Abstract
Background:
The relative importance of predictive factors for advanced non-small cell lung cancer (NSCLC) patients on epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor (TKI) treatment remains unclear.
Materials and methods:
We retrospectively enrolled advanced NSCLC patients with single first-generation EGFR-TKI treatment for ⩾5 years (Y) in Taiwan. Clinical data was collected and compared with those of another cohort with single first-line EGFR-TKI treatment for <5 Y. Plasma cell-free DNA (cfDNA) samples were collected from patient subsets, pre- and post-TKI, in the >5 Y group.
Results:
Overall, 128 and 278 patients were enrolled in the ⩾5 Y and <5 Y groups, respectively. Significant factors in the multivariate analysis of patients’ characteristics including Eastern Cooperative Oncology Group performance status 0–1, postoperative recurrence, without brain metastasis, oligometastasis (each score of 2), female sex, erlotinib use, and without bone metastasis (each score of 1), were incorporated into a risk scoring system. The area under the receiver operating characteristic curve was 0.82 [95% confidence interval (CI): 0.78–0.86]. Of the plasma cfDNA samples from 33 patients in the ⩾5 Y group, only 1 had a T790M in 25 patients without progressive disease. In 27 patients with single agent use for ⩾96 months, 22 (81.5%) received local treatment (surgery or radiotherapy) for the primary lung tumor before and during TKI treatment.
Conclusion:
For NSCLC patients with single first-generation EGFR-TKI use for ⩾5 Y, factors with different relative importance exist and the risk-scoring model is feasible with modest accuracy. The role of local treatment for primary tumors in patients with long-term TKI use requires further investigation.
Keywords
Introduction
The discovery of epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) has resulted in a dramatic change in the treatment strategy of advanced EGFR-mutant non-small cell lung cancer (NSCLC) patients.1,2 Although most EGFR-mutant NSCLC patients initially respond to gefitinib, erlotinib, and afatinib, patients usually develop a progressive disease within 1 year after the initiation of treatment. 3 However, a subgroup of patients experience a longer clinical benefit from EGFR-TKIs. There are few reports about this phenomenon. In a recent study, treatment with afatinib was reported to provide a significantly higher 24-month progression-free survival (PFS) rate than gefitinib in a first-line setting (17.6% versus 7.6%). 4 Some case studies report that complete remission with EGFR-TKI treatment was maintained for more than 2 years.5–8 However, the real predictive factors for single EGFR-TKI long-term use in advanced EGFR-mutant NSCLC patients remains unclear. Therefore, we investigated the relative importance of predictive factors by studying the differences in the characteristics between patients with single EGFR-TKI use for ⩾5 years (⩾5 Y) and patients with <5 years (<5 Y) use.
Materials and methods
Patients
We retrospectively enrolled advanced NSCLC patients with single EGFR-TKI use for ⩾5 Y in nine medical centers in Taiwan under the ‘TKI Prolonged Single Agent Use over 5 Years’ (TIPS-5) study from September 2005 to September 2014. To be eligible for the study, patients were required to have cytological- or pathological-confirmed NSCLC, at inoperable advanced stage IIIB to stage IV, to have undergone single EGFR-TKI treatment for ⩾5 Y without progressive disease (PFS of EGFR-TKI ⩾5 Y), and a clear survival status during follow-up. Patients were excluded if they had undergone EGFR-TKI treatment for ⩾5 Y due to treatment beyond progression, another active malignancy, concomitant use of another anticancer medicine during the period of EGFR-TKI use, or if they had Eastern Cooperative Oncology Group performance status (ECOG PS) 4.
After forming the cohort of eligible patients with single EGFR-TKI use for ⩾5 Y, we enrolled another cohort with advanced NSCLC patients with first-line single EGFR-TKI use for <5 Y (PFS of EGFR-TKI < 5 Y) from January 2011 to June 2014 from the Taichung Veterans General Hospital. The development and validation of a prognostic risk score model was performed for the NSCLC patients with single EGFR-TKI use for ⩾5 Y. Another independent group of NSCLC patients who met the study criteria were enrolled as the validation cohort for the risk score model established for EGFR-TKI use for ⩾5 Y from February 2012 to August 2015 from the National Taiwan University Hospital. The study was conducted ethically in accordance with the World Medical Association Declaration of Helsinki, and was approved by the Institutional Review Board (IRB) of each hospital (Supplemental Table A.1). The informed consent documents for clinical data records and genetic testing were written by patients or were waived according to the requirement of each IRB.
Data records
The patients’ clinical characteristic and demographic data, including age, sex, family history, tumor stage, smoking status, ECOG PS, baseline EGFR mutation status, treatment history, metastatic site, and outcome variables were collected for analysis. We defined oligometastasis as no more than five metastatic lesions. The lung cancer tumor-node-metastasis staging was conducted according to the 8th edition of the American Joint Committee on Cancer staging system. 9 Computed tomography of the chest was performed every 3 months for National Health Insurance reimbursement. The treatment response of EGFR-TKIs was confirmed using the Response Evaluation Criteria in Solid Tumors (Version 1.1). 10
EGFR mutation test for tumor tissue
The methods of EGFR mutation-detection in tumor tissues of the patients in the single EGFR-TKI use for ⩾5 Y group were different in each hospital. The methods used for EGFR testing included direct sequencing, mutant type-specific sensitive methods [e.g., matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS)], the scorpion amplification refractory mutation system (ARMS), and Cobas EGFR Mutation Test. For the patients with single EGFR-TKI use for <5 Y, the EGFR mutation detection method was MALDI-TOF MS, performed as previously described. 11 A rebiopsy of some patients in both groups after disease progression was conducted.
EGFR mutation detection of liquid biopsy in pre- and post- EGFR-TKI treatment plasma samples
A subset of patients in the single EGFR-TKI use for ⩾5 Y group had available pre- and post-EGFR-TKI treatment plasma samples. The post EGFR-TKI treatment plasma samples were collected, close to the latest follow-up dates in patients without progression or the dates of disease progression.
Plasma cell-free DNA (cfDNA) extraction was performed according to our previous study. 12 EGFR mutation-detection including L858R, exon19 deletion, and T790M, was performed based on our previous studies with modifications.12–14 Briefly, the combination of peptide nucleic acid (PNA) and MALDI-TOF nucleotide mass spectrometry was utilized for experiments in the ISO15189-certified clinical center laboratory. PNA oligos used to specifically lock the wild-type allele were synthesized using PanaGene (Daejeon, Korea). All nucleotide MALDI-TOF MS assays were adapted from the MassARRAY System (Agena Bioscience, USA) according to the manufacturer’s instructions. Data were collected and analyzed using Typer 4 software (Agena Biosciences, USA). The tests were conducted at the National Center of Excellence for Clinical Trial and Research of the National Taiwan University Hospital.
Statistical methods
Univariate analysis using the Fisher’s exact test was performed to assess the differences in the characteristics between both groups. Logistic regression models were used to estimate the odds ratios (ORs) and 95% confidence intervals (CIs) of the PFS of lung cancer for each variable. The variables showing significant associations with the 5-year PFS (p < 0.05) in the univariate analyses were included in a stepwise selection procedure to generate a final multiple model. The full model was used to develop a risk-scoring system for the prediction of the probability of PFS of lung cancer. The regression coefficients of the variables were converted into an integer risk score by rounding up the quotient of the regression coefficient by a single constant. The constant was the sex regression coefficient, defining the integer risk score for female sex to be one. Risk estimates for the 5-year PFS of lung cancer according to the sum of scores could be calculated. 15 To evaluate the predictive accuracy of the risk score model, the receiver operating characteristic (ROC) curve was derived and the area under the ROC curve (AUROC) was calculated. All the statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC). Two-tailed tests and p values < 0.05 for significance were used.
Results
Patients’ characteristics and demographic data
A total of 128 patients were enrolled in the ⩾5 Y use group, and 278 patients in the <5 Y use group. The baseline characteristics are shown in Table 1. A total of two hundred and one (49.5%) patients were ⩾65 years old, while two hundred and fifty-two patients were female (62.1%). A total of sixteen patients had stage IIIB to IIIC disease, and three hundred and eight patients had stage IV disease. The number of patients who had postoperative recurrence was eighty-two, while five patients had recurrence after concurrent chemoradiotherapy (CCRT). After the initial response to EGFR-TKI, radiotherapy for the primary tumor lesion only including stereotactic surgery was done for nine patients (three with oligometastasis and six with non-oligo-metastasis; eight in the ⩾5 Y use group). The characteristics of these nine patients are listed in Supplemental Table A.2. A total of one hundred and thirty-five patients had oligometastatic lesions (33.3%). The metastatic sites were the lungs (54.4%), brain (27.6%), bone (38.2%), liver (7.9%), and adrenal glands (3.0%).
Univariate analysis of factors associated with 5-year progression-free survival of lung cancer.
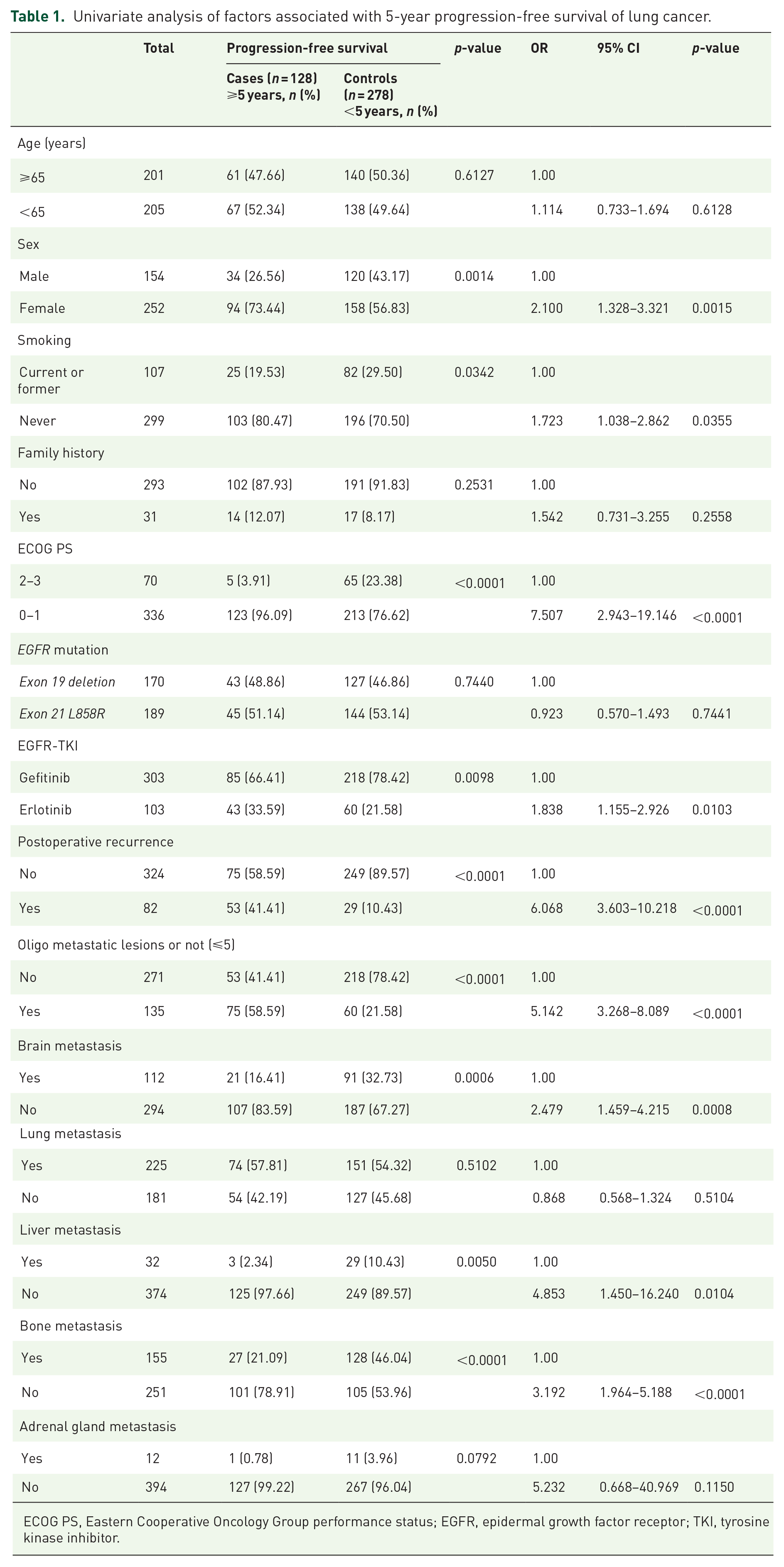
ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor.
The Kaplan–Meier curves of the PFS of both groups are shown in Figure 1a. The PFS distribution in the ⩾5 Y use group is shown in Figure 1b. There were 27 patients with single-agent use for ⩾96 months, amongst whom, 22 (81.5%) were administered a local treatment for the primary lung tumor (16 with postoperative recurrence, two with CCRT recurrence, and four with radiotherapy) during treatment with TKIs.

The PFS of the ⩾5 year and the <5 year use group. (a) The difference between the ⩾5 year and the <5 year use groups in Kaplan–Meier curve of PFS (b) The distribution of PFS in the ⩾5 year use group.
Univariate analysis of the clinical factors showed a significantly greater prevalence of the female sex, never-smokers, PS 0-1, erlotinib use, postoperative recurrence, and oligometastases (⩽5), but a lower prevalence of brain, liver, and bone metastases in the ⩾5 Y use group. There were no differences in age, family history, EGFR mutation subtypes, lung-to-lung, and adrenal metastases (Table 1).
In the multivariate analysis, female sex, ECOG PS 0-1, erlotinib use, postoperative recurrence, no brain metastasis, no bone metastasis, and with oligometastatic lesions (⩽5) showed a greater prevalence in the ⩾5 Y use group. There were no differences in never-smokers and liver metastasis (Table 2).
Multivariate analysis of factors associated with 5-year progression-free survival of lung cancer.

ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor.
Risk score model of TKI use for more than 5 years
For the survey of the relative importance of each predictive factor, the risk scores of each factor for the 5-year EGFR-TKI single agent use of lung cancer are shown in Table 3. Predictors for EGFR-TKI use for ⩾5 Y in the derivation dataset were ECOG PS 0-1, postoperative recurrence, the absence of brain metastasis, and oligometastasis (each with a corresponding score of 2); female sex, erlotinib use, and without bone metastasis (each with a corresponding score of 1).
Risk scores of each factor for the 5-year progression-free survival of lung cancer.

ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor.
The nomogram of the multivariate model of the risk scores is shown in Figure 2. The chances of ⩾5 Y EGFR-TKI single-agent use in lung cancer patients ranged from 20% (score 0) to 95% (score 11). We used the ROC curve and the AUROCs to evaluate the predictive accuracy of the risk score model. The risk score model in the study using each factor had an AUROC of 0.82 (95% CI: 0.78–0.86, Supplemental Figure A.1A).

Nomogram of the multivariate model of the risk scores. The nomogram of the multivariate model of the risk scores were constructed to predict the chances of PFS ⩾5 years with EGFR-TKIs treatment in non-small cell lung cancer patients.
The baseline characteristics of the validation cohort are shown in Supplemental Table A.3. A total of 177 patients were enrolled: 11 in the ⩾5 Y use group, and 166 in the <5 Y use group. A total of eighty-two (46.3%) patients were ⩾65 years old, while one hundred and twenty-three patients were female (69.5%). Six patients had stage IIIB to IIIFC disease, and one hundred and thirty-nine patients had stage IV disease. Thirty-two patients (18.1%) had postoperative recurrence. Fifty-six patients had oligometastatic lesions (31.6%). The proportions of the brain, lung, liver, bone, and adrenal gland metastases were 68.9%, 42.4%, 10.2%, 41.2%, and 8.5%, respectively. There were no patients with recurrence after CCRT or radiotherapy only for the primary tumor lesion after the response to EGFR-TKIs. When we applied the risk score model from the discovery to validation dataset, the predictive performance was AUROC 0.75 (95% CI: 0.64–0.86, Supplemental Figure A.1B).
EGFR mutation status
In the discovery dataset, the EGFR mutation test was not conducted in 30 patients in the ⩾5 Y use group, largely because the EGFR mutation test was previously not available in Taiwan. A total of one hundred and seventy patients harbored the exon 19 deletion mutation, 189 patients harbored the exon 21 L858R point mutation, and 15 patients had other mutations, including mixed mutations. There were two patients with undetected EGFR mutation.
In the ⩾5 Y use group, 26 patients had progressive disease after EGFR-TKIs treatment. Among them, eight patients underwent tissue rebiopsy, and T790M was detected in four patients. In the <5 Y use group, 72 patients underwent tissue rebiopsy, and 36 patients had T790M mutation (50%). The T790M mutation rates among patients with EGFR exon 19 deletion mutation, exon 21 L858R point mutation, and other mutations were 56.8%, 40.7%, and 0%, respectively (Supplemental Figure A.2).
Regarding the cfDNA analysis in the group with ⩾5 Y single agent EGFR-TKI use, there were 33 patients with post-EGFR-TKI treatment plasma samples collected close to the dates of latest follow-up in patients without progression or the dates of disease progression (Figure 3). Among these 33 patients, 11 (33%) patients with pre-EGFR-TKI plasma samples were analyzed; seven had non-shedding tumors and four had shedding tumors (three with exon 19 deletion and one with L858R). Of these 33 patients with post-TKI plasma samples, 24 (72.7%) had a tumor tissue biopsy available for analysis before EGFR-TKI treatment; 14 had an exon 19 deletion, eight had L858R, and two had other mutations. There were nine patients who were not tested for EGFR mutation due to unavailability of the test.

The swimmer plot for PFS in 33 patients of the ⩾5 year use group. The detailed data included treatment PFS, pre-treatment and post-treatment EGFR subtype of cfDNA and tissue biopsy, and the status of progressive disease.
Of the 33 patients with post-EGFR-TKI treatment plasma samples for analysis, two were positive for EGFR mutation among 25 (75.8%) patients without progressive disease who were still under treatment with TKIs; one patient harbored T790M, and the other had EGFR exon 19 deletion. Eight (24.2%) patients had progressive disease under TKIs treatment, and T790M mutation was detected in four of them, including two with T790M alone and two with combined exon 19 deletion and L858R (Figure 3).
Discussion
Advanced NSCLC patients who were administered single first-generation EGFR-TKIs for ⩾5 Y without disease progression were uncommon. From this study, predictive factors with different relative importance exist; with ECOG PS 0-1, postoperative recurrence, without brain metastasis, and oligometastasis (each with a corresponding score of 2); and female sex, erlotinib use, and without bone metastasis (each with a corresponding score of 1). A risk-score model for EGFR-TKI ⩾5 Y use was established and verified in the validation dataset. In addition, local treatment of the primary tumor, including surgery, CCRT, and radiotherapy during EGFR-TKI use may provide further clinical benefit. From the cfDNA data, either shedding or non-shedding tumors can have a good outcome with long-term TKI use. For those patients with persistent disease control, most of them were without detectable EGFR mutation in plasma.
Several factors, such as female sex, good PS, oligometastasis, absence of brain metastasis, etc., are all well-known important prognostic factors for EGFR-mutant lung cancer patients treated with EGFR-TKIs. However, the relative importance of these factors has not been elucidated. As disease progression occurs about 1 year after the initiation of first-generation EGFR-TKIs in EGFR-mutant patients, an exaggerated emphasis on prolonged single EGFR-TKI use for ⩾5 Y could differentiate the relative importance of each factor clearer, especially through the risk score model.
The effect of local treatment of the primary lung tumor in advanced EGFR-mutant patients treated with EGFR-TKIs remains unclear. In a randomized phase III trial (ClinicalTrial.gov identifier: WJTOG 3405) that compared gefitinib with chemotherapy in patients with advanced EGFR-mutant NSCLC, postoperative recurrence was the only independent prognostic factor for overall survival (OS) analysis and was statistically better than de novo stage IIIB/IV disease. Using the Cox proportional hazards model, the hazard ratio (HR) was 0.459 (95% CI 0.312–0.673, p < 0.001). 16 The OS of recurrent metastatic disease after treatment with curative intent may be better than that of de novo metastatic disease, as shown in several retrospective studies.17–20 In our study, besides postoperative recurrence, five patients had recurrence after CCRT for curative intent in the ⩾5 Y group.
In a study by Al-Halabi et al., 21 the initial progression of EGFR-TKI-treated cancers occurred in about half of the original disease sites, with-or-without distant metastasis. There were reports of survival improvement with radiotherapy for the primary and oligometastatic sites in EGFR-TKI-treated lung cancer patients.22,23 Furthermore, two reports found that primary tumor resection improved survival in stage IV NSCLC patients in the Surveillance, Epidemiology, and End Results database.24,25 Therefore, concerning the effect of the local treatment of primary tumors, consolidation stereotactic body radiation therapy (SBRT) and curative dose of radiotherapy only to the primary tumor in a subset of patients following TKI response seemed feasible. Here, there were nine patients who received radiotherapy, including SBRT for the primary tumor lesion after the initial response to EGFR-TKIs (eight patients in the ⩾5 Y use group and one patient in the <5 Y use group) even in the non-oligometastatic disease. Furthermore, 22 (81.5%) out of 27 patients with single agent use for ⩾96 months in the ⩾5 Y use group received a local treatment for the primary lung tumor. Regardless of the recurrence after operation or CCRT, or radiotherapy to the primary tumor during the response to EGFR-TKI use, a lower tumor burden could be one of the possible explanations for this difference in prognosis for local treatment to the primary lung tumor. Another hypothesis could be that after the primary tumor eradication, immune competence is restored, even if a metastatic tumor is present. 26 More prospective studies are needed to investigate the effect of local treatment for primary tumors and its association with long-term EGFR-TKI use.
In the present study, there was no difference in the use of EGFR-TKIs for ⩾5 Y between patients with exon 19 deletion and those with L858R. However, the association between EGFR exon 19 deletions and prolonged survival in patients with advanced NSCLC treated with EGFR-TKIs has been reported in several studies.27–31 The phenomenon may be partly explained by more co-occurring mutations in patients with L858R mutation.32,33 The occurrence of concomitant mutations in patients with lung cancer using EGFR-TKIs for ⩾5 Y with sensitizing EGFR mutations needs to be investigated.
In this study, the risk score model using each factor had an AUROC of 0.82 (95% CI: 0.78–0.86) in the discovery dataset. It was a good tool for the prediction in patients with first-generation EGFR-TKI single agent use of more than 5 years. In the validation dataset, there were no patients with recurrence after CCRT or radiotherapy only for the primary tumor lesion after the response to EGFR-TKIs; this could reduce the predictive ability of our risk score model. However, even with this limitation, our risk score model still had a modest predictive ability (AUROC, 0.75). A larger sample size is needed in the future study.
According to the cfDNA data obtained in this study, EGFR mutation was detected in two out of 25 patients with persistent tumor control for ⩾5 Y. There is a need for further investigation as previous studies show that recurrences occur early in patients with detectable plasma EGFR mutation. 34 In patients with disease progression, T790M was detected in 50% (4/8), compared with 36.9% in our previous study after treatment with first- and second-generation EGFR-TKIs. 12
Our study had some limitations. Firstly, it was a retrospective study; therefore, it had more bias when compared with a prospective study. Secondly, all patients in this study were Taiwanese; therefore, our findings may not be generalizable to other ethnic populations. Thirdly, the cohort of the <5 Y TKI use group was from a single center, and the enrolment period was not the same as that of the ⩾5 Y TKI use group. Finally, in Taiwan, afatinib was reimbursed since May 2014, and osimertinib was reimbursed since April 2020. In the present study, we enrolled patients with single first-generation EGFR-TKI use for ⩾5 years. Therefore, few patients with afatinib or osimertinib as first-line treatment could be enrolled for analysis while we collected the data.
Conclusion
In conclusion, in advanced EGFR mutant NSCLC patients, predictive factors with different relative importance exist in single first-generation EGFR-TKI use for ⩾5 Y and were integrated into an established and verified risk-score model, which had a modest accuracy. According to the cfDNA data, both shedding and non-shedding tumors can have good outcomes with long-term TKI use. For patients with persistent disease control, most of them had an indetectable EGFR mutation in plasma. In addition, the role of local treatment for primary tumors in patients with long-term TKI use needs further investigation. Further prospective research is required to confirm these results.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359211018022 – Supplemental material for The relative importance of predictive factors for single first-generation EGFR-TKI use for more than 5 years in patients with advanced non-small cell lung cancer: Taiwan multicenter TIPS-5 study
Supplemental material, sj-pdf-1-tam-10.1177_17588359211018022 for The relative importance of predictive factors for single first-generation EGFR-TKI use for more than 5 years in patients with advanced non-small cell lung cancer: Taiwan multicenter TIPS-5 study by Yen-Hsiang Huang, Jen-Yu Hung, How-Wen Ko, Po-Lan Su, Chun-Liang Lai, Huang-Chih Chang, Te-Chun Hsia, Sheng-Hao Lin, Kuan-Li Wu, Cheng-Ta Yang, Wu-Chou Su, Yi-Chun Chu, Chin-Chou Wang, Wei-Yu Liao, Yen-Ting Lin, Ching-Hsiung Lin, Meng-Chih Lin, Kuo-Hsuan Hsu, Jeng-Sen Tseng, Tsung-Ying Yang, Kun-Chieh Chen, Mei-Hsuan Lee, Sung-Liang Yu, Chao-Chi Ho and Gee-Chen Chang in Therapeutic Advances in Medical Oncology
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
