Abstract
Background:
Non-small-cell lung cancer (NSCLC) with additional nodule(s) located in the same lobe or ipsilateral different lobe were designated as T3 and T4, respectively, which was merely defined by anatomical location of additional nodule(s), regardless of other prognostic factors.
Methods:
A total of 4711 patients with T1-4, N0-2, M0 NSCLC undergoing complete resection were identified between 2009 and 2014, including 145 patients with additional nodule(s) in the same lobe (T3-Add) and 174 patients with additional tumor nodule(s) in ipsilateral different lobe (T4-Add). Overall survival (OS) was compared using multivariable Cox regression models and propensity score matching analysis (PSM).
Results:
T3-Add patients [T3-Add versus T3, hazard ratio (HR), 0.695; 95% confidence interval (CI), 0.528–0.915; p = 0.009] and comparable OS with T2b patients through multivariable Cox analysis, and further validated by PSM. T4-Add patients carried a wide spectrum of prognosis, and the largest diameter of single tumor was screened out as the most effective indicator for distinguishing prognosis. T4-Add (⩽3 cm) patients had better OS than T4 patients [T4-Add (⩽3 cm) versus T4, HR, 0.629; 95% CI, 0.455–0.869; p = 0.005] and comparable OS with T3 patients. And T4-Add (>3 cm) patients had comparable OS with T4 patients.
Conclusion:
NSCLC patients with additional nodule(s) in the same lobe and ipsilateral different lobe (maximum tumor diameter ⩽ 3 cm) should be further validated and considered restaging as T2b and T3 in the forthcoming 9th tumor, node, and metastasis staging system.
Introduction
Precise staging is crucial for predicting prognosis and guiding treatment strategies. In 2016, the American Joint Committee on Cancer (AJCC) and International Association for the Study of Lung Cancer (IASLC) published the 8th edition tumor, node, and metastasis (TNM) staging system.1–3 According to 8th staging system, additional tumor nodules (referred to as intrapulmonary metastasis [IPM] in the pathology community), which was defined as having a classic lung cancer and at least one solid separate lung nodules, located in the same lobe and ipsilateral different lobe were classified as T3 and T4 categories, respectively.2,4 However, the 8th edition classification of T3/T4 non-small-cell lung cancer (NSCLC) with additional nodule (s) remains the same as the 7th edition without any modifications although the classification of other T descriptors has been modified considerably.2,5 The latest classification change of NSCLC with additional nodule(s) was in the 7th edition staging system: tumors with additional nodule(s) in the same or the ipsilateral different lobe were reclassified as T3 and T4 from T4 and M1, respectively.5,6 It was worth noting that the classification of additional nodule(s) was merely based on anatomical location of additional nodule (s) regardless of other factors.2,5 For this potential vulnerability, previous studies had manifested that tumor size and histologic type, which were not proposed as factors in the 8th classification, were associated with prognosis of patients with T4 NSCLC with additional nodule(s) in ipsilateral different lobe.7,8 Indeed, the survival curves also suggested that patients with additional nodule(s) in the same lobe seemed to have a relatively favorable prognosis than other T3 patients in the 8th IASLC staging project. 9 A recent study using National Cancer Data Base (NCDB) also proposed that patients with additional nodule(s) in the same lobe with the primary tumor, which was staged as T3 according to 8th staging system, should be down-staged as T2b. 10 Hence, it is necessary to develop a more precise T classification to describe the tumor burden of T3/T4 NSCLC with additional nodule(s).
In IASLC database, 1122 patients had additional nodule(s) with complete pathologic information between 1999 and 2010, which was used for formulating the classification of additional nodule(s) in 8th TNM staging system, were of high heterogeneity in prognosis over time (5-year overall survival (OS) rates: patients during 1999–2003, 19%; patients during 2004–2006, 43%; and patients during 2007–2010, 71%). 9 The increase in OS over time may result from improvement of adjuvant treatment strategies recent years. Moreover, patients derived from Japan (accounting for 93% Asian patients) were observed to have relatively better prognosis than patients contributed from other regions. 9 Notably, the 2015 IASLC study specifically mentioned the lack of analysis with respect to extra nodules due to a lack of cases and there were few studies concentrating on these patients since the publication of 8th TNM classification. 9
Therefore, we conducted this study to evaluate the accuracy of the current staging and propose a modified classification for additional nodule(s) in the same and ipsilateral different lobe using a large independent NSCLC cohort from East Asia.
Methods and materials
Patients and methods
The Institutional Review Boards of the two hospitals approved this study (IRB NO. K22-263) on behalf of the Surgical Thoracic Alliance of Rising Star group. Between January 2009 and December 2014, we retrospectively evaluated 6955 patients with completely resected NSCLC in our center. Complete resection was defined as combination of negative surgical margin and systematic lymphadenectomy. In all, 653 patients with multiple primary lung cancers (MPLCs), 366 patients with metastasis in contralateral lung or extra-thoracic metastasis, 206 patients receiving neoadjuvant treatment, 387 patients with history of other tumors, and 632 patients with unknown characteristics were excluded (Supplemental Figure 1). Finally, 4711 patients who were pathologically diagnosed with T1-4, N0-2, M0 NSCLC and received complete resection were analyzed in this study, including 319 patients with metastasis in the same lobe (T3-Add, 145 patients) or ipsilateral different lobe (T4-Add, 174 patients). An independent cohort, including 55 T3-Add patients and 83 T4-Add patents, from Chinese Academy of Sciences Ningbo No. 2 Hospital, was used for external validation.
Histological evaluation
All resected specimens were fixed in formalin, and sliced at 5–10 mm intervals for sampling. Multiple lung cancers were evaluated microscopically by conventional hematoxylin and eosin staining. Discrimination of additional nodule(s) from MPLCs was generally based on the Martini–Melamed standard and IASLC criteria.4,9,11 Additional nodule(s) was defined as an independent mass isolated from a primary malignant tumor which histopathological characteristics are the same as those of the primary tumor and lack microscopic features that indicate the primary tumor as previous study. 12 First, multiple foci of lepidic-predominant adenocarcinoma, minimally invasive adenocarcinoma, and adenocarcinoma in situ were not diagnosed with additional nodule(s). Then, histologic type of resected tumors was compared. Tumors with different histologic types, for example a tumor was adenocarcinoma and the other was squamous cell carcinoma (SCC), were evaluated as MPLCs and tumors with similar histologic type were further evaluated. 4 For adenocarcinomas, tumors were evaluated as additional nodule(s) when the major and other histologic subtypes were similar between tumors. Molecular status was also used to distinguish IPM from multiple primary lung adenocarcinomas. Lung adenocarcinomas with at least two same mutations in common and lung adenocarcinomas with one same rare mutation were evaluated as IPM. 13 For multiple SCCs and those adenocarcinomas with similar major histologic subtype but different other histologic subtypes, multiple tumors were evaluated as additional nodule(s) when the cytologic, stromal features, and expression level of tumor transcription factor-1 were similar.4,14
Two experienced pathologists (L.H. and X.X.) distinguished additional nodule(s) and MPLC independently. When the result from one pathologist was not consistent with the other result, a third pathologist (C.W.) would evaluate the slides again and proposed a result agreed by all three pathologists.
Follow-up
OS was defined as the time between date of surgery and the date of death or the last follow-up. Follow-up was conducted through outpatient examinations or telephone calls. Chest computed tomography (CT) scans and abdominal ultrasound/CT were performed on follow-up visits every 6 months within 2 years after operation and annually thereafter. Magnetic resonance imaging and bone scan were annually performed for 5 years or when the patient had signs or symptoms of recurrence.
Statistical analysis
Pearson’s χ2 test and Fisher’s exact test were applied to analyze categorical data, while Student’s t-test and the Mann–Whitney U-test were applied to analyze numerical data. Univariable and multivariable Cox proportional hazards models were used to identify independent prognostic factors for OS. 15 Characteristics with p values less than 0.05 in univariable analyses were selected and then included into multivariable analyses. Besides, propensity score matching (PSM) analyses were conducted to balance patients’ baseline characteristics between different groups. Kaplan–Meier and log-rank tests were conducted to evaluate OS rates. The impact of the largest tumor size was evaluated in binary fashion as follows. For each specific whole centimeter of largest tumor size, survival was incrementally compared between T4-Add patients with greater than that tumor size, to those T4-Add with less than or equal to that tumor size using a cox proportional hazards regression model that adjusted for gender, histology type, and adjuvant therapy. In the derivation of tumor size cutpoint, the curve of hazard ratios (HRs) of each specific whole centimeter of largest tumor size was fitted by locally estimated scatterplot smoothing (LOESS) method, and the tumor size that coincided with the highest HR was chosen as the optimal cutpoint. Statistical analyses were accomplished via R software, version 3.5.3 (http://www.R-project.org) and SPSS, version 23.0 (IBM, Armonk, NY, USA). All analyses were double-tailed. A p value <0.05 was considered statistically significant.
Results
Study cohort
Between January 2009 and December 2014, 4711 patients underwent complete resection and were diagnosed with T1-4, N0-2, M0 NSCLC were selected into study. In all, 1674 (35.5%), 1956 (41.5%), 681 (14.5%), and 400 (8.5%) patients were diagnosed with T1, T2, T3, and T4N0-2M0 NSCLC, respectively. Based on the 8th AJCC TNM staging system 4 , 145 and 174 patients were diagnosed with T3-Add (additional nodule[s] in the same lobe) and T4-Add (additional nodule[s] in the ipsilateral different lobe). The average primary tumor size of T3-Add and T4-Add tumors was 3.19 cm and 3.84 cm, respectively. Among 414 T2b patients, 138 and 276 patients were T2b NSCLC with and without visceral pleural invasion, which tumor size greater than 4 cm but less than or equal to 5 cm, respectively. Among 536 T3 patients, 356, 91, and 89 patients were T3 NSCLC with tumor size greater than 5 cm but less than or equal to 7 cm, invading T3 structures (parietal pleura, chest wall, pericardium, or phrenic nerve) or combination of tumor size and T3 structures, respectively. Among 226 T4 patients, 191, 29, and 6 patients were T4 NSCLC with tumor size greater than 7 cm, invading T4 structures (mediastinal fat, great vessels, trachea, or esophagus), or combination of tumor size and T4 structures, respectively. Median follow-up time was 80.4 months for the entire cohort. Figure 1 shows the OS of patients stratified by T stage regardless of N stage, which results demonstrated that T3-Add patients had a comparable prognosis with T2b patients (T3-Add versus T2b, p = 0.70), and T4-Add patients had an intermediate prognosis between T3 and T4 patients (T4-Add versus T3, p = 0.91; T4-Add versus T4, p = 0.069). Baseline characteristics of T2b-4, N0-2, M0 patients are shown in Table 1. And details of involved sites are shown in Supplemental Table 1.

OS of patients with T1-4, N0-2, M0 NSCLC stratified by T stage in the entire cohort.
Baseline characteristics of T2b-4, N0-2, M0 patients included in the study.

NA, not available; SCC, squamous cell carcinoma; SLR, sublobar resection; SD, standard deviation; p1 indicates difference between T3 and T3-Add patients; p2 indicates difference between T4 and T4-Add patients. Significant p values are in bold form.
Comparison with IASLC staging project
To strengthen the reliability of the results, the numeric results between our study and the IASLC staging project study were compared.2,9 In the current study, the 5-year OS of T2bN0M0, T3N0M0, and T4N0M0 patients were 66.1%, 54.8%, and 45.2%, respectively, which was consistent with results of the 2015 IASLC study of lung cancer staging project (T2bN0M0, 65.0%; T3N0M0, 57.0%; and T4N0M0, 47.0%). The 5-year OS of 56.1% for T3-Add patients in the current study seems to be a little higher than approximately 47% in the IASLC study, 9 but the survival was comparable with 52.9–55.0% in previous studies.7,16,17 The 5-year OS for T4-Add patients was 43.4% in our study, which was comparable to nearly 40% in the IASLC study and 37.2% of Ucvet and his colleagues’ study9,18
Comparison of T3-Add versus T3 and T3-Add versus T2b tumors
Using univariable Cox analysis, T stage of T3, higher age, marriage status of single/divorced/widowed, histology type of others, positive pathologic N status, and adjuvant therapy of none or not available (NA) were found to be associated with poor OS (Table 2). In further multivariable Cox analysis, T stage [T3-Add versus T3, HR, 0.695; 95% confidence interval (CI), 0.528–0.915; p = 0.009] was identified as an independent prognostic factor when T3 as reference. Besides, marriage status (single/divorced/widowed versus married, HR, 1.418; 95% CI, 1.098–1.830, p = 0.007), pathologic N status (positive versus negative, HR, 1.684; 95% CI, 1.361–2.084, p < 0.001), and adjuvant therapy (yes versus none/NA, HR, 0.705; 95% CI, 0.569–0.875, p = 0.001) were also identified as independent prognostic factors for OS. Figure 2(a) demonstrated that patients in T3-Add group had a significantly better prognosis than patients in T3 group (p = 0.006). Clinicopathological characteristics were well balanced through PSM analysis, and the results are manifested in Supplemental Table 2. After PSM, Figure 2(b) shows that patients in T3-Add group still had a significantly better prognosis than patients in T3 group (p = 0.003).
Univariable and multivariable Cox regression analyses of OS for patients with T2-3, N0-2, M0 NSCLC satisfying the criteria for T3-Add versusT3 and T3-Add versus T2b.
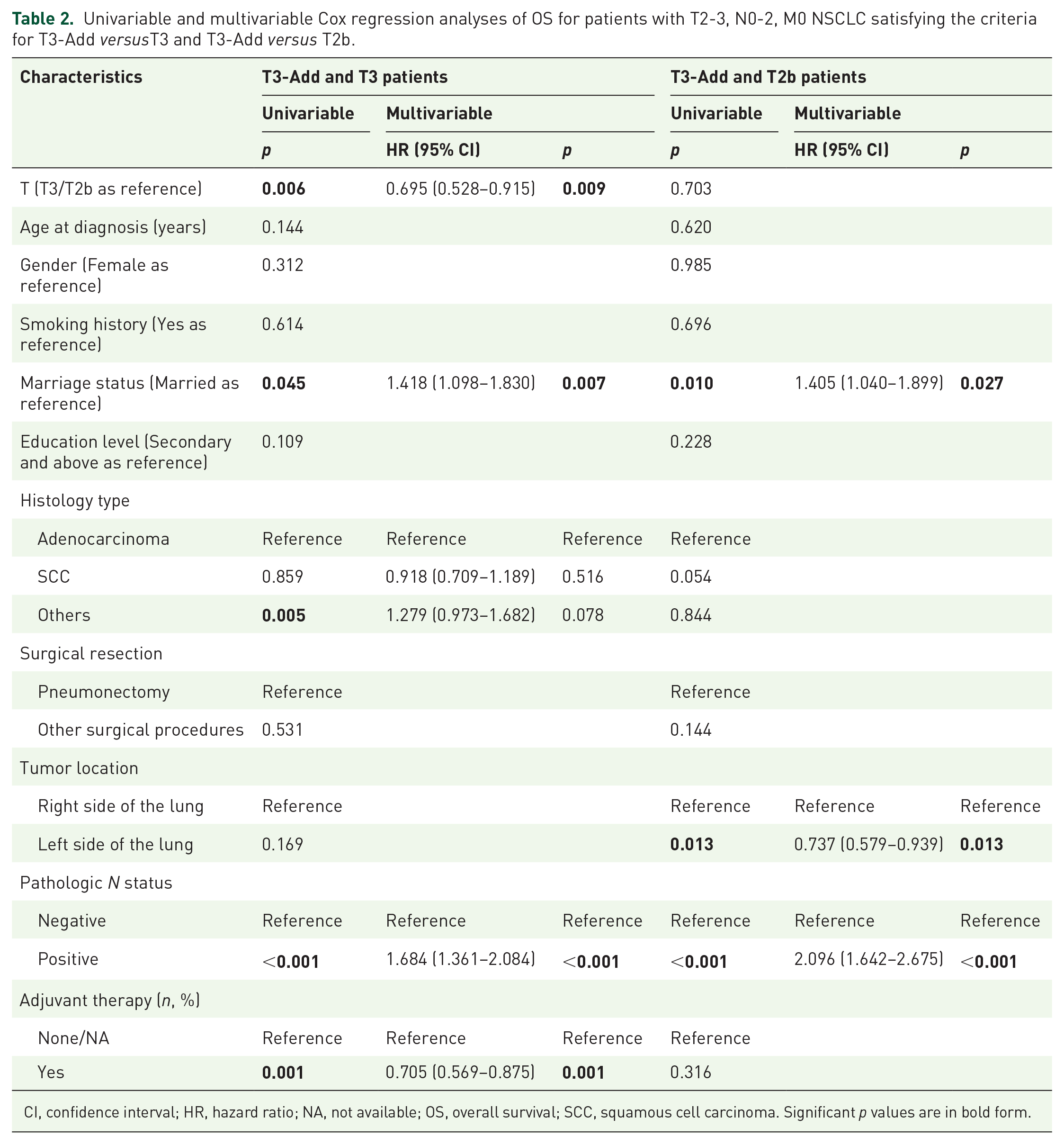
CI, confidence interval; HR, hazard ratio; NA, not available; OS, overall survival; SCC, squamous cell carcinoma. Significant p values are in bold form.

OS of patients with T2b-3, N0-2 M0 NSCLC for T3-Add versus T3 (a and b) and T3-Add versus T2b (c and d) before PSM (a and c) after PSM (b and d).
Then, the prognoses of T2b and T3-Add patients were compared. No significant differences of prognoses between patients in these two groups were identified in survival curves (p = 0.70, Figure 2(c)). Similarly, clinicopathologic characteristics were balanced and shown in the Supplemental Table 2. After PSM, patients in T3-Add and T2b groups still had comparable prognoses (p = 0.75, Figure 2(d)).
Comparison of T4-Add versus T4 and T4-Add versus T3 tumors
In Supplemental Table 3, T stage (T4 versus T4-Add, p = 0.070; T3 versus T4-Add, p = 0.911) was not significantly associated with prognosis in univariable analysis. Also, survival curves showed that T4-Add patients had an intermediate prognosis between T4 and T3 patients (T4-Add versus T4, p = 0.069; T4-Add versus T3, p = 0.91) in Figure 3(a) and (b). Nevertheless, as shown in Figure 1, T3 patients had a significantly favorable prognosis than T4 patients. Therefore, a hypothesis was proposed that some patients of T4-Add may have a comparable prognosis with T3 patients and the other patients may have a comparable prognosis with T4 patients.

OS of patients with T3-4, N0-2 M0 NSCLC for T4-Add versus T4 (a), T4-Add versus T3 (b), T4-Add (⩽3 cm) versus T4 (c and d), T4-Add (⩽3 cm) versus T3 (e and f), and T4-Add (>3 cm) versus T4 (g and h) before PSM (a, b, c, e, and g) after propensity score matching (d, f, and h).
Identification of independent prognostic factors for OS of T4-Add patients
T4-Add patients were reported to have high heterogeneity in prognosis, even though in node negative T4-Add patients, with 5-year survival ranges from 0% to 43%.7,8,19–21 Therefore, to identify the most prognostic factor which can be used to distinguish T4-Add patients with different prognoses, univariable and multivariable Cox analyses was conducted for T4-Add patients and results are shown in Supplemental Table 4. Gender (male versus female, p = 0.029), largest tumor size (as a continuous variable, p = 0.033), and histology type (others versus adenocarcinoma, p = 0.003) were associated with prognosis of T4-Add patients. As illustrated in Supplemental Figure 2, using LOESS method, the highest HR statistic coincided with a pathologic largest tumor size of 3.0 cm in T4-Add patients. Then, T4-Add patients with largest tumor size of a single tumor less than and equal to 3 cm (n = 94) were selected and grouped as T4-Add (⩽3 cm). T4-Add patients with largest tumor size of a single tumor larger than 3 cm (n = 80) were selected and grouped as T4-Add (>3 cm). Univariable analysis manifested that largest tumor size was significantly associated with OS of T4-Add patients (>3 cm versus ⩽ 3 cm, p = 0.004), which was consistent with a previous study. 7
Furthermore, multivariable Cox analysis confirmed largest tumor size of a single tumor (>3 cm versus ⩽ 3 cm, HR, 1.701; 95% CI, 1.166–2.482, p = 0.006), gender (male versus female, HR, 1.483; 95% CI, 1.007–2.184, p = 0.046), and adjuvant therapy (yes versus none/NA, HR, 0.661; 95% CI, 0.444–0.986, p = 0.042) were independent prognostic factors for OS. Notably, compared with patients with adenocarcinoma, patients with SCC had a comparable prognosis (SCC versus adenocarcinoma, HR, 1.012; 95% CI, 0.456–2.248, p = 0.976) and patients with other histologic types had a significantly worse prognosis (others versus adenocarcinoma, HR, 1.691; 95% CI, 1.074–2.661, p = 0.023).
Comparison of T4-Add (⩽3 cm) versus T4 and T4-Add (⩽3 cm) versus T3 tumors
Clinicopathological characteristics of T3, T4, and T4-Add (⩽3 cm) patients were compared and shown in Supplemental Table 5. In comparison with T4 patients, T4-Add (⩽3 cm) patients were more likely to be female (51.1% versus 15.5%, p < 0.001), adenocarcinoma (79.8% versus 26.1%, p < 0.001), lack of smoking history (63.8% versus 38.9%, p < 0.001), and not receive pneumonectomy (3.2% versus 13.7%, p = 0.005). Figure 3(c) demonstrated that T4-Add (⩽3 cm) patients had a significantly better prognosis than T4 patients (p = 0.004). Univariable analyses revealed that T stage [T4-Add (⩽3 cm) versus T4, p = 0.004], pathologic N status (positive versus negative, p = 0.022), and adjuvant therapy (yes versus none, p < 0.001) were associated with prognosis. Furthermore, T stage [T4-Add (⩽3 cm) versus T4, HR, 0.629; 95% CI, 0.455–0.869; p = 0.005] and adjuvant therapy (yes versus none/NA, HR, 0.628; 95% CI, 0.464–0.850; p = 0.003) were identified as independent prognostic factors for prognosis through multivariable analysis in Table 3. Then, clinicopathological characteristics were balanced via PSM analysis and shown in Supplemental Table 6. As shown in Figure 3(d), T4-Add (⩽3 cm) patients still had a significantly better prognosis than T4 patients after PSM (p = 0.008).
Univariable and multivariable Cox regression analyses of OS for patients with T4-Add (⩽3) versus T4 and T4-Add (⩽3) versus T3 tumors.

CI, confidence interval; HR, hazard ratio; OS, overall survival; SCC, squamous cell carcinoma. Significant p values are in bold form.
For T3 and T4-Add (⩽3 cm) patients, Supplemental Table 5 demonstrated that T4-Add patients were more likely to be female (51.1% versus 17.2%, p < 0.001), adenocarcinoma (79.8% versus 25.0%, p < 0.001), be located in right lung (66.0% versus 54.3%, p = 0.036), lack of smoking history (63.8% versus 35.4%, p < 0.001), and not receive pneumonectomy (3.2% versus 12.3%, p = 0.009). Figure 3(e) shows that T3 and T4-Add (⩽3 cm) patients had a comparable prognosis (p = 0.12). Also, Table 3 demonstrated T stage [T4-Add (⩽3 cm) versus T3] was not significantly associated with prognosis of patients with T3 and T4-Add (⩽3 cm) patients in univariable analysis (p = 0.119). Moreover, T4-Add (⩽3 cm) patients had a comparable prognosis with T3 patients after balancing baseline characteristics well (Figure 3(f), p = 0.56).
Besides, prognoses of T4-Add (>3 cm) and T4 patients were evaluated and shown in Figure 3(g) and (h). No significant differences were observed between prognoses of T4-Add (>3 cm) and T4 patients regardless of PSM (before PSM, p = 0.78; after PSM, p = 0.74).
Validation of restaging by subgroups analyses
Several additional subgroup analyses were conducted to further validate the reliability of our results. First, pathologic N stat`us was an important independent prognostic factor identified by multivariable analysis in the present study and previous studies.7,8 Therefore, we conducted PSM analyses for T2b versus T3-Add and T3 versus T4-Add (⩽3 cm) patients stratified by pathologic N status. The proportion of N0 patients were 59.3% (86/145) and 42.0% (73/174) in T3-Add and T4-Add patients, respectively. In T4-add (⩽3 cm) patients, the proportion of N0 patients was 46.8% (44/94) while the proportion of N0 patients was merely 36.3% (29/80) in T4-add (>3 cm) patients. Supplemental Figure 3 demonstrated that patients had a comparable prognosis no matter pathologic N status was negative [T2b versus T3-Add, p = 1.00; T3 versus T4 (⩽3 cm), p = 0.79] or positive [T2b versus T3-Add, p = 0.96; T3 versus T4 (⩽3 cm), p = 0.15] after baseline characteristics were balanced well. Then, subgroup analyses stratified by histology type were also conducted. Supplemental Figure 4 demonstrated that T2b and T3-Add patients had a comparable prognosis with the histology type of adenocarcinoma (Supplemental Figure 4A, p = 0.40), SCC (Supplemental Figure 4B, p = 0.68), and others (Supplemental Figure 4C, p = 0.94) after PSM. Also, T3 and T4-Add (⩽3 cm) patients had a comparable prognosis with the histology type of adenocarcinoma (Supplemental Figure 4D, p = 0.28) and others (Supplemental Figure 4E, p = 0.40) after PSM [subgroup analysis of SCC was not conducted because there were only 2 T4-Add (⩽3 cm) patients with SCC]. Besides, to exclude influence of T3/T4 descriptors (multiple descriptors or possible diagnostic mistake), T3 and T4 patients diagnosed only by tumor size other than T3/T4 descriptors were selected and grouped as T3-Size and T4-Size, respectively. As shown in Supplemental Figure 5, patients also had a comparable prognosis before and after PSM [before PSM: T2b-Size versus T3-Add, p = 0.80; T3-Size versus T4-Add (⩽3 cm), p = 0.21; after PSM: T2b-Size versus T3-Add, p = 0.96; T3-Size versus T4-Add (⩽3 cm), p = 0.44].
External validation
To strengthen the results, an independent cohort from Chinese Academy of Sciences Ningbo No. 2 Hospital was collected and shown in Supplemental Table 6. In the independent cohort, T3-Add patients had a comparable prognosis with T2b patients (p = 0.703) and a better prognosis than T3 patients (p = 0.053), although the difference failed to reach significance due to relatively small sample size (Supplemental Figure 6). Similarly, T4-Add (⩽3 cm) patients had a comparable prognosis with T3 patients (p = 0.948) and a better prognosis than other T4 patients while the difference also failed to reach significance (p = 0.256).
Discussion
In this study, patients with additional nodule(s) in the same lobe (T3-Add) were found to have a significantly better prognosis than other T3 patients and a comparable prognosis with T2b patients. Also, patients with additional nodule(s) in ipsilateral different lobe (T4-Add) with largest tumor size less than or equal to 3 cm were found to have a significantly favorable prognosis than other T4 patients and a comparable prognosis with T3 patients. The above results were validated to be reliable through subgroup analyses stratified by pathologic N status and histology type. Given the results of this study, we proposed that additional nodule(s) in the same and ipsilateral different lobe, which are currently classified as T3 and T4, should be considered for restaging as T2b and T3 in the upcoming 9th edition of the AJCC Staging Manual for NSCLC, respectively.
It was reported that patients with IPM had a significantly unfavorable prognosis than patients with MPLC.14,22–24 In 1975, Martini and Melamed proposed the most practical criteria (MM criteria) used to distinguish additional nodule(s) from MPLC. 11 In 2009, Girard and his colleagues proposed a comprehensive histologic assessment used to distinguish IPM and MPLC, which was concordant with a five-gene mutation panel molecular assessment and reported to predict prognosis better than molecular or MM criteria. 14 In 2016, based on histologic type, major and other histologic subtypes, and clinical status, IASLC proposed a criteria to distinguish MPLC and additional nodule(s).4,25 Usually, additional nodule(s) were defined as an independent mass isolated from a primary malignant tumor which histopathological characteristics are the same as those of the primary tumor and lack microscopic features that indicate the primary tumor. In 2020, based on clinical data and imaging variables, a novel algorithm differentiating between MPLC and additional nodule(s) was proposed by Suh and his colleagues and overall accuracy was approximately 88.9%. 26
In the current study, the 5-year OS for T3-Add patients of 56.1% was similar to previous studies and much better than 42.5% of NCDB database, which may result from that merely 55.8% T3-Add patients in NCDB database received surgery.7,9,10,16,17 It was reported in IASLC database that pathologically confirmed T3-Add patients had a relatively better prognosis than other T3 patients no matter pathologic N status is negative (5-year OS status: T3-Add, 58%; T3 diagnosed with single other T3 descriptor, 53%; T3 diagnosed with multiple other T3 descriptors, 44%) or positive (deaths /number of patients: T3-Add, 409/832, 49.2%; T3 diagnosed with single other T3 descriptor, 1001/1944, 51.5%; T3 diagnosed with multiple other T3 descriptors, 199/351, 56.7%) although the differences did not reach significance. 9 Also, Kumar and his colleagues proved that T3-Add patients had significantly favorable OS rates than T3-size patients who were diagnosed only by tumor size larger than 5 cm but less than or equal to 7 cm (T3-Add, 5-year OS, 37.8%; T3-Size, 5-year OS, 36.3%; p < 0.001) and a comparable prognosis with T2b-size patients (T3-Add, 5-year OS, 53.4%; T2b-Size, 5-year OS, 52.3%; p < 0.30). 10 However, recurrence-free survival was not evaluated and pathological N stage was not used to conduct PSM analysis. In addition, patients with additional nodule(s) in ipsilateral different lobe were not included in this study.
The 5-year OS for T4-Add patients of 43.4% in the present study was comparable to approximately 40.0% of T4-Add patients in several previous studies.9,18,27 Similar to the results of the current study, in IASLC database, T4-Add patients were also reported to have a similar prognosis with all T3 patients, T3 patients diagnosed by descriptors other than additional nodule(s), and T4 patients diagnosed by descriptors other than additional nodule(s) (deaths/number of patients: T4-Add, 51/102, 50%; T3, 1693/3263, 51.8%; T3-other descriptors, 52.3%; and T4-other descriptors, 113/206, 54.8%). 9 However, no further validation of these patients with metastases in ipsilateral different lobe stratified by tumor size or histology type was conducted in IASLC database. In our study, tumor size of 3 cm was identified as an independent prognostic factor for OS and used as a cutoff value for selecting T4-Add patients who should be considered restaging as T3. Also, Okamoto and his colleagues 7 found that tumor size less than and equal to 3 cm was associated with better prognosis of patients with metastases in ipsilateral different lobe but differences failed to reach significance due to relatively small sample size (n = 21; >3 cm versus ⩽ 3 cm, HR, 1.64; 95% CI, 0.94–9.17, p = 0.064).
There were two reasons that may explain why patients with IPM (T3-Add and T4-Add) had significantly better prognosis than other T3 or T4 patients. First, histologic types may result in the difference of survival. The proportions of SCC and other of NSCLC in other T3 and T4 patients (SCC, 49.4% and 46.9%; other histologic types, 25.6% and 27.0%) were significantly higher than that in T3-Add and T4-Add patients (SCC, 19.3% and 5.7%; other histologic types, 17.9% and 18.4%). Patients with SCC and other histologic types of NSCLC were reported to have worse survival than patients with adenocarcinomas.28–30 Second, primary tumor size of T3-Add and T4-Add (T3-Add, 3.19 cm; T4-Add, 3.84 cm) was smaller than other T3 or T4 patients (T3, 5.57 cm; T4, 7.87 cm) and tumor size may determine prognosis more than other variables. Similar result that T3-Add patients had better prognosis than other T3 patients was also observed in Kumar and his colleagues’ research. 10
In addition, in the 2015 IASLC study, the comparison of one versus two or more nodules was limited due to a paucity of patients with two or more nodules. 9 In several previous studies, the number of additional nodules did not show a significant effect on prognosis of patients.8,16,31 Only in Ucvet and his colleagues’ study, T4-add patients with two or more nodules were reported to have a significantly worse prognosis (p = 0.01) but the sample size of this study was relatively low (N = 27). 18 In the current study, there were 25.5% (37/145) and 16.7% (29/174) patients with two or more additional nodules in the T3-Add and T4-Add patients, respectively. And the number of additional nodules did not show a significant effect on prognosis of T3-Add patients (two or more nodules versus one nodule, HR, 1.153, 95% CI, 0.694–1.918, p = 0.582) and T4-Add patients (two or more nodules versus one nodule, HR, 1.230, 95% CI, 0.758–1.997, p = 0.402). Future study containing more patients may provide stronger evidence.
According to the latest National Comprehensive Cancer Network guidelines for NSCLC, surgery was the first choice for additional nodule(s) in the same and ipsilateral different lobe. 32 After surgery, adjuvant chemotherapy or chemoradiotherapy should be offered to these patients based on the pathologic N status and residual tumor classification.33,34 Nevertheless, in clinical practice, we found that patients who were suspected to be additional nodule(s) were less likely to receive surgery as previous studies proposed (resected T3-Add: 55.8–86.6%; resected T4-Add: approximately 54.3%).10,25 These patients were not usually treated as candidates for surgery by doctors even though metastasis was limited in the lung, especially for patients with metastases in ipsilateral different lobe. Indeed, patients with additional nodule(s) did benefit from surgery followed by systemic therapy.32,35,36 Based on these findings, we believed that down-staging patients with additional nodule(s) would be more likely to change the doctors’ consideration, get these patients treated with surgery, and improve prognosis of these patients.
There are several limitations existing in the present study. First, selection bias was inevitable due to nature of retrospective study. Confounding variables, including gender, histology, gene mutation, and so on, may affect survival of patients although multivariable Cox analyses, PSM, and subgroup analyses were conducted to decrease the influence of these confounding variables as much as possible. Second, 4711 patients, only including 319 patients with additional nodule(s), were included in this study although our hospital was a high-volume institution in the world. Third, EGFR-mutated, stage IB to IIIA patients were found to benefit from adjuvant targeted therapy significantly 37 but few patients in our study cohort received adjuvant targeted therapy.
Conclusions
In this large-cohort study, NSCLC patients with additional nodule(s) in the same lobe and ipsilateral different lobe (tumor size ⩽ 3 cm), which were staged as T3 and T4 according to AJCC 8th TNM staging system, were analyzed to have a significantly favorable prognosis than other T3 and T4 patients and comparable survival with T2b and T3 patients, respectively. Based on these results, we proposed that NSCLC patients with additional nodule(s) in the same lobe and ipsilateral different lobe (largest tumor size ⩽ 3 cm) should be further validated and considered restaging as T2b and T3 in the forthcoming 9th TNM classification, respectively. (Supplemental Figure 7)
Supplemental Material
sj-docx-1-tam-10.1177_17588359221130502 – Supplemental material for Reconsidering T component of cancer staging for T3/T4 non-small-cell lung cancer with additional nodule
Supplemental material, sj-docx-1-tam-10.1177_17588359221130502 for Reconsidering T component of cancer staging for T3/T4 non-small-cell lung cancer with additional nodule by Fang Wang, Hang Su, Haoran E, Likun Hou, Minglei Yang, Long Xu, Jiani Gao, Mengmeng Zhao, Junqi Wu, Jiajun Deng, Xiaofeng Xie, Yifan Zhong, Yingze Li, Tingting Wang, Dong Xie, Chunyan Wu and Chang Chen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
