Abstract
Tumor-associated macrophages (TAMs), the most abundant inflammatory cell group in the tumor microenvironment, play an essential role in tumor immune regulation. The infiltration degree of TAMs in the tumor microenvironment is closely related to tumor growth and metastasis, and TAMs have become a promising target in tumor immunotherapy. Molecular imaging is a new interdisciplinary subject that combines medical imaging technology with molecular biology, nuclear medicine, radiation medicine, and computer science. The latest progress in molecular imaging allows the biological processes of cells to be visualized
Introduction
Molecular imaging combines the characteristics of molecular biology technology and modern medical imaging with fluorescence tools, and small molecules and metals can be illuminated to observe the biological processes of cells and subcells
In recent years, the role of the tumor microenvironment (TME) in cancer progression and metastasis has garnered much interest. New targeted therapies are now focused on tumor cells themselves and on disrupting the interactions between tumor and stromal cells. 9 Inhibitors targeting immune checkpoints (ICs), such as anti-PD-1/L1 and anti-CTLA-4 monoclonal antibodies (mAbs), have been used to treat non-small cell lung cancer, 10 melanoma, 11 and breast cancer. 12 Tumor-infiltrating lymphocytes (TILs) 13 and macrophages 14 have also been studied in preclinical or clinical trials.
Tumor-associated macrophages (TAMs) play particular functionals in tumor progression, including cancer initiation and promotion, local immunosuppression, metastasis, and drug resistance.15,16 At present, targeted TAM immunotherapy in (pre)clinical applications is mainly focused on decreasing the TAM population, promoting macrophage phagocytic activity, and reprogramming TAMs to an antitumor M1 type. There is an urgent need for methods that can be used to evaluate the presence, subtypes, and density of macrophages in the TME. The gold standard for assessing macrophages in tumor tissues is immunohistochemistry (IHC), flow cytometry, or gene expression analysis. These techniques can provide detailed molecular and morphological information. However, they require the sacrifice of animals and cannot be used when biopsy samples are not available. The development of molecular imaging provides a starting point for solving these problems. Optical imaging, PET/SPECT, MRI, and US have been used to track and monitor the dynamic changes in TAMs noninvasively. Individual or combined use of these imaging strategies might help stratify patients before or during the early stages of immunotherapy. 17 This review mainly discusses the application of TAM-based tumor immunotherapy and highlights the latest advances in molecular imaging technology for macrophage tracking.
TAMs in cancer immunotherapy
TAMs represent the main immune cell population of the TME and have been mainly classified into the antitumor type M1 phenotype (classically activated state) and the protumor M2 phenotype (alternatively activated state). 18 In terms of protein expression, M1 macrophages are characterized by the expression of proinflammatory cytokines (tumor necrosis factor alpha, TNF-α; interleukin 6, IL-6; or interleukin 12, IL-12), major histocompatibility complex (MHC) molecules, and inducible nitric oxide synthase (iNOS). M2 macrophages decrease the expression of these molecules and recognize various landmarks such as Arg1, mannose receptor, and scavenger receptor. 19 Macrophages are plastic cells that can adopt different phenotypes depending on the immune context. Under the stimulation of CSF-1 (colony-stimulating factor-1), interleukin 10 (IL-10), or interleukin 13 (IL-13), macrophages differentiate into M2 phenotype, which promotes tumor development mainly through the following points: (1) suppress tumor immunity by (i) secreting effector molecules (IL-10, ARG1, indoleamine 2, 3-dioxygenase (IDO) and transforming growth factor-β (TGF-β), (ii) expressing inhibitory receptors (MHC-I, PD-L1, and CD86) to reduce the activities of T cells and natural killer (NK) cells, or (iii) secreting chemokines CCL5, CCL22, and CCL20 to recruit immune suppressive cells 20 ; (2) promotes tumor angiogenesis by producing pro-angiogenic cytokines and growth factors such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), angiopoietin 2, CXCL1, and FGF-2 21 ; and (3) promotes tumor cell invasion and metastasis by (i) releasing effector molecular cathepsin, MMP2, MMP9, CCL18, and CYP4A, which are involved in extracellular matrix (ECM) destruction, 22 (ii) various factors such as TNF-α, epidermal growth factor (EGF), CCL18, osteonectin, and cathepsin are able to promote tumor cell intravasation, 23 and (iii) chemokines and cytokines of CXCL8, 24 CSF-2, TNF-α, ICAM-1, and IL-6 25 contribute to epithelial-mesenchymal transition (EMT). In contrast, lipopolysaccharide (LPS) and interferon-gamma (INF-γ) induced macrophages to differentiate into the M1 phenotype, which has the antitumor activity of scavenging and destroying phagocytic tumor cells secreting IL-12, TNF-α, and iNOS and initiating Th1.26,27
Increased TAM infiltration correlates with an inert response to cancer treatment and poor prognosis in multiple cancer models. 28 Hence, targeting TAMs is a potential and promising strategy for cancer immunotherapies. Immunotherapeutic strategies fall into three major categories: (1) decreasing the TAM population, (2) promoting macrophage phagocytic activity, and (3) reprogramming TAMs to an antitumor M1 phenotyope (Figure 1). TAM-targeted therapies can also function as combination therapies to promote traditional or IC treatments’ antitumor effect.
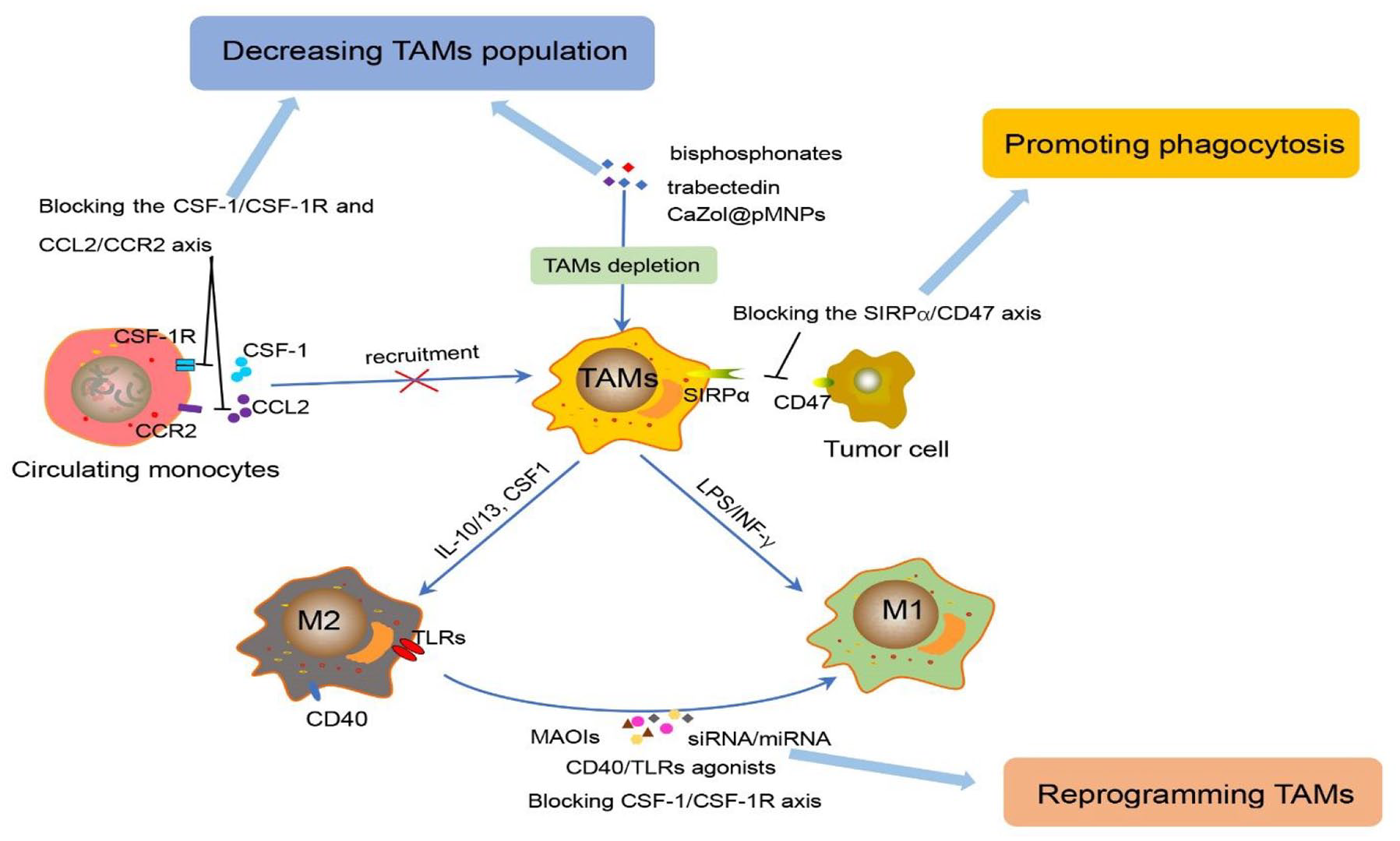
Immunotherapeutic strategies of targeting TAMs. The immunotherapeutic strategies fall into three main points: (1) decreasing the TAM population by blocking the CSF-1/CSF-1R axis and CCL2/CCR2 axis to inhibit the monocyte recruitment, and depleting TAMs with drugs (bisphosphonates, trabectedin, and
Decreasing the TAM population
TAMs have mainly two different origins: much greater of them, termed bone marrow–derived macrophages (BDMs), are derived from circulating monocytes and recruited to the tumor site
Limiting the recruitment of circulating monocytes into the TME is a promising strategy to reduce protumor TAMs. Some trials targeting chemoattractants secreted by malignant and stromal cells have been investigated such as macrophage CSF-1 31 and C–C motif chemokine ligand 2 (CCL2) 32 and their receptors. CSF-1/CSF-1R (colony-stimulating factor-1 receptor) is critical for monocyte recruitment to the tumor site and differentiation into M2 macrophages. PLX3397 (an inhibitor of CSF-1R) has been found to effectively decrease the number of TAMs and circulating monocytes in mesothelioma mouse models. It can augment survival synergistically, reduce TAMs, and increase the number and function of CD8+ T cells when combined with dendritic cell vaccination. 33 In addition to solid tumors, targeting CSF-1R has been proven effective in hematopoietic malignancies. It has been demonstrated that BLZ945 (CSF-1R inhibition) could block leukemia-associated monocyte-derived cell (LAM) polarization and deplete LAMs in a mouse model of T-cell acute lymphoblastic leukaemia (T-ALL). 34
CCL2, a chemokine released by monocytes and tumor cells, and its receptor C–C chemokine receptor type 2 (CCR2) play a critical role in mediating monocyte egress from the bone marrow into the TME.
35
Blocking the CCL2/CCR2 axis has effectively reduced tumor growth in several animal models.36,37 Wang
Depleting macrophages already extant in tumor tissue has also shown an antitumor effect. Some compounds, including trabectedin,
41
bisphosphonates,
42
and clodronate liposomes,
43
have been confirmed to scavenge TAMs by inducing their apoptosis effectively. Lipid-coated calcium zoledronate-encapsulated complexes (
Promoting macrophage phagocytic activity
The successful growth and maturation of tumors need to evade all kinds of immune surveillance activities effectively, for example, to escape the phagocytosis of macrophages. One key mechanism of tumor cell immune escape is through upregulation of the immunosuppressive signaling molecule CD47, which interacts with signal regulatory protein alpha (SIRPα) on macrophages to transmit the ‘don’t eat me’ signal.
46
Blocking the CD47-SIRPα axis is an exciting strategy to enhance TAM phagocytic properties. Several CD47-SIRPα blocking agents have been developed such as fully human anti-CD47 antibodies, anti-CD47 single-chain variable fragments, anti-SIRPα antibodies, and high-affinity monomeric SIRPα devoid of the Fc portion.
47
In
Reprogramming TAMs to antitumor M1 phenotyope
In light of this characteristic plasticity, reprogramming TAMs to antitumor M1 is an ideal strategy for cancer immunotherapy, including TLR agonists, 54 blockade of the CSF-1/CSF-1R axis, 55 interference RNAs – small interfering RNA (siRNA) or microRNA (miRNA), 56 monoamine oxidase inhibitors (MAOIs), 57 and CD40 agonists. 58
As a type of pathogen pattern recognition receptor (PRR), TLRs are innate immunity PRRs expressed by antigen-presenting cells (APCs), including macrophages. The activation of TLRs can induce the immune response, reprogramming TAMs to the M1 phenotype, which has been proven in preclinical or clinical trials with TLR3, TLR7, TLR8, and TLR9 agonists in many tumor models.54,59 Recently, to better target macrophages and reduce side effects, TLR agonists such as CpG oligodeoxynucleotides (CpG ODNs) have been targeted for delivery with the nanoparticle vector murine M2 macrophage-targeting peptide M2pep. In four T1 tumor-bearing mice, these M2pep-rHF-CpG nanoparticles repolarized M2 TAMs to the M1 type and inhibited tumor growth after intravenous injection. 60
miRNAs have been demonstrated to participate in myeloid differentiation and macrophage activation in previous studies by suppressing the expression of target genes. 61 In orthotopic HCC or subcutaneous Lewis lung cancer (LLC) mice, miR-99b and/or miR-125a were delivered into TAMs, and the authors found that miR-99b or miR-125 treatment increased the expression of M1 markers (such as IL-6, TNF-α, and iNOS) while decreasing the M2-specific marker Arg1, which contributed to inhibiting tumor growth. 56
Monoamine oxidase A (MAO-A) is an enzyme best known for its function in the brain, and correlation studies of clinical data show that high intratumoral MAO-A expression in a wide range of cancers is associated with poor patient survival. Sikic
The Cluster of Differentiation 40 (CD40) is a member of the tumor necrosis factor (TNF) receptor family and is widely expressed by tumor cells and APCs. Studies have found that treatment with CD40 agonists alone or with anti-CSF-1R antibodies in preclinical tumor models can decrease immunosuppressive macrophages and increase maturation and differentiation of proinflammatory TAMs. 62 Two agonistic anti-CD40 antibodies (CP-870,893 and RO7009789) are being tested in clinical trials. CP-870,893 was administered in combination with gemcitabine chemotherapy in a phase I trial for patients with pancreatic cancer; the results showed it was well tolerated, and partial responses (PRs) were identified in approximately 20% of patients. 63
Overview on molecular imaging
Molecular imaging is a technique based on radionuclides or optical probes to observe tumor biomarkers in real time and accurately monitor the dynamic changes in target genes.
64
Among the imaging modalities, each technique has its inherent strengths and weaknesses (Table 1). PET/SPECT, a radionuclide-based molecular imaging technology, is used extensively in preclinical and clinical researches due to its high sensitivity and total body penetrance. It can realize the early diagnosis, treatment guidance, and study of the molecular mechanisms of diseases
Characteristics of targeting TAM imaging techniques.

CSF-1R, colony-stimulating factor-1 receptor; DN-ICG, dextran-indocyanine green; FLI, fluorescence imaging; HA-FOL-NBs, hyaluronic acid folate-conjugated nanobubbles; IONPs, iron oxide nanoparticles; MPI, magnetic particle imaging; MRI, magnetic resonance imaging; NB, nanobubble; NIR, near-infrared; PET, positron emission tomography; PFC, perfluorocarbon compound; SPECT, single-photon emission computed tomography; TAM, tumor-associated macrophage; US, ultrasound.
Tracking TAMs by molecular imaging
Taking advantage of the characteristics of these imaging techniques allows us to expand our knowledge of TAM-based immunotherapies and provide bridges to the clinical applications of these therapies. Despite advances in immune-oncology, most patients still exhibit primary or acquired therapeutic resistance mediated by immunosuppressive macrophages. 70 The preclinical and clinical data showed that patients with intense TAM infiltration could benefit from TAMs targeting immunotherapies, highlighting the need for specialized techniques to detect and quantify TAM populations to improve treatment outcomes for cancer patients. Molecular imaging can screen suitable patients, and monitor and evaluate immunotherapeutic efficacy and prognosis. 71 Therefore, molecular imaging is exceedingly helpful in the development of TAM-based immunotherapies. The characteristics of targeting TAM imaging techniques and TAM-labeling strategies are illustrated in Tables 1 and 2. Several specific targets of TAMs for imaging have been demonstrated such as the macrophage mannose receptor (MMR or CD206), translocator protein (TSPO), phagocytic activity, FR-β, CSF-1R, F4/80, TLRs, and SIGN-R1 (specific ICAM-3-grabbing nonintegrin-related 1) (Figure 2(A)).
Molecular imaging strategies for TAM tracking.

CSF-1R, colony-stimulating factor-1 receptor; CT, computed tomography; DM, deoxymannose; DN-ICG, dextran-indocyanine green; DPA, (N,N-diethyl-2-(2-(4-(2-fluoroethoxy)phenyl)-5,7-dimethylpyrazolo[1,5-a]pyrimidin-3-yl)acetamide; FB, fluorobenzoate; FR-β, folate receptor β; HA-FOL-NBs, hyaluronic acid folate-conjugated nanobubbles; HCC, hepatocellular carcinoma; IONPs, iron oxide nanoparticles; MAN-LIPs, mannosylated liposome; MMR, macrophage mannose receptor; MPI, magnetic particle imaging; MMTV-PyMT, the mammary specific polyomavirus middle T antigen overexpression mouse model; MRI, magnetic resonance imaging; NB, nanobubble; NIRF, near-infrared fluorescence; PET, positron emission tomography; PFC, perfluorocarbon compound; SIGN-R1, specific ICAM-3-grabbing nonintegrin-related 1; SPECT, single-photon emission computed tomography; TAM, tumor-associated macrophage; TLRs, toll-like receptors; TSPO, translocator protein; US, ultrasound; WHO, World Health Organization.
MMR, CD206
MMR, CD206, is an intracellular C-type lectin receptor. As an immune adhesion molecule, MMR is mainly expressed on the surface of APCs such as specific macrophages and immature dendritic cells. 100 Some studies have shown that MMR is abnormally expressed in various tumors (pancreatic, colorectal, and gastric cancer), and its expression level correlates with the prognosis of patients.101–103
Approximately 10 years ago, MMR was confirmed to be a special target for TAM imaging. A report by intravenous injection of 99mTc-labeled anti-MMR nanobodies successfully targeted M2 macrophages

Molecular imaging of TAMs. (A) Some techniques have been applied to target TAM molecular imaging such as PET, SPECT, MRI, US, and optical imaging. The main imaging targets that have been used in clinical or preclinical studies include MMR, TSPO, CSF-1R, TLRs, SIGN-R1, FR-β, F4/80, and its phagocytic activity. (B) Examples of TAM molecular imaging by targeting MMR. Coronal PET/CT images of WT
MMR has also been investigated in optical imaging and MRI. Zhang
Given the inevitable shortcomings of nuclear imaging and optical imaging, to better provide information on the biological dynamics of macrophages, a recent study reports an approach of using anti-biofouling PEG-b-AGE polymer-coated iron oxide nanoparticles (IONPs) to target imaging of MMR-expressing M2-like TAMs. In this case, at 48 h after intravenous injection of mannose iron oxide nanoparticles (Man-IONPs) in mice bearing orthotopic four T1 mammary tumors, MRI showed a more significant IONP-induced decrease in transverse relaxation time (T2) in tumors compared with nontargeted IONP probes. More importantly, this study found no nonspecific uptake cellular of IONPs by M1-like macrophages and cancer cells, and there was no nonspecific interaction between IONPs and serum proteins. 80
Mannose receptors targeting radiotracers are currently applied to the imaging of macrophages in the TME and proven to have important clinical significance for detecting tumor sentinel lymph nodes, 105 assessing atherosclerotic plaque stability 106 and detecting rheumatoid arthritis early. 107 Hence, MMR is a promising target for the noninvasive detection of macrophages to obtain diagnostic and prognostic information on inflammatory diseases and investigate targeted macrophage immunotherapy.
TSPO
TSPO is an 18-kDa mitochondrial outer membrane protein mainly expressed in activated microglia, astrocytes, and infiltrating macrophages. TSPO has become a promising imaging target in some inflammatory diseases, especially in neuroinflammatory diseases (such as multiple sclerosis, 108 Parkinson’s disease, 109 and glioma). 81
Different studies have shown that the expression of TSPO is positively correlated with the degree of malignancy and negatively correlated with survival.110,111 Glioma-associated microglia/macrophages (GAMs) were identified as TSPO sources, and preclinical imaging studies suggested that PET imaging of TSPO could be a potential tool to improve tumor detection and track glioma cell infiltration. The first-generation TSPO radiotracer [
11
C]PK11195 was successfully used in glioma imaging. However, as a 11C-labeled tracer, [
11
C]PK11195 has a short half-life and requires a cyclotron to produce the tracer in the field, and a low signal-to-noise ratio limits its use.112,113 These limitations have led to the development of new TSPO ligands for PET imaging such as [
18
F]DPA-714,81,82 [
18
F]DPA-713,
114
[
18
F]PBR06,
83
and [
18
F]GE-180.84,85 Among the current (pre)clinical tracers used, [
18
F]DPA-714, a second-generation tracer, has been highlighted as a suitable imaging marker in glioma models.115,116 Pigeon
Although some results have indicated that TSPO expression levels are positively correlated with the grade of malignancy, it is not clear to what extent the upregulation of TSPO reflects a proinflammatory or anti-inflammatory phenotype. Recently, Pannell
Third-generation TSPO receptor ligands (
18
F-GE-180) were subsequently produced to improve the target-background contrast. Nathalie
In an experimental study of a mouse model of pancreatic ductal adenocarcinoma (PDAC), Lanfranca
Phagocytic activity
Macrophages are an essential component of innate immunity, and serve as the first line of defense against infection. Phagocytosis is an eponymous function and remarkably stable property of the macrophage lineage. It is able to engulf various substances such as pathogens, nanoparticles, and liposomes through surface receptors and cytoplasmic recognition systems.119,120
At present, tracers based on the phagocytic function of macrophages are mainly used for MRI, and some involve radionuclide imaging. Scanning is usually performed 24 h after intravenous injection of the contrast agents to allow macrophages to phagocytize and infiltrate into the tumor area while allowing the unengulfed contrast agent to be cleared from the tumor. Several studies previously demonstrated that TAMs could be tracked with MRI contrast agents such as gadolinium (Gd), manganese (Mn) chelates, IONPs, and fluorine 19 (
19
F)-incorporated perfluorocarbon compounds (PFCs).121,122 Macrophages and Kupffer cells mainly engulf IONPS, which have been more widely used in cancer images because of their easy synthesis, high surface-to-volume ratio, and therapeutic ability.123,124 Ferumoxytol, a US Food and Drug Administration (FDA)–approved ultra-small superparamagnetic iron oxide (USPIO) nanoparticle, has been proven for the first time to be able to be used as a contrast agent to label TAMs by MRI in a mouse breast cancer model.
88
Recent clinical studies by Aghighi
MPI is a tracer method based on tomographic imaging technology to detect the spatial distribution of superparamagnetic iron oxide (SPIO). It has the characteristics of three-dimensional imaging, high resolution and sensitivity, and no electric radiation hazard. Its signal strength is proportional to the tracer concentration and is an inspection method that can obtain quantitative data.
126
MPI has been used to detect iron-labeled TAMs in a murine model of breast cancer. In this study, the authors found that ferumoxytol (USPIO) was a superior iron nanoparticle for this application than ferucarbotran (SPIO)
19
F-based MRI is emerging as a useful tool for imaging cells. PFC nanoparticles are preferentially phagocytosed by monocytes and lack any fluorine background in the body.
127
19
F signal intensity (SI) is directly proportional to the tissue amount of the PFC, which makes 19F MRI a highly sensitive and specific quantitative tool.
128
Makela
Given macrophage phagocytosis imaging characteristics, the isotope for radionuclide imaging needs to have a long half-life such as 64Cu (
Other targets
SIGN-R1 is a calcium-dependent lectin highly expressed on macrophage surfaces.
129
Luo
TLRs are receptors of the innate immune system that recognize pathogens and are primarily expressed on macrophages and dendritic cells. TLR4 has been most closely connected to inflammation-mediated carcinogenesis and tumor progression.
130
Lee
FR-β is a cell surface receptor that is significantly upregulated on activated macrophages during inflammation but not on resting or other immune cells.
131
For this reason, it has been used as a macrophage-based therapeutic and imaging agent. For example, [Ga]Ga-NOTA-folate (Ga-FOL) was verified to be specific for targeting FR-β in a study of an atherosclerotic mouse model.
132
Another targeted FR tracer, aluminum F-labeled 1,4,7-triazacyclononane-triacetic acid conjugated folate (F-FOL), demonstrated specific uptake in inflamed myocardium containing macrophages expressing FR-β, and the nonlabeled FR-β ligand folate glucosamine could efficiently block this response
F4/80, a 160-kDa cell surface glycoprotein, is highly restricted in mouse tissue macrophages. It is used as a pan-macrophage marker to detect the distribution, phenotypic heterogeneity, and activation state of TRMs. F4/80 was critical in the macrophage–NK interaction and participates in the induction of peripheral immune tolerance.
134
Terry
Given that the CSF-1R has been found to be restrictively expressed by TAMs and monocytes which more prefer to locate at the boundary of HCC. A CSF-1R-conjugated nanobubble CSF-1R (NBCSF-1R) was developed using the biotinylation method and demonstrated that it might be a promising noninvasive diagnostic modality to detect the margin and residual of HCC after radiofrequency ablation (RFA).
99
In addition to the biomarkers and tracers for the
Monitoring tumor immunotherapy with TAM imaging
Among the existing targeted TAM imaging methods, encouragingly, some of them have been investigated in clinical trials (Table 3). The combination of molecular imaging and tumor immunotherapy for real-time detection of the therapeutic response is the focus of clinical research. CD47 represents a promising new target for osteosarcoma immunotherapy, and its mAbs have been confirmed to inhibit the interaction between CD47 and SIRPα, increasing the number of M1-like macrophages and activating phagocytes.137–139 The Mohanty group used ferumoxytol-enhanced MRI to detect CD47 mAb therapeutic responses in osteosarcoma-bearing mice and showed significant hypo-intense (dark) ferumoxytol enhancement on post-contrast T2-MR images compared with pre-contrast images in the tumor area. CD47 mAb-treated tumors were significantly smaller than control IgG-treated tumors on day 10 of therapy. This verified the usefulness of ferumoxytol-MRI for detecting TAM response to CD47 mAb, and this imaging model is expected to be applied to monitor the therapies’ response in clinical trials. 139
Examples of TAM-targeted imaging in clinical trials.

DPA, (N,N-diethyl-2-(2-(4-(2-fluoroethoxy)phenyl)-5,7-dimethylpyrazolo[1,5-a]pyrimidin-3-yl)acetamide; MRI, magnetic resonance imaging; PET, positron emission tomography; TAM, tumor-associated macrophage; TSPO, translocator protein.
Increasing evidence suggests that the TME, particularly tumor-infiltrating immune cells, including TAMs, changes significantly after radiotherapy and chemotherapy. One study used three nanoparticles, QD710-Dendron quantum dots (QD710-D), ferumoxytol, and PG-Gd-NIR813, for NIRF, T2W MRI, and dual optical/T1W MRI (Figure 3(A)) in an MDA-MB-435 tumor model.

Monitoring immunotherapy by molecular imaging of TAMs. (A) Optical imaging and T1W MRI
By NIRF, DN-ICG has been demonstrated to target TAMs with good sensitivity and specificity. The authors further designed experiments to confirm that this imaging technique could also monitor the treatment response dynamically. Low-dose radiotherapy (5 Gy) and chemotherapy (zoledronic acid) were designed elaborately to regulate the number of TAMs in the subcutaneous model of the pancreatic tumor. They observed that the number of TAMs was twice as high as before low-dose radiotherapy (5 Gy) treatment by flow cytometry. However, after a 3-day treatment with zoledronic acid, TAMs were 50% lower than that in the control group. NIRF analysis of TAMs revealed that the fluorescence signal in the tumor region increased twofold in the low-dose radiotherapy group and decreased by 50% in the zoledronic acid-treated group at 6, 12, and 24 h after the injection of DN-ICG (Figure 3(B)), which was consistent with the flow cytometric analysis. 95 These studies indicate that noninvasive imaging techniques can observe the quantitative changes in TAMs and assess the early treatment response using imageable nanoparticles.
Discussion and conclusion
Given the crucial roles of TAMs in orchestrating tumor progression, preclinical and clinical trials have demonstrated that targeting TAMs is a novel and promising approach to improving antitumor therapy. The classification, distribution, and density of TAMs in the TME vary among individuals and tumor types and change dynamically during treatment. There is an urgent need to have an accurate understanding of TAMs in the TME. This review shows that molecular imaging techniques can provide better means for the noninvasive, real-time, and quantitative
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grant from the National Natural Science Foundation of China (No. 81972837, No. 82071986, No. 81771827, and No. 81971721).
