Abstract
Background:
Axillary node status is used in clinical practice to guide the selection of axillary surgery in breast cancer patients. However, to date, the optimal axillary management following neoadjuvant therapy (NAT) for breast cancer remains controversial. Our study aimed to investigate the association of molecular subtype, clinical stage, and ypN status after NAT in breast cancer patients, especially those achieving breast pathological complete remission (pCR).
Patients and methods:
Patients receiving ⩾4 cycles of NAT were retrospectively included between January 2009 and January 2020. ypN status was compared among patients with different breast pCR statuses, clinical stages, and molecular subtypes in univariate and multivariate analyses.
Results:
A total of 1999 patients were included: 457 (22.86%), 884 (44.22%), and 658 (32.92%) patients with cT1-2N0, cT1-2N1, and locally advanced breast cancer (LABC), respectively. Altogether, 435 (21.8%) patients achieved breast pCR: 331 with ypN– and 104 with ypN+ status. Patients achieving breast pCR had a significantly lower ypN+ rate than those without pCR [23.9%
Conclusion:
In breast pCR patients after NAT, clinical stage and molecular subtype were significantly associated with ypN status. Patients with cT1-2N0 and HER2-positive disease who achieved breast pCR had a very low ypN+ rate, possibly indicating the possibility for de-escalation of axillary surgery in this patient subgroup.
Keywords
Introduction
Over the past decades, neoadjuvant therapy (NAT) has emerged as a preferred option for locally advanced breast cancer (LABC) and has been increasingly used in operable patients, especially those with a relatively large tumor or axillary lymph node (ALN)-positive, human epidermal growth factor receptor-2 (HER2)-positive, or triple-negative disease.1–3 NAT has a demonstrated ability to downstage the primary tumor, increase operability, facilitate breast conservation, and test
Patients who achieve pathological complete remission (pCR) after NAT have significantly superior clinical outcomes compared with those with residual disease.4–7 Therefore, achieving pCR is the foremost goal of NAT. As reported in previous studies, the total pCR rate is estimated to be 18–28% and to vary across different molecular subtypes, ranging from 8% to 11% in hormone receptor (HR)-positive/HER2-negative disease and 17–32% in HR-positive/HER2-positive disease to 33–51% in HR-negative/HER2-positive disease and 31–36% in triple-negative disease.1,2,8 The neoadjuvant treatment regimen can impact the pCR rate. For example, the addition of taxanes to doxorubicin and cyclophosphamide improved response to neoadjuvant therapy from 13% to 27%. 9 The combination of dual anti-HER2-targeted therapy with standard chemotherapies significantly increased the pCR rate, subsequently ranging from 45% to 62%. 10 Besides, clinical tumor stage is also an important predictor for pCR. 11
In general, breast pCR can be achieved in 23–28% of patients and can reach a rate of around 70% or higher in HER2-positive/HR-negative patients treated with standard chemotherapy plus trastuzumab and pertuzumab.2,12,13 Of note, breast pCR was demonstrated to be associated with statistically significantly lower axillary residual burden.12,14 The rate of axillary ypN0 in breast pCR patients was twice higher than that in patients without breast pCR.12,14 In specific cN0 patients with HER2-positive or triple-negative disease, Barron
While axillary node status is used to guide the subsequent selection of axillary surgery, optimal axillary management following NAT remains controversial. Currently, sentinel lymph node biopsy (SLNB) after NAT in cN0 patients is reliable and has a similar accuracy to that of upfront SLNB. 17 Nevertheless, the use of SLNB in cN+ patients still lacks consensus, even though it shows reliable accuracy when dual tracers are used or ⩾3 sentinel nodes are removed.17,18 NAT has been shown to increase the rates of ypN0 compared with upfront surgery without NAT by 1.5–3.6-fold in cN0 patients and to convert 20–61% of cN+ patients with different molecular subtypes to ypN0 status. 19 Given the decreased ypN+ rate after NAT, especially in breast pCR patients, the potential for reducing or eliminating axillary surgery warrants further research. Therefore, our study was designed to investigate the association of clinical stage and molecular subtype with ypN status after NAT and to identify potential predictors for ypN status in patients with different breast response to NAT, with the view to guide an optimal ALN surgical approach.
Patients and methods
Study population
Consecutive patients diagnosed with invasive breast cancer between January 2009 and January 2020 were retrospectively reviewed. Data were derived from the Shanghai Jiao Tong University Breast Cancer Database (SJTU-BCDB), which included more than 30,000 breast cancer cases from 32 medical centers in China. Patients meeting the following criteria were enrolled: female patients; invasive breast cancer diagnosed by core needle biopsy (CNB) before NAT with complete clinicopathological information; receiving ⩾4 cycles of neoadjuvant chemotherapy with or without anti-HER2 targeted therapy (trastuzumab ± pertuzumab); undergoing standard breast and axillary surgery with complete histo-pathological data after NAT. Exclusion criteria were as follows: male patients; patients receiving neoadjuvant endocrine therapy alone; patients who underwent tumor excision biopsy before NAT; patients diagnosed with occult breast cancer; patients with unknown neoadjuvant regimens; patients without accurate pathological data or surgery information; and patients who received <4 cycles of NAT (Figure 1).
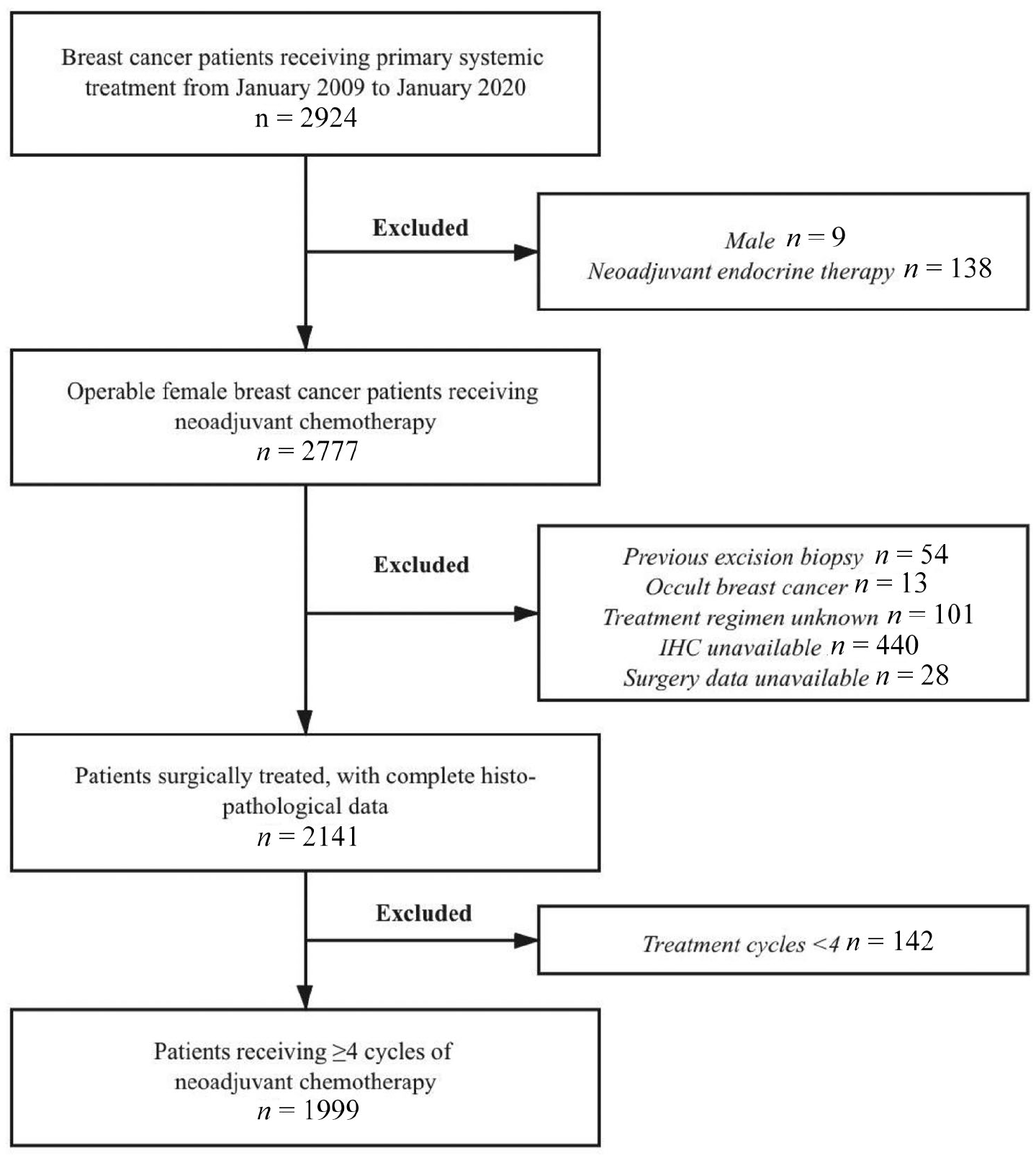
Study population flowchart.
Neoadjuvant regimens were determined at the physicians’ discretion and classified into three categories: regimens containing anthracyclines, such as CEF [cyclophosphamide (C), epirubicin (E), and fluorouracil (F)] and EC; regimens containing taxanes, such as TC [docetaxel (T)], PCb [paclitaxel (P), carboplatin (Cb)] and TCb; and regimens combining anthracyclines and taxanes, such as EC-T, TEC, dose-dense EC-weekly P, and ET. HER2 positive breast cancer patients were also recommended to receive trastuzumab ± pertuzumab treatment as anti-HER2 targeted therapy.
This study was approved by the independent Ethical Committees of Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (2020-309). Patients identity remained anonymous, and the requirement for informed consent was waived due to the observational nature of the study. All human-related procedures were in concordance with the 1964 Helsinki Declaration and its later amendments.
Clinical and pathological assessment
Clinical tumor and nodal status before NAT were determined through physical examination (PE) by experienced physicians and diagnostic ultrasound by at least two independent radiologists. No suspicious lymph node under PE and ultrasound or a negative cytological test result confirmed by ultrasound-guided fine-needle aspiration was defined as cN0. Clinical TNM staging of breast cancer was based on the American Joint Committee on Cancer cancer staging manual (eighth edition, 2017). Patients with clinical stage III or cT3N0 disease were classified as LABC. 20 Comparisons were done among the cT1-2N0, cT1-2N1, and LABC groups.
Diagnosis of breast cancer was made by CNB before NAT. Histopathological and immunohistochemical (IHC) evaluation of tumors was accomplished by at least two independent experienced pathologists. The applied positivity criteria followed the 2018 American Society of Clinical Oncology/College of American Pathologists guidelines. Samples were defined as positive for estrogen receptor (ER) or progesterone receptor (PR) if no less than 1% of the invasive tumor cells stained positive by IHC. A 3+ IHC result for CerbB-2 or HER2 gene amplification confirmed by florescent
Statistical analysis
Univariate analysis with two-sided Pearson Chi-square test was applied to compare categorical variables among tumors with different clinical stages, molecular subtypes, and pathological response to NAT. Multivariable logistic regression models were used to determine the impact factors for pathological nodal response and variables with
Results
Baseline characteristics
Altogether, 1999 breast cancer patients were included in the study (Figure 1). Patients’ clinicopathological characteristics are shown in Table 1. The median age was 50 (ranging from 21 to 83) years old. Invasive ductal carcinoma was diagnosed in 1880 (94.05%) of the enrolled population. Grade I–II and III tumors were found in 633 and 346 patients, respectively. A total of 540 patients were classified as cN0 cases. cT1-2N0, cT1-2N1, and LABC disease were found in 457 (22.9%), 884 (44.2%), and 658 (32.9%) patients, respectively. The Luminal A, Luminal B (HER2–), Luminal B (HER2+), HER2-amplified, and triple-negative molecular subtypes represented 5.7%, 43.0%, 21.9%, 14.1%, and 15.3% of the study population, respectively.
Baseline characteristics of study population.

A, anthracyclines; ALN, axillary lymph node; ALND, axillary lymph node dissection; BCS, breast conserving surgery; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; LABC, locally advanced breast cancer; NA, not available; NAC, neoadjuvant chemotherapy; PR, progesterone receptor; SLNB, sentinel lymph node biopsy; T, taxanes; TNBC, triple negative breast cancer.
In terms of treatment regimens, 1425 (71.3%) patients were treated with NAT using a combination of anthracyclines and taxanes. Targeted therapy was given in 24.0% of the whole patient population, accounting for 66.8% of the HER2-positive patients. Moreover, 1284 (64.2%) patients received 4–6 cycles of NAT and 715 (35.8%) received more than six cycles. After NAT, mastectomy and axillary lymph node dissection were carried out in 90.2% and 94.1% of patients, respectively.
Univariate analysis showed that histologic type (
Pathological response in the whole study population
Upon completion of NAT, 435 (21.8%) patients achieved breast pCR and 917 (45.9%) achieved ypN0. Breast pCR rates for cT1-2N0, cT1-2N1, and LABC disease were 20.6%, 24.2%, and 19.3%, respectively (
Univariate and multivariate analyses identified clinical stage (
Multivariate analysis of impact factors for ypN+ in the whole population.

A, anthracyclines; CI, confidence interval; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; LABC, locally advanced breast cancer; NAC, neoadjuvant chemotherapy; OR, odds ratio; pCR, pathological complete remission; PR, progesterone receptor; T, taxanes; TNBC, triple negative breast cancer.
Axillary pathological response in breast pCR patients
In patients with breast pCR, the ypN+ rate was 6.4%, 25.7%, and 33.9% in cT1-2N0, cT1-2N1, and LABC patients, respectively (
Pathologic axillary lymph node status stratified by clinical stage and molecular subtype between patients with breast pathological complete remission (pCR) and non-pCR.
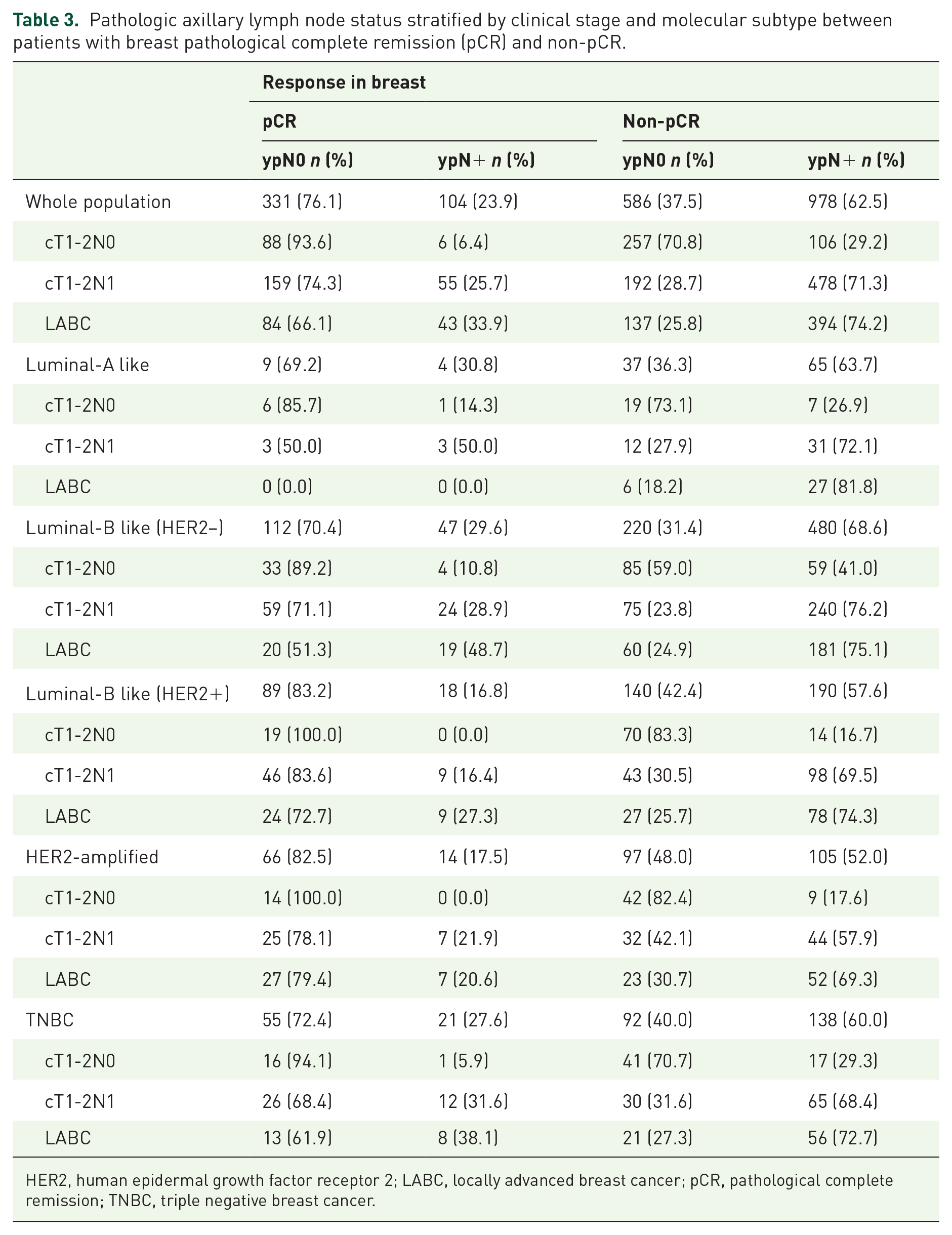
HER2, human epidermal growth factor receptor 2; LABC, locally advanced breast cancer; pCR, pathological complete remission; TNBC, triple negative breast cancer.

Pathologic nodal residual burden in patients with breast pathological complete remission (pCR) and non-pCR.
When stratified by molecular subtype, the ypN+ rate was 30.8%, 29.6%, 16.8%, 17.5%, and 27.6% in the Luminal A, Luminal B (HER2–), Luminal B (HER2+), HER2-amplified, and triple-negative subtypes, respectively (
Multivariate analyses found that initial clinical stage (
Multivariate analysis of impact factors for ypN+ in breast pathological complete remission patients.

CI, confidence interval; HER2, human epidermal growth factor receptor 2; LABC, locally advanced breast cancer; OR, odds ratio; PR, progesterone receptor; TNBC, triple negative breast cancer.
Axillary pathological response in breast non-pCR patients
The total ypN+ rate was 62.5% in breast non-pCR patients and 29.2%, 71.3%, and 74.2% in the cT1-2N0, cT1-2N1, and LABC subgroups (
Multivariate analyses showed that only initial clinical stage (
Discussion
In our large cohort of breast cancer patients who received NAT, the overall breast pCR rate was 21.8% and the ypN0 rate was 45.9%. Our results suggest that clinical stage and molecular subtype are significantly associated with ypN status in breast pCR patients. Furthermore, in breast pCR patients with cT1-2N0 and HER2-positive disease, 100% of patients were found to achieve ypN0. Our findings thus support that elimination of axillary surgery should be considered in these sub-populations.
Both breast pCR and ypN0 status were significantly associated with survival in breast cancer patients.
2
Many studies have investigated possible factors related to pCR with the view to further improve treatment efficacy. Tumor biology has been an important factor for predicting pathological response in both the breast and axilla. The American College of Surgeons Oncology Group Z1071 study found that the pCR rate was the highest in the HER2-positive subtype (45.4%) and the lowest in the HR-positive/HER2-negative subtype (11.4%), with an intermediate value for the triple-negative subtype (38.2%) (
In clinical practice, the status of nodal residual disease is used to guide the choice of subsequent axillary surgery upon the completion of NAT. Currently, SLNB is recommended in cN0 patients after NAT, with a high detection accuracy and relatively low false-negative rate (FNR).23,24 The ypN+ rate was 24.5% in cT1-2N0 patients in our study, which was relatively higher than other studies.14,15 The reason might be attributed to insufficient treatment for pathological response assessment in these patients, as 68.1% of cT1-2N0 patients in our study received only 4–6 cycles of NAT. A retrospective study from Tadros
Optimal axilla management following NAT in cN+ patients is still controversial. Although the use of SLNB has increased since 2013,18,28,29 several prospective studies have indicated that a low SLNB FNR (<10%) would be difficult to achieve, unless more than two sentinel lymph nodes are examined or dual tracers are applied.30–32 In the current study, all cN+ patients received upfront axillary dissection, with a ypN0 rate of 74.3% in the cT1-2N1 subgroup and of 66.1% in the LABC patients with breast pCR. Nearly two-thirds of axillary dissections might be spared if SLNB was properly done, while elimination of axillary surgery was not supported, even for those HER2+ or TNBC subtype. A study from Piltin
Molecular subtypes and clinical stages also influenced the subsequent adjuvant therapies significantly (Supplemental Table S5). Among the 1532 patients with adjuvant treatment data, anti-HER2 targeted therapy was administrated in 68.8% of HER2 positive patients. Use of adjuvant chemotherapy was significantly associated with NAT cycles, 57.8% in these treated with 4–6 cycles and only 23.5% in patients receiving >6 cycles of NAT (OR = 0.30,
This study has some limitations. First, as a retrospective study, not all of the patients who were included had finished their scheduled NAT regimens. Second, the data in our study were derived from different medical centers, with the lack of a commonly defined protocol for the evaluation of clinical stage and pathological response. Third, some patients who received NAT were excluded due to missing data on treatment, IHC, or surgery, which may have affected the results.
Conclusions
Our study demonstrated that in breast pCR patients after NAT, clinical stage and molecular subtype were significantly associated with ypN status. More importantly, 100% of breast pCR patients with cT1-2N0 and HER2-positive disease achieved ypN0, indicating the possibility of omitting axillary surgery in those selected patients. For other patients with cT1-2N0 disease or breast pCR, SLNB was a recommended procedure under certain conditions. Meanwhile, in breast non-pCR patients with cT1-2N1 or LABC disease, axillary dissection should not be spared.
Supplemental Material
sj-pdf-1-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-1-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-2-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-3-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-4-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-4-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-5-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-5-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-6-tam-10.1177_1758835921996673 – Supplemental material for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery
Supplemental material, sj-pdf-6-tam-10.1177_1758835921996673 for Association between tumor molecular subtype, clinical stage and axillary pathological response in breast cancer patients undergoing complete pathological remission after neoadjuvant chemotherapy: potential implications for de-escalation of axillary surgery by Jin Hong, Yiwei Tong, Jianrong He, Xiaosong Chen and Kunwei Shen in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors are grateful to all the patients who participated in this study. The authors also want to thank all the doctors across the country who contributed patients’ data to the SJTU-BCDB for their work and support for this study.
Author contributions
KWS and XSC developed the main concept and contributed to the study design. JH, YWT, and JRH performed the data analysis and interpretation. JH and YWT drafted the manuscript. XSC and KWS contributed to the editing and critical revision of the manuscript. All authors reviewed and approved the final draft.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81772797); the Shanghai Municipal Education Commission (20172007); and Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (GCQN-2017-A18, GCQN-2018-B11). All these financial sponsors had no role in the study design, data collection, analysis, or interpretation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
