Abstract
Aim:
More than half of patients with breast, lung, or prostate cancer who have bone metastases have evidence of skeletal-related events (SREs). Denosumab is a fully human monoclonal antibody that binds to and neutralizes receptor activator of nuclear factor kappa-B ligand (RANKL) on osteoblasts and their precursors. The United States Food and Drug Administration (FDA)-approved dose of denosumab is 120 mg every 4 weeks; however, other schedules have been used in practice for patient convenience. Evidence for the safety and efficacy of alternative dosing intervals is lacking.
Patient & Methods:
Adult patients with solid cancers and bone metastases who received at least two doses of denosumab 120 mg were reviewed. Patients were grouped based on an average denosumab dosing interval of <5 weeks (short-interval) versus 5–11 weeks (medium-interval) versus ⩾12 weeks (long-interval). The primary outcome was the time to first SRE while on denosumab between the short- and medium-interval groups. The secondary outcomes were overall survival (OS), efficacy comparisons between the other groups, and safety events.
Results:
There was no significant difference in median time to first SRE between the short- and medium-interval denosumab groups [33.2 versus 28.4 months, hazard ratio (HR): 1.13, 95% confidence interval (CI): 0.66–1.92, p = 0.91] or the medium- and long-interval dosing groups (28.4 versus 32.2 months, HR: 1.15, 95% CI: 0.66–2.01, p = 0.62). Median OS was not found to differ significantly between any of the groups. There were significantly more hospitalizations in the short-interval dosing group than the other groups (55.2% versus 33.8% versus 30.4%, p < 0.001).
Conclusion:
Extending denosumab dosing intervals does not appear to negatively impact time to first SRE and is associated with fewer hospitalizations in real-world patients with solid cancers and bone metastases.
Keywords
Introduction
A majority of patients with advanced solid cancers have metastases to the bones. 1 Bone metastases compromise the structural integrity of the bones by disrupting the homeostasis between osteoclasts and osteoblasts. Tumor cells in the bone lead to increased expression of receptor activator of nuclear factor kappa-B ligand (RANKL) – the main driver of osteoclast formation, function, survival, and subsequent bone destruction in metastases. 2 Osteoblast/osteoclast dysregulation and bone destruction leads to an increased risk of skeletal-related events (SREs) including spinal cord compression, pathological fracture, and radiotherapy or surgery to the bone. More than half of patients with breast, lung, or prostate cancer who have bone metastases have evidence of SREs. 1 SREs lead to increased pain, decreased overall survival (OS), and decreased health-related quality-of-life (HRQoL).1,3 Therefore, national guidelines recommend the use of bone-modifying agents in patients with metastatic breast cancer who have bone metastases, and are commonly utilized in other solid cancers who have evidence of bone metastases.4–7
Denosumab is a fully human monoclonal antibody that binds to and neutralizes RANKL on osteoblasts and their precursors. Neutralization of RANKL inhibits osteoclast function, prevents generalized bone resorption, prevents tumor-induced bone destruction, and ultimately decreases SREs.2,8–10 Three large randomized phase III trials have examined the efficacy of denosumab versus zoledronic acid on prevention of SREs in patients with solid tumors and bone metastases.
The dosing strategy used in three landmark clinical trials for the prevention of SREs, and subsequently United States Food and Drug Administration (FDA) approved, was 120 mg subcutaneously every 4 weeks.2,8–10 However, other schedules are frequently utilized in the clinic for patient convenience in conjunction with chemotherapy or hormone therapy schedule. Other denosumab dosing intervals, specifically 12 weeks, have been examined, but studies to date have failed to address a middle dosing interval that is often observed in clinical practice or examine OS.11,12 Alternative dosing of denosumab could be convenient for the patient but may affect efficacy of preventing SREs when given in a different dosing interval than studied. The safety and clinical outcomes of alternative denosumab dosing intervals in patients with solid cancers and bone metastases are unknown in a real-world population.
Methods
The primary objective of this retrospective cohort study was to assess outcomes in patients with solid cancers and bone metastases who received an average denosumab dosing interval of <5 weeks (short-interval) versus 5–11 weeks (medium-interval). A third group was included for patients who received an average dosing interval of ⩾12 weeks. The primary outcome was time to first SRE while on denosumab. SREs are defined as pathologic fracture (exclusive of major inciting trauma), any radiation or surgery to bone, or spinal cord compression. This includes patients who have received radiation and denosumab as treatment for their bone metastases. The secondary objectives were to explore differences in OS between the groups, as well as safety parameters including the incidence of hypocalcemia, osteonecrosis of the jaw (ONJ), and hospitalizations.
Adult patients 18 years or older with solid cancers and bone metastases who received at least two doses of denosumab 120 mg at Emory Healthcare from 1 November 2010 (date of FDA approval of denosumab for prevention of SRE) to 27 July 2018 were identified via DataWarehouse and included in this retrospective analysis. The date of data cutoff was 18 June 2019, allowing for a minimum time of follow up of 11 months. Patients who received denosumab for another indication (e.g., hypercalcemia of malignancy, osteoporosis, giant cell neoplasm, or multiple myeloma) were excluded. Patients who received a dose other than 120 mg, who did not have bone metastases, or who received denosumab outside of our health system were excluded. Patients who changed dosing intervals during denosumab treatment were also excluded. Patients’ electronic medical records were reviewed for data. Patient chart review was truncated when power was met in the primary comparison group. A tumor registry was utilized for dates of death or date of last follow up and cross referenced with patient charts. The Emory University Clinical and Translational Research Committee and the Emory University Institutional Review Board approved this study (IRB00106032 Approved 29 August 2018).
Statistical analysis
Descriptive statistics were used to summarize patient characteristics and clinical factors. The Chi squared test or Fisher’s exact test was used to analyze differences in safety events and other categorical variables. To assess the correlations between categorical clinical factors and numerical variables, the t test or an ANOVA test was conducted when data followed normal distribution, otherwise the Wilcoxon rank sum test or the Kruskal–Wallis test was used. Time to SRE survival while on denosumab was defined as the time from date of first dose of denosumab to date of skeletal-related event, death from any cause, or date of last follow up – whichever came first. OS was defined as time from date of diagnosis of bone metastases to date of death or last follow up. 13 Patients were censored at date of last follow up for analyses of time to SRE and OS analyses. Time to SRE and OS were estimated with the Kaplan–Meier method and compared using the log-rank test at a two-sided significance level of 5%, for comparisons between different groups stratified by cohort (short-, medium-, and long-dosing intervals), type of primary cancer, performance status, prior SRE, prior radiation, prior surgery, prior systemic therapy, presence of visceral disease, and presence of brain metastases, respectively. In order to achieve a minimum power of 80%, 125 patients were needed in the primary comparison groups. A Cox proportional hazards model was used to determine the effects of denosumab dosing schedules on clinical outcomes after adjustments for covariates at the significance level of a p value <0.05 in a multivariable analysis, and was used to estimate hazard ratio (HR) with 95% confidence interval (CI). Backward variable selection with an alpha level of removal of 0.1 was used to identify the best predictive models. The proportional hazards assumption was evaluated graphically and analytically with regression diagnostics. All data management and statistical analysis were conducted using SAS Version 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
There were 432 patients who met inclusion criteria and were part of the analysis. Reasons for exclusion are listed in Figure 1. The median time of follow up for all three groups was 70.8 months. The median time of follow up for the short-interval group was 72 months (3.12–95.25 months), the medium-interval group was 54 months (4–84.63 months), and the long-interval group was 58 months (7.87–80.41 months). The average denosumab dosing interval amongst all patients was 8.3 weeks, and was 4.6 weeks, 7.3 weeks, and 13.1 weeks in the short-, medium- and long-interval groups, respectively. The average number of doses of denosumab received was 10.6, 8.3, and 4.5 in the short-, medium-, and long-interval groups, respectively. Patient characteristics are listed in Table 1. Overall, the population was heavily pretreated, with 36% of patients receiving more than two lines of previous therapy for metastatic disease at time of first dose of denosumab. A majority of patients had either breast cancer (26.9%) or prostate cancer (38.7%), and 18.7% of patients had brain metastases. Other tumor types are detailed in Table S1 (Supplemental Appendix). There was a higher number of prostate cancer patients in the long interval versus breast and other cancer types.

CONSORT diagram.
Patient characteristics by denosumab dosing interval.

ECOG, Eastern Cooperative Oncology Group; SD, standard deviation; SRE, skeletal-related event.
There was no significant difference in the median time to first SRE between the primary comparison groups (short-interval versus medium-interval) or between the other groups (Table 2; Figure 2; Figures S1–S3, Supplemental Appendix). The multivariate analysis showed that patients had a significantly shorter median time to first SRE if they had prior radiation for their primary disease, received prior bisphosphonate therapy, or had a previous SRE (Table S2, Supplementary Appendix). Prior bisphosphonate use was not distributed evenly between groups.
Efficacy outcomes.

For the comparison between the short- and medium-interval groups, power = 88%.
For the comparison between the medium- and long-interval groups, power = 84%.
For the comparison between the short- and long-interval groups, power = 45%.
CI, confidence interval; mOS, median overall survival.

Kaplan–Meier-estimate time to first SRE short interval versus medium interval.
Similarly, there was no significant difference in median OS (mOS) between the primary comparison groups or between the other groups (Table 2; Figure 3; Figures S4–S6, Supplemental Appendix). The multivariate analysis showed that patients who were hospitalized while receiving denosumab had a significantly shorter mOS than patients who were not, and patients who had breast or prostate cancer had a longer mOS than other tumor types (Table S3, Supplemental Appendix).
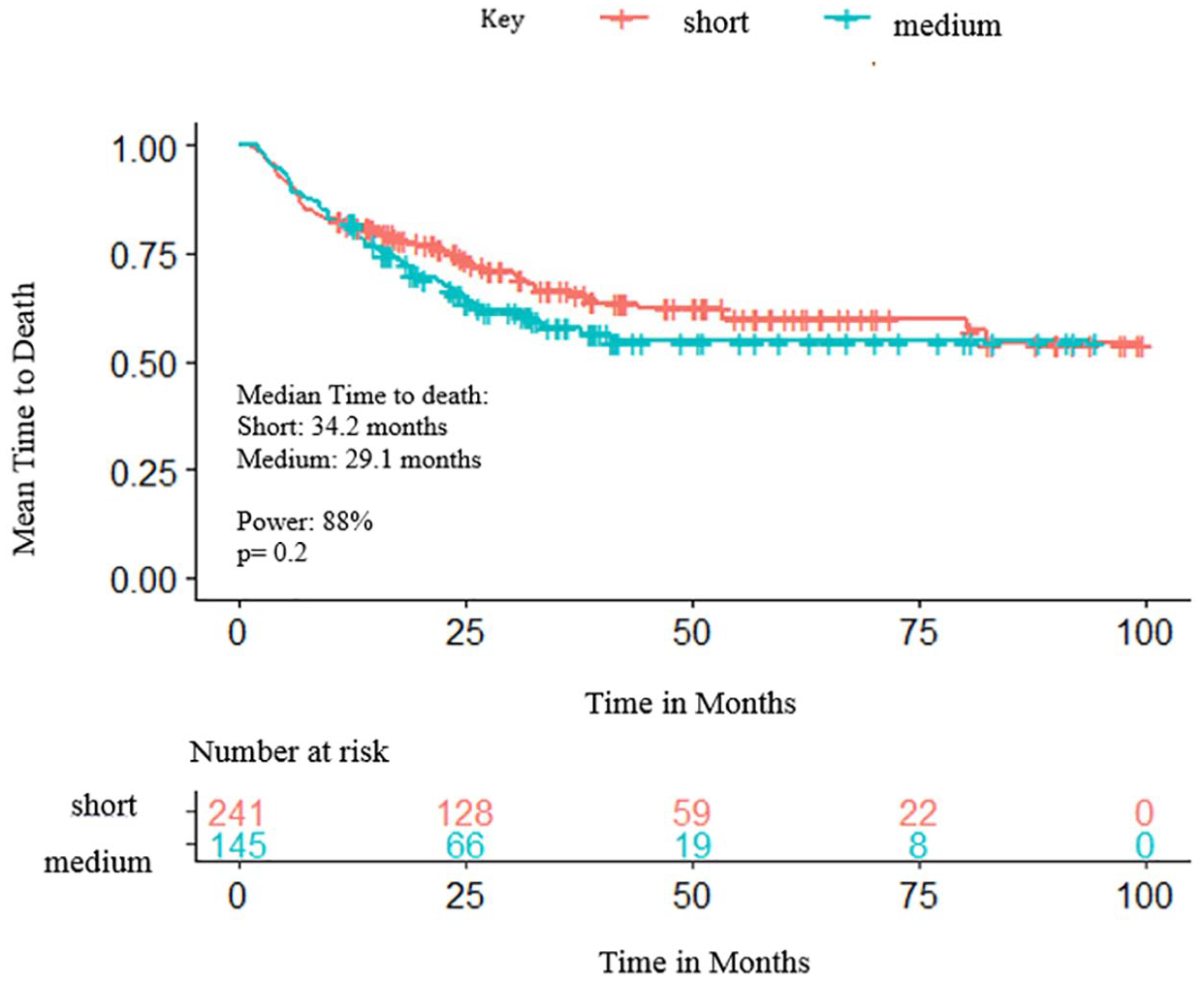
Kaplan–Meier-estimate time to death <5 weeks versus 5–11 weeks.
There were significantly more hospitalizations associated with short-interval denosumab dosing compared with extended dosing intervals (Table 3). Abdominal pain, hematuria and fever were the three most common reasons for hospitalization (Table S4, Supplementary Appendix). There was no significant difference in the incidence of any hypocalcemia or ONJ while on denosumab between the three groups; however, the study was not powered to detect a difference (Table 3). Both asymptomatic and symptomatic hypocalcemia were included. Whether the ONJ was spontaneous or after a dental procedure was difficult to assess due to the retrospective nature of the study.
Incidence of safety events while on Denosumab.

Based on corrected calcium.
ONJ, osteonecrosis of the jaw confidence interval.
Discussion
In this retrospective real-world cohort, we demonstrated that extended dosing schedules of denosumab beyond the recommended 4 weeks was not significantly different with time to first SRE or mOS. The decision to use average frequency was to incorporate a real-world population where patients may not receive their doses at a precise frequency. Patients who switched frequency (e.g., every 4 weeks to every 12 weeks dosing interval) were excluded to eliminate confounding factors. Each group encompasses the three most common dosing frequencies of every 4 weeks, every 6 weeks (for patients given therapy every 3 weeks, every other cycle), and every 12 weeks, which align with the administration of patients’ primary anti-cancer therapy in a majority of the cohort (breast and prostate cancer). The interval frequencies outside of labeling recommendations are used for patient convenience, adherence, and scheduling issues such as holidays. The primary comparison groups of short-interval (<5 weeks) versus medium-interval (5–11 weeks) were chosen to account for current gaps in the literature and real-world experience in this patient population. While power was achieved for the primary outcome comparison of the short- and medium-interval groups and also for the comparison between the medium- and long-interval groups, power was not achieved for the comparison between the short- and long-interval groups or for the comparison between the three arms as a whole for median time to SRE or OS due to a relatively small number of patients in the long-interval arm. However, observationally, there did not appear to be a difference in time to SRE or OS.
A systematic review and meta-analysis of every 4 weeks versus every 12 weeks denosumab or bisphosphonate administration in patients with bone metastases from breast cancer showed no difference in the incidence of SREs with extended interval dosing, and OS was not studied. 8 Though we focused on denosumab and not only patients with breast cancer, our results support these findings and add insight into survival outcomes associated with different dosing intervals. Moreover, a small phase II trial by Fizazi et al. examined denosumab 180 mg every 4 or 12 weeks in solid tumor patients who previously received bisphosphonates versus bisphosphonate continuation. 12 Fizazi et al. included multiple different tumor types with mostly breast or prostate cancer. Multiple myeloma was included in the study, which was excluded in our study. Of note, all patients in this study were exposed to prior intravenous bisphosphonates, whereas, in our study, only a small proportion of patients were previously exposed to intravenous bisphosphonates. Fizazi et al. found both dosing intervals decreased bone turnover markers, which was the primary endpoint, and the incidence of SRE in the every 4 weeks versus every 12 weeks was similar, with 5% versus 11%, respectively as a secondary endpoint. The length of the study was 6.25 months versus our study with a length of 9 years retrospectively. An average time to first on-study SRE was not reported but patients normalized their bone turnover markers while on denosumab despite already being on intravenous bisphosphonates. Fewer patients receiving denosumab experienced on-study SREs than those receiving intravenous bisphosphonates. Another phase II, randomized, active control trial by Lipton et al. comparing denosumab every 4 weeks (30, 120, 180 mg) and every 12 weeks (60, 180 mg) with intravenous bisphosphonate found no difference in time to first on-study SRE between all groups. 14 Lipton et al. studied specifically breast cancer patients and found no difference in on-study time to first SRE or bone marker turnover, but did not report an average. Only a small minority of patients in our study received prior bone-modifying therapy; however, our results are similar, with no difference with SRE incidence and time to SRE with those of the phase II trials, yet focuses on the approved denosumab dosing of 120 mg and includes a middle-interval cohort. The REaCT-BTA trial by Clemmons et al. was a randomized non-inferiority trial comparing every 12 weeks versus every 4 weeks intervals in bone-targeting agents including denosumab. Breast and prostate cancer patients with bone metastases were randomized to receive long- or short-interval treatment. There was no significant difference in HRQoL, which was the primary endpoint, and any of their secondary endpoints, including pain, global health status, and symptomatic skeletal events, although this study did not meet power. 15 There is also the current SWISS REDUCE trial that is planned to be completed by December 2022, which is a non-inferiority phase III trial also in breast and prostate cancer patient with bone metastases [ClinicalTrials.gov identifier: NCT02051218]. 16
Recently, it has been shown that denosumab may have antitumor efficacy and may impact mOS in patients with bone metastases, which is why we felt it necessary to include mOS as an endpoint. 17 Tumor cells stimulate production of parathyroid hormone-related peptide, which leads to the release of RANKL from osteoblasts. RANKL then binds to RANK on osteoclasts, leading to its maturation, activation, and survival. The release of growth factor from the bone due to osteolysis leads to stimulation of tumor growth by indirectly inducing angiogenesis and osteoclastogenesis that alter the bone microenvironment. 18 There are cancer cells that express RANK/RANKL, which allows denosumab to directly inhibit migration and tumor enlargement seen in vitro with non-small cell lung cancer. 19 Denosumab acts in this cycle by inhibiting RANKL, leading to decreased osteolysis, and, therefore, decreased release of growth factor, leading to a potential antitumor effect.17,20,21 Denosumab might also inhibit RANKL from cells that express RANK, which has been shown in vivo in several tumor lines.22,23 There is a significant interplay with the immune system and cytokine release, leading to bone remodeling and metabolism centered around RANK/RANKL, mTOR (mammalian target of rapamycin) and others (IL-33/IL31).18,24–27 Scagliotti et al. reported a subgroup analysis from a randomized phase III trial on any lung cancer patient and found a benefit in OS with denosumab 120 mg every 4 weeks versus IV zoledronic acid (8.9 versus 7.7 months; HR 0.80, p = 0.01). 28 This study also found improved OS in patients with visceral metastases, which could explain our findings that the medium-interval group did not have a worse OS despite having statistical significant difference with visceral metastases. A retrospective review that evaluated non-squamous NSCLC with bone metastases found an OS benefit of 21.4 months with denosumab versus zoledronic acid (12.7 months) and no treatment (10.5 months) (p < 0.01). 29 While denosumab may have direct antitumor efficacy, from our data this efficacy does not appear to be interval-dependent. Our data showed no difference in mOS between the groups across a variety of different tumor types, prior treatments, and severity of disease. Breast and prostate cancer patients had longer mOS versus the other cancer types, consistent with natural disease course and efficacious therapies for these tumor types. Of note, there has been a risk of secondary malignancies in patients receiving denosumab dosing every 4 weeks, although our study did not evaluate this. 30
Although there were statistically significant differences in patient characteristics between the three groups, amongst those variables in the multivariate analyses only prior bisphosphonate use was found to be associated significantly with time to SRE, although not distributed evenly between dosing intervals. The differences in cancer types between the three groups may be due to the frequency with which treatments are given, which would correlate with when patients could receive denosumab for convenience and less frequent visits to the infusion center. Of note, there was a higher number of prostate cancer patients, who are less likely to develop a SRE, in the long-interval arm, which could influence the results. Typically, these patients are receiving androgen deprivation therapy every 12 weeks, and this therefore influences the frequency of denosumab. This could be a consideration that potentially influences our results. There was also a difference with presence of visceral metastases being higher in the medium-interval group, indicating a possibly sicker population. This could explain an observational trend towards shorter mOS in that group, though not significant. However, this did not correlate with any differences in safety, with greater hospitalizations being reported in the short-interval group. Provider selection bias could have influenced hospitalization by choosing the short-interval group in higher risk or more aggressive disease, leading to the observational higher rates seen in this group but not affecting OS. Baseline renal function, calculated using the Cockcroft-Gault Equation, was also different between groups. The medium-interval group had a slightly worse renal function but did not correlate with safety. The dietary supplementation data was obtained based on patients’ electronic medication list and history and is subject to inherent limitations on omission, accuracy, and compliance. This could impact SRE risk and development of hypocalcemia while on denosumab. Supportive care is recommended to help prevent side effects while on denosumab, and guidelines recommend patients take vitamin D 400–1000 international units daily and calcium 1000–1200 mg daily.31,32
The multivariate analysis in our study showed that any prior SRE or radiation to the bone decreases time to first SRE despite being on denosumab. The studies that brought denosumab to approval versus zoledronic acid with different tumor types did not report whether prior SRE or radiation to bone increased time to first SRE.2,8,9 The median time to first SRE in the studies was 20.6 months (Henry et al.), 20.7 months (Fizazi et al.), and not reached (Stopek et al.). There was no difference in OS between denosumab and zoledronic acid.2,8,9
There were significantly more hospitalizations in the short-interval group versus the extended interval groups; however, reasons for hospitalization can be attributed to many different factors in cancer patients, and the most common reasons found in our cohort were unlikely related directly to denosumab administration. As mentioned previously, this could also be due to provider selection bias. Although there was no statistically significant difference in the incidence of hypocalcemia or ONJ between the groups, the study was not powered to detect a difference, and, observationally, the incidence trended in favor of the extended-interval groups, decreasing these adverse effects. The DRONJ study was a single-center retrospective observational study that looked at risk factors associated with ONJ in metastatic solid cancer patients on denosumab 120 mg every 4 weeks. 33 The authors found a correlation with patients on hormone therapy, chemotherapy/molecular targeted treatment, and apical periodontitis. The incidence of ONJ was 14% (14/123 patients) versus our study, which had an incidence of 1.6% (7/432). Our study better aligns with what is found in prospective trials, and the DRONJ study was in Japanese patients, which may influence the results.34–36 The lack of statistical significance could be skewed due to the limited number of patients in the long-interval group and not meeting power when comparing all three groups. The most common adverse event associated with denosumab within the phase III studies and meta-analysis and systemic review was hypocalcemia with very low rates of ONJ.2,8,9,11 The most common adverse event in the phase II Fizazi et al. study was hypocalcemia, nausea, and anemia, no cases of ONJ were reported. 12
In conclusion, extending denosumab dosing intervals beyond 4 weeks did not appear to negatively impact time to first SRE or mOS in solid tumor patients with bone metastases. Extending dosing intervals make logistical sense in the real-world setting. Impacts of extended dosing intervals of denosumab on HRQoL and cost analysis warrants further study.
Supplemental Material
sj-pdf-1-tam-10.1177_1758835920982859 – Supplemental material for Safety and efficacy of extended dosing intervals of denosumab in patients with solid cancers and bone metastases: a retrospective study
Supplemental material, sj-pdf-1-tam-10.1177_1758835920982859 for Safety and efficacy of extended dosing intervals of denosumab in patients with solid cancers and bone metastases: a retrospective study by Aseala I. Abousaud, Meagan S. Barbee, Christine C. Davis, Sarah E. Caulfield, Zeyuan Wang, Alexa Boykin, Bradley C. Carthon and Keerthi Gogineni in Therapeutic Advances in Medical Oncology
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in the submitted article are the own words of the authors and not an official position of the institution.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
