Abstract
Background:
The benefit of chemotherapy in colon cancer patients is well documented but depends largely on whether patients complete the planned treatment regimen. We evaluated predictors of early discontinuation (EDChemo) and dose reduction of chemotherapy, especially the role of adverse treatment effects, in stage III patients who received adjuvant chemotherapy.
Methods:
Stage III colon cancer patients who were diagnosed in 2003–2014 and recruited into a population-based study in Germany and received FOLFOX [5-fluorouracil (5-FU), leucovorin (LV), and oxaliplatin], capecitabine monotherapy (CapMono), or 5-FU/LV were included. We assessed determinants of EDChemo and dose reduction using multivariable logistic regression. Also, we estimated proportions of EDChemo and dose reduction that are attributable to adverse effects using attributable fractions.
Results:
EDChemo and dose reduction rates were 52% and 17% for FOLFOX, 28% and 9% for CapMono, and 45% and 6% for 5-FU/LV, respectively. Predictors of EDChemo were low-grade tumor and treatment in a medium-volume hospital (for FOLFOX), obesity (for CapMono), and increasing age, T4 stage, and treatment in a medium-volume hospital (for 5-FU/LV). Adverse effects were particularly strongly associated with EDChemo and contributed to about 63%, 51%, and 32% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively. Of the various adverse effects, gastrointestinal events showed the strongest associations with EDChemo and accounted for about 7%, 26%, and 20% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively. Adverse effects were, moreover, a strong determinant of dose reduction and accounted for about 82% of all cases.
Conclusions:
EDChemo is common in stage III colon cancer patients receiving chemotherapy and more than half of the cases of EDChemo and dose reduction are due to adverse treatment effects. Further research should address the potential for reducing EDChemo and dose reduction rates by close monitoring of patients for early signs and enhanced management of adverse effects, especially gastrointestinal events.
Background
Colorectal cancer (CRC) is the third most commonly diagnosed malignancy in the world, with an annual incidence of about 1.8 million cases. 1 About two-thirds of these tumors are located in the colon. 1 Several treatment regimens for colon cancer have emerged in the past decades. Results from the MOSAIC, 2 XELOXA, 3 and X-ACT trials, 4 in particular, have influenced treatment for colon cancer. Oxaliplatin-based therapy [e.g. FOLFOX, 5-fluorouracil (5-FU), leucovorin (LV), and oxaliplatin] and capecitabine monotherapy (CapMono, a 5-FU prodrug) are the main regimens for postoperative treatment of stage III colon cancer. 5 Even though these newer combinations (e.g. FOLFOX) improve patient survival, they tend to have more adverse treatment effects and lower completion rates.5,6
The benefit of chemotherapy in stage III colon cancer patients is well documented but depends in part on whether patients complete the planned regimen. 7 Prior data suggest that about one-third of chemotherapy recipients discontinue their treatments prematurely.8–11 Results from the IDEA7,12 and SCOT trials 13 suggest that patients receiving FOLFOX for 3 (versus 6) months, especially those with T4 and/or N2 stage, have poorer survival outcomes. Multiple studies have investigated factors associated with early discontinuation of adjuvant chemotherapy (EDChemo),6,8–11,14,15 but many referred to patients who were diagnosed before 20068–10,15 or received 5-FU/LV only.8,9,15 Besides EDChemo, dose reduction is also common in recipients of chemotherapy and negatively affects patient survival, especially those aged <70 years 16 or with body mass index (BMI) ⩾30 kg/m2. 17
An obvious determinant of EDChemo and dose reduction could be adverse treatment effects, 6 but the extent to which various adverse effects contribute to EDChemo and dose reduction is unclear. In particular, quantitative evidence on the proportions of EDChemo and dose reduction that are attributable to adverse effects is lacking but could be useful for refining risk stratification for EDChemo and dose reduction. In a cohort of stage III colon cancer patients, we aimed to thoroughly assess predictors of EDChemo and dose reduction, with a particular focus on the role of adverse effects.
Methods
Study design and population
Our patient cohort analysis is based on data from colon cancer patients who were diagnosed in 2003–2014 and recruited into the DACHS (Darmkrebs: Chancen der Verhütung durch Screening) study. The DACHS study is a population-based case–control study conducted in southern Germany, which was initiated to assess the potential for endoscopic screening to reduce CRC risk. Cases are also followed up in order to evaluate treatment-related and prognostic factors for CRC. Patients with first time diagnosis of CRC (International Classification of Diseases, 10th Revision, codes C18–C20) and aged ⩾30 years were eligible. Participants were recruited from all 22 hospitals providing first-line treatment for CRC in the study region of about 2 million inhabitants. In the recruiting hospitals, eligible patients were informed about the study by their physicians and were recruited either during or shortly after their hospital stay for CRC surgery. Data from a population-based cancer registry indicated that about 50% of the eligible patients in the study region were successfully recruited. Incomplete recruitment was mainly due to work overload of the clinicians involved in patient recruitment. Further details of the DACHS study have been described previously.18,19 The DACHS study was approved by the ethics committees of the Medical Faculty of Heidelberg University (ID: 310/2001) and the state medical boards of Baden-Wuerttemberg (ID: M-198-02) and Rhineland-Palatinate [ID: 837.419.02 (3637)]. All participants also gave written informed consent.
Inclusion criteria
Our sample comprised all surgically resected stage III colon cancer patients who received adjuvant chemotherapy (Figure 1). Because the main regimens received by the participants were FOLFOX, CapMono, and 5-FU/LV (including MAYO, Roswell Park, ARDALAN, and AIO regimens) in almost all the cases (N = 589, 94%), we focused our analyses on patients receiving these treatments.

Flow diagram showing selection of the study population.
Data collection
At baseline (during or shortly after hospital stay for colon cancer surgery), trained interviewers conducted interviews with the participants to collect information on sociodemographic (e.g. age at diagnosis and sex) and lifestyle factors (e.g. BMI at diagnosis) and medical history, using a standardized questionnaire. Detailed information on tumor (e.g. tumor stage and site) and medical characteristics (e.g. comorbidities) were also recorded in hospital records. We used the Charlson comorbidity index (CCI) 20 to quantify overall comorbidity, using information on comorbidities that were diagnosed either prior to or at the time of colon cancer diagnosis, as described previously.21,22 CCI score was categorized into three groups, namely CCI 0 (no comorbidity), 1, and 2+ (moderate-to-severe comorbidity).
About 3 years after diagnosis, detailed information on colon cancer treatment was collected retrospectively from medical records that had already been saved electronically. Treatment information extracted for our analyses included date of chemotherapy initiation and completion, number of chemotherapy cycles, type of treatment regimen, adverse treatment effects, whether patients completed all planned chemotherapy cycles, and whether there were dose reductions of chemotherapy during the course of treatment. Dose reduction was defined as administration of chemotherapy with a dose <75% of the starting dose. EDChemo was defined by treating physicians/oncologists and was obtained with the question, “did the patient discontinue the chemotherapy regimen earlier than planned?” Of patients having data on chemotherapy cycles (n = 436, 75%), those who had EDChemo of FOLFOX, CapMono, and 5-FU/LV received <10, <8, and <6 cycles, respectively.
Information on vital status and cause of death was ascertained from population registries and public health authorities about 3, 5, and 10 years after colon cancer diagnosis.
Statistical analysis
Because EDChemo is particularly common in oxaliplatin-based therapies, 6 we conducted the analyses separately for FOLFOX, CapMono, and 5-FU/LV. We first compared baseline characteristics (e.g. age and CCI score) among FOLFOX, CapMono, and 5-FU/LV recipients and differences were tested for statistical significance using Chi-square tests. We then used multivariable logistic regression to estimate odds ratios (ORs) and 95% confidence intervals (CIs) for the associations of baseline characteristics with EDChemo (yes/no). Next, we compared the frequency of adverse treatment effects in the three regimens in all patients and in subgroups according to age and comorbidity level. We used multivariable logistic regression to determine factors associated with adverse effects (any, yes/no).
Also, associations of adverse effects (individual and any) with EDChemo (yes/no) were assessed using multivariable negative binomial regression. Here, we also calculated population-attributable fractions (PAFs) to quantify proportions of EDChemo in the population that are statistically attributable to overall and specific adverse effects. In brief, PAFs were calculated using the Miettinen’s formula, 23 which uses adjusted relative risk (RR) and thus provides a more valid estimate in the presence of confounding.
where cF denotes case fraction (proportion of patients experiencing adverse effects who had early discontinuation).
We moreover compared treatment duration and number of chemotherapy cycles according to EDChemo, adverse effects (any), as well as specific adverse effects such as neuropathy, and gastrointestinal effects. Furthermore, we used multivariable logistic regression to determine factors associated with dose reduction during chemotherapy administration (yes/no, for all regimens combined due to small case numbers). Here, we also estimated proportions of dose reduction that are statistically attributable to adverse effects using PAFs, as described above.
Finally, we assessed the associations of EDChemo and number of chemotherapy cycles with all-cause mortality (mortality from any cause) and colon cancer mortality (mortality from colon cancer) using Cox proportional hazards regression. Because few patients received CapMono and 5-FU/LV, we conducted this analysis among recipients of FOLFOX only. Time was calculated from colon cancer diagnosis to the respective endpoints or end of follow-up, whichever occurred first. Two adjustment levels, defined a priori, were applied: (1) adjustment for sex, age, T-stage, and N-stage and (2) additional adjustment for living with a partner, BMI, tumor grade, tumor location, year of diagnosis, surgical volume, and CCI score. Regarding model 2, we used backward selection to select covariates with p < 0.5 for the adjustment (all variables in model 1 were forced into the model). With the exception of age, which was treated as continuous variable, all covariates were included in the models as categorical variables, as shown in Table 1. We checked the proportional hazards assumption for all covariates, by assessing whether their interaction with follow-up time was statistically significant. Time-dependent covariates (interaction terms of BMI and tumor grade with follow-up time) were added to the models because of violation of the proportional hazards assumption. Also, we addressed time differences between colon cancer diagnosis and patient recruitment by adding “delayed entry time” to the models.
Characteristics of FOLFOX, CapMono, and 5-FU/LV recipients.

Including MAYO (n = 77), Ardalan (n = 24), Roswell Park (n = 25), AIO (n = 12), and other regimens (n = 13).
Number of colon cancer surgeries performed per year.
In patients with available data only (n = 467, 87%; two hospitals did not report any information on dose reductions hence patients recruited in those hospitals were excluded from the analysis).
p-values were calculated from Pearson’s Chi-square test (*calculated from analysis of variance test).
5-FU, 5-fluorouracil and oxaliplatin; 5-FU/LV, any combination of 5-FU and LV; BMI, body mass index; CapMono, capecitabine monotherapy; FOLFOX, combination 5-FU, LV and oxaliplatin; LV, leucovorin; SD, standard deviation.
Statistical tests were two-sided, with a significance level of 5%. All analyses were conducted with SAS software, version 9.4 (SAS Institute, Cary, NC, USA).
Results
Characteristics of the study participants
Of 4916 CRC patients diagnosed in 2003–2014, 2926 had tumors located in the colon and 830 were in stage III (Figure 1). Of these, those who were not operated for colon cancer (n = 1), did not receive chemotherapy (n = 201), received chemotherapy other than FOLFOX, CapMono, and 5-FU/LV (n = 39) or had missing information on any of the variables of interest (n = 5) were excluded. A total of 584 colon cancer patients were included in the analysis. The median age was 68 (interquartile range, 61–74) years, and about 55% of the patients were men. FOLFOX was the most frequently applied regimen (n = 322, 55%; Table 1). For patients diagnosed in 2003–2006, 5-FU/LV had been the most frequently applied regimen, but this regimen was rarely applied among patients diagnosed in later years. Compared to CapMono and 5-FU/LV recipients, recipients of FOLFOX were younger, had lower comorbidity, and were more likely to be treated in high-volume hospitals.
About 52% of FOLFOX recipients discontinued their treatment prematurely. This proportion was higher than in CapMono (28%) or 5-FU/LV recipients (45%, p < 0.001). However, in patients not experiencing any adverse effect [Figure 2(b)], the proportion of EDChemo in FOLFOX recipients (18%) was comparable to that of CapMono recipients (14%, p = 0.559) and was even lower than in 5-FU/LV recipients (30%, p = 0.086). Of patients having data on dose reduction (n = 467, 80%), dose reduction rates were substantially higher in FOLFOX (17%) than in CapMono (9%) or 5-FU/LV (6%, p = 0.009, Table 1). EDChemo rates were also slightly higher in patients having versus not having dose reduction of chemotherapy (52% versus 46%), but the difference was not statistically significant (p = 0.391).

Frequency of early discontinuation of chemotherapy by treatment regimen according to the presence or absence of adverse effects.
Associations of baseline characteristics with EDChemo
Table 2 summarizes the associations of baseline characteristics with EDChemo of FOLFOX, CapMono, and 5-FU/LV. Among FOLFOX recipients, low-grade tumor (OR: 1.75, 95% CI: 1.06–2.86) and treatment in medium-volume hospital (versus high; OR: 1.83, 95%CI: 1.10–3.05) were associated with higher odds of EDChemo, respectively. EDChemo rates were also nonsignificantly higher in patients with CCI score 2+ (versus 0), those diagnosed in 2003–2006 (versus 2011–2014), and those in T1/2 (versus T4) stage. For CapMono, the frequency of EDChemo was 17% in patients with BMI < 25 kg/m2 and 46% in obese patients (OR = 4.31, 95% CI = 1.32–14.06). Also, patients not living with a partner, those who were overweight, and those with tumors located in the proximal colon and T4 (versus T1/2) stage had nonsignificantly 1.72-, 2.08-, 2.29-, and 3.70-times higher odds of EDChemo. Among 5-FU/LV recipients, there was a 32% increased odds of EDChemo per 5 years increase in age (OR: 1.32, 95% CI: 1.06–1.65). Treatment in medium-volume hospital (versus high; OR: 2.45, 95% CI: 1.08–5.55) and T4 stage (versus T1/2; OR: 4.35, 95% CI: 1.05–16.67) were also associated with much higher odds of EDChemo. Patients diagnosed in 2003–2005 (versus 2007–2014) had 2.45-times higher odds of EDChemo, but the difference was not statistically significant.
Associations of baseline characteristics with early discontinuation of chemotherapy (yes versus no).
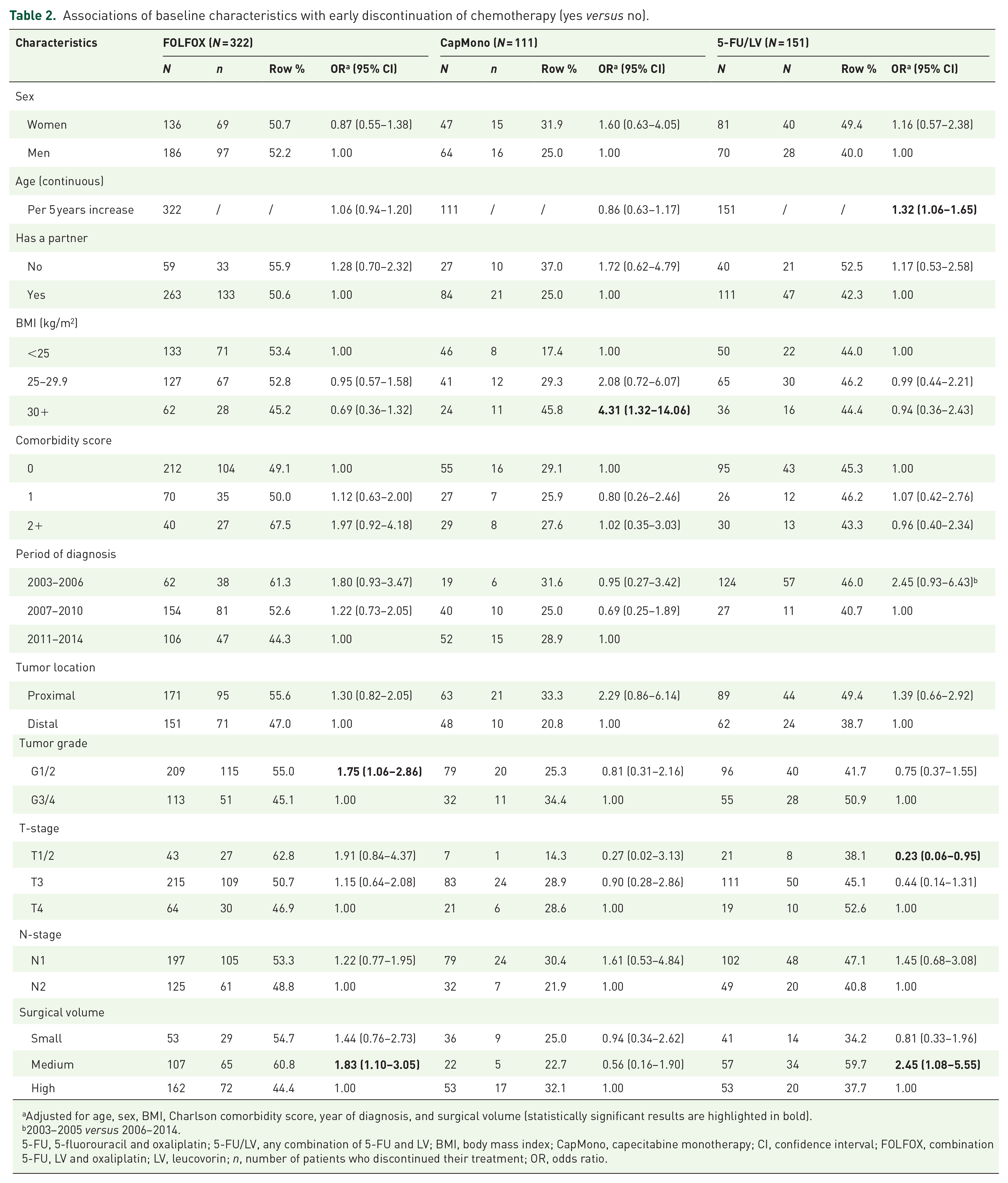
Adjusted for age, sex, BMI, Charlson comorbidity score, year of diagnosis, and surgical volume (statistically significant results are highlighted in bold).
2003–2005 versus 2006–2014.
5-FU, 5-fluorouracil and oxaliplatin; 5-FU/LV, any combination of 5-FU and LV; BMI, body mass index; CapMono, capecitabine monotherapy; CI, confidence interval; FOLFOX, combination 5-FU, LV and oxaliplatin; LV, leucovorin; n, number of patients who discontinued their treatment; OR, odds ratio.
Frequency and determinants of adverse effects
Adverse treatment effects were reported for almost four out of five patients treated with FOLFOX (79%) compared to approximately one out of two patients treated with CapMono (49%) or 5-FU/LV (50%, p < 0.001; Table 3). Also, the average number of adverse effects was higher in patients treated with FOLFOX (1.25) than those treated with CapMono (0.63) or 5-FU/LV (0.78, p < 0.001). The four most common adverse effects were neuropathy (31%), gastrointestinal effects (30%), cardiovascular/hematological effects (13%), and dermatological/allergic effects (11%), but they differed by treatment regimen (Table S1). For example, neuropathy (52%) and cardiovascular/hematological effects (21%) were particularly common in patients treated with FOLFOX, while gastrointestinal effects (40%) and dermatological/allergic effects (mainly hand–foot syndrome, 16%) were more prevalent in patients receiving 5-FU/LV and CapMono, respectively.
Adverse treatment effects and reasons for early discontinuation of adjuvant chemotherapy reported by treating physicians.
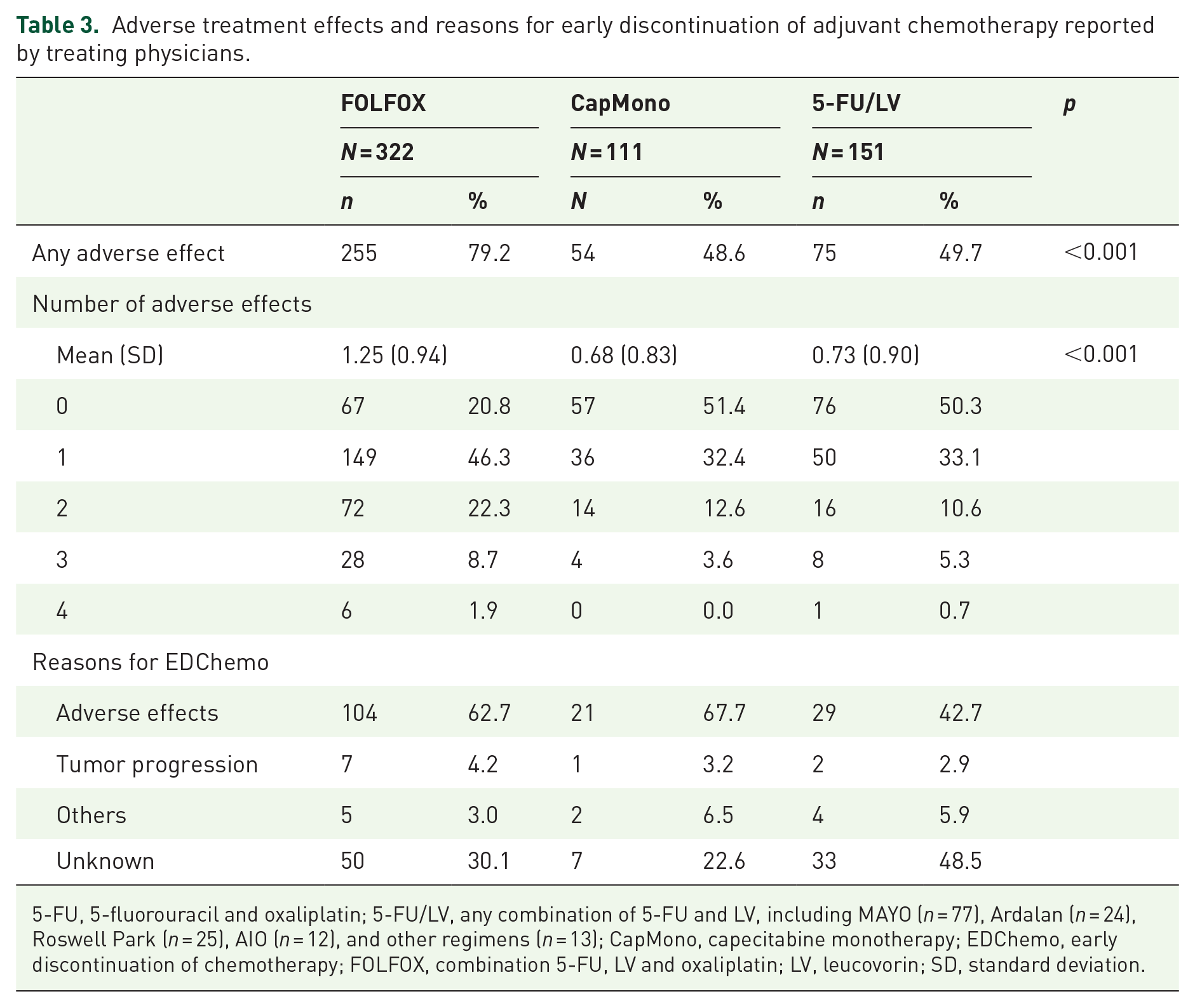
5-FU, 5-fluorouracil and oxaliplatin; 5-FU/LV, any combination of 5-FU and LV, including MAYO (n = 77), Ardalan (n = 24), Roswell Park (n = 25), AIO (n = 12), and other regimens (n = 13); CapMono, capecitabine monotherapy; EDChemo, early discontinuation of chemotherapy; FOLFOX, combination 5-FU, LV and oxaliplatin; LV, leucovorin; SD, standard deviation.
In multivariable analysis (Table S2), female sex (OR: 2.48, 95% CI: 1.33–4.63), diagnosis in 2003–2006 (versus 2011–2014; OR: 3.24, 95% CI: 1.23–8.50), and low-grade tumor (OR: 1.97, 95% CI: 1.09–3.58) were associated with increased odds of adverse effects of FOLFOX, respectively. For CapMono, patients in N2 stage had 73% lower odds of adverse effects (OR: 0.27, 95% CI: 0.10–0.76). Women and patients with low-grade tumors had nonsignificantly 1.99- and 2.35-higher odds of adverse effects, respectively. Among 5-FU/LV recipients, adverse events tended to be more common in women and in patients diagnosed in 2003–2006 (versus 2007–2014), but the differences were not statistically significant.
Associations of adverse effects with EDChemo
Table 4 shows RRs and PAFs for the associations of adverse effects with EDChemo of FOLFOX, CapMono, and 5-FU/LV. In FOLFOX recipients, gastrointestinal effects (RR: 1.25, 95% CI: 1.03–1.52), dermatological/allergic effects (RR: 1.62, 95% CI: 1.32–1.99), and ‘other’ adverse effects (RR: 1.66, 95% CI: 1.35–2.05) were associated with higher risk of EDChemo. Approximately 7%, 6%, and 7% of EDChemo of FOLFOX were statistically attributable to these adverse events, respectively. For CapMono, patients experiencing gastrointestinal effects (RR: 2.62, 95% CI: 1.52–4.50) and ‘other’ adverse effects (RR: 3.15, 95% CI: 1.70–5.86) had substantially higher risk of EDChemo, with corresponding PAFs of 26% and 15%. Among 5-FU/LV recipients, gastrointestinal effects were associated with about 1.60-fold increased EDChemo (RR: 1.62, 95% CI: 1.15–2.27), with a PAF of about 20%.
Associations of adverse treatment effects with early discontinuation of chemotherapy.

Adjusted for age, sex, body mass index, Charlson comorbidity score, year of diagnosis, and surgical volume (backward selection of variables with p < 0.5).
Statistically significant results are highlighted in bold.
PAF = cF × (RR−1)/RR, where cF denotes case fraction (proportion of patients experiencing adverse effects who had early discontinuation).
5-FU, 5-fluorouracil and oxaliplatin; 5-FU/LV, any combination of 5-FU and LV, including MAYO (n = 77), Ardalan (n = 24), Roswell Park (n = 25), AIO (n = 12), and other regimens (n = 13); CapMono, capecitabine monotherapy; CI, confidence interval; CV, cardiovascular; FOLFOX, combination 5-FU, LV and oxaliplatin; LV, leucovorin; NC, not calculated because all patients who experienced adverse effects discontinued their treatment; PAF, population-attributable risk fraction; RR, relative risk.
The proportions of EDChemo in patients experiencing versus not experiencing any adverse treatment effect were 60% and 18% for FOLFOX (p < 0.001), 43% and 14% for CapMono (p = 0.001), and 60% and 30% for 5-FU/LV (p < 0.001), with corresponding RRs of 3.16, 3.23, and 1.96. Overall, about 63%, 51%, and 32% of EDChemo of FOLFOX, CapMono, and 5-FU/LV were statistically attributable to adverse effects, respectively.
Treatment duration and number of chemotherapy cycles
The distributions of treatment duration and number of chemotherapy cycles according to EDChemo and adverse effects, separately for FOLFOX, CapMono, and 5-FU/LV, are shown in Table S3. In all the regimens, mean treatment duration and number of cycles were significantly lower in patients who discontinued their treatments prematurely and in patients who experienced adverse effects (any) and gastrointestinal effects. By contrast, no significant differences in treatment duration and chemotherapy cycles were observed among patients with and without neuropathy. Among FOLFOX recipients, the mean treatment duration (15.3 versus 22.9 weeks) and chemotherapy cycles (6.2 versus 9.0) were significantly lower in patients experiencing gastrointestinal effects (without neuropathy) than those experiencing neuropathy (without gastrointestinal effects, p < 0.001). Also, among patients having EDChemo, the mean number of chemotherapy cycles was significantly lower in those experiencing than those not experiencing gastrointestinal effects (Table S4).
Determinants of dose reduction
Table 5 shows ORs for the determinants of dose reduction during chemotherapy administration (all regimens combined due to small case numbers). In the model adjusted for age, sex, BMI, CCI score, year of diagnosis, hospital volume, and treatment regimen, treatment in small (versus high) volume hospital was associated with lower odds of dose reduction (OR = 0.42, 95% CI = 0.18–0.95). Men, overweight patients, patients with high-grade (versus low-grade) tumors, patients treated in medium (versus high)-volume hospitals, and those receiving CapMono or 5-FU/LV regimen (versus FOLFOX) also had dose reductions less frequently, but the associations were not statistically significant. Dose reduction rates were also much higher in patients experiencing versus not experiencing any adverse effect (18% versus 2% p < 0.001). In multivariable analysis, adverse effects were particularly strongly associated with dose reduction (OR: 8.36, 95% CI: 2.47–28.34) and accounted for about 82% of all dose reduction cases.
Factors associated with dose reduction of adjuvant chemotherapy after initiation (in patients with available data, n = 467).

Adjusted for age, sex, BMI, Charlson comorbidity score, year of diagnosis, surgical volume, and chemotherapy regimen.
Additional adjustment for adverse effects.
5-FU, 5-fluorouracil and oxaliplatin; 5-FU/LV, any combination of 5-FU and LV (including MAYO, Ardalan, Roswell Park, AIO, and other types); BMI, body mass index; CapMono, capecitabine monotherapy; CI, confidence interval; FOLFOX, combination 5-FU, LV and oxaliplatin; LV, leucovorin; n, number of patients who discontinued their treatment; OR, odds ratio. Statistically significant results are highlighted in bold.
After additionally adjusting for adverse effects, the associations of sex, tumor grade, hospital volume, and treatment regimen with dose reduction were attenuated sharply, but that for overweight became slightly stronger and reached statistical significance (OR: 0.50, 95% CI: 0.26–0.98).
Associations of EDChemo and chemotherapy cycles of FOLFOX with mortality
During a median follow-up of 5.2 (interquartile range, 4.3–9.6) years, 79 (25%) patients died, out of which 62 (78%) died from colon cancer. In the comprehensively adjusted models (Table 6), patients having EDChemo of FOLFOX had 38% higher risk of all-cause mortality (HR: 1.38, 95% CI: 0.86–2.21) and 42% higher risk of colon cancer mortality (HR: 1.42, 95% CI: 0.82–2.47), but the associations were not statistically significant. There was an inverse relationship between number of FOLFOX cycles and mortality, and the association seemed stronger for colon cancer mortality. For example, an additional FOLFOX cycle was associated with 9% lower risk of colon cancer mortality (HR: 0.91, 95% CI: 0.84–1.00). Among patients having EDChemo, a 14% lower risk of colon cancer mortality was observed per additional FOLFOX cycle (HR: 0.86, 95% CI: 0.74–0.99).
Associations of early discontinuation and chemotherapy cycles with mortality among recipients of FOLFOX (N = 322).

114 patients who received FOLFOX did not have data on chemotherapy cycles and hence were excluded from the analysis.
Crude estimates.
Adjusted for age, sex, T-stage, and N-stage.
Additional adjustment for living with a partner, BMI, grade, tumor location, year of diagnosis, surgical volume, Charlson comorbidity score, BMI × log(follow-up time), and grade × log(follow-up time) (backward selection of variables with p < 0.5).
Statistically significant results are highlighted in bold.
BMI, body mass index; CI, confidence interval; FOLFOX, combination 5-FU, LV and oxaliplatin; HR, hazard ratio; LV, leucovorin.
Discussion
The benefit of chemotherapy in stage III colon cancer patients is well documented. 24 The magnitude of this benefit is, however, dependent in part on whether patients complete the planned chemotherapy cycles. In a cohort of colon cancer cases receiving adjuvant chemotherapy, we assessed predictors of EDChemo and dose reduction, paying particular attention to the role of adverse treatment effects. We found that low-grade tumor and treatment in medium-volume hospital (for FOLFOX), obesity (for CapMono), and increasing age, T4 stage, and treatment in medium-volume hospital (for 5-FU/LV) were associated with increased EDChemo. Adverse effects were particularly a strong predictor of EDChemo and contributed to about 63%, 51%, and 32% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively. Adverse effects were, moreover, a strong determinant of dose reduction and accounted for about 82% of all cases of dose reduction.
Multiple studies have investigated factors associated with EDChemo in colon cancer patients6,8–10,14,15 but mostly for 5-FU/LV only8–10,15 or patients diagnosed before 2006.8–10,15 However, treatment regimen for colon cancer has changed since 2006, 25 calling for timely ascertainment of predictors of EDChemo in the current treatment regimens. The reported risk factors for EDChemo varied in the previous studies 26 and included female sex,9–11 older age,8,10,11,15 being single,8,10,15 higher comorbidity,8,10 poorly differentiated tumor,8,10 oxaliplatin-based regimen, 6 and receipt of treatment in higher volume hospitals. 9
Our study expands prior knowledge by providing separate analyses for FOLFOX, CapMono, and 5-FU/LV, unlike the vast majority of the previous studies that analyzed different regimens together even though EDChemo rates and patient characteristics differ by treatment regimen. 6 In our study, older age was not associated with EDChemo of CapMono, but older patients receiving FOLFOX and 5-FU/LV tended to have higher EDChemo risk. The absence of an association between older age and EDChemo in CapMono recipients corroborates the preferential use of CapMono as an adjuvant chemotherapy in older patients, given its less toxicity compared to oxaliplatin-based therapies, whose efficacy and safety in older patients are still debatable.2,27 However, another possible reason might be due to older patients receiving lower starting doses of CapMono likely due to comorbidities and/or poor renal function, 28 but this information was not available in our study. Our study also identified BMI < 25 kg/m2 as a potential risk factor for dose reduction, which is plausible as lower BMI correlates with low muscle mass or frailty. 29
An obvious determinant of EDChemo could be adverse treatment effects. 6 In the Eindhoven Cancer Registry, patients experiencing any grade III–IV adverse effect had over four-fold increased odds of EDChemo of CapMono. 6 However, the extent to which various adverse effects contribute to EDChemo is unclear. In our study, adverse effects that were independent predictors of EDChemo were gastrointestinal, dermatological/allergic (FOLFOX only), and ‘other’ adverse effects. In all the investigated regimens, gastrointestinal effects (e.g. diarrhea, nausea, and vomiting) had the strongest association with EDChemo and contributed to 7%, 26%, and 20% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively. Although chemotherapy-induced neuropathy negatively affects quality of life of CRC patients, 30 it played a minor role in EDChemo in our study. For example, no significant differences in treatment duration and number of chemotherapy cycles were observed among patients with and without neuropathy. Reasons for these observations are unclear, but a possible explanation is that neuropathy, unlike cardiovascular and gastrointestinal effects, may be less life threatening to warrant treatment discontinuation.
Our study provides evidence of differential association of adverse effects with EDChemo. It identified especially gastrointestinal events as relevant risk factors for EDChemo and could thus be recommended to receive further attention in routine care for colon cancer patients. The findings that about 82% of dose reduction and approximately 63%, 51%, and 32% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively, are due to adverse effects suggest that comprehensive screening for early signs and enhanced management of adverse effects, especially gastrointestinal events, might improve colon cancer care. However, development of scoring algorithms that incorporate information on specific adverse effects and other key predictors of EDChemo and dose reduction could also be beneficial for enhanced identification of high-risk patients and thereby also improve colon cancer care.
In agreement with results of the IDEA7,12 and SCOT trials, 13 patients having EDChemo of FOLFOX tended to have poorer prognosis in our study, even though the association was not statistically significant, likely due to limited statistical power. Also, an inverse association between number of chemotherapy cycles and mortality, especially colon cancer mortality, was observed, which agrees with prior evidence. 31 However, large, prospective studies are still needed to clarify whether EDChemo or shorter chemotherapy cycle is associated with poorer outcomes and in which specific patient groups, taking into consideration adverse treatment effects and quality of life.
Major strengths of our study include ‘real life’ administration of chemotherapy and detailed evaluation of determinants of EDChemo and dose reduction. Our study also has limitations. First, even though great efforts were made to include patients of all ages in the DACHS study, there was age gradient in patient recruitment, with higher recruitment of younger patients, in whom administration of chemotherapy, especially oxaliplatin-based therapies, is higher. Second, it is possible that the associations of adverse effects with EDChemo vary also by their severity, but we were not able to use standardized tools such as the Common Toxicity Criteria 32 to grade the adverse effects, as data on severity were not available in our study. Third, patients receiving chemotherapy for a longer duration might experience more adverse effects, potentially biasing the association of adverse events with EDChemo towards null. We, however, lacked information on when adverse events occurred. Fourth, although we ascertained several determinants of EDChemo and dose reduction, other relevant factors such as frailty and functional status could not be evaluated, as such data were not available in our study. Fifth, we classified any 5-FU/LV combination into one group because of small case numbers, but adverse effects might vary between specific 5-FU/LV regimens. Also, we combined cardiovascular and hematological effects into one group because of limited case numbers. Potential variations in their associations with treatment duration and/or discontinuation should be kept in mind when interpreting results of the combined group. Lastly, due to limited sample size, a number of potential associations could not reach statistical significance. Also, because few patients received CAPOX (n = 15), we could not assess determinants of EDChemo of CAPOX in our study. Future, large studies providing separate analyses for the currently used regimens and using the concept of PAFs are thus needed for further evidence on the roles of adverse effects and other relevant factors in EDChemo and dose reduction.
Conclusions
Among stage III colon cancer patients receiving adjuvant chemotherapy, low-grade tumor and treatment in medium-volume hospital (for FOLFOX), obesity (for CapMono), and increasing age, T4 stage, and treatment in medium-volume hospital (for 5-FU/LV) were associated with premature discontinuation of chemotherapy. Our study also suggests that over half of the cases of early discontinuation and dose reduction of chemotherapy are due to adverse treatment effects, with gastrointestinal effects showing the strongest associations and accounting for about 7%, 26%, and 20% of EDChemo of FOLFOX, CapMono, and 5-FU/LV, respectively. Further research should address the potential for reducing early discontinuation and dose reduction rates by close monitoring of patients for early signs and enhanced management of adverse effects, especially gastrointestinal events. Development of scoring algorithms that incorporate information on specific adverse effects and other relevant factors would also be valuable for enhanced identification of patients at increased risk of early discontinuation and dose reduction of chemotherapy.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359211006348 – Supplemental material for Early discontinuation and dose reduction of adjuvant chemotherapy in stage III colon cancer patients
Supplemental material, sj-pdf-1-tam-10.1177_17588359211006348 for Early discontinuation and dose reduction of adjuvant chemotherapy in stage III colon cancer patients by Daniel Boakye, Lina Jansen, Niels Halama, Jenny Chang-Claude, Michael Hoffmeister and Hermann Brenner in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank Ute Handte-Daub, Ansgar Brandhorst, Petra Bächer, and Dr. Viola Walter for their excellent technical assistance. We are particularly grateful to the study participants, as well as the interviewers who assisted in the data collection. We also gratefully appreciate the cooperation of the below-listed clinics and institutions: Chirurgische Universitätsklinik Heidelberg, Klinik am Gesundbrunnen Heilbronn, St. Vincentiuskrankenhaus Speyer, St. Josefskrankenhaus Heidelberg, Chirurgische Universitätsklinik Mannheim, Diakonissenkrankenhaus Speyer, Krankenhaus Salem Heidelberg, Kreiskrankenhaus Schwetzingen, St. Marienkrankenhaus Ludwigshafen, Klinikum Ludwigshafen, Stadtklinik Frankenthal, Diakoniekrankenhaus Mannheim, Kreiskrankenhaus Sinsheim, Klinikum am Plattenwald Bad Friedrichshall, Kreiskrankenhaus Weinheim, Kreiskrankenhaus Eberbach, Kreiskrankenhaus Buchen, Kreiskrankenhaus Mosbach, Enddarmzentrum Mannheim, Kreiskrankenhaus Brackenheim, and Cancer Registry of Rhineland-Palatinate, Mainz.
Author contributions
The author contributions were as follows: Conception and design of the study, D.B., L.J., and H.B.; Data acquisition and coordination, D.B., L.J., J.C-C., M.H., and H.B.; Data analysis and interpretation, D.B., L.J., M.H., and H.B.; Drafting of manuscript, D.B., L.J., and H.B.; and Critical revision of manuscript for important intellectual content, D.B., L.J., N.H., J.C-C., M.H., and H.B. All authors approved the final version of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the German Research Council (BR 1704/6-1, BR 1704/6-3, BR 1704/6-4, CH 117/1-1); German Federal Ministry of Education and Research (01KH0404, 01ER0814, 01ER0815, 01ER1505A, 01ER1505B); and the Ministry of Science, Research and Arts of Baden-Wuerttemberg.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

