Abstract
Metastatic vulvar melanoma is a rare and aggressive disease and survival is usually poor. Vulvar melanomas harbor BRAF V600 mutations only infrequently; consequently, target therapy is a rare therapeutic option and immunotherapy usually has only a weak effect. On the other hand, KIT mutations are rare in cutaneous melanomas, but relatively frequent in mucosal melanomas, particularly in vulvar-vaginal melanomas, and can be a therapeutic target. Herein, we report a clinical case of a patient with metastatic vulvar melanoma, harboring an exon 17 c-KIT mutation, treated with avapritinib (BLU-285) – a highly potent and selective oral kinase inhibitor designed to treat imatinib-resistant gastro-intestinal stromal tumors (GIST) by targeting KIT/PDGFRα activation loop mutants (exons 17/18). After failure of the combination of ipilimumab + nivolumab first and then nivolumab alone, the patient received avapritinib 300 mg/daily for central nervous system (CNS), lymph-nodal, right adrenal gland, lung, and subcutaneous metastases. Best response was partial remission, according to RECIST 1.1 criteria. Time to treatment progression was 11 months. Main toxicities were grade 2 cutaneous vasculitis that required avapritinib discontinuation, and grade 2 uveitis of unknown origin, treated by vitrectomy and empiric antibiotic and antiviral therapy due to negative cultural tests. Uveitis was detected at the time of progression and therapy was definitively discontinued. In conclusion, avapritinib proved to be effective even in the presence of a pretreated disease, a high tumor burden, and brain metastases. In our experience, treatment was feasible and toxicity manageable. Considering the lack of effective therapies and the poor outcome of the disease, determination of c-KIT mutations should be performed routinely in cases of metastatic mucosal melanoma.
Introduction
Vulvar melanoma is a rare disease, with an incidence of 1.3/1,000,000 persons per year, 1 and is the most frequent mucosal melanoma in the Swedish and Dutch populations.2,3
Vulvar melanoma can arise from the hairy or from the glabrous part of the vulva. The significance of tumor location, although controversial, 4 could account for the particular behavior and the molecular characteristics of this disease that make it a distinct entity from cutaneous and other mucosal melanomas. 5
In the metastatic disease, survival is usually poor, due either to the aggressive biological behavior or to the lack of efficacy of novel therapeutic strategies. Vulvar melanoma only seldomly presents BRAF V600 mutations (3–26% of cases),4,5 and, consequently, target therapy with BRAF and MEK inhibitors is a rare therapeutic option. Immunotherapy with anti-PD1 antibodies seems to have a weak effect on mucosal melanoma, with a median progression-free survival (PFS) and overall survival (OS) of 3.9 and 12.4 months, respectively. 6 A combination of anti PD-1 and anti CTLA4 antibodies seems to be more effective, with a median PFS of 5.9 months and an overall response rate (ORR) of 37.1%, despite the high rate of grade 3–4 toxicity (40%). 7 Interestingly, there is another possible pathway to target mucosal melanoma through mutations occurring in the c-KIT gene.
KIT is a class III receptor tyrosine kinase (RTK) that has a crucial role in sustaining growth and maintenance of cells and tissues. KIT is expressed by hematopoietic progenitor and stem cells,8,9 mast cells, 10 melanocytes, 11 and primordial germ cells. 12 KIT-activating mutations are involved in the pathogenesis of several cancers, such as most of the mastocytoses, 13 mast cell leukemias, 14 gastro-intestinal stromal tumors (GISTs), 15 and melanomas. 16 c-KIT activation is followed by receptor auto-phosphorylation and binding of substrate and adaptor proteins. Downstream molecules then drive the transcription of genes involved in influencing the tumor mechanisms of proliferation, survival, adhesion, invasion, metastasis, and angiogenesis. The most frequent c-KIT mutations involve the juxta-membrane (JM) zone of the receptor (exons 9–11) and are usually point mutations.
In GISTs, the presence of exons 9–11 c-KIT mutations is usually associated with the response to imatinib, an inhibitor of tyrosine-kinases like KIT, PDGFRA, ABL, and Fms-like tyrosine kinase-3 (FLT3), and of colony stimulating factor-1 receptor (CSF1R). 17 On the other hand, some acquired KIT mutations are responsible for imatinib resistance, such as exon 17–18 mutations, which activate the loop domain of the receptor. 18
KIT alterations, such as mutations or amplifications, are rare in cutaneous melanoma, but relatively frequent in mucosal melanoma, particularly in vulvar–vaginal melanoma, where they are present in 20–44% of analyzed specimens.16,19
In several phase II trials, imatinib proved effective in patients with metastatic melanoma mostly harboring JM KIT point mutations, with an ORR (complete + partial remission) ranging from 5 to 29%.20,21 The benefit of imatinib seems to be limited in the presence of only KIT amplifications,20,21 or mutations of the loop domain of the receptor. Both alterations represent mechanisms of resistance to treatment and occur very rarely in melanoma. 5
Avapritinib (BLU-285) is a highly potent and selective oral kinase inhibitor, designed to treat imatinib-resistant GISTs by targeting KIT/PDGFRα activation loop mutants (exons 17/18). Avapritinib was demonstrated to be effective in GIST and in systemic mastocytosis with a good toxicity profile.22,23 There are several phase II–III clinical trials currently going on with avapritinib on GISTs and myeloid malignancies. The United States Food and Drug Administration (FDA) recently approved avapritinib for the treatment of unresectable or metastatic PDGFRA exon 18 mutant GISTs.
Case description
Here, we report the clinical case of a patient with metastatic vulvar melanoma, harboring an exon 17 c-KIT mutation, treated with avapritinib, following disease progression after two lines of treatment. The patient’s consent for publication was obtained in accordance with local regulations and laws, and is archived in the patient’s hospital record.
This 47-years-old patient came to our attention with stage pT4bN2b vulvar melanoma arising from the mucosa of the right labium minor, after wedge resection of the vulva and right inguinal-iliac-obturator lymph-nodes dissection, performed in December 2015. In September 2016, the patient presented with vulvar recurrence, with lymph-nodal and lung metastases. 17 A p.N822K c-KIT mutation was detected by next-generation sequencing (NGS) on vulvar recurrence. KIT was not amplified, while NRAS and BRAF were wild type. In November 2016, the patient started ipilimumab and nivolumab combination immunotherapy and discontinued it after four cycles due to severe toxicity (G4 hyperglycemia with development of type I diabetes mellitus). Best response was stable disease, lasting about 10 months. Progression was observed in November 2017, because of the onset of central nervous system (CNS), lymph nodal, right adrenal gland, lung, and subcutaneous metastases. The patient received nivolumab in combination with cyber-knife radiotherapy on three brain metastases. After radiotherapy, she experienced hemiparesis with seizures due to hemorrhage and radio-necrosis of the irradiated lesions. All symptoms regressed after steroid administration. Despite treatment, after 4 months of therapy, the patient had a further progression due to CNS, right breast, lymph nodal, right adrenal gland, lung, gastric, gallbladder, and subcutaneous metastases. At that time performance status (PS) was 1 [Eastern Cooperative Oncology Group (ECOG)] and lactate dehydrogenase (LDH) 635 international units (IU)/L (UPN 220 IU/L).
In June 2018, the patient started avapritinib 300 mg/daily. Assessment of response was performed by means of computed tomography (CT) scan, every 8 weeks of therapy, according to RECIST criteria 1.1. Best response was partial remission at the 16th week, which was already evident at the 8th week (Figure 1). LDH normalized after 8 weeks. Site by site responses according to RECIST 1.1 criteria are reported in Table 1. Reduction in tumor burden was evident in all metastases, including CNS metastasis, where the patient only achieved stable disease. She maintained her partial response for 11 months. Afterwards, CT scan showed CNS, liver, and adrenal gland disease progression.

CT scan at baseline, after 8 and 16 weeks focused on right adrenal gland (A), subcutaneous/lymphnodal (B) and CNS (C) metastases.
Pattern of response according to RECIST 1.1 criteria after 12 weeks of therapy.
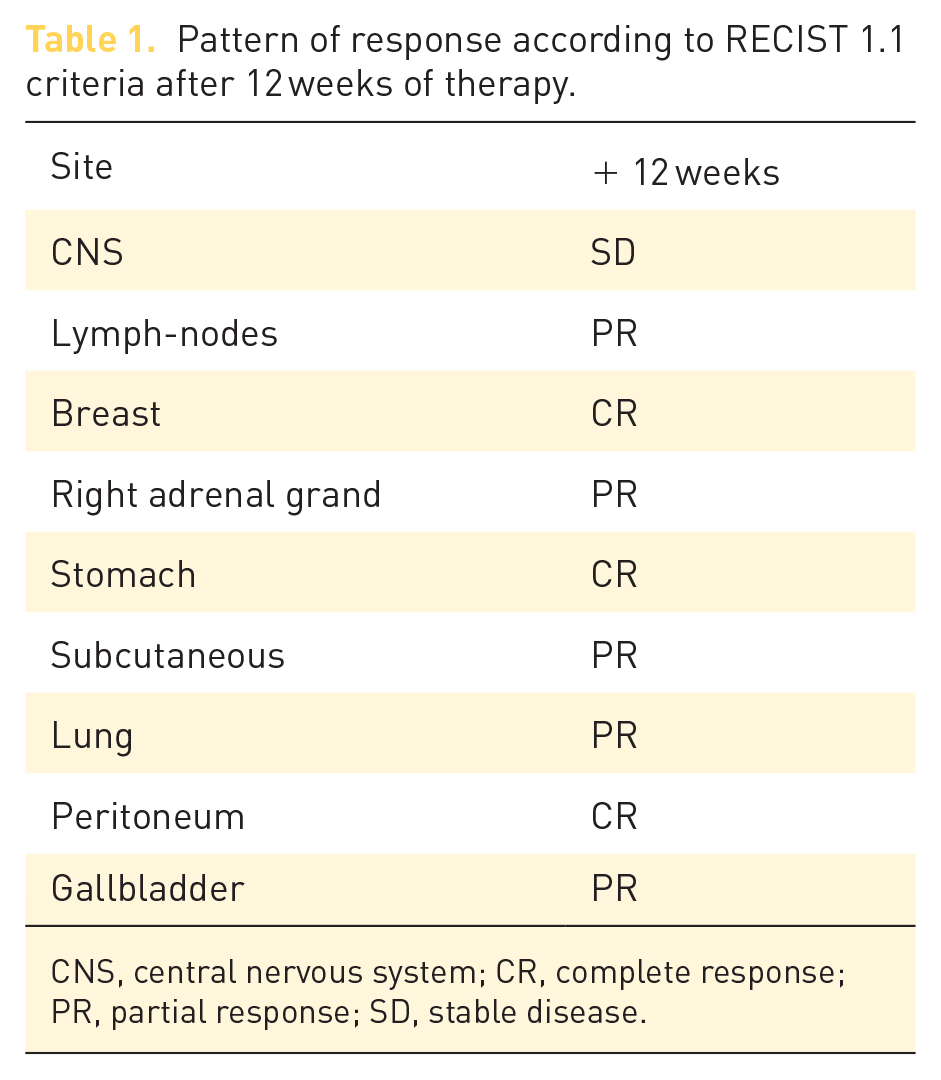
CNS, central nervous system; CR, complete response; PR, partial response; SD, stable disease.
Major adverse events were grade 2 cutaneous vasculitis that required avapritinib discontinuation for 3 weeks and high dose steroid administration, and grade 2 uveitis of unknown origin, treated by vitrectomy and empiric antibiotic and antiviral therapy due to negative cultural tests. Uveitis was detected at the time of progression and therapy was definitively discontinued. The patient died after 4 months from further melanoma progression.
Discussion
The role of KIT as an oncogene and the role of its driven mutations is well known.
GISTs were the first solid tumor in which KIT was targeted by imatinib, which is the standard therapy in both adjuvant and advanced settings. In GISTs, imatinib is usually effective in the presence of exon 9–11 c-KIT mutations, while imatinib resistance is often associated with acquired gene mutations, such as V654A, D820Y, N822K, and A829P.18,24
Imatinib showed activity also in patients with melanoma harboring c-KIT mutations, while c-KIT amplifications seem to select imatinib-refractory patients. 21
c-KIT N822K mutation is infrequent in melanoma, is often a primary mutation and may predict imatinib resistance. Few data are available on the activity of imatinib in this subgroup of patients. Carvajal et al. described a phase II study in which one patient with metastatic melanoma harboring the c-KIT N822K mutation achieved stable disease during imatinib treatment. 25 Thus, the presence of exon 17 mutations activating the loop domain of the receptor provides a strong rationale in giving avapritinib also in metastatic melanoma in the presence of these uncommon mutations.
To our knowledge, this is the first patient who received avapritinib for metastatic melanoma. The clinical benefit was evident in terms of disease control, in terms of the quality and duration of the response obtained. Time to treatment progression was 11 months, which is reminiscent of the median PFS observed for BRAF and MEK inhibitor combinations in patients with metastatic melanoma harboring BRAFV600 mutation.
The main toxicities recorded were cutaneous vasculitis and uveitis, which should be very rare with avapritinib, 26 and more frequent with immunotherapy. 27 Thus, we speculate on the role of previous immunotherapies in the appearance of these more commonly considered immune-related adverse events. Avapritinib proved effective even in the presence of a pretreated disease, a high tumor burden, and CNS metastases. In our experience, treatment was feasible and toxicity manageable. The activity of avapritinib should be evaluated prospectively in patients with metastatic melanoma harboring exon 17–18 KIT mutations. Considering the lack of effective therapies and the poor prognosis of the disease, the determination of c-KIT mutations should be performed routinely in the presence of metastatic mucosal melanoma to explore all therapeutic options in this subgroup of patients.
Footnotes
Acknowledgements
Thanks go to Lucia Stavolone for English language support.
Conflict of interest statement
Cocorocchio E.: paid consultant for Roche, Novartis, BMS Ferrucci PF: paid consultant for Roche, Novartis, BMS, Amgen, MSD.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
