Abstract
Background:
In metastatic urothelial cancer (mUC), bone metastasis (BM) are associated with significant morbidity and mortality, yet their role as an independent prognostic variable remains unclear. We aimed to determine the impact of BM on overall survival (OS) in patients with mUC treated with first-line platinum-based chemotherapy (PBC).
Methods:
mUC patients receiving PBC at the Princess Margaret Cancer Center, Tom Baker Cancer Center, or Cross Cancer Institute from January 2005 to January 2018 were identified retrospectively using central pharmacy database records. Patient disease, treatment, and response characteristics were collected. Progression-free survival (PFS) and OS were estimated using the Kaplan–Meier method. Variables reaching significance (p < 0.05) in univariable analysis (UVA) of survival (OS) were included in multivariable analysis (MVA) (Cox).
Results:
Overall, 376 patients with a median follow-up of 16.8 (range: 2.2–218.3) months were included. Median age was 67 (range: 28–91) years, 76% were male, 63% had an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0–1, and 41% had BM. All patients received first-line PBC. Patients with BM had inferior median PFS (4.9 months (95% CI 3.6–6.2) versus 6.5 months (95% CI 5.4–7.6), p = 0.03) and median OS (8.8 months (95% CI 7.8–9.7) versus 10.8 months (95% CI 9.1–12.5), p = 0.002). In UVA, ECOG PS 2–3 (p < 0.001), presence of BM (p = 0.002), and WBC count ⩾ 11,000 cells/mm3 (p = 0.001) were associated with inferior survival. Prior cystectomy (p < 0.001) and lack of progression (stable disease, partial or complete response) on treatment was associated with improved OS (p < 0.001). These variables maintained significance in MVA.
Conclusion:
In this retrospective study, mUC patients with BM had worse OS suggesting that BM may be an independent negative prognostic factor and including BM as a stratification factor in future mUC clinical trial designs may be warranted. A greater focus must be placed on novel therapeutic strategies to better manage BM to reduce both morbidity and mortality.
Introduction
Urothelial carcinoma is a principal contributor to global cancer-related mortality, 1 accounting for 160,000 deaths annually. 2 At first diagnosis, 70% of cases are non-muscle-invasive, 25% have evidence of muscle invasion, and 5% present with de novo metastatic disease. Nearly half of the cases with muscle-invasive disease will develop incurable metastatic disease with a low 5-year survival of ~5%. 3 Platinum-based chemotherapy (PBC) is a mainstay in the first-line treatment of metastatic urothelial carcinoma (mUC), extending median overall survival (OS) by 3 months.4,5 The addition of maintenance avelumab for patients without disease progression on first-line PBC has further improved survival in the first-line setting. 6 Several studies by Galsky et al., 7 Apolo et al., 8 and Bajorin et al. 9 have assessed how clinical variables affect survival in mUC and developed nomograms to predict patient outcome. Although these nomograms were developed in the pre-immune checkpoint inhibitor (ICI) era, clinical factors, such as Eastern Cooperative Oncology Group (ECOG) performance status (PS); laboratory values, such as hemoglobin, white blood cell (WBC) count, and albumin level; number of metastatic sites (1, 2, ⩾3 sites); visceral metastasis (lung, liver, and bone metastasis (BM)); and response to first-line PBC were shown to be prognostic.7 –9 Reflecting on the trials used to develop these nomograms, BM have generally been included in the definition of visceral metastasis along with lung and liver disease, which has limited the ability to evaluate their independent prognostic impact. 10 Data from studies in second-line mUC also suggest that the location of metastases may impact survival. 11
In mUC, BM occur in up to 47% of patients and in approximately 15% of patients, is the only site of metastatic disease. 12 Despite significant morbidity and mortality, BM and their optimal management have been understudied in mUC. The role of bisphosphonates in mUC has been evaluated in a small prospective trial showing decreased incidence of skeletal-related events (SRE) with improved 1-year survival; 13 however, larger trials are needed to better understand their role in mUC. Radiotherapy is commonly used for symptom control in patients with BM from mUC with recent evaluation in the oligometastatic setting; 14 yet its prognostic effect remains inconclusive.
In this multi-institutional retrospective study, we aimed to investigate the impact of BM on survival in patients with mUC receiving first-line PBC
Materials and methods
Study design and data collection
Patients receiving first-line PBC between January 2005 and January 2018 at the Princess Margaret Cancer Center (PMCC) in Toronto, Ontario, or Tom Baker Cancer Center (TBCC), or Cross Cancer Institute (CCI), in Alberta, Canada were identified retrospectively through central pharmacy chemotherapy databases and electronic medical records. Patients with incomplete records, lost to follow-up, or concomitant cancers were excluded from the chart review. Patients with indeterminant bone lesions on conventional CT imaging were not considered to have BM. Data collection was performed by genitourinary medical oncology fellows at all participating centers within a common password-protected excel spreadsheet. Patient and tumor characteristics, including age, gender, ECOG PS, laboratory parameters, and metastatic sites at diagnosis, were collected. Treatment details (chemotherapy regimen, prior radical surgery, and palliative radiotherapy) were also collected. Individual patient radiology reports (CT and/or bone scans) at baseline to determine metastatic location, and serially to determine response, were evaluated to first-line PBC. This study was approved by the PMCC Research Ethics Board (REB) and the Health and Research Ethics Board of Alberta. Informed consent was exempted for this retrospective study through REB review.
Statistical analysis and study endpoints
Clinical variables were tabulated using descriptive statistics. Cohorts were compared using chi-square/Fisher’s exact test for categorical variables and Student’s t-test for continuous variables compared to categorical variables.
The primary endpoint of the study was the impact of BM on OS. OS was estimated from the date of initial diagnosis of mUC to the date of death from any cause. The secondary endpoint was the impact of BM on progression-free survival (PFS). PFS was estimated from the date of initial diagnosis of mUC to the date of disease progression or death. PFS and OS were analyzed using the Kaplan–Meier (KM) method with log-rank analyses for BM versus non-BM. Univariate analysis (UVA) for individual clinical variables followed by multivarible analysis (MVA) using Cox-proportional hazard regression evaluated association between clinical variables and OS. We applied a backward selection algorithm where variables with a p value less than 0.05 in UVA stayed in the model and were included in the MVA. To avoid dependent variable interaction, the number of metastatic sites (1, 2, ⩾3 sites) were excluded from the MVA to evaluate the prognostic significance of organ-specific (e.g. BM) metastatic involvement. For all statistical tests, a p value of less than 0.05 was considered significant. Analyses were performed using SPSS® software v24.0 (IBM, New York, NY) and PRISM v9.1.0 (GraphPad, San Diego, CA).
Results
Patient demographics
In total, 640 patients with urothelial carcinoma were identified from PMCC (n = 333) and TBCC or CCI (n = 307) (Figure 1). Overall, 376 mUC patients receiving first-line PBC were included in the final analyses. The median age was 67.4 (range: 28–91) years of which 75.5% were male and 62.8% had an ECOG PS of 0–1, 24.0% had elevated baseline WBC, 40.9% had BM, and 59.1% had no BM (Table 1). A majority of patients received first-line gemcitabine and cisplatin (GC) chemotherapy (60.1%), with 36.7% receiving gemcitabine and carboplatin and 3.2% having other PBC. Overall, 43.0% had prior radical cystectomy, and 46.0% received palliative radiation therapy to any lesion (bone, lymph node, bladder) for pain or symptomatic management.
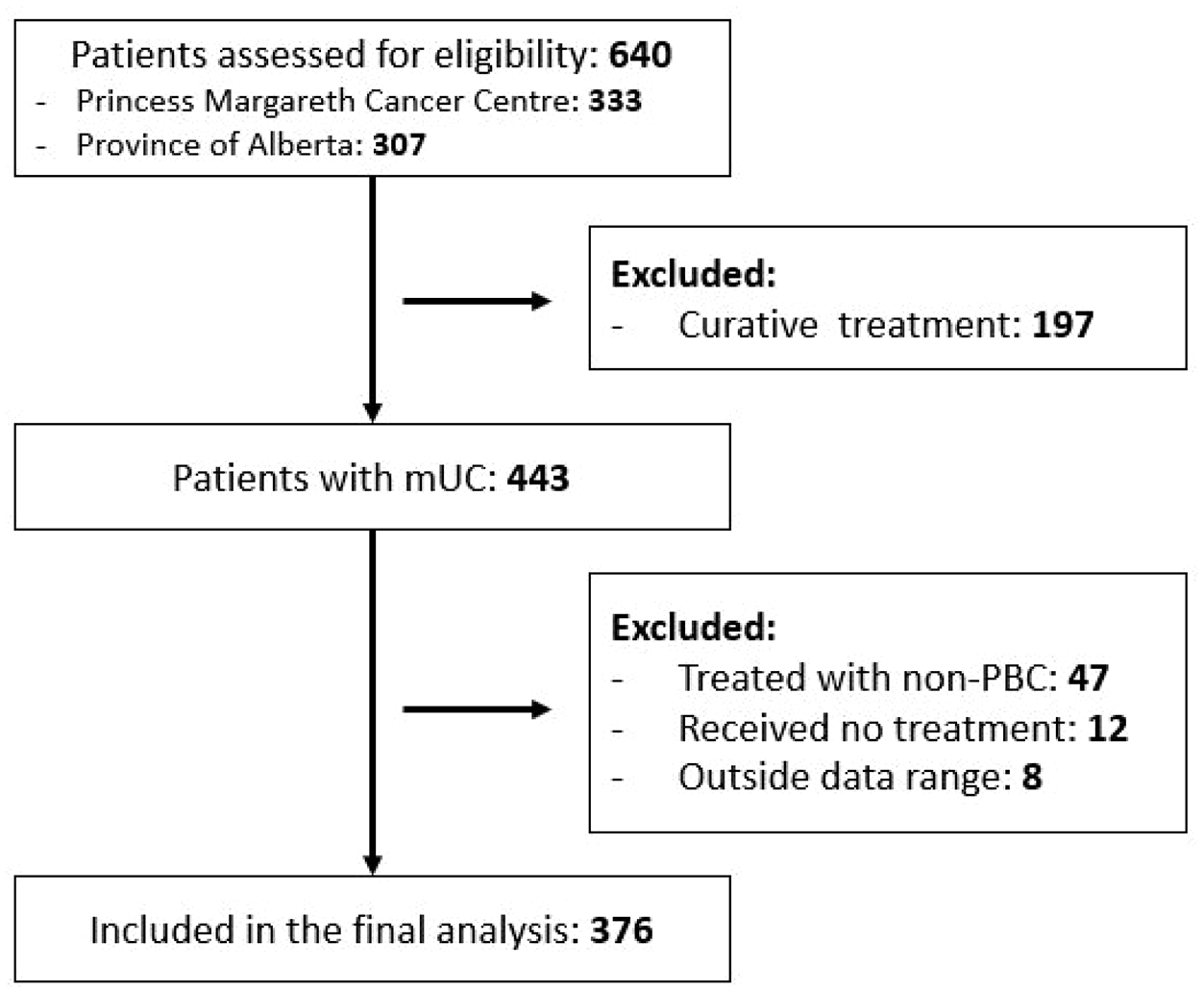
CONSORT diagram.
Clinical characteristics.

CR, complete response; ECOG PS, Eastern Cooperative Oncology Group Performance Status; PBC, platinum-based chemotherapy; PD, progressive disease; PR, partial response; SD, stable disease.
Survival outcomes and prognostic factors
For the entire cohort, median follow-up was 16.8 (range: 2.2–218.3) months. The mPFS in the entire cohort was 5.8 months (95% CI 4.9–6.7) and the median OS was 9.7 months (95% CI 8.6–10.8). The mPFS for patients with BM was 4.9 months (95% CI 3.6–6.2) compared with 6.5 months for patients without BM (95% CI 5.4–7.6; p = 0.03) (Figure 2). The median OS for patients with BM was 8.8 months (95% CI 7.8–9.7) compared with 10.8 months (95% CI 9.1–12.5; p = 0.002) for patients without BM (Figure 2).

(a) PFS and (b) KM OS analysis for patients with BMs relative to no BMs.
In UVA of OS, ECOG PS 2–3 (p < 0.001), presence of BM (p = 0.002), and WBC count ⩾ 11,000 cells/mm3 (p = 0.001) were associated with poor OS. Prior radical cystectomy (p < 0.001) and response to first-line PBC (partial response (PR) and complete response (CR)) (p < 0.001) or stable disease (SD; p < 0.001) correlated with better survival (Figure 3(a)). In multivariable analysis of OS, ECOG 2–3 (p < 0.001), presence of BM (p = 0.007), and WBC count ⩾ 11,000 cell/mm3 (p = 0.008) maintained association with inferior survival. Prior radical cystectomy (p = 0.003) and response to first-line PBC (PR (p < 0.001), CR (p = 0.002)) or stable disease (SD; p < 0.001), again correlated with improved survival (Figure 3(b)). Of note, ECOG PS 2–3 (p < 0.001), progressive disease (PD) on PBC (p < 0.001), and presence of BM (p = 0.02) were also independent predictors of PFS (Supplementary Tables 1 and 2).
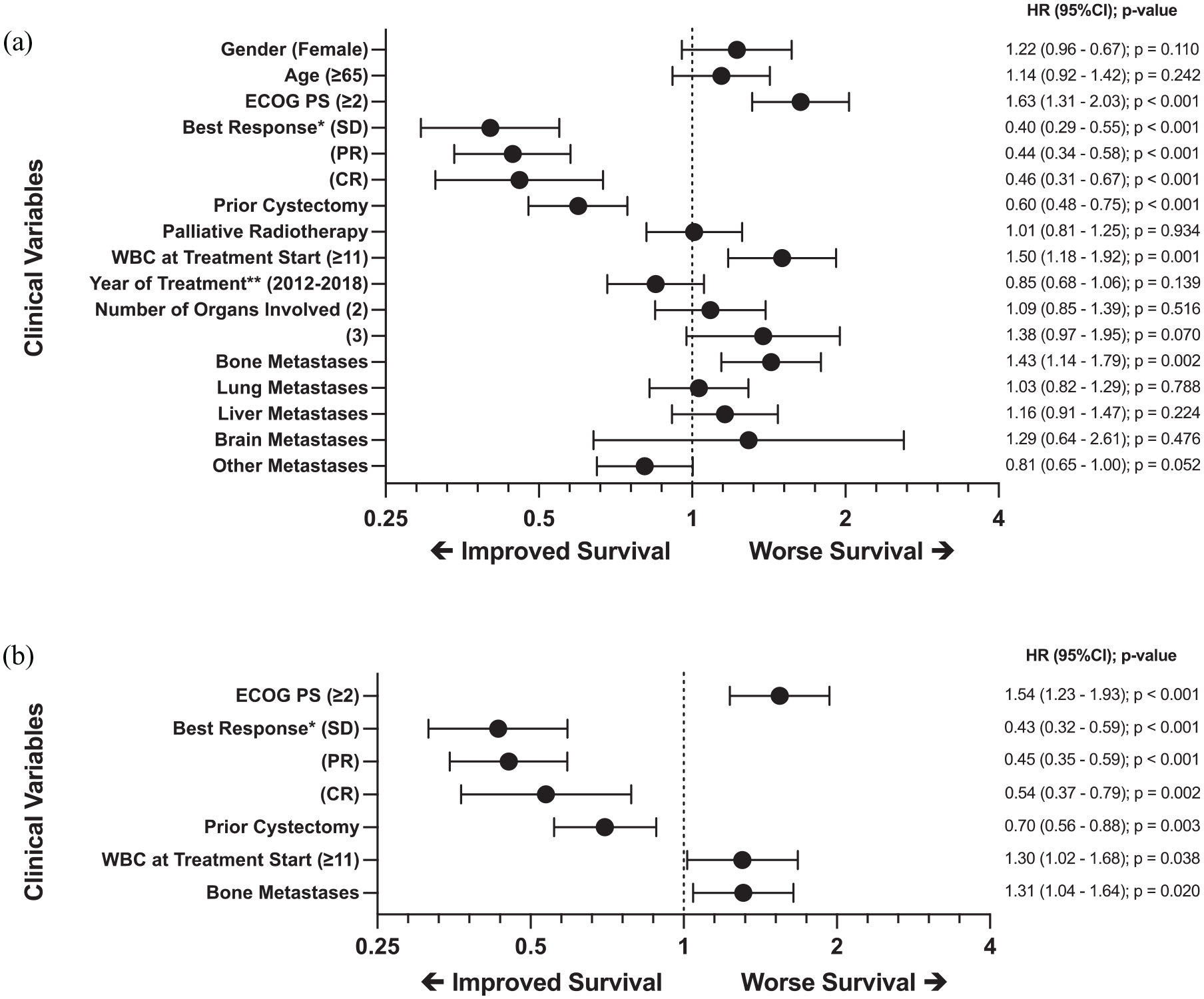
(a) Forest plot of OS for clinical variables in UVAs and (b) forest plot of OS for clinical variables in stepwise MVAs.
Discussion
In this retrospective multi-institutional study, we identified that mUC patients with BM treated with first-line PBC had worse OS. This is a novel finding as the impact of BM on survival outcomes in metastatic bladder cancer has not been well studied to date. Current prognostic nomograms include BM in visceral metastatic disease subgroups, attenuating its independent impact on survival. 10 In our study, BM remained an independent risk factor for worse survival in MVA among known prognostic factors, such as ECOG, WBC count at baseline, and poor response to PBC. These findings are concordant with a non-comparative retrospective study of 1223 mUC patients demonstrating decreased survival for patients with BM. 15 A similar international retrospective study by Necchi et al. 10 in 128 patients additionally demonstrated that patients with BM treated with first-line PBC had worse outcomes than patients without BM not withstanding additional metastatic sites.
Our data are also consistent with a recent retrospective study by Nelson et al., 16 which evaluated 270 mUC patients with BM prior to starting systemic treatment, demonstrating inferior survival and less benefit from subsequent ICIs. BM also appears to portend a worse prognosis in the platinum-refractory setting. The URO-1 bone study, for example, compared 208 mUC patients with and without BM treated with a second-line ICI. Patients with BM had inferior survival compared to those without BM. 17 In our study, the impact of BM on subsequent treatment with ICI was not evaluated. Taken together, these data would suggest that BM may be an independent predictor of survival both in patients receiving chemotherapy and those receiving second-line ICI. Thus, incorporating BM as a stratification factor in future clinical trial designs may warrant further discussion.
Our results reinforce the rationale for studying bone-targeted agents and radiotherapy in mUC patients with BM, which would represent an important therapeutic advance in this disease. In a prospective randomized study of 40 mUC patients treated with palliative radiotherapy and randomized to receive zoledronic acid or placebo for 6 months, zoledronic acid decreased incidence of SREs, delayed onset, and reduced SRE-related pain, with a trend to improved 1-year OS. 13 Results of a phase II study (NCT03520231) assessing the role of denosumab in association with chemotherapy are awaited and if positive, will provide a rationale for larger prospective trials to confirm the role of bone-targeting agents in mUC.
Further studies by Maing et al. 14 have evaluated the role of radiotherapy in treating BM in mUC and demonstrated a survival advantage when radiation doses were given as an equivalent dose in 2-Gy fractions of ⩾20 Gy. In regard to oligometastatic disease (⩽3 lesions), durable disease control in a select group of patients with metastasis-directed stereotactic body radiation therapy (SBRT) 18 was seen, although a paucity of data exists in this space. Multikinase inhibitors (e.g. cabozantinib) have also shown some activity in heavily pre-treated mUC patients with BM. 19 Enhanced molecular understanding of mUC patients with BM is warranted and should bring vital information to help guide future therapeutic strategies.
Aside from the inherent obstacles of retrospective analyses, this study’s limitations include missing data and clinician-assessed response rather than central, standardized review by response evaluation criteria in solid tumors (RECIST) 20 criteria. To overcome this barrier, the uniform handling of conflicting and missing data was ensured by medical oncologists with training in genitourinary malignancies. Although recent advances in mUC have established ICI in first- and second-line treatment approaches,6,21 –23 we could not assess the impact of BM on ICI in our analysis. However, this study was conducted at a time predating the routine incorporation of ICI into general practice. Thus, this reflects the strength of our study making it a useful comparator for future retrospective analyses that may include immunotherapeutic agents. It should also be highlighted that this study constitutes a relatively large sample size, treated similarly across three institutions and confirms our hypothesis that the presence of BM leads to a worse overall prognosis.
In conclusion, our study suggests that in mUC patients being treated with first-line PBC, the presence of BM is a poor prognostic factor that negatively impacts OS. Although this in itself is not a novel finding, the concept of collecting information on BMs specifically and using BMs as an independent stratification factor in clinical trial design in mUC is novel. This will ensure that when new therapies are evaluated in randomized trials that the arms are well balanced between treatment groups and conclusions are both accurate and generalizable. Imbalances in BM between arms, may lead to novel treatments being deemed ineffective when in fact they are, ultimately limiting therapeutic progress. Our study also shows that BM in mUC is an area of significant unmet need, and further study into bone-targeted therapies in conjunction with other novel therapeutic strategies will be the key to reducing morbidity and mortality. Recognizing the critical importance of BM in mUC represents an important therapeutic advance in this disease.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221094879 – Supplemental material for The prognostic impact of bone metastasis in patients with metastatic urothelial carcinoma treated with first-line platinum-based chemotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359221094879 for The prognostic impact of bone metastasis in patients with metastatic urothelial carcinoma treated with first-line platinum-based chemotherapy by Husam A. Alqaisi, Carlos Stecca, Zachary W. Veitch, Jamila Riromar, Jeenan Kaiser, Nazanin Fallah-Rad, Di Maria Jiang, Scott North, Sunil Samnani, Nimira Alimohamed and Srikala S. Sridhar in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Princess Margaret Cancer Center and Cancer Care Alberta thank the patients and families for their contribution to this research.
Ethics statement
This study was approved by the Princess Margaret Cancer Center Research Ethics Board (CAPCR 21-5101) and the Health and Research Ethics Board of Alberta (Cancer Committee). Informed consent was exempted for this retrospective study through REB review.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ZWV has received honoraria from Pfizer, Exact Sciences, Knight Pharmaceuticals, Novartis, Gilead pharmaceuticals, and AstraZeneca. DMJ has received honoraria from Janssen, Ipsen, Bayer, Amgen, EMD Serono, and Pfizer and consulting fees from Bayer. SSS has received consulting fees from Astellas, AstraZeneca, Bayer, Bristol Myers Squibb, Eisai, EMD Serono, Hoffmann-La Roche, Immunomedics, Ipsen, Janssen, Merck, Pfizer, and Seagen; she has also received research funding from Bayer and Janssen pharmaceuticals. The remaining authors declare no conflicts of interest.
Supplemental material
Supplemental material for this article is available online.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical restrictions.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
