 3000.
The costs of CH (US$ 12897.15) were based on regimens with and without anthracyclines according to clinical practice and their approximate proportional distribution in TAILORx and RxPONDER trials: 50% of dose-dense AC-T [4 cycles of doxorubicin/cyclophosphamide (with Pegfilgrastrim) every 2 weeks followed by 4 cycles of paclitaxel every 2 weeks] and 50% of TC × 4 [4 cycles of docetaxel and cyclophosphamide every 3 weeks (with Pegfilgrastrim)]. Dose-dense AC-T was chosen because it is the preferred anthracycline-based regimen for patients with HR+/HER2– eBC by NCCN guidelines version 2.2022. Detailed information on all costs is presented in Supplemental Tables S1–S3. The costs related to adverse event management were not included in this cost-effectiveness analyses because of high hypothetical level of calculation regarding most clinical management cost data were not available. Also, work absenteeism following chemotherapy (CT) and costs associated with disease recurrence were not included in the model.
Uncertainty was addressed using deterministic and probabilistic sensitivity analyses (PSAs).
The estimate of 5-year follow-up visits as well as the monitoring exams and costs of implanted catheter for administration of CT used in the cost-effectiveness analyses are detailed in Supplemental Tables S2–S3.
Sensitivity analyses
We performed deterministic one-way sensitivity and PSAs on the variables within clinically plausible ranges of our baseline characteristics. Deterministic one-way sensitivity analyses explore the impact that individual parameter uncertainty has on the model results. PSAs explore the impact of joint uncertainty of all parameters on the model results.
Both clinical and costs parameters were varied. The former was varied based on 95% confidence intervals (CIs) and the latter were varied at ±20% of the mean (Table 1). The results were presented by Tornado diagram. PSAs were conducted using beta distributions for probability parameters and utility estimates, and gamma distributions for cost estimates. The distributions of input parameters were drawn 1000 times, and acceptability curves were created.
Model parameters.

CT, chemotherapy
Results
Baseline characteristics
A total of 645 patients were included in this study. Their demographic and clinicopathological characteristics are summarized in Table 2. The median age was 54 (range: 24–78 years) and 58.3% were >50 years. Most tumors were T1c (51.5%), N0 (80.5%), and histologic grade 2 (67.1%). Using the modified Adjuvant! Online criteria for clinical risk classification, 411 tumors (63.7%) had low-risk and 234 (36.3%) had high-risk disease.
Baseline characteristics of participants.
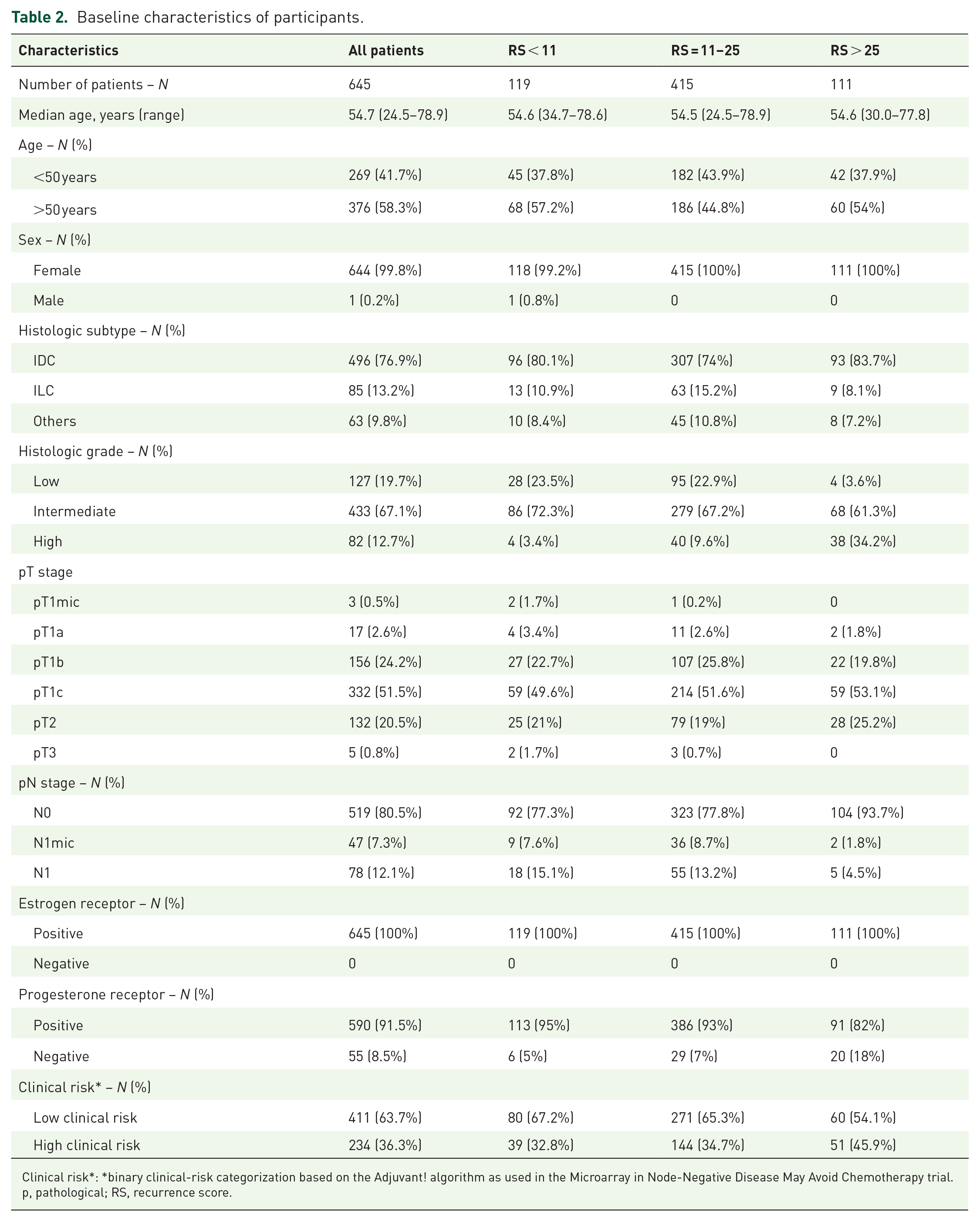
Clinical risk*: *binary clinical-risk categorization based on the Adjuvant! algorithm as used in the Microarray in Node-Negative Disease May Avoid Chemotherapy trial.
p, pathological; RS, recurrence score.
The ODX indicated low (<11), intermediate (11–25), and high (>25) genomic risk in 119 (18.4%), 415 (64.3%), and 111 (17.2%) tumors, respectively.
In the clinical practice 168 patients received ACT: 2 patients in the low genomic risk group (1.6%), 75 patients in the intermediate genomic risk (18%), and 91 patients in the high genomic risk (81.9%).
Clinical impact on ACT recommendation
Of 645 patients from the overall population, 234 (36.3%) had high clinical risk and, therefore, clinical indication of ACT. Of these same 645 patients, only 198 (30.7%) had genomic indication of ACT – corresponding therefore to a potential 5.6% absolute reduction in the use of ACT. Among 411 tumors with low clinical risk, 99 had genomic indication of ACT, corresponding to a potential 24.8% absolute increase in the use of ACT in this group.
Of 234 (36.3%) patients with high clinical risk, only 99 (42.3%) had genomic indication of ACT – corresponding therefore to a potential 57.7% absolute reduction in ACT indication.
Cost-effectiveness analyses in the overall population
Of 645 patients from the overall population, 234 [36.3%; 95% confidence interval (CI): 32.6–40.0%) had high clinical risk and would have ACT indicated as a default. In terms of genomic risk, 119 (18.4%; 95% CI: 15.5–21.4%), 415 (64.3%; 95% CI: 60.6–68.0%) and 111 (17.2%; 95% CI: 14.3–20.1%) had low, intermediate, and high risk, respectively. These cases represent the probability of risk included on decision tree (first model in the Figure 1).

Decision tree in the overall population.
In this model, the scenario considered as usual care comparator was one of all women with high clinical or genomic risk being referred to ACT and all women with low clinical risk (without ODX) not being referred to ACT. Of 534 patients with low and intermediate genomic risk, 87 (16.3% 95% CI: 13.4–19.1%) had indication of ACT (patients ⩽50 years with pN0, pT1b-2, HR+/HER2– and either RS 16–20 with high clinical risk or RS 21 to 25; patients ⩽50 years with N1, HR+/HER2– eBC and any RS ⩽ 25).
As previously mentioned, ODX led to a potential 5.6% absolute reduction in ACT indication in the overall population; however, this was associated with an increase of US$ 2288.87 per patient in the ODX group (Table 3).
Results of economic model comparing cost in the overall population.

CT, chemotherapy; ICER, incremental cost-effectiveness ratio; ODX, Oncotype DX.
In the univariate sensitivity analysis, which allows a reviewer to assess the impact that changes in a certain input (parameter) will have on the output results of an economic evaluation, the most important parameter was the ‘probability of high clinical risk’. Changing the probability of high clinical risk between its lowest and highest values has an impact on the incremental cost-effectiveness ratio (ICER), ranging from US$ 19,544.67 to US$ 147,619.33 (Figure 2).

Tornado diagram summarizing changes in the ICER of genomic stratification (ODX) versus clinical stratification strategies for overall population.
Based on a PSA of 1000 simulated cases, the ODX group was more expensive and more effective than clinical stratification in 100% and 98.5% of simulations, respectively (Figure 3).

ICER scatterplot per CT spared in the overall population.
The cost-effectiveness acceptability curve depicts the likelihood of being cost-effective based on the willingness to pay to avoid one CT (Figure 4).

Cost-effectiveness acceptability curve in the overall population.
Cost-effectiveness analyses in the high clinical risk population
Of 234 (36.3%) patients who had high clinical risk and would as a default be referred to ACT, 51 (21.8%; 95% CI: 18.6–25.0%) had high genomic risk and would therefore retain indication for ACT. Of 183 patients with low/intermediate genomic risk, 48 (26.3%; 95% CI: 22.8–29.6%) would still retain indication of ACT (according to the aforementioned criteria) and 135 patients (57.7%) would avoid unnecessary CT (second model in the Figure 5).

Decision tree in high clinical risk population.
In this model, ODX was effective (57.7% absolute reduction in ACT indication), with cost-savings of US$ 4350.66 per patient (Table 4). According to the univariable analysis, CT costs were the most important variable with ICER ranging from US$ -10,120.58 to US$ -4961.72 (Figure 6). The results were robust in the 1000 probabilistic simulations being the dominant option (less expensive and more effective) in 100% of the simulations (Figure 7).
Results of economic model comparing in the high clinical risk population.
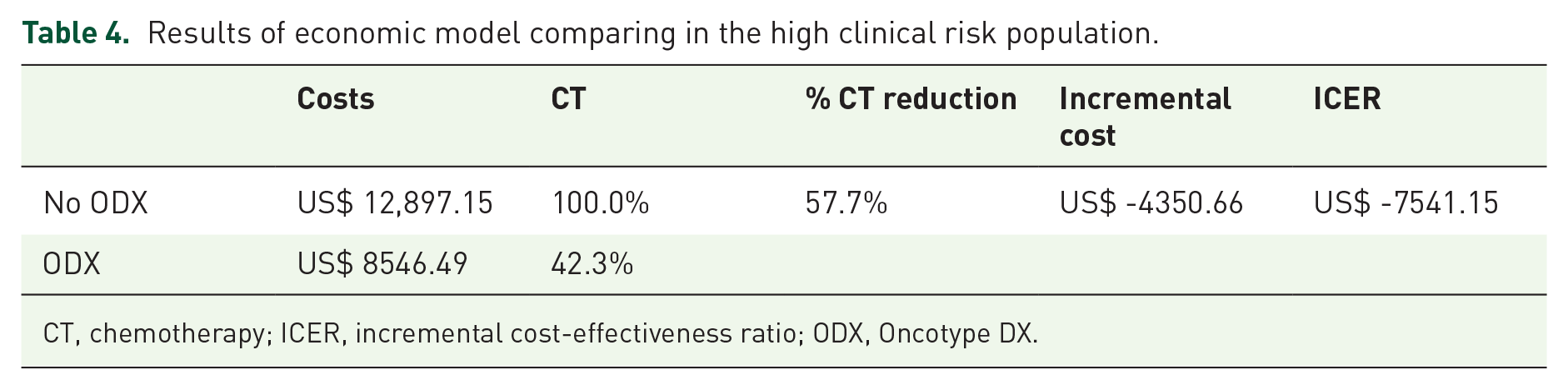
CT, chemotherapy; ICER, incremental cost-effectiveness ratio; ODX, Oncotype DX.

Tornado diagram summarizing changes in the ICER of genomic stratification (ODX) versus clinical stratification strategies for high clinical risk population.

ICER scatterplot per CT spared in the high clinical risk population.
Discussion
To the best of our knowledge, this is the largest study ever conducted in Brazil evaluating the clinical impact and cost-effectiveness of ODX testing in patients from the private health system diagnosed with HR+/HER2– eBC. As main findings of the study, among patients with N0-1, ODX testing is cost-saving for those with a high clinical risk (binary clinical risk categorization based on the Adjuvant! Algorithm). Furthermore, ODX – despite not being found cost-saving in the overall population – appears to be more effective than clinicopathological features in terms of its clinical utility (i.e. 5.6% absolute reduction in ACT). Our study highlights a greater accuracy of ODX for prognostic stratification and to avoid overtreatment in the high clinical risk group (57.7% decrease in ACT indication) and undertreatment in the low clinical risk group (24.8% increase in ACT indication) with a potential improvement in results. In TAILORx trial, 8.9% of patients with low clinical risk had high genomic risk (RS > 25). 30
To estimate the prevalence of genomic risk by ODXRS, population-based studies outweigh randomized controlled trials because of the external validity consideration. 32 In the TAILORx trial, 17%, 69%, and 14% of the patients, respectively, had low (RS < 11), intermediate (RS = 11–25), and high (RS > 25) genomic risk and 70.2% and 29.8% of the patients, respectively, had low and high clinical risk. Among patients with high and low clinical risk, only 27.3% and 8.9%, respectively, had a genomic high risk. 26 Our population-based data (including 19.4% of N1mic/N1 patients) largely mirrors that of TAILORx, with low (RS < 11), intermediate (RS = 11–25), and high (RS > 25) genomic risk found in 18.4%, 64.3%, and 17.2% of the patients, respectively, and 14.5% and 21.8% of the patients classified as low and high clinical risk, respectively, having a RS > 25.
Post hoc analyses of a subset of participants enrolled in two NSABP clinical trials (N0 patients) found that approximately 15% of the low clinical risk group and 45.2% of the high clinical risk group (based on Adjuvant! Algorithm) had a RS ⩾ 31. 33 However, these results contrast substantially with those of the Connecticut Tumor Registry, in which 3% of patients classified as low clinical risk patients and 30.4% of patients classified as high clinical risk (according to the PREDICT model, a risk calculator developed by the National Health Service in the United Kingdom) had a RS ⩾ 31. 34 These data highlight the limitations of clinicopathological features in predicting genomic-grounded risk and benefit of CT.
Despite their high cost, multiple studies – most of them using Adjuvant! Online tool as clinical usual care comparator – have shown that MMP and ODX (level of evidence I for both) are indeed cost-effective in guiding decisions regarding the use of ACT in patients with HR+/HER2– eBC, and the results have been consistent across a wide range of healthcare systems and regions such as the USA, Canada, United Kingdom, and The Netherlands.17,18,35,19,20,36,22 The optimal allocation of ACT and an overall reduction in the use of CT are equally important drivers of the extent of economic benefit.
A recent European systematic review addressed the clinical utility and economic value of four key GES, namely, MMP, ODX, Prosigna, and EndoPredict. A total of 28 studies evaluating the clinical utility of GES was identified (22 of them referring to ODX). Reassuringly, the pooled analysis showed a 45.7% and 32.2% reduction of ACT indication for ODX and MammaPrint, respectively. In all, 40 original economic evaluations were found (32 of them referring to ODX) which largely demonstrated that GES resulted in better patient outcome with a positive impact on QALYs and genomic testing was cost saving in 14 (35%) evaluations and cost increasing in 26 (65%) of the evaluations. Considering the improvement in patient outcomes, GES were found to be cost-effective in 90% of the studies, below the dashed 40,000 euro-per-QALY line. 37 A budget impact analysis of the same four GES in Germany showed that only ODX was associated with reduced costs compared to standard clinical practice in the base case of the healthcare payer and societal perspectives – EUR 296 and EUR 12,656 per woman, respectively. Also, cost-savings, from the societal perspective, were observed in N0 and N+ patients as results of reduction of CT use in both subpopulations. 38
For ODX, multiple studies carried out in several countries have supported the cost-effectiveness of this assay. For these studies, the cost of utilizing ODX was compared with the cost of CT, QALYs, as well as societal costs.18,23,24,25,39,35,40 Most of the cost-effectiveness appears to be primarily attributable to the cost of CT and life-years lost for patients with disease relapse. 40 In our study, we did not perform the cost-effectiveness analysis by QALYs, which are defined as life-year gains multiplied by life-quality; however, we performed an analysis by CT spared. There are some reasons for that: first, even though ODX provides robust prognostic information, it does not ‘produce’ a survival impact – as we see with interventional treatments; second, estimating the impact of not receiving CT on quality of life would rely on dubious assumptions, such as limiting the impact of CT to the disutility of its side effects. The physical and emotional impact of knowing that the risk of relapse is low enough to avoid CT would be difficult to quantify. As a result, we chose a cost-effectiveness analysis based on CT avoided rather than a cost-utility analysis, preserving the rationale of the benefit brought by ODX while avoiding the need for utility assumptions
An Italian prospective, multicenter, real-life study that included patients with HR+/HER2–eBC with clinicopathological ‘intermediate risk’ of recurrence (derived from the St. Gallen Consensus Guidelines combining nuclear grade, tumor size, Ki67, nodal involvement and ER level) demonstrated a substantial reduction of ACT recommendation from 24.6% to 15.2% after ODX testing in the entire population. Also, it showed approximately a 50% rate of sparing ACT for patients initially recommended this treatment. Of interest, changes were more frequent in N1 (18.1% sparing) than in N0 (5.2% sparing). There was also a reduction of the total budget leading to a net saving of EUR 81,017. 41
ODX is currently publicly funded in many countries such as the UK, Ireland, Switzerland, Spain, Israel, Greece, Canada, and Japan (some of them highly restrictive welfare states).42,43 All major insurers in the United States are also expected to cover the cost of the ODX assay. 42
In a previously mentioned study that prospectively investigated the clinical utility of ODX in 179 women with eBC treated at two public hospitals in Brazil showed a 63% reduction of ACT indication – which is in line with data generated in other countries. This study included a lower number of patients younger than 50 years of age than our study (22% versus 41.7%), a higher number of tumors >2 cm (62% versus 21.3%), and of N1 tumors (32% versus 12.1%). 16 A small prospective study from Brazil also suggested that the adoption of ODX in private medicine is cost-effective – with ICER of R$ 92.403,25 (US$ 18.480,65) per CT spared (real-world scenario: 36% of conventional AC-T and 64% of TC × 4). 44
The present analysis has some limitations. First, treatment costs may be underestimated as costs incurred over patients’ lifetime due to short- and long-term toxicities and work absenteeism following CT and recurrence were not included. Second, the paucity of data on costs associated with the management of CT side effects also precluded the inclusion of these information in the model. Therefore, for the cost-effectiveness analysis, our results should be considered rather conservative. Third, because of the very different reality of the Brazilian public system, our results should not be extrapolated to patients treated in the public realm – that is, currently most of the population. Finally, in TAILORx age was used as criteria (⩽50 years and >50 years) while in RxPONDER the menopausal status was used (pre- and postmenopausal) in the stratification. In our retrospective study, since documentation of menopausal status was not reliably documented, we standardized the age criteria for both NO and N+ patients. This approach may have underestimated the CT indication for premenopausal patients >50 years.
On the other hand, our study has several strengths. First, our patient population closely resembles that of the pivotal ODX clinical trials, and our results are consistent with many other clinical utility and cost-effectiveness studies performed in other contexts. Second, our sample size was robust enough to allow for the analyses and should also be considered diversified and inclusive from a geographic and socio-economical perspective.
In conclusion, this study suggests that, in the perspective of the Brazilian private health system, ODX is a dominant strategy (more effective and cost-saving) for patients with N0-1 HR+/HER2– eBC and high clinical risk and a cost-attractive strategy in relation to clinicopathological features in the overall population, avoiding approximately six unnecessary CT treatments per 100 women. Therefore, its incorporation into routine practice should be strongly considered by healthcare providers.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221141760 – Supplemental material for Cost-effectiveness analysis of Oncotype DX from a Brazilian private medicine perspective: a GBECAM multicenter retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359221141760 for Cost-effectiveness analysis of Oncotype DX from a Brazilian private medicine perspective: a GBECAM multicenter retrospective study by Leandro Jonata Carvalho Oliveira, Thais Baccili Cury Megid, Daniela Dornelles Rosa, Carlos Alberto da Silva Magliano, Daniele Xavier Assad, Daniel Fontes Argolo, Solange Moraes Sanches, Laura Testa, José Bines, Rafael Kaliks, Maira Caleffi, Debora de Melo Gagliato, Marina Sahade, Romualdo Barroso-Sousa, Tatiana Strava Corrêa, Andrea Kazumi Shimada, Daniel Negrini Batista, Daniel Musse Gomes, Marcelle Goldner Cesca, Débora Gaudêncio, Larissa Matos Almeida Moura, Julio Antonio Pereira de Araújo, Artur Katz and Max Senna Mano in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
