Abstract
Purpose:
Dabrafenib and trametinib combination therapy (dab + tram) is indicated to treat BRAF V600 mutation–positive unresectable/metastatic melanoma and as adjuvant treatment for resected stage III disease. Dab + tram–related pyrexia may require early therapy discontinuation. A modified Delphi panel was conducted to develop consensus on the optimal management of dab + tram–related pyrexia in patients with melanoma.
Methods:
In all, 10 UK oncologists experienced in melanoma management participated in a three-round modified Delphi study (Round 1: one-to-one interview; Rounds 2 and 3: email survey). In each round, participants rated the extent of their agreement with statements about defining and managing dab + tram–related pyrexia. Consensus was defined as >80% agreement for critical management (CM) and >60% for non-critical management (NCM) statements.
Results:
All 10 participants completed Round 1; 9 completed Rounds 2 and 3. Consensus was reached on 42/66 statements (20 CM and 22 NCM). Drug-related pyrexia was agreed as being strictly an elevation of body temperature, although other symptoms may be present (89% agreement). Panelists agreed on the need for simple and generic guidance on dab + tram–related pyrexia management that does not differentiate between patient groups (100%), and that management of first and second dab + tram–related pyrexia episodes should be the same regardless of treatment intent (100%). Regarding CM, participants agreed that both dab and tram should be interrupted for pyrexia (100%) without considering the use of steroids (89%); patients on dab + tram presenting to non-oncology services with pyrexia should be directed to an oncology-specific service as soon as possible and assessed for infection (100%). NCM statements on steroid use following dab + tram interruption and when to restart dab + tram did not reach consensus.
Conclusions:
These consensus statements provide a framework on optimal management of dab + tram–related pyrexia in patients with melanoma which should inform future guidelines.
Introduction
The development and use of immune and targeted therapies have changed the way melanomas are treated, with durable survival benefits in select groups of patients. 1 The goal of treatment is to prolong survival with minimal impairment of quality of life. Therefore, drug-related adverse events require prompt and effective management to ensure that patients derive optimal benefit from therapy. 2
Dabrafenib (TAFINLAR®, Novartis Pharmaceuticals Corporation) plus trametinib (MEKINIST®, Novartis Pharmaceuticals Corporation) combination therapy (dab + tram) is an anti-BRAF/MEK–targeted therapy indicated for the treatment of adult patients with unresectable or metastatic melanoma with a BRAF V600 mutation and for the adjuvant treatment of adult patients with stage III melanoma with a BRAF V600 mutation, following complete resection.3,4 In patients with metastatic melanoma treated with dab + tram, durable and long-term survival was seen, with approximately one-third of clinical trial patients remaining alive at 5 years. 5 However, pyrexia symptoms commonly appear during treatment, with 51–59% of clinical trial patients experiencing pyrexia.5–7 Patients receiving treatment with dab + tram who experience pyrexia may require treatment modification and permanent therapy discontinuation.6,7
Pyrexia resolves in most patients receiving dab + tram following treatment interruption and usually is not an indicator of possible neutropenic sepsis. 6 To optimize patient care and support healthcare providers (HCPs), consensus on the management of pyrexia is needed, particularly for HCPs outside an oncology-specific service. To develop consensus on the optimal management of dab + tram–related pyrexia in patients with melanoma, a modified Delphi panel was conducted. A Delphi panel allows for the systematic collection and aggregation of informed judgments from experts and is an iterative technique characterized by repeated rounds of controlled feedback to achieve consensus. In healthcare research, Delphi panels are widely used and are proven to be a rigorous and feasible way to obtain consensus. 8
Methods
Study design
The Delphi method allows for anonymous, expert input that aims to achieve consensus through three rounds of controlled feedback. The traditional Delphi method starts with an open-ended set of questions, allowing all panel members complete freedom in their responses. As a result, the number of items requiring consideration can be extremely high, leading to very lengthy second-round questionnaires, which can discourage panel members from participating and lead to difficulties in sustaining experts’ interest in the study. 8 The modified Delphi technique substitutes the initial open-ended round of statements of the classical Delphi method with input from a steering committee (SC) of therapy area experts to develop the first draft of consensus statements. These statements are further expanded and refined during three rounds of feedback with a panel of experts. 8
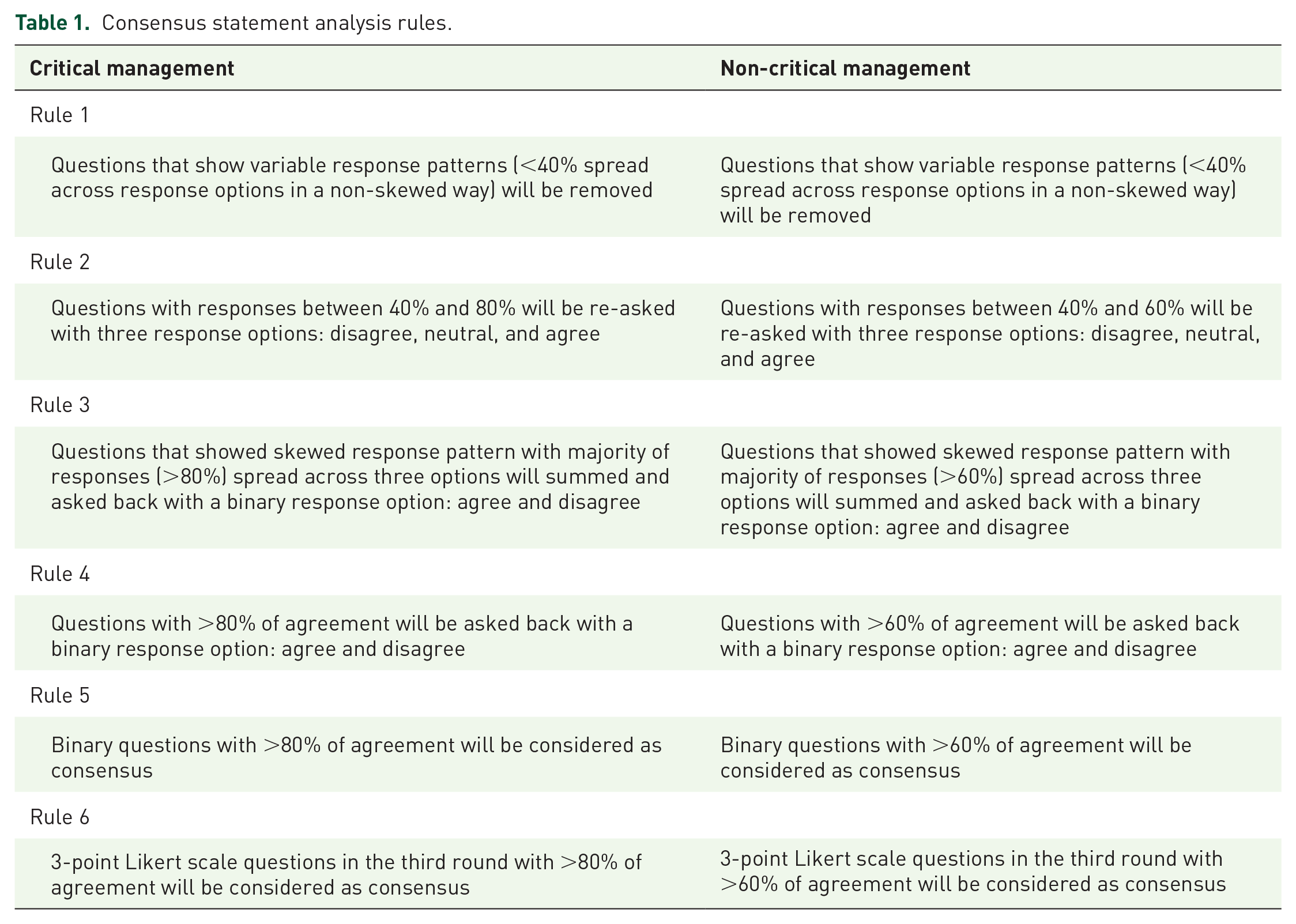
In this modified Delphi panel, HCPs specializing in oncology in the National Health Service (NHS) from the United Kingdom (UK) were invited to provide input on consensus statements regarding the optimal management of dab + tram–associated pyrexia in patients with melanoma. This selection of panelists reflects individuals who provide daily management of melanoma patients in the UK. Four clinical experts were asked to form the Delphi panel SC. As members of the SC, these experts outlined the Delphi panel statement framework (including eligibility criteria for Delphi panelists and number of panelists), set the analysis rules for critical and non-critical management consensus statements (Table 1), and invited additional experts to participate as panelists. All panelists provided written consent to participate in the Delphi panel and complete the survey.
Consensus statement analysis rules.
According to the governance arrangements for research ethics committees, studies conducted with NHS staff who are recruited by virtue of their professional role are exempt from research ethics committee review in the UK; therefore, no ethics committee reviewed this study. 9
Critical management statements were classified as statements that are directly related to pyrexia management and considered critical in the management of patients with pyrexia; non-critical management statements were those that are not considered critical or essential for the management of patients with drug-related pyrexia. Consensus statements were developed and shared with the panelists in three rounds of review.
Survey development
The surveys administered to the Delphi panelists aimed to develop consensus on four main topics: (1) to define the characteristics, signs and symptoms, and clinical features of drug-related pyrexia; (2) to define patient populations requiring different approaches to pyrexia management; (3) to define pyrexia management strategies, outcomes, and resolution; and (4) to define the continuation of treatment after successful management of drug-related pyrexia. The surveys also aimed to obtain feedback on general questions related to treatment and/or management guidelines.
Within each of the four topics, questions addressed critical and non-critical management of drug-related pyrexia. During each survey round, panelists were asked to rate the extent to which they agreed with each question using Likert scales or binary (disagree/agree) responses (Table 2).
Consensus statement response options.

During the first round, panelists participated in one-to-one telephone interviews to complete a paper-based survey. Statements were both quantitative (Likert scales) and qualitative (open-ended/free response) to allow panelists to provide response rationales. The first-round answers were analyzed qualitatively (content analysis) and quantitatively (mean, mode, median, interquartile range, and frequency). For the second round, the qualitative results from the first round were used to refine statements and to develop new ones to be asked during the second-round survey. The statements for the second round were reviewed and approved by the SC. Questions for the second and third rounds were quantitative, and the results were analyzed according to the analysis rules; binary responses or 3- or 5-point Likert scales were selected by the SC depending on the level of consensus achieved for the same statement in the prior survey round. The second and third (final) rounds of the Delphi panel were conducted via PDF survey, which was sent by email to panelists. After each round, the SC shared the results with panelists, including the panelist’s previous answer and the mean, median, mode, interquartile range, and frequency of answers for each consensus statement.
Data analysis
The SC agreed that critical management statements required consensus (i.e. panelists answered the same way) of ⩾80% of panelists and that non-critical management statements required consensus of ⩾60%. Survey responses were extracted for each question into a Microsoft Excel database, and responses were assigned a score (i.e. 1–5, 1–3, or 1–2) corresponding to each response scale.
Levels of dispersion (interquartile range), central tendencies (mean, median, and mode), and percentage frequencies of responses to each statement were calculated between each round for Likert scales. Frequencies were calculated for binary options. Qualitative data from the panelists’ interviews were collated to analyze the content. The qualitative content analysis focused on similar opinions and terms of reference inductively identified. All comments and answers from the panelists were addressed either to refine existing statements or to create new statements for the subsequent rounds.
Panelist selection
Purposive sampling was used to recruit panelists who are especially knowledgeable about the subject of interest, as recommended for qualitative and mixed-methods methodologies. 10 The SC agreed to recruit 10 panelists; it was decided that this sample size would be sufficient for reasonable geographic consensus. During the first SC meeting, members compiled a list of potential clinicians to invite as panelists. Clinicians were selected to participate using the following pre-specified inclusion criteria: practicing oncologist in the UK, experience managing patients with melanoma, and an interest in research of drug-related pyrexia.
Results
In all, 10 practicing oncologists in the UK participated as panelists in the three-round modified Delphi panel. In all, 66 statements were evaluated by the panel over three rounds of surveys. All 10 panelists completed Round 1, and 9 panelists completed Rounds 2 and 3 of the study.
A total of 42 statements reached consensus (Figure 1), 36 of which reached positive consensus, meaning that panelists agreed on the statement in question. Negative consensus, where the panelists disagreed with the statement, was reached for six statements. Of the 42 statements that reached consensus, 20 concern critical management, and the remaining 22 regard non-critical pyrexia management issues. Critical management statements achieving consensus were related to two of the four main topics, with most statements related to pyrexia management strategies.

Delphi consensus statements by review round.
Topic 1: Defining drug-related pyrexia
Several critical management statements were identified for topics related to defining drug-related pyrexia (Table 3). The expert panel agreed on ‘drug-related pyrexia’ as a term that could be used to refer to the type of pyrexia associated as a side effect of medical treatment (100% agreement) and recommended that patients presenting with pyrexia be assessed for infection before receiving antibiotics (100% agreement). The panel also agreed (89% agreement) with a statement defining pyrexia as strictly an elevation of body temperature, and that other symptoms such as chills, rigors, night sweats, flu-like symptoms, and/or hypotension are not critical to the definition of drug-related pyrexia.
Statements reaching consensus on topic 1: Defining drug-related pyrexia.

Topic 2: Patient populations requiring different approaches to pyrexia management
The Delphi panel did not define any patient populations that would require a different approach to pyrexia management. The panelists reached consensus on three non-critical management statements (Table 4). The panelists unanimously agreed that treatment of first and second dab + tram–related pyrexia episodes should be the same regardless of treatment intent. The panelists agreed that the common terminology criteria (CTC) grading of pyrexia is not particularly relevant to the overall management of dab + tram–related pyrexia (78% agreement) and that management of the first or second episode of dab + tram–related pyrexia in the adjuvant setting should consider the impact on the patient’s quality of life and their willingness to accept side effects (89% agreement).
Statements reaching consensus on topic 2: Patient population group(s) requiring different approaches to pyrexia management.

Topic 3: Pyrexia management strategies, outcomes, and resolution
In all, 12 statements regarding definitions of pyrexia management strategies, outcomes, and management resolution achieved positive consensus; three statements reached negative consensus (Table 5). The panel reached 100% agreement on multiple statements regarding critical management. First, they agreed that for patients receiving dab + tram in the adjuvant or metastatic setting, it is preferable to interrupt both drugs rather than just one of the therapies. Second, patients receiving dab + tram who present to a non-oncology service should be put in contact with oncology staff as soon as possible, along with assessing for infection. Third, if patients receiving dab + tram present to a non-oncology service with pyrexia and the oncology-specific service is not available, the course of action is to interrupt dab + tram, assess for infection, and communicate with oncology service as soon as possible. Fourth, if patients are feeling worse after 24 h of having to interrupt dab + tram following an episode of drug-related pyrexia, the patients should be advised to contact their oncology triage service or clinical nurse specialist as soon as possible. Fifth, if patients receiving dab + tram experience a second episode of drug-related pyrexia, they should be advised to interrupt dab + tram, take regular paracetamol and/or ibuprofen, and expect the symptoms to resolve within 24–72 h. Finally, the panel unanimously agreed that for patients receiving dab + tram who experience pyrexia, management with antibiotics before an infection has been confirmed could be an option if neutropenic sepsis is suspected. In addition, 89% of the panelists agreed with the following statements: For patients receiving dab + tram in the adjuvant setting who experience severe drug-related pyrexia, it is preferable to interrupt dab + tram without considering the use of steroids. The role of steroids in the management of drug-related pyrexia remains controversial, and among the expert group there was some difference of opinion. Furthermore, a review of the literature reveals that other consensus documents have included the use of steroids as a management strategy. 11 For patients receiving dab + tram in the metastatic setting who are not tolerating their current dose, it is advisable to consider switching to an alternative regimen after two or more treatment interruptions. Finally, for patients receiving dab + tram in the adjuvant setting who are not tolerating their current dose, it is advisable to consider suspending the treatment after two treatment interruptions with dose reductions.
Statements reaching consensus on topic 3: Pyrexia management strategies, outcomes, and defining resolution.

GP, general practitioner.
Three critical management statements achieved negative consensus (89% disagreement for all statements). The panelists disagreed with statements that dab + tram should not be suspended until drug-related pyrexia has been confirmed, or that it is preferable to interrupt only dabrafenib in patients experiencing dab + tram–related pyrexia in either the adjuvant or metastatic setting.
Regarding non-critical management statements, there was 100% agreement among panel members that if patients receiving dab + tram experience other side effects in addition to drug-related pyrexia, the duration of interruption of dab + tram should be affected. It was also agreed that treatment guidance for drug-related pyrexia management in patients receiving dab + tram should be simple and generic.
Topic 4: Continuation of treatment after successful management of drug-related pyrexia
The panelists unanimously disagreed (100% disagreement) with the statement that restarting dab + tram at full dose after a first interruption for drug-related pyrexia requires the patient to be an inpatient (Table 6). Among other statements, Delphi panelists did not reach consensus on statements on steroid use following dab + tram interruption and the continuation of dab + tram treatment after discontinuation for pyrexia (Supplemental Table 1). As described earlier, there remains debate and a degree of equipoise among the clinical community on the role steroids may play in the management of drug-related pyrexia.
Statements reaching consensus on topic 4: Continuation of treatment after successful management.
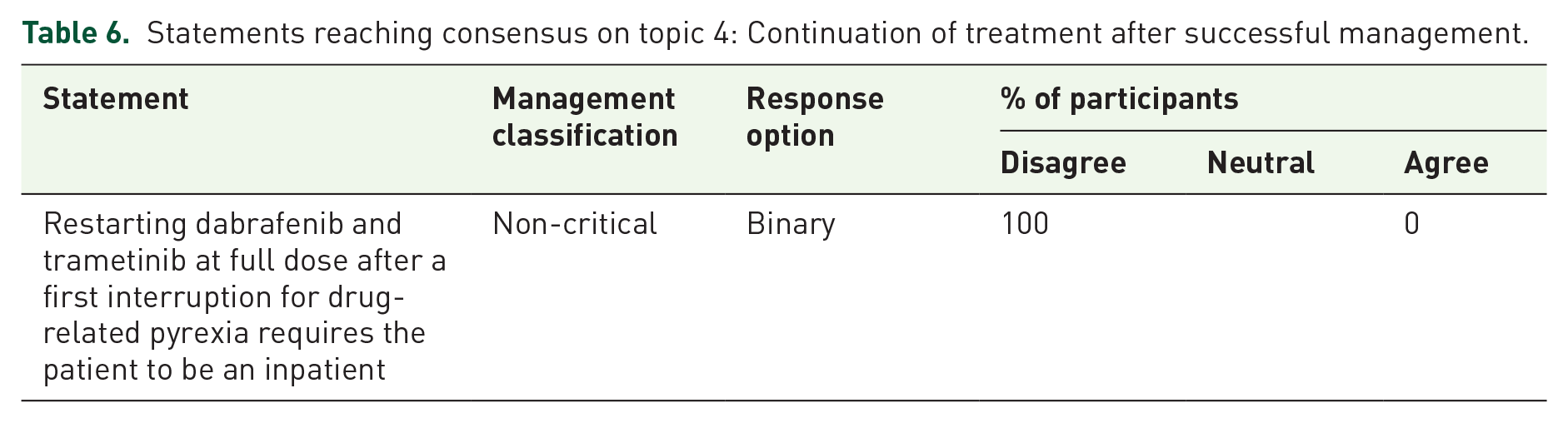
No statements reached a positive consensus related to the continuation of treatment after successful management of drug-related pyrexia.
General questions on guidelines
Most non-critical management statements that achieved consensus were related to treatment guidelines (Table 7). The panel agreed (89% agreement) that patients would be able to self-manage drug-related pyrexia episodes if provided with appropriate guidance and information. The panel unanimously agreed that if oncology services are not available, the following statement should be included as patient-held information for HCPs about the appropriate management of dab + tram–related pyrexia: ‘I am on targeted treatment for my melanoma. This is not a chemotherapy. Two out of 3 patients receiving this treatment will develop a non-infective (drug-related) fever. My risk of neutropenia is very low (X %); please ensure I have interrupted my dabrafenib and trametinib and then assess me for any evidence of infection as any other patient presenting with a fever’. Finally, the panel of experts agreed that an alert card (100% agreement), a phone app (100% agreement), or a keyring (100% agreement) would be appropriate for displaying patient-held information for HCPs about the management of pyrexia associated with dab + tram treatment.
Statements reaching consensus for general questions on guidelines.

AE, adverse event; GP, general practitioner.
Discussion
Results from several studies show that dab + tram offers the possibility of durable and long-term efficacy in patients with advanced, BRAF v600-mutant metastatic melanoma.5,12,13 However, drug-related pyrexia can occur during dab + tram treatment. Results from a combined analysis of adverse event data from the COMBI-d and COMBI-v studies show that 6% of patients treated with dab + tram experienced grade ⩾3 pyrexia and 4% permanently discontinued therapy. 5 These results highlight the need for the prompt and effective management of pyrexia in patients treated with dab + tram.
As described above, we convened a modified Delphi panel to gain consensus on the optimal management of dab + tram–related pyrexia in patients with melanoma.
In terms of translation to real-life clinical practice, the expert panel agreed that the term ‘drug-related pyrexia’ could be used to refer to the type of pyrexia that is a side effect of medical treatment. Although ‘pyrexia’ is strictly an elevation of body temperature, patients who are receiving dab + tram presenting with this side effect may exhibit a pyrexia prodrome that includes symptoms not critical to the strict definition of pyrexia, including chills, rigors, night sweats, and flu-like symptoms. Patients presenting with drug-related pyrexia could, but may not always, look sweaty and tired/fatigued in their presentation. The panel noted that it was important for all HCPs who are involved in managing toxicities in melanoma patients to understand that pyrexia in patients treated with dab + tram may be related to the treatment itself and usually resolves upon interruption of drug treatment and is not usually an indicator of possible neutropenic sepsis.
Consensus on the management of pyrexia in patients on dab + tram was reached, including on the use of antibiotic therapy, treatment of multiple drug-related pyrexia episodes, and the role of treatment interruption and use of alternative treatment regimens. In particular, it was agreed that both dab and tram should be interrupted rather than one of the drugs alone. This is important, as there appears to be a difference of practice among oncology professionals.
The Delphi panel did not define any patient populations that would require a different approach to pyrexia management. The panel noted that patients receiving dab + tram who present to a non-oncology service should be discussed with the oncology service as soon as possible and be assessed for infection.
Recommendations for patients experiencing a second episode of drug-related pyrexia include treatment interruption and use of paracetamol and/or ibuprofen. Upon restarting dab + tram therapy at full dose following treatment interruption, the panel did not recommend hospital admission.
The panel did not reach consensus on statements regarding specific dosages and use of steroids for the management of drug-related pyrexia.
Lastly, the panel agreed that guidelines for the management of drug-related pyrexia could be beneficial for several healthcare services including oncologists, oncology triage services, acute physicians/general medics/adverse event services, and oncologist registrars.
Results from this modified Delphi panel align with the adapted pyrexia adverse event management algorithm used in the COMBI-i and COMBI-APlus studies. In both the COMBI-i study, which enrolled patients with metastatic melanoma receiving dab + tram versus dab + tram + immunotherapy, and the COMBI-APlus study, which enrolled patients receiving dab + tram in the adjuvant setting, an adapted pyrexia adverse event management algorithm was utilized which promptly interrupted dab + tram treatment at the onset of pyrexia (temperature ⩾ 38°C) or the associated prodrome (COMBI-i only; chills, rigors, night sweats, or influenza-like symptoms). In these studies, patients who remained symptom free for 24 or more hours could restart dab + tram at the same dose level. Overall, 52.7% and 67.8% of patients receiving dab + tram experienced pyrexia in COMBI-i and COMBI-APlus, respectively, with the rate of grade ⩾3 pyrexia 3.0% in COMBI-i and the rate of grade 3/4 pyrexia 3.8% in COMBI-APlus. Hospitalizations and treatment discontinuations due to pyrexia were 5.3% and 1.5% in COMBI-i and 4.3% and 2.4% in COMBI-APlus, suggesting that implementation of an adapted pyrexia treatment algorithm is effective in reducing severe pyrexia outcomes, such as high-grade pyrexia, hospitalization due to pyrexia, and discontinuation of treatment early due to pyrexia, while the overall safety profile and early efficacy data in COMBI-APlus appear consistent with those previously observed.12,14
As the number of treatments available to physicians increases for patients with melanoma, a clear understanding of the risks and benefits of therapy should be discussed with patients as the goal of therapy is to prolong survival without negatively affecting quality of life. Through the use of a modified Delphi panel, consensus was obtained on methods of managing drug-related pyrexia in patients with melanoma. In addition, this panel endorsed the use of simple treatment recommendations that were similar across adjuvant and metastatic patient populations. Outcomes from this Delphi panel should be used as a framework to aid in the development of guidelines that maximize the clinical benefits of dab + tram treatment and minimize the effects of drug-related pyrexia by providing clear strategies for its prompt identification and management.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221127681 – Supplemental material for Delphi panel for consensus on the optimal management of dabrafenib plus trametinib-related pyrexia in patients with melanoma
Supplemental material, sj-docx-1-tam-10.1177_17588359221127681 for Delphi panel for consensus on the optimal management of dabrafenib plus trametinib-related pyrexia in patients with melanoma by Ricky Frazer, Avinash Gupta, Christopher Herbert, Miranda Payne, Sergio Diaz-Mendoza, Sally-Anne Vincent and Elena Kovaleva in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank the Delphi panelists for their contribution to this work, including Dr Pippa Corrie, Dr Helen Winter, Dr Guy Faust, Dr Toby Talbot, Dr Nicholas Coupe, Dr Patricio Serra-Bellver, Dr Sarah Westwell, Dr Shobha Silva, Dr Satish Kumar, and Dr Heather Shaw. Medical writing and editorial support were provided by Beth Lesher, PharmD, BCPS, and Catherine Mirvis, BA, of OPEN Health (Bethesda, MD) and funded by the study sponsor. Analysis and review support were provided by Laura Baldock and Navdeep Sahota of OPEN Health (Marlow, UK).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
