Abstract
The insulin-like growth factor (IGF) pathway is a complex pathway involving interactions between membrane-bound receptors, ligands, binding proteins, downstream effectors, and other receptor tyrosine kinase signaling cascades. The IGF pathway has been identified as a potential therapeutic target in non-small cell lung cancer (NSCLC) based on the following provocative factors. Preclinical observations in NSCLC have shown that this pathway is involved in tumor cell proliferation, survival, and invasiveness. In addition, IGF-1R protein expression is found in a significant number of non-small cell tumor specimens. Initial therapeutic efforts involved the development of monoclonal antibodies and tyrosine kinase inhibitors that target IGF-1R, a transmembrane receptor tyrosine kinase. Enthusiasm for targeting this pathway increased when a randomized phase II study showed that combining an anti-IGF-1R monoclonal antibody (figitumumab) with a platinum doublet resulted in a higher response rate and trends for superior progression-free survival and overall survival. Subsequently, a phase III study failed to confirm the promising results observed in the phase II trial. Currently, investigators are studying different monoclonal antibodies and tyrosine kinases targeting IGF-1R. In unselected patients, results presented thus far do not suggest efficacy of this agent. However, retrospective subgroup analyses suggest that circulating IGF-1 levels might identify patients who could benefit from treatment with an IGF-1R monoclonal antibody and may warrant further exploratory studies for predictive molecular markers. The purpose of this paper is to briefly discuss the IGF pathway and its relationship with other signaling pathways in lung cancer and to review the ongoing IGF clinical trials and efforts to identify predictive molecular markers.
Keywords
Introduction
Despite advances in medical treatment, lung cancer remains the leading cause of cancer mortality in the United States with 222,520 new cases and 157,300 deaths predicatively described for 2010 [Jemal et al. 2010]. Parallel therapeutic efforts in pathway and biomarker discovery have redefined traditional treatment protocols and added novel targeted therapies to the oncologist’s armamentarium for advanced stage non-small cell lung cancer (NSCLC). Based on preclinical observations and molecular marker studies in patient specimens, the insulin-like growth factor (IGF) pathway has been identified as a promising therapeutic target in lung cancer. Monoclonal antibodies and small molecule inhibitors targeting the IGF-1 receptor (IGF-1R) being tested in both NSCLC and small cell lung cancer (SCLC) are reviewed.
The insulin-like growth factor pathway
The insulin-like growth factor (IGF) pathway is intricate, involving interactions between receptors, ligands, binding proteins, and downstream enzymes. A simian virus 40 transformation model first implicated its role in tumorigenesis in 1993 and since then the IGF pathway components have increasingly been linked to the deregulation of cell growth and metastasis [Douglas et al. 2010; Renehan et al. 2004; Schairer et al. 2004; Sell et al. 1993; Rosendahl et al. 2010; Sarkissvan et al. 2011]. The pathway’s major components consist of IGF-1 and IGF-2 with respective receptors IGF- 1R and IGF-2R as well as IGF binding proteins (IGFBPs) 1–6, and has extensive external regulatory measures (see previous reviews [Firth and Baxter, 2002; Grimberg and Cohen, 2000; Rajaram et al. 1997; Rosenzweig and Atreya, 2010]).
IGF-1 and IGF-2 are structurally related to insulin and function to regulate cell growth, proliferation, transformation, differentiation, migration, and are powerful anti-apoptotic factors [Yu et al. 2000; Lee et al. 2002]. IGF-1 is the systemic mediator of growth hormone (GH) in adults and is secreted by hepatocytes. In neonatal life, IGF-2 is a more important factor for cell differentiation, growth, and maturation, but by adulthood, IGF-1 plays a more significant role despite lower serum concentrations than IGF-2. The two factors are also synthesized at a local tissue level by GH-independent mechanisms and act in a paracrine and autocrine fashion to control cellular growth [Juul et al. 1995; Durai et al. 2005]. Ninety per cent of IGF-1 and IGF-2 is found complexed with IGFBP-3, the most prevalent serum IGF binding protein, and a function-deficient acid labile subunit (ALS) to form a 150 kDa ternary complex [Baxter et al. 1989]. Several research groups have linked increased serum IGF expression as a risk factor for ovarian, pancreatic, prostate, colorectal, lung, and premenopausal breast cancers [Douglas et al. 2010; Lann and LeRoith, 2008; Renehan et al. 2004; Schairer et al. 2004]. In addition, decreased levels of IGFBP-3 have also been associated with the development of malignancy and the malignant phenotype [Douglas et al. 2010; Lee et al. 2002; Zhang et al. 2010].
IGF-1R is a glycoprotein composed of two extracellular alpha subunits that bind IGF-1 preferentially and with lesser affinity to IGF-2 and insulin. The two intracellular beta subunits contain the tyrosine kinase domain responsible for activating the IRS/PI3K/AKT/mTOR pathway and the Ras/Raf/MAPK pathways that promote cell growth, transformation, migration, and survival [Desbois-Mouthon et al. 2009; Ariga et al. 2000; Parrizas et al. 1997; Peruzzi et al. 1999]. Owing to homology to the insulin receptor (IR), most small molecule inhibitors of IGF-1R also target IR, whereas monoclonal antibodies are able to be more IGF-1R specific. Conversely, IGF-2R is a mannose-6-phosphate receptor, has no functional intracellular domain, and ultimately serves to clear IGF-2 from the system.
IGF-1R expression by immunohistochemistry (IHC) and fluorescent in situ hybridization (FISH) has been detected in 39–84% in patients with advanced NSCLC [Cappuzzo et al. 2006, 2009; Dziadziuszko et al. 2009; Ludovini et al. 2009; Wynes et al. 2009]. This increased expression appears to occur more frequently in squamous cell NSCLC [Cappuzzo et al. 2009; Gualberto et al. 2010]. In early stage NSCLC, the expression of IGF-1R may be less prevalent, found using IHC in only 12.7% of the 125 surgical series examined by Lee and colleagues [Lee et al. 2008]. The prognostic significance of IGF-1R expression by IHC remains unclear. In two independent studies of early stage, resected NSCLC, no statistical association of IGF-1R expression by IHC with survival was found [Cappuzzo et al. 2009; Lee et al. 2008].
IGFBPs 1–6, as well as serum and tissue proteases, are thought to play a role in the IGF pathway and may be markers of tumor activity. IGFBPs are mainly found in free or binary form where they exit capillaries and act at a local tissue level. Most major IGFBPs have both IGF-dependent and independent actions, the former exerted by binding and sequestering IGF and the latter by binding directly to cell membrane and nuclear receptors. IGFBP-3, in particular, displays antiproliferative effects and pro-apoptotic effects both through and independently of the p53 downstream pathway [Buckbinder et al. 1995; Butt et al. 2000; Devi et al. 2000; Santer et al. 2006; Kim et al. 2004]. Levels of serum IGFBPs appear to be prognostic in NSCLC patients whether they are treated with an epidermal growth factor receptor (EGFR) tyrosine kinase receptor (TKI) drug or systemic chemotherapy [Fidler et al. 2009; Karmali et al. 2010]. Similar prognostic significance of IGFBPs was also recently recorded for early stage, surgically resected NSCLC [Shersher et al. 2011]. The intricacies of the IGF-independent action of IGFBPs are beyond the scope of this review but have been described in several cancer models [Chang et al. 2002; Dunlap et al. 2007; Firth and Baxter, 2002; Hoeflich et al. 2001; Li et al. 2010; Migita et al. 2010; Sureshbabu et al. 2009; Walker et al. 2007; Wolpin et al. 2007].
Rationale for combining IGF-1R and EGFR inhibitors
Prognostic evaluation of IGF-1R and EGFR by IHC reveals mixed results depending on the investigator and antibodies used for analysis. IHC analyses in two small retrospective studies suggest that patients with high IGF-1R expression may have improved outcomes on gefitinib therapy compared with those with lower IGF-1R expression [Cappuzzo et al. 2006; Fidler et al. 2008]. In a study by Ludovini and colleagues in surgically treated patients, high co-expression of both IGF-1R and EGFR significantly correlated with worse disease-free survival [Ludovini et al. 2009]. The inconsistency of results for reported IGF-1R expression and outcomes may be related to antibody heterogeneity, bias of scoring systems by IHC, statistically underpowered studies, or the presence of other poorly understood pathways and regulators in an already complex signaling lattice between IGF, EGF, and insulin. Nevertheless, cross-talk between the EGFR, IGF-1R, and IR pathways is supported by preclinical data. Cell-line data suggest that the IGF-1R receptor and downstream pathways are a mechanism of resistance to EGFR TKIs and monoclonal antibodies targeting EGFR, and that targeting both pathways leads to synergistic decreases in downstream signaling [Barnes et al. 2007; Chakravarti et al. 2002; Desbois-Mouthon et al. 2009; Huang et al. 2009; Knowlden et al. 2008; Morgillo et al. 2007; Slomiany et al. 2007]. See Figure 1 for an illustration of interactions between IGF-1R, IR, and EGFR signaling pathways.
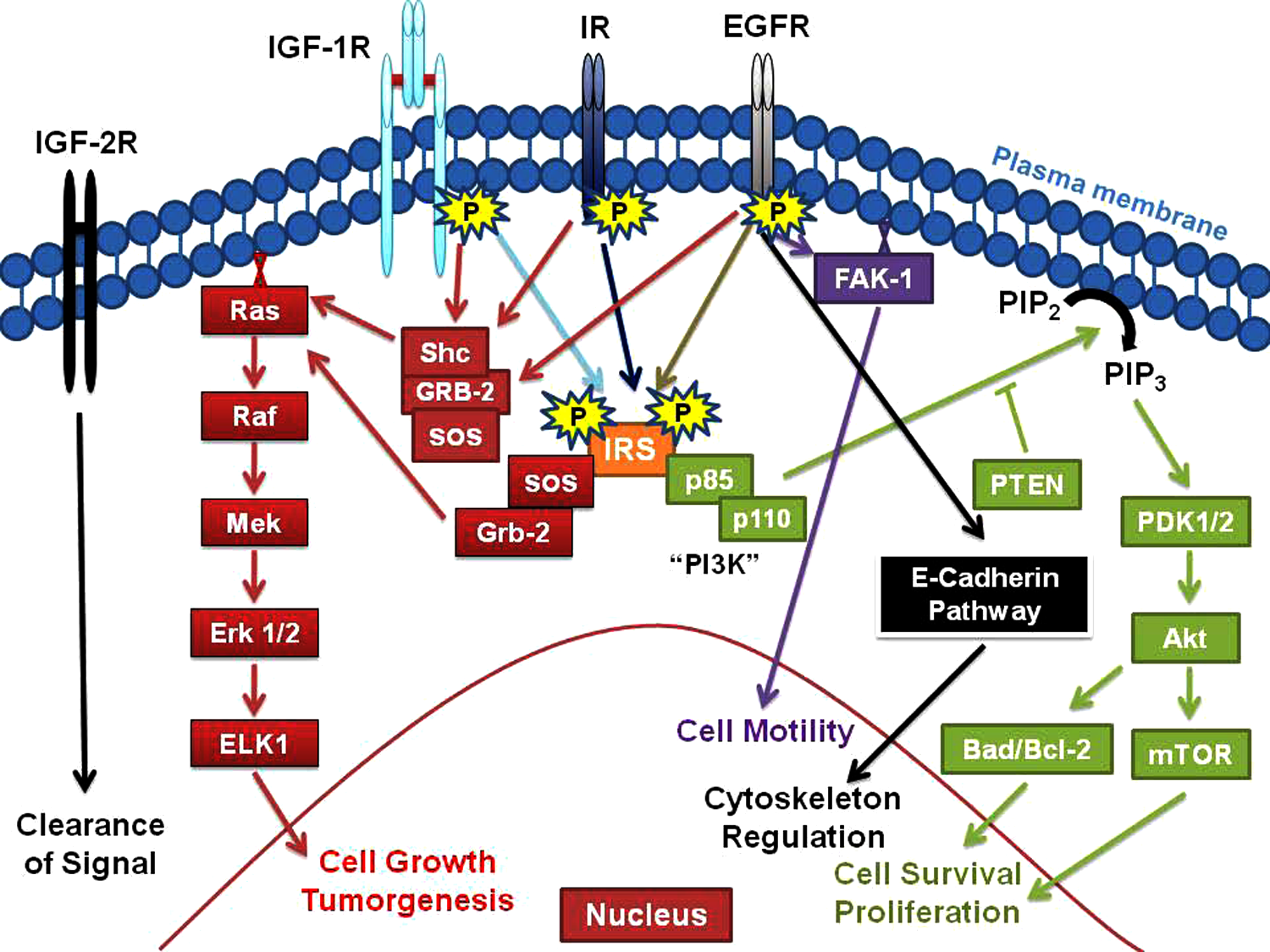
Interactions between insulin-like growth factor-1 receptor (IGF-1R), insulin receptor (IR), and epidermal growth factor receptor (EGFR) signaling pathways. IGF-2R, insulin-like growth factor-2 receptor. The various abbreviations in the diagram refer to known kinases involved in each of the pathways.
The hypothesis that upregulation of IGF-1R signaling in some NSCLC patients contributes mechanistically to EGFR TKI resistance is being tested in clinical trials. In one of these studies, 171 patients were randomized to erlotinib plus one of two doses of R1507 (monoclonal antibody against IGF-1R) versus erlotinib plus placebo. The addition of R1507 failed to show an improvement in 12-week progression-free survival (PFS) in unselected patients. However, circulating levels of IGF-1 were analyzed in 165 patients, and patients whose IGF-1 levels were higher than median had a 46% PFS rate at 12 weeks compared with an 18% PFS rate in patients with IGF-1 levels below the median [Habben et al. 2011]. KRAS mutation analysis was performed in 132 tumor specimens. The KRAS mutation rate was 27% and the 12-week PFS rate for patients with KRAS mutation was 37% with R1507 versus 0% for placebo-treated patients with a hazard ratio (HR) of 0.35, p = 0.012 [Ramalingan et al. 2011]. Despite the interesting results in patient subsets defined by these molecular parameters, it appears that there are no plans for further development of R1507 in NSCLC.
There are several other ongoing trials testing the strategy of inhibiting both the EGFR and IGF-1R pathways. In one study, cetuximab is combined with cixutumumab (an anti-IGF-1R monoclonal antibody). Patients are randomized to gemcitabine–cisplatin or carboplatin–cetuximab with or without cixutumumab. An IGF-1R tyrosine kinase (OSI-906) is being combined with erlotinib in two randomized trials. In the first study, chemotherapy naïve patients with EGFR mutations are randomized to erlotinib combined with OSI-906 or to erlotinib and placebo. In the second study, patients who are eligible for erlotinib maintenance are assigned to the EGFR TKI plus OSI-906 versus placebo.
IGF-1R inhibition and cytotoxic chemotherapy
Preclinical observations which showed that IGF-1R inhibition potentiated the cytotoxic effect of chemotherapeutic agents provided the basis for combinatorial clinical trials [Buck et al. 2008; Casa et al. 2008; Duan et al. 2009]. A randomized, first-line NSCLC phase II study comparing paclitaxel–carboplatin with or without figitumumab (an IgG2 human monoclonal antibody against IGF-1R) showed higher response rate and longer PFS in figitumumab-treated patients with the best results being observed in squamous cell patients [Karp et al. 2009]. Serum samples were available for 110 of 156 patients in the randomized phase II trial, and, like the phase II trial of R1507 with erlotinib, superior PFS was observed in figitumumab-treated patients who had higher levels of circulating free IGF-1. In addition, circulating levels of IGF-1 were inversely related to tumor tissue expression of E-cadherin and directly related to vimentin expression. Although further validation of these findings is needed, these preliminary observations suggest that circulating free IGF-1 might be a predictive biomarker for figitumumab sensitivity and surrogate for an epithelial-to-mesenchymal transition (EMT) in the tumor, which is implicated in NSCLC therapeutic resistance [Buck et al. 2008; Gualberto et al. 2011].
The phase III trial of carboplatin–paclitaxel with or without figitumumab was disappointing as it was discontinued for futility and there were safety concerns with early fatal toxicities in patients randomized to receive figitumumab. Particularly, early toxicity was noted in patients with low circulating levels of serum IGF. Again, however, in the subset of patients with high IGF serum levels, the addition of figitumumab appeared to offer benefit over carboplatin–paclitaxel [Jassem et al. 2010]. The issue is further complicated as the endocrinology literature notes that low levels of IGF-1 have been correlated with increased mortality [Brugts et al. 2008]. Table 1 lists IGF-1R targeting agents with recently completed and ongoing trials in NSCLC and includes dose-limiting toxicities for these agents. Table 2 summarizes published trials with IGF-1R targeted agents in NSCLC.
Agents in development that target insulin-like growth factor-1 receptor (IGF-1R) in non-small cell lung cancer (NSCLC).

EGFR, epidermal growth factor receptor; Ig, immunoglobulin; IR, insulin receptor; TKI, tyrosine kinase inhibitor.
Reported clinical trials of insulin-like growth factor-1 receptor (IGF-1R) targeted agents in non-small cell lung cancer (NSCLC).

EGFR, epidermal growth factor receptor; OS, overall survival; PC, paclitaxel-carboplatin; PCF, paclitaxel-carboplatin-figitumumab; PFS, progression-free survival.
The strategy of combining an IGF-1R inhibitor with cytotoxic agents is also being tested in extensive stage SCLC. The Eastern Cooperative Oncology Group is comparing etoposide– cisplatin–IMC-A12 with the chemotherapy doublet alone. This randomized phase II trial is nearing completion. In the relapsed SCLC setting, OSI-906 is being compared with a single agent, topotecan, in an ongoing study.
IGF-1R and mTOR inhibition
Single-agent trials of mammalian target of rapamycin (mTOR) inhibitors in NSCLC showed these agents were well tolerated but study designed endpoints of response rates were not met [Soria et al. 2009; Molina et al. 2007]. Preclinical data show mTOR inhibition can activate AKT through IGF-1R resulting in a synergistic interaction with IGF-1R inhibitors [Wan et al. 2007; Shi et al. 2005; Kurmasheva et al. 2009; O’Reilly et al. 2006]. This observation led investigators to initiate a phase I trial studying cixutumumab in combination with temsirolimus. The results of the combination showed stable disease in 47% of patients treated [Naing et al. 2011]. With time on therapy, levels of IGF-1 and IGFBP-3 were found to increase although this did not seem to correlate with response rates. Only 1 of 42 patients enrolled in the study had NSCLC and patients with adrenal corticoid carcinoma and Ewing’s sarcoma had the best outcomes. A small trial primarily in sarcoma patients showed a remarkable rate of disease stability (15/18 patients plus one partial response) and no apparent increase in toxicity when an mTOR inhibitor was combined with figitumumab [Quek et al. 2011].
Future directions
The IGF pathway has been implicated in NSCLC pathogenesis for many years, and advancement in research has identified the IGF-1R as a putative target. The negative results in currently reported trials in unselected patients are disappointing, but they do not exclude the possibility that inhibition of the IGF-1R pathway could provide benefit in patients with specific molecular signatures. The preliminary results of presented trials suggest that high circulating levels of IGF-1 may be a potential, easy to test biomarker that may identify patients who may benefit from this class of agents. Currently, the monoclonal antibody IMC-A12 is being tested in combination with a platinum doublet in stage IV NSCLC and in extensive stage SCLC. Although submission of tissue and blood specimens is encouraged in both of these trials, it is unclear whether a sufficient number of specimens will be collected to derive meaningful molecular marker results from these relatively small studies and it is doubtful that significant clinical benefit will be observed with an IGF-1R monoclonal antibody in unselected patients. Ongoing randomized phase II trials will provide the first results for an IGF-1R (OSI-906) TKI combination in NSCLC. In each trial, OSI-906 is combined with erlotinib and submission of blood and tumor specimens for molecular marker testing is mandatory. Considering the large number of novel therapies available for testing and the disappointing results with figitumumab, it seems likely that a strong signal will be required from the ongoing small studies to warrant the continued study of inhibitors of the IGF pathway in lung cancer.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
MJF has served as an advisor for Bristol Myers Squibb and Genentech and received speaking honoraria from Lilly and Genentech. PB has served as an advisor for Genentech, Lilly, Bristol Myers Squibb.
