Abstract
Background:
Breast cancer (BC) in young women merits a specific approach given the associated fertility, genetic and psychosocial issues. De novo metastatic breast cancer (MBC) in young women is an even more serious condition, with limited data available.
Methods:
We evaluated management of women aged ⩽40 years with de novo MBC in a real-life national multicentre cohort of 22,463 patients treated between 2008 and 2016 (NCT0327531). Our primary objective was to compare overall survival (OS) in young women versus women aged 41–69 years. The secondary objectives were to compare first-line progression-free survival (PFS1) and to describe treatment patterns.
Results:
Of the 4524 women included, 598 (13%) were ⩽40 years. Median age at MBC diagnosis was 36 years (range = 20–40). Compared with women aged 41–69 years, young women had more grade III tumours (49% versus 35.7%, p < 0.0001), human epidermal growth factor receptor 2 amplified (HER2+) disease (34.6% versus 26.4%, p < 0.0001) and HR–/HER2– disease known as “triple negative breast cancer” (TNBC) (17.1% versus 12.7%, p < 0.0001). BRCA testing was performed for 260 young women, with a BRCA1/2 mutation in 44 (17% of those tested) In young HR+/HER2– patients, chemotherapy (CT) was given as the frontline treatment more frequently compared with older ones (89.6% versus 68.8%, respectively, p < 0.0001). After median follow-up of 49.7 months (95% confidence interval, CI = 48.0–51.7), the median OS of young women was 58.5 months, 20.7 months and not attained in HR+/HER2–, TNBC and HER2+ subgroups, respectively. After adjustment for histological subtype, tumour grade, and number and type of metastasis, young women had significantly better OS compared with older ones, except for the TNBC subgroup, for which the outcome was similar. PFS1 was statistically different only in the TNBC subgroup, with 7.8 months for young women and 6.3 months for older women (p = 0.0015).
Conclusion:
De novo MBC affects a significant proportion of young women. A subgroup of these patients achieves long OS and merits multidisciplinary care.
Introduction
Although breast cancer (BC) mostly affects postmenopausal women, a significant proportion of women with BC are ⩽40 years old (7% of all cases of BC).1 This age cut-off is acknowledged in the fourth consensus guidelines for BC in young women, recognizing that they have specific health issues (fertility, genetics, psychosocial concerns) that often merit a different approach.2 A certain number of particularities have been reported in young BC patients, including more aggressive biology and a higher proportion of triple-negative and human epidermal growth factor receptor 2 amplified (HER2+) forms of cancer.3,4 Younger patients usually present with a more advanced clinical stage than their older counterparts, which further explains the worse outcomes.5,6 Given its low prevalence, characterizing this disease in young women has been tremendously challenging, and current management largely follows treatment algorithms for later life BC. The Advanced Breast Cancer (ABC) guidelines state that treatment decisions in young women should not be driven by their age but rather by the biology of their BC to ensure appropriate and tailored treatment and to avoid overtreatment that often occurs when decisions are driven by age alone.7
Most of our knowledge of BC in young women relies on cases of early BC. Limited evidence is available on metastatic breast cancer (MBC) in young women and data on de novo metastatic disease in this population are even more scarce.8 Only small retrospective series with incomplete data on tumour type, management and prognostic factors have been published so far. The POSH (Prospective study of Outcomes in Sporadic versus Hereditary breast cancer) cohort study conducted at 127 hospitals in the United Kingdom is the largest multicentre prospective observational cohort on invasive BC in women ⩽40 years.9 Of the 2956 patients selected between 2000 and 2008, only 74 (2.5%) had de novo metastatic disease. No details of the type of treatment in the metastatic setting are reported. Given increasing incidence of advanced BC in young women according to the US National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database,10 and the specific issues affecting these women, we aimed to provide a comprehensive analysis of de novo MBC management in young women in a real-life setting. Better knowledge of epidemiology, response to treatment and outcomes in this specific population may improve both management and prognosis.
Materials and methods
Study design
This noninterventional, retrospective, comparative study was conducted to describe the outcome of predefined MBC patients selected from the ESME (Epidemiological Strategy and Medial Economics) database. The ESME-MBC database (NCT03275311) is an ongoing unique national cohort collecting real-life individual retrospective data from all consecutive patients, male or female, ⩾18 years, who started cancer treatment for MBC in one of the 18 cancer centres participating in the ESME research programme from 1 January 2008 to 31 December 2016. Patient-related data, hospitalization-related data and pharmacy-related data are collected, including the patients’ demographic characteristics, pathologies and outcomes. Treatment strategies are also recorded, including CT, targeted agents, endocrine therapy (ET), radiotherapy (RT) and other local treatments, as well as supportive therapies such as bone-targeted agents.
In this study, patient selection focused on female de novo MBC patients, aged <70 years at diagnosis, with full immunohistochemistry (IHC) data available on HR and HER2 status and on first-line treatment. We excluded 56 men, 295 women with missing data concerning first-line treatment or IHC status, and 1473 women aged ⩾70 years at diagnosis. Data were compiled until the cut-off date (15 October 2018), death (if this occurred before the cut-off date) or date of last contact (if lost to follow-up). An independent ethics committee (Comité de Protection des Personnes Sud Est II 2015-79) approved our analysis. No formal informed consent was required, but all patients had approved the reuse of their electronically recorded data. In compliance with French regulations, the ESME-MBC database was authorized by the French data protection authority (Registration ID 1704113 and Authorization N° DE-2013.-117) and managed by Unicancer in accordance with current best practice guidelines.11 Moreover, in compliance with the applicable European regulations, complementary authorization was obtained on 14 October 2019 regarding the ESME research Data Warehouse.
Objectives and endpoints
The study’s primary objective was to compare the overall survival (OS) of de novo MBC in young women versus patients aged 41–69 years, adjusted for other prognostic factors, globally and in the three major MBC subtypes. The secondary objectives were to compare the first-line progression-free survival (PFS1) in young women with de novo MBC versus patients aged 41–69 years, describe treatment patterns and estimate the prognostic factors associated with OS.
OS was the primary endpoint and was defined as the time from the initiation of first-line treatment (or diagnosis in the sensitivity analysis) to death from any cause or last contact (censored data). PFS1 was defined as the time from the initiation of first-line treatment to first disease progression or death from any cause. Patients who were progression-free and still alive at the time of the analysis were censored at the last contact. A treatment line was defined as a given therapeutic strategy followed until progression and could therefore involve several treatments, including CT, targeted agents or ET. De novo metastatic disease was defined as the presence of metastasis at the time or within 3 months (90 days) of primary tumour diagnosis. Disease progression was defined as the appearance of a new metastatic site or progression of pre-existing metastases at least 1 month after starting treatment.
Tumour subtype assessment and treatments
Standard guidelines were applied to any analysis performed within the ESME database. HER2 and hormone receptor (HR) statuses were derived from existing results on metastatic tissue sampling if available or, if unavailable, from last sampling on early disease. BC was HR+ if oestrogen receptor (ER) or progesterone receptor (PR) expression was ⩾10% (IHC), as per European guidelines. An HER2 IHC score of 3+ or an IHC score of 2+ with positive fluorescence in situ hybridization (FISH) or chromogenic in situ hybridization (CISH) classified the tumours as HER2+. BRCA testing was recorded and used for subgroup analysis. Systemic therapies were classified into the following four groups: ET, CT, immunotherapy and targeted therapy (including anti-HER2 therapy, CDK4/6 inhibitors and mTOR inhibitors).
Statistical analysis
Continuous variables were described using median and range (minimum–maximum), and qualitative variables were presented as frequencies and percentages. Patient and clinicopathological characteristics were compared between age groups for the overall population and IHC subgroups, using chi-square or Fisher’s exact test for qualitative variables and the Mann–Whitney test for continuous variables. All statistical tests were two-sided, and a p value <0.05 was considered statistically significant.
Survival data (OS and PFS1) were calculated from the initiation of first-line treatment and estimated using the Kaplan–Meier method, and groups were compared using a two-sided logrank test. We conducted univariable and multivariable analyses to identify the prognostic factors associated with OS and PFS1, using the logrank test and Cox proportional hazards model, respectively. Hazard ratios (HRs) and their 95% confidence intervals (CIs) were estimated using the Cox proportional hazards model. A sensitivity analysis was performed by calculating OS from the diagnosis. All statistical analyses were carried out using Stata (version 16) software.
Results
Patient characteristics
We identified 6348 patients with de novo MBC in the ESME database; 4524 of these patients met the inclusion criteria, with 598 (13%) women ⩽40 years. The flow chart is shown in Figure 1. The median age for young women was 36 years (range = 20–40), and, as expected, the vast majority were premenopausal (97%). Table 1 summarizes the patient characteristics by age group. HR and HER2 status were distributed as follows: HR+/HER2– (48.3%; n = 289 patients), HER2+ (34.6%; n = 207) and HR–/HER2– (17.1%; n = 102). Lobular carcinoma was rare in young women (n = 15; 2.5%). BRCA testing was performed in 260 young women, with a BRCA1/2 mutation identified in 44 (17% of those tested). Other mutation screening was performed in 67 young women, with 12 positive results (=18%), including seven TP53 mutations. Compared with patients aged 41–69 years, young women more frequently had grade III tumours (49% versus 35.7%, p < 0.0001), ER– status (30.1% versus 25.1%, p = 0.0090), HER2+ tumours (34.6% versus 26.4%, p < 0.0001) and TNBC tumours (17.1% versus 12.7%, p < 0.0001). Surgery of the primary tumour was performed more frequently in young women (41.7% versus 31.6%, p < 0.0001). Visceral metastases were found in around 54% of patients, with no difference between the two groups. However, liver metastases at diagnosis were more frequent in young women (38.1% versus 30.7%, p = 0.0003). Ten percent of young women were included in clinical trials versus 8.4% of older women (p = 0.1864). Median time between metastatic diagnosis and treatment initiation was 0.7 months (range = 0–42 months).

Flow chart.
Patient characteristics.

ER, oestrogen receptor; HER2, human epidermal growth factor receptor 2 amplified; HR, hormone receptor; NA, not available; PR, progesterone receptor.
Patient management
HR+/HER2– disease
The vast majority of young patients (89.6%) received frontline CT, and strikingly only 10.4% received ET-based treatment alone as initial treatment. The pattern of treatment was significantly different in women aged 41–69 years (CT = 68.9% and Et alone = 31.1%). The majority of patients treated with CT also received ET given sequentially after achieving initial response for CT. Given the inclusion period for the database, only 1.3% received CDK4-6 inhibitors during the course of the disease in the total population. Frontline CT in young women included taxane (27.4%), anthracycline (34%), poly-CT with taxane + anthracycline (37%) or other CT regimens (1.5%). ET was mainly tamoxifen (81.1%), and almost half of the patients had ovarian suppression with gonadotropin-releasing hormone (GnRH) analogues (data on oophorectomy rate were not available). In contrast, for women aged 41–69 years, the most commonly used ET was aromatase inhibitor (80%).
HR–/HER2– disease
First-line treatment included CT alone in 64.7% or combined with bevacizumab in 29.4% of young patients. Young women more frequently received poly-CT (taxane + anthracycline) compared with women aged 41–69 years: 47.7% versus 35%, respectively. Few women received platinum CT as the first line (13.7% for young women and 6.5% for older women, p = 0.0116). As the cut-off for HR negativity was <10%, few women in both groups received ET (2% in young women and 2.6% in older women).
HER2+ disease
Of the 207 young patients with HER2+ disease, 196 (94.7%) received frontline anti-HER2-based treatment, of whom 80 (40.8%) received double blockade anti-HER2 (trastuzumab plus pertuzumab), while 113 (57.7%) received trastuzumab alone. This was similar to older women, with 92.6%, 39.8% and 59.3%, respectively. The most common CT partner was taxane, for almost three-quarters of the patients.
Table 2 summarizes the different treatments received as the first line for each histological subtype.
Treatment modalities in first line.

ET, endocrine therapy; GnRH, gonadotropin-realizing hormone; HER2, human epidermal growth factor receptor 2 amplified; HR, hormonal receptor; TT, target therapy.
OS analysis
After median follow-up of 49.7 months (95% CI = 48–51.7), 2144 deaths were reported (249 in the ⩽40 years group versus 1895 in the 41- to 69-year-old group). Median OS was 50.5 months (95% CI = 48.5–52.9 months), 59.9 months (95% CI = 52.7–66.1 months) and 49.1 months (95% CI = 47.2–52.1 months) in the whole population, young women and women aged 41–69 years, respectively. Table 3 summarizes patients’ outcome per tumour subtype and age group. Young women had a numerically better 5-year OS rate compared with women aged >40 years, reaching 50%, 63% and 24% in the HR+/HER2–, HER2+ and TNBC subtypes, respectively. Figure 2 shows the Kaplan–Meier curves for OS. The results were similar in the sensitivity analysis by calculating OS from the MBC diagnosis (Supplementary Figure 1).

Kaplan–Meier curves for overall survival (OS) per age group in the entire patient cohort and in each tumour subtype group.
Patient outcome per age group and tumour subtype.

CI, confidence interval; HER2, human epidermal growth factor receptor 2 amplified; HR, hormone receptor.
In the univariable analysis, young age, the HR+/HER2– and HER2+ subtypes, low grade (I/II versus III), number of metastatic sites <3 and absence of visceral metastases were associated with better OS in the whole de novo MBC population (Table 4). In the multivariable analysis, age 41–69 years remained an independent risk factor for OS in the whole (HR = 1.28, 95% CI = 1.11–1.47), HR+/HER2– (HR = 1.29, 95% CI = 1.06–1.58) and HER2+ populations (HR = 1.37, 95% CI = 1.03–1.83) (see Table 5 and Figure 3). Conversely, young age was not associated with better OS in the TNBC subgroup.

Forest plot for multivariable subgroup analysis of OS according to tumour subtype and treatment pattern. The diamonds in the forest plot represent the HRs comparing age groups (41–69 versus ⩽40 years) adjusted for histological grade, number and type of metastases; tumour subtype in the overall population; or HR status in the HER2+ subgroup. The horizontal lines represent the 95% CIs.
Univariable analysis of the factors associated with overall survival in the population.
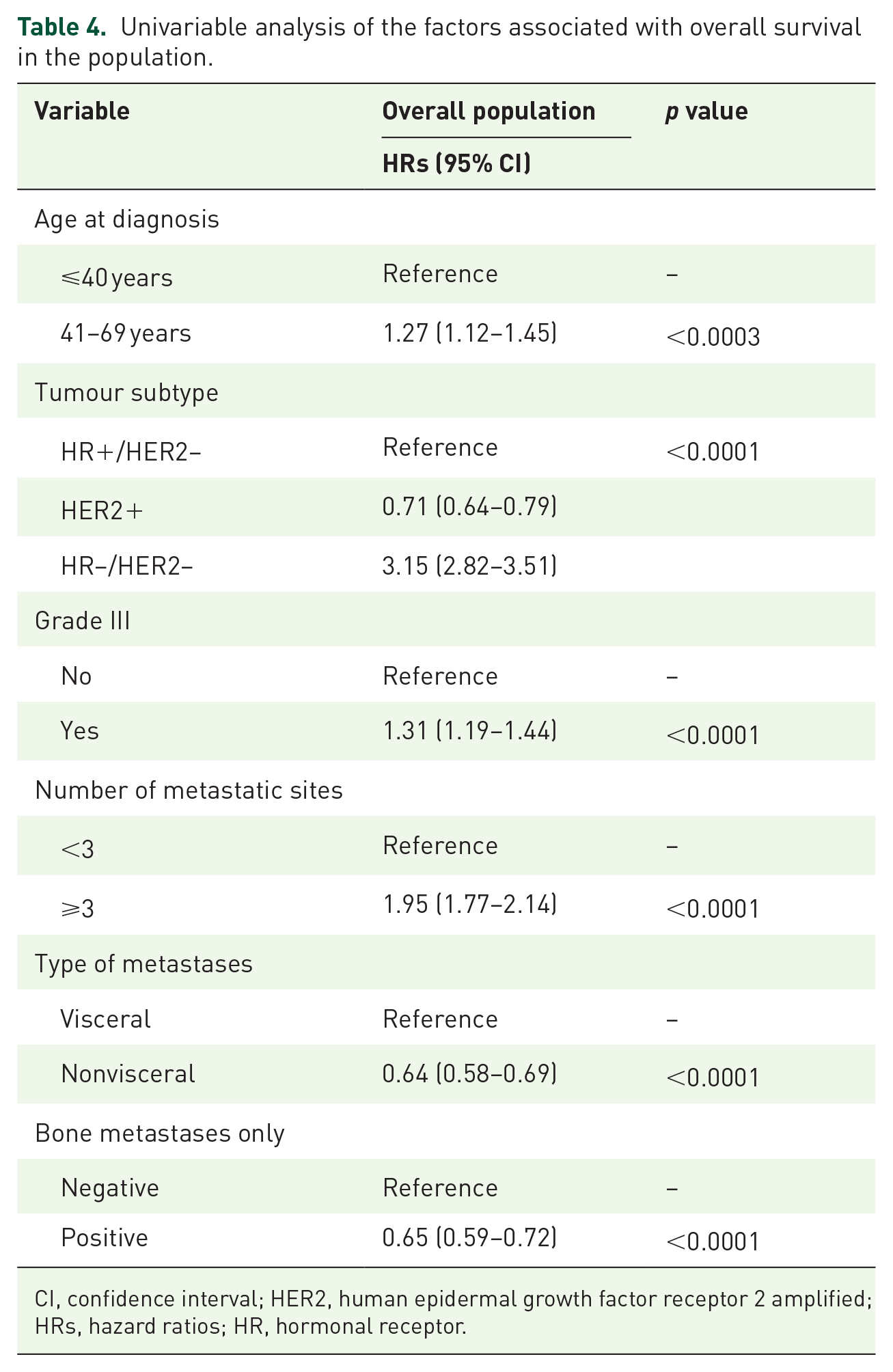
CI, confidence interval; HER2, human epidermal growth factor receptor 2 amplified; HRs, hazard ratios; HR, hormonal receptor.
Multivariable Cox model analysis of overall survival.

CI, confidence interval; HRs, hazard ratios; HER2, human epidermal growth factor receptor 2 amplified; HR, hormonal receptor; IHC, immunohistochemistry.
PFS1 and systemic treatment
Globally, women aged ⩽40 years had no statistical difference in PFS1 – median PFS1: 14.8 months (95% CI = 13.3–17.0) – compared with older women aged 41–69 years – median PFS1: 13.8 months (95% CI = 13.1–14.4), p = 0.0591 (Table 3). In the TNBC subgroup, however, PFS1 was statistically different between the two age groups. Figure 4 shows the Kaplan–Meier curves for PFS1. Of note in the HR+/HER2-patients, PFS1 in young patients with frontline CT +/– ET was 14.4 months compared with 7.6 months for young women treated with frontline ET. For TNBC, PFS1 with CT + bevacizumab was 4.9 versus 6.4 months in young women and in women aged 41–69 years, respectively.

Kaplan–Meier curves for progression-free survival (PFS) per age in the entire patient cohort and in each tumour subtype group.
Management of BRCA patients and outcome
In the whole population, 96 patients had a BRCA1/2 mutation, of whom 44 were ⩽40 years. Young BRCA patients frequently had grade III tumours (63.4%, compared with 49% in the overall population of young women). The majority had HR+/HER2– and TNBC tumours (52.3% and 43.2%, respectively), and only two patients (4.5%) had HER2+ tumours. In comparison with older BRCA patients, young BRCA women had fewer HR+/HER2– tumours (52.3% versus 71.2%) and twice as many TNBC tumours (43.2% versus 17.3%). They had few metastatic sites (<3 for 95.5%), with 50% of visceral metastases and nonvisceral metastases. Older BRCA women had more bone metastases than younger ones (73.1% versus 43.2%, p = 0.0030). The vast majority of both young and older BRCA women received CT as first-line treatment (97.7% versus 86.5%, p = 0.07).
Discussion
Our study, involving around 600 young women with de novo MBC, is one of the widest and most comprehensive published so far. Young women with de novo MBC have better OS compared with their older counterparts. However, this OS advantage is restricted to women with HR+/HER2– and HER2 + BC, unlike the TNBC subgroup, which maintains a poor prognosis regardless of the age of the patients at diagnosis. Our analysis gives significant insight into a clinical situation concerning 13% of women with de novo MBC.
The better survival observed in young patients with de novo MBC differs somewhat from the data on young women with early BC. In a recent analysis based on the SEER database from 2004 to 2008, patients aged <30 and 30–39 years had significantly lower overall and BC-specific survival rates than patients aged 40–49 and 50–59 years.12 There are several possible explanations for this less favourable prognosis, including the higher proportion of grade III tumours or triple-negative subtype, diagnostic delay and decreased adherence to adjuvant ET.13,14
Several studies have reported an OS advantage for young women in the context of de novo MBC. Analysing the SEER database focusing on the de novo MBC highlighted an advantage in OS for young women with de novo MBC with the exception of the TNBC subgroup.15–18 Similar results have been reported in various cohort studies after adjustment for tumour subtypes. In a study by Ogiya, median OS was significantly better in young women (45 versus 33 months, p < 0.0001). This difference remained statistically significant after adjustment for HR and HER2 status: adjusted HRs are 0.75 (95% CI = 0.64–0.88), 0.5 (95% CI = 0.37–0.67) and 0.65 (95% CI = 0.46–0.93) in the HR+/HER2–, HR+/HER2+ and HR–/HER2+ subtypes, respectively. Again, young patients with de novo metastatic TNBC had the same prognosis as their older counterparts: adjusted HR = 0.99 (95% CI = 0.8–1.22), p = 0.91.15 The study by Freedman et al.18 showed similar results with an adjusted HR for BC-specific death of 0.78 (95% CI = 0.72–0.85) and 0.80 (95% CI = 0.75–0.84) in the metastatic setting for women aged ⩽35 and 35–44 years compared with women aged 55–64 years. The reasons for this better outcome, despite a more aggressive tumour phenotype, are not yet fully known and are probably multifactorial. Young women have fewer comorbidities and are probably able to receive more, and more aggressive, lines of treatment. Staging modalities may play an additional role. Conventional imaging procedures for disease staging include contrast-enhanced computed tomography, and bone scans in women with clinically positive axillary nodes, large tumours or clinical signs/symptoms.19 Young women may benefit from increasing use of imaging for staging (even in early-stage BC) and improvements in diagnostic imaging technology – with more positron emission tomography (PET)/computed tomography performed – leading to stage migration as described by Will Rogers.20,21
As metastatic disease remains mostly incurable, accurate management of these patients is crucial for making possible the longest OS with acceptable treatment toxicity. Approximately 40% of the young women in our cohort underwent surgery of the primary tumours, which seems higher than in reported series. This may reflect a more aggressive strategy in these patients, given their age. Managing the primary tumour site in patients with MBC remained controversial, with mixed results regarding the impact on outcome. Some meta-analyses of retrospective studies with limitation bias suggested that breast surgery may improve the survival of women with MBC.22–24 However, more recently, a randomized study of de novo MBC and two new meta-analyses reported contradictory results,25–27 with no clear benefit for locoregional treatment. Moreover, as shown in another ESME study,28 patients who undergo surgery are often those with favourable prognostic factors (low number of metastatic sites, histological subtype and age).
Our analysis has highlighted another divergence in the management of young patients. Contrary to their older counterparts, only a few young women with HR+/HER2– MBC received frontline endocrine-based treatment. The guidelines for BC in young women repeatedly state that young age alone should not be a reason for prescribing more aggressive treatment. CT in HR+/HER2– disease must be used only in cases of visceral crisis. The Monaleesa-7 study has shown that ET + ribociclib provided median progression-free survival (PFS) of 23.8 months in premenopausal women and increased OS compared with Et alone.29 This study was one of the first dedicated to premenopausal patients, and out of the 672 randomized patients, 186 were <40 years. In the subgroup of de novo MBC patients, the results were even more impressive, with an HR of 0.43 (95% CI = 0.29–0.64). In our study, the median PFS of patients receiving CT +/– ET was 14.4 months and was similar to the placebo arm of the Monaleesa-7 study with tamoxifen alone (13.0 months). In addition, the young Pearl study has demonstrated the superiority of ET + palbociclib over capecitabine in premenopausal women with HR+/HER2– MBC in terms of PFS.30 Our patients were treated between 2008 and 2016, a pre CDK4-6 era, explaining the low rate of patients treated with CDK4-6 inhibitors. Only 30 patients received frontline ET with a modest PFS of 7.6 months (95% CI = 6.2–16.3) without clear explanations.
This difference in terms of management according to age was also observed in the TNBC subgroup. Young women received more poly-CT than older women, although combination regimens have not shown any benefit to OS compared with monotherapies, including our study. In the HER2 subgroup, 25% of the young women also received poly-CT in combination with anti-HER2 target therapy, while the reference studies involved mono-CT.31,32 As shown in previous ESME studies, the median OS of patients with HER2+ disease was higher than the HR+/HER2– population even in young patients.33 The OS advantage is probably explained by the several breakthrough therapies over the 10 past years in the HER2+ disease, including pertuzumab and trastuzumab emtansine (TDM1). The real impact of CDK4-6 inhibitors on OS in the HR+/HER2– population will be measurable in the future in this database with the widespread of CDK4-6 inhibitor use.
The ESME-MBC programme represents a very large-scale ongoing multicentre cohort, with one of the largest numbers of MBC patients ever included in a retrospective analysis for outcome estimates. The centralized real-life data are both exhaustive and of high quality, providing consistency with the real world. However, the limitations of this study must be considered when interpreting its results, the main one being its retrospective and observational nature. Information on subtype classification and treatment was collected retrospectively, which can mean missing data. Moreover, this was a multicentre study with 18 Comprehensive Cancer Centres in a single country, making this cohort representative of de novo MBC patients across France, but not necessarily across other countries.
Conclusion
Our comprehensive retrospective study is the largest report on young women diagnosed with de novo MBC. This population has specific features, with a distinct presentation and better prognosis than in older women, especially in the HR+/HER2– and HER2+ subgroups. A significant proportion of young women are treated more aggressively because of their age, while the intensity of the treatment received does not seem to be a decisive factor for improving OS.
Supplemental Material
sj-jpg-1-tam-10.1177_17588359211070362 – Supplemental material for Real-world evidence of the management and prognosis of young women (⩽40 years) with de novo metastatic breast cancer
Supplemental material, sj-jpg-1-tam-10.1177_17588359211070362 for Real-world evidence of the management and prognosis of young women (⩽40 years) with de novo metastatic breast cancer by Amélie Mallet, Amélie Lusque, Christelle Levy, Barbara Pistilli, Etienne Brain, David Pasquier, Marc Debled, Jean Christophe Thery, Anthony Gonçalves, Isabelle Desmoulins, Thibault De La Motte Rouge, Christelle Faure, Jean Marc Ferrero, Jean Christophe Eymard, Marie Ange Mouret-Reynier, Anne Patsouris, Paul Cottu, Florence Dalenc, Thierry Petit, Olivier Payen, Lionel Uwer, Séverine Guiu and Jean Sébastien Frenel in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank the 18 French Comprehensive Cancer Centres (FCCC) for providing the data and each ESME local coordinator for managing the project at the local level. Moreover, we thank the ESME Scientific Committee members for their ongoing support. The 18 participating FCCC are as follows: I. Curie, Paris/Saint-Cloud; G. Roussy, Villejuif; I. Cancérologie de l’Ouest, Angers/Nantes; C. F. Baclesse, Caen; ICM Montpellier; C. L. Bérard, Lyon; C. G.-F. Leclerc, Dijon; C. H. Becquerel, Rouen; I. C. Regaud, Toulouse; C. A. Lacassagne, Nice; Institut de Cancérologie de Lorraine, Nancy; C. E. Marquis, Rennes; I. Paoli-Calmettes, Marseille; C. J. Perrin, Clermont-Ferrand; I. Bergonié, Bordeaux; C. P. Strauss, Strasbourg; I. J. Godinot, Reims; C. O. Lambret, Lille.
ESME central coordinating staff
Programme director: Anne-Laure Martin.
Data management team: Coralie Courtinard, Emilie Nguyen, Olivier Payen, Irwin Piot, Dominique Schwob and Olivier Villacroux.
Operational team: Nathalie Bouyer, Michaël Chevrot, Daniel Couch, Patricia D’Agostino, Pascale Danglot, Levent Dinc, Tahar Guesmia, Elodie Kupfer, Harmonie Oulie, Frédéric Roy, Gaëtane Simon and Toihiri Said.
Software designer team: Matou Diop, Blaise Fulpin, Fréja Messo, José Paredes and Alexandre Vanni.
ESME local coordinators
Thomas Bachelot, Delphine Berchery, Etienne Brain, Mathias Breton, Loïc Campion, Emmanuel Chamorey, Sandrine Dabakuyo, Valérie Dejean, Stéphanie Delaine, Anne-Valérie Guizard, Anne Jaffré, Lilian Laborde, Carine Laurent, Marie-Paule Lebitasy, Agnès Loeb, Damien Parent, Geneviève Perrocheau, Marie Ange Mouret-Reynier, Michel Velten.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by R&D UNICANCER. The ESME-MBC database is supported by an industrial consortium (Roche, Pfizer, AstraZeneca, MSD, Eisai and Daiichi Sankyo). Data collection, analysis and publication are fully managed by R&D UNICANCER independently of the industrial consortium.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
