Abstract
Background:
99mTechnetium labeled methylene diphosphonate bone scans (BSs) are commonly used to monitor disease progression in bone for patients with metastatic breast cancer (MBC). However, new BS lesions may represent osteoblastic bone healing, which we now define as bone pseudoprogression. In this study, we aimed to assess the clinical significance and determination methods of bone pseudoprogression.
Methods:
This retrospective analysis was conducted among 48 patients with hormone receptor-positive MBC treated with first-line endocrine therapy. Four months after initiating therapy, all the participants did not show extraosseous disease progression. Participants were divided into two groups according to the presence of new BS lesions. All the patients continued on treatment until explicit disease progression (extraosseous disease progression or progressive lysis on bone lesions). Explicit progression-free survival (PFS) and extraosseous objective response rate were analyzed between the two groups.
Results:
New BS lesions were observed in 11 of 48 (22.9%) patients. All the new BS lesions appeared as osteoblastic bone lesions on computed tomography. For patients with new BS lesions, the median PFS was 26.57 months [95% confidence interval (CI) 15.46–37.68], which was similar to that (29.57 months; 95% CI 19.24–39.90) in patients without new BS lesions [hazard ratio: 1.098 (95% CI 0.482–2.503), p = 0.818]. Notably, 82.9% of patients without new BS lesions showed an extraosseous objective response, whereas 85.7% of patients with new BS lesions demonstrated an extraosseous objective response [odds ratio: 0.806 (95% CI 0.061–5.682), p = 0.999]. The median interval between bone pseudoprogression and true disease progression was 21.26 months (95% CI 10.11–32.42).
Conclusions:
Osteoblastic new BS lesions detected on follow-up BSs may represent bone pseudoprogression. Clinicians should raise awareness of bone pseudoprogression, thereby avoiding premature discontinuation of therapy and maximizing the opportunity to benefit from endocrine therapy. Due to the small sample size and retrospective nature of the study, large prospective clinical trials are needed to confirm our findings.
Keywords
Introduction
Bone is the predominant metastatic site for breast cancer. Between 39.0% and 71.4% of patients with metastatic breast cancer (MBC) develop bone metastasis during the course of the disease. 1 Complications of bone metastasis include pathological fractures, spinal cord compression, and hypercalcemia, 2 all of which can greatly impair the quality of life of the patient. Breast cancer with bone metastases remains virtually an incurable disease. However, endocrine therapy, chemotherapy and radiotherapy could be used to delay disease progression.
99mTechnetium labeled methylene diphosphonate bone scans (BSs) are commonly used for screening new bone lesions and monitoring disease progression in bone. However, the results of BSs can sometimes be misleading. In other words, the BS could exhibit false-positive results. Ryan et al. 3 showed that, among patients with metastatic prostate cancer who received abiraterone acetate treatment, 48% patients showed new lesions or increased tracer uptake on post-treatment BSs, which were discordant with the decline in prostate-specific antigen (PSA) levels. Coleman et al. 4 showed that, for patients with MBC who received systemic therapy for bone metastases, 30.2% patients showed healing of osteolytic bone lesions on X-ray, with a paradoxical deterioration on the BS. As a result, the Prostate Cancer Clinical Trials Working Group 3 gave the following recommendations: in the absence of extraosseous disease progression, new BS lesions alone do not constitute disease progression, and patients should continue with the prior therapy. 5 Progression of bone metastasis is only considered when two more new lesions are detected relative to the previous BS.
The false-positive results on BSs could be mistakenly diagnosed as disease progression, which might lead to premature discontinuation of therapy and negatively affect clinical outcomes. It is of great need to differentiate false-positive results from true disease progression. BSs do not directly show bone lesions, but instead visualize skeletal vascularity and osteoblastic metabolism. 6 An effective therapy of bone metastases would lead to increased osteoblastic bone healing, thus causing the false-positive results on BSs. 7 Accordingly, for the first time, we defined osteoblastic (confirmed by computed tomography) new BS lesions, without additional new lesions on subsequent BSs, as bone pseudoprogression.
For patients with hormone receptor (HR)-positive MBC, endocrine therapy has always been the preferred option. Recently, several cyclin-dependent kinase 4/6 inhibitors (CDK4/6i) achieved satisfactory results in HR-positive MBC. Pivotal randomized clinical trials have shown that the efficacy of CDK4/6i plus endocrine therapy is better than that of endocrine therapy alone, including palbociclib-based PALOMA-1, 8 PALOMA-2, 9 and PALOMA-3 trials; 10 ribociclib-based MONALEESA-2, 11 MONALEESA-3, 12 and MONALEESA-7 trials; 13 and abemaciclib-based MONARCH-214 and MONARCH-3 trials. 15
In this study, we assessed the determination methods and clinical significance of bone pseudoprogression in HR-positive MBC, with the goal of avoiding the premature discontinuation of therapy and maximizing the benefit of endocrine treatment with or without CDK4/6i.
Methods
Cohort population
From 1 September 2015 to 30 September 2020, we conducted a retrospective analysis of 60 patients mainly from one phase III, randomized controlled CDK4/6i trial (NCT02763566, NCT02297438) at Fudan University Shanghai Cancer Center (FUSCC). NCT02297438 was a randomized controlled trial studying palbociclib (125 mg once daily for 21 days followed by 7 days off) plus letrozole (2.5 mg per day) versus placebo plus letrozole. NCT02763566 was a randomized phase III trial, which compared the efficacy of abemaciclib (150 mg twice daily) plus endocrine therapy (letrozole 2.5 mg/day or fulvestrant 500 mg/day) versus placebo plus endocrine therapy. All the patients were postmenopausal women with HR-positive, human epidermal growth factor receptor 2 (HER2)-negative MBC who received first-line treatment.
Computed tomography (CT) and magnetic resonance imaging (MRI) were performed at baseline and every two to three cycles thereafter to assess the extraosseous disease. Extraosseous disease was evaluated according to RECIST version 1.1 criteria. 16 Complete response (CR) was defined as the disappearance of all target lesions. Partial response (PR) was defined as the sum of diameters of target lesions that reached 30% or more decrease, taking the baseline sum diameters as reference. Progressive disease (PD) was defined as the sum of diameters of target lesions that reached 20% or more increase (absolute increase of at least 5 mm), taking the smallest sum as reference. The appearance of new lesions was also considered disease progression. Stable disease (SD) was defined as neither sufficient decrease to reach the standard of PR nor sufficient increase to reach the standard of PD. Patients with extraosseous disease progression before or at the time of the first post-treatment BS were excluded from the study. Patients with insufficient follow-up (<3 months), and patients who discontinued treatment because of the new BS lesion were also excluded. In total, 48 patients were included in our study.
All procedures performed in studies involving human participants were carried out in accordance with the ethical standards of the FUSCC Ethics Committee (approval no. 1606161-7-1811G) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Data were collected from electronic patient records. All patient demographics and disease characteristics were retrieved retrospectively, including age, Eastern Cooperative Oncology Group (ECOG) status, histological subtype, Tumor-node-metastasis (TNM) stage at initial diagnosis of cancer, prior neoadjuvant or adjuvant chemotherapy (yes or no), previous endocrine therapy, disease-free survival, metastatic organ sites and the presence of visceral metastasis. Written informed consents about the reuse of their electronically recorded data were obtained from all patients.
Analysis of bone pseudoprogression
For all the included patients, 99mtechnetium labeled methylene diphosphonate BSs were performed at baseline and every 4 months thereafter. The new BS lesion would be reexamined by CT (bone window). We defined osteoblastic (confirmed by CT) new BS lesion, without additional new lesions on subsequent BSs, as bone pseudoprogression. Participants were divided into two groups according to the presence of bone pseudoprogression. All the patients continued on treatment until explicit disease progression (extraosseous disease progression or progressive lysis on bone lesions). Explicit progression-free survival (PFS) and extraosseous objective response rate (ORR) were analyzed between the two groups.
Statistical analysis
Explicit PFS was defined as the interval between treatment initiation and the date of explicit disease progression (extraosseous disease progression or progressive lysis on bone lesions) or death for any reason. The median PFS and the 95% confidence intervals (CIs) were calculated using the Kaplan–Meier method and compared using log-rank tests. Hazard ratios (HRs) with two-sided 95% CIs were calculated by Cox proportional hazards models. Additional stratified multivariate Cox regression models of PFS were adjusted according to CDK4/6i treatment (yes versus no), presence of visceral metastasis (yes versus no), and metastatic organ sites (1 versus ⩾2). Extraosseous ORR was defined as the percentage of patients who had a CR or PR on extraosseous disease. The differences of extraosseous ORR between the two groups were compared using the Pearson’s chi-squared test (or Fisher’s exact test). All tests were two-sided, and results with p values less than 0.05 were considered statistically significant.
Results
Patient characteristics
This retrospective analysis was conducted among 60 patients with HR-positive MBC who received first-line endocrine therapy. BSs and CT (bone window) were used to assess bone lesions every 4 months. CT or MRI were used to assess extraosseous disease every two to three cycles. Four months after initiating therapy, 50 patients did not show extraosseous disease progression. Of these 50 patients, 13 (26%) patients had new BS lesions. All the new BS lesions appeared as osteoblastic bone lesions on CT (bone window). Eleven of these patients continued with the treatment. As a result, 48 patients who did not show extraosseous progression, with or without new BS lesions, were included in this study (Figure 1).
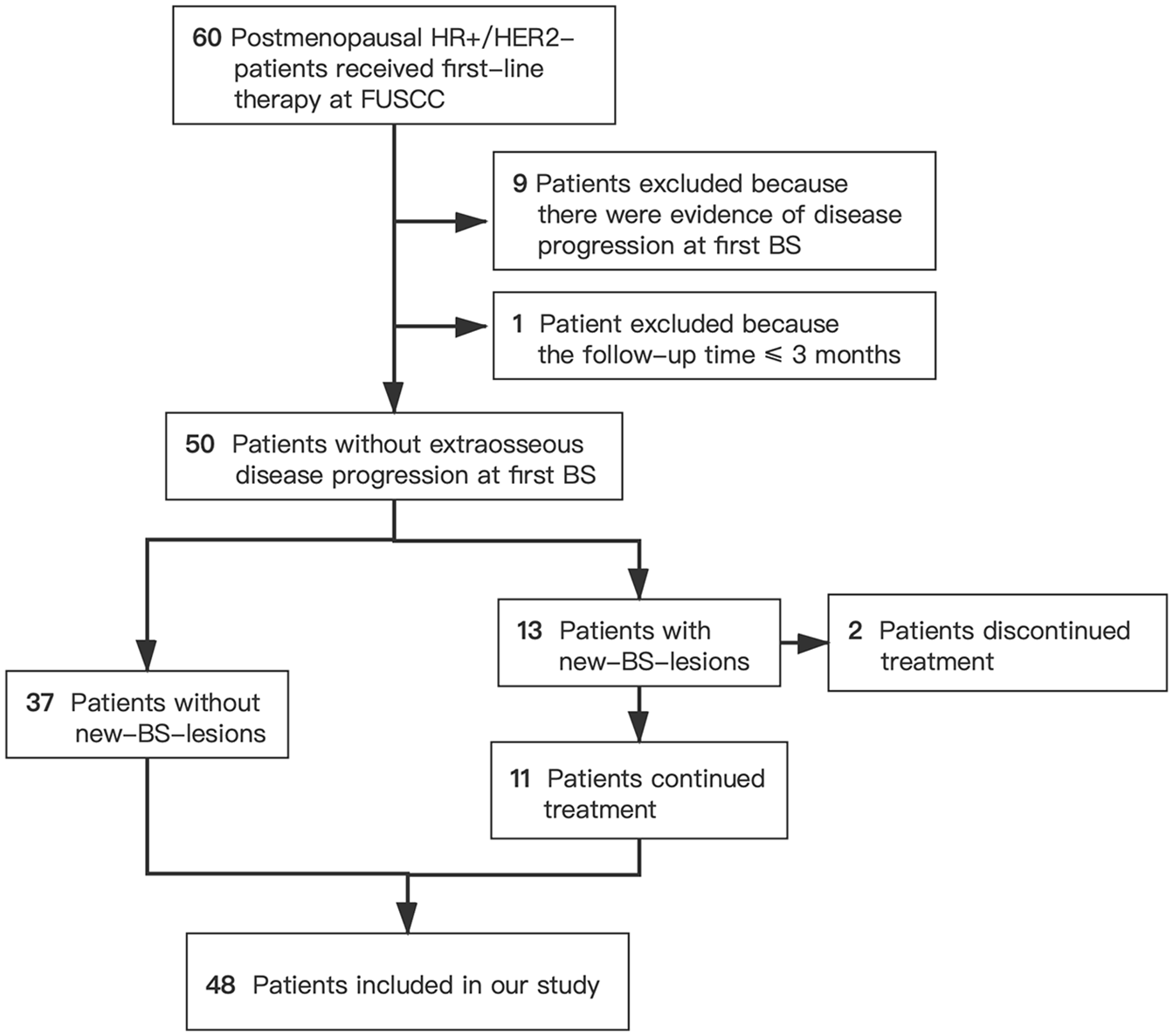
Patient selection flowchart.
Baseline patient characteristics, including age, ECOG status, histological subtype, TNM stage at initial diagnosis of cancer, prior neoadjuvant or adjuvant chemotherapy (yes or no), previous endocrine therapy, disease-free survival, metastatic organ sites and the presence of visceral metastasis, were similar between patients without and with new BS lesions (Table 1). All patients were postmenopausal women with HR-positive MBC. The first-line therapy regimens were also well balanced between the two groups. Among patients without new BS lesions, 21 (56.8%) patients received CDK4/6i plus endocrine therapy as first-line therapy. Among patients with new BS lesions, seven (63.6%) patients received CDK4/6i plus endocrine therapy as first-line therapy.
Patient baseline demographics and disease characteristics.

Based on the American Joint Committee on Cancer Staging Manual, 8th ed.
AI, aromatase inhibitor; ALP, alkaline phosphatase; BS, bone scan; CDK4/6i, cyclin-dependent kinase 4/6 inhibitor; ECOG, Eastern Cooperative Oncology Group; FULV, fulvestrant; LDH, lactate dehydrogenase; NSAI, non-steroidal aromatase inhibitor; SD, standard deviation; TAM, tamoxifen; TNM, Tumor-node-metastasis.
Effects of new BS lesion on efficacy
The median follow-up was 25.4 months (interquartile range 16.9–31.8), at which time three (27.3%) patients in the pseudoprogression group and 14 (37.8%) patients in the non-pseudoprogression group remained on the treatment. The median explicit PFS was 26.57 months (95% CI 15.46–37.68) in the pseudoprogression group, which was similar to that (29.57 months; 95% CI 19.24–39.90) in the non-pseudoprogression group [HR: 1.098 (95% CI 0.482–2.503), p = 0.818]. A stratified multivariate Cox regression model was used to account for imbalanced baseline characteristics, and a similar HR was observed [HR: 1.366 (95% CI 0.577–3.236), p = 0.478]. In addition, extraosseous ORRs did not significantly differ between patients with and without new BS lesions. For patients with measurable disease, 82.9% of patients in the non-pseudoprogression group showed an extraosseous objective response (PR or CR), and 85.7% patients in the pseudoprogression group demonstrated an extraosseous objective response [odds ratio (OR): 0.806 (95% CI 0.061–5.682), p = 0.999]. The efficacy data are summarized in Table 2 and Figure 2. The median interval between bone pseudoprogression and true disease progression was 21.26 months (95% CI 10.11–32.42; Supplemental Figure 1).
Summary of efficacy analyses.

BS, bone scan; CDK4/6i, cyclin-dependent kinase 4/6 inhibitor; ET, endocrine therapy.

Progression-free survival in patients with lack of extraosseous disease radiographic progression, with or without new BS lesions: (a) all patients, (b) patients who received CDK4/6 inhibitors plus endocrine therapy, and (c) patients who received placebo plus endocrine therapy.
Subgroup analyses were performed in patients who received CDK4/6i plus endocrine therapy as first-line therapy and patients who received placebo plus endocrine therapy. For patients who received CDK4/6i plus endocrine therapy, PFS tended to improve in patients with bone pseudoprogression (56.23 months) compared with that in patients without bone pseudoprogression [29.57 months; 95% CI 16.01–43.05; HR: 0.835 (95% CI 0.278–2.512), p = 0.752]. After adjusting for imbalanced baseline characteristics, a consistent HR was observed [HR = 0.765 (95% CI 0.231–2.536), p = 0.661]. Among patients who received placebo plus endocrine therapy, the PFS was 18.06 months (95% CI 7.42–25.18) in patients with pseudoprogression and 24.05 months (95% CI 11.10–33.84) in patients without pseudoprogression [HR = 2.711 (95% CI 0.564–13.02), p = 0.061]. The adjusted HR was 2.724 (95% CI 0.694–10.687; p = 0.151).
Morphology of the new BS lesions
New BS lesions were most frequently detected in the ribs (36.4%) and long bones (36.4%), followed by the spine, pelvis, and sternum. Four (36.4%) patients had more than two new BS lesions. The characteristics of the new BS lesions are summarized in Table 3.
Characteristics of the new BS lesions.

BS, bone scan.
For 11 patients with new BS lesions on the first post-treatment BS, no progression was detected on the second post-treatment BS. For example, a 60-year-old patient with HR-positive MBC was treated with CDK4/6i plus letrozole as first-line therapy. The baseline BS showed metastasis to the spine, ribs, skull, and sternum. Four months after initiating the first-line CDK4/6i combined therapy, a BS revealed two new BS lesions in the pelvis (Figure 3). However, concomitant CT (bone window) analysis showed osteoblastic bone lesions that represented bone healing. The patient continued with the regimen, and the second post-treatment BS showed no new BS lesions. After 31.5 months, the patient remained on treatment and did not show explicit disease progression.

Case report of one patient showing bone pseudoprogression in (a) bone scans and (b) computed tomography.
Discussion
Whether new BS lesions represent true disease progression or osteoblastic bone healing can be challenging to determine. Unfortunately, few studies have been conducted to answer this clinically relevant question. In this study, we systematically evaluated the association between osteoblastic new BS lesions and treatment efficacy. The findings of our study suggested that osteoblastic (confirmed by CT) new BS lesions, without additional new lesions on subsequent BSs, might represent bone pseudoprogression. To our knowledge, this was the first study to propose the specific definition of bone psedoprogression in MBC.
The false-positive result of BSs was first described in 1972. 17 Subsequently, Schneider et al. 18 conducted a phase II trial of taxol chemotherapy in patients with MBC and found that 14.3% of patients with clinical response (⩾50% reduction in the size of the solid tumor) showed paradoxical new BS lesions. In addition, Vogel et al. 19 retrospectively studied patients with MBC who received first-line hormonal therapy in the clinical trial setting and showed that 29% patients had a possible false-positive result of BSs. These studies all helped confirm the real existence of bone pseudoprogression. However, none of these studies systematically compared the treatment efficacy (PFS, ORR) between patients with and without bone pseudoprogression, thence showing less credibility.
In our study, several findings were notable. First, 22.9% of patients showed bone pseudoprogression on follow-up BSs. Clinicians should raise awareness of the bone pseudoprogression phenomenon, thereby avoiding premature discontinuation of the therapy. Second, we recommended using 99mtechnetium labeled methylene diphosphonate BSs for early detection of bone metastasis, and CT (bone window) for evaluating treatment efficacy on early bone metastases, because CT (bone window) can yield superior detail regarding skeletal abnormalities. The sclerosis of osteolytic bone lesions on CT generally suggests bone healing and response to treatment, and the progressive lysis generally suggests disease progression. 20 Moreover, CT could detect medular bone metastases 21 (even though they are more apparent on MRI) at the beginning of the treatment, which could help discriminate bone pseudoprogression. Third, when the follow-up BS produces equivocal findings (bone pseudoprogression or true disease progression), a second post-treatment BS may be taken to re-examine bone pseudoprogression after another 3 months. The first post-treatment BS could be used as the baseline. A lack of new bone lesions on the second post-treatment BS could also help distinguish pseudoprogression from true progression.5,22
For patients who received endocrine therapy alone, PFS tended to be improved in patients showing non-pseudoprogression (mPFS of 24.05 months) versus that in patients showing pseudoprogression [mPFS of 18.06 months, HR = 2.711 (95% CI 0.564–13.02), p = 0.061]; however, these findings should be interpreted with caution. The difference in PFS between the two groups could be attributed to imbalances in baseline characteristics. For example, the four patients with bone pseudoprogression had visceral metastasis and more than one metastatic organ site at baseline. After a stratified multivariate Cox regression model was used to account for imbalances in baseline characteristics, the HR was 2.724 (95% CI 0.694–10.687, p = 0.151).
Our novel research shed light on the direction of future research, so as to differentiate bone pseudoprogression accurately from true progression. First, improved functional imaging of the bone metastasis is needed. The whole-body MRI with diffusion weighted imaging (DWI) showed high accuracy in the detection of bone metastasis. 23 However, bone pseudoprogression can appear on MRI because of marrow reconversion. 24 With high bone uptake and rapid blood clearance, 18 F-sodium fluoride (NaF) positron emission tomography (PET) is an ideal tool for bone metastasis imaging. 25 However, several studies still showed bone pseudoprogression on 18 F-NaF PET.26,27 Second, potential biomarkers should be studied to improve pseudoprogression diagnostic accuracy, such as serum alkaline phosphatase (ALP), tumor-specific marker CA153, and collagen type I pyridine crosslinking peptide (ICTP). 28 Third, the mechanisms of the bone pseudoprogression are still elusive and need further elucidation. Fourth, in this study, all the new BS lesions presented as osteoblastic lesions on CT. Whether osteolytic/mixed new BS lesions represent bone pseudoprogression or disease progression need further research. Fifth, our study pointed out the unmet need to study bone pseudoprogression on CT and MRI.
This study has some limitations. The relatively small sample size is the main limitation of our study, yielding a limited statistical power. Large prospective clinical trials may be warranted to confirm the findings of our study. The retrospective nature is another limitation of our study. Moreover, the initial post-treatment BS was not performed until the completion of 4 months of treatment and therefore may have underestimated the incidence of bone pseudoprogression. In one prospective study, 75% of patients with MBC showed bone pseudoprogression during the first 3 months after treatment. Gradishar et al.29 also demonstrated that the incidence of early pseudoprogression (week 9 in the PREVAIL trial and week 13 in the AFFIRM trial) in patients with metastatic castration-resistant prostate cancer (mCRPC) was 17.6–23.8%. In contrast, the incidence of late pseudoprogression (week 17 or later in the PREVAIL trial and week 25 or later in the AFFIRM trial) was only 0.5–3.7%. However, according to National Comprehensive Cancer Network (NCCN) guidelines, the suggested BS interval for follow-up of patients with MBC is 4–6 months. 30 Accordingly, this bias is not expected to have an impact on clinical practice.
Conclusion
In summary, our findings showed that osteoblastic new BS lesions commonly represented bone pseudoprogression rather than disease progression in HR-positive MBC. Correlation of BS and CT findings (conversion of osteolytic lesions to sclerotic areas) may help differentiate bone pseudoprogression, thereby avoiding the premature discontinuation of therapy and maximizing the benefit of endocrine treatment.
Supplemental Material
sj-jpg-1-tam-10.1177_17588359211022881 – Supplemental material for Determination and clinical significance of bone pseudoprogression in hormone receptor-positive metastatic breast cancer
Supplemental material, sj-jpg-1-tam-10.1177_17588359211022881 for Determination and clinical significance of bone pseudoprogression in hormone receptor-positive metastatic breast cancer by Mingxi Lin, Yizi Jin, Ziyi Yang, Xichun Hu and Jian Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank all the doctors, nurses, patients, and their family members for their support throughout this study.
Author contributions
Mingxi Lin: data curation, methodology, formal analysis, writing – original draft and editing. Yizi Jin: data investigation, visualization, and editing. Ziyi Yang: data curation, formal analysis. Xichun Hu: project administration, supervision, and editing. Jian Zhang: conceptualization, funding acquisition, project administration, resources, supervision, validation, and editing.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science and Technology Major Project (grant no. 2020ZX09201-013), the National Natural Science Foundation of China (grant no. 82072915), the Shanghai Municipal Science and Technology Commission Guidance Project, P.R. China (contract no. 18411967800), a research grant from Shanghai Hospital Development Center (grant no. SHDC12018X03), and CSCO-ROCHE Cancer Research Fund 2019 (grant no. Y-2019Roche-171).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
