Abstract
Background:
Stomatitis is a frequent dose limiting toxicity of everolimus, an approved therapy for patients with metastatic breast cancer. No randomized trials of a prophylactic measure to prevent mucositis have been reported.
Methods:
We conducted a phase II, open-label trial in which patients with metastatic breast cancer starting everolimus were randomized to best supportive care (BSC) versus prophylactic use of an oral mucoadhesive, non-steroid containing mouth wash. The primary endpoint was rate of any grade stomatitis as reported by the treating physicians. Secondary endpoints were severity of stomatitis according to the Oral Mucositis Assessment Scale (OMAS) and rates of everolimus dose reduction or discontinuation due to mucositis.
Results:
Of 61 evaluable patients, 32 were randomized to and treated with oral mucoadhesive and 29 with BSC. Any grade stomatitis developed in 46.9% (15/32) of study arm and 65.5% (19/29) of BSC arm patients (p = 0.14). The difference between the two arms was significantly in favor of the mucoadhesive arm when mucositis was scored according to the OMAS with average score of 0.3 in study arm versus 0.5 in the control arm (p = 0.03). There were fewer dose adjustments or therapy discontinuations in the study arm compared with BSC (16% versus 31%, respectively) but the difference did not reach statistical significance.
Conclusion:
Here we provide early evidence from the first randomized trial supporting the use of oral prophylactic mucoadhesive for everolimus-associated stomatitis. A trial comparing prophylactic oral mucoadhesive to steroid mouth wash may be warranted.
Introduction
Endocrine therapy is the mainstay treatment for patients with metastatic or unresectable hormone receptor (HR) positive breast cancer. With the approval of targeted agents such as CDK4/6 inhibitors, progression free survival for patients with HR positive breast cancer treated with combination of these agents and endocrine therapy has almost doubled.1–3 However, treatment resistance to endocrine therapy, whether de novo or acquired, will ultimately affect all patients. One well characterized pathway of resistance is activation of the PI3K/AKT/mTOR pathway.
Everolimus, an mTORC1 inhibitor, is thought to directly and indirectly inhibit tumor growth by inhibiting the proliferative signals of the PI3K pathway. After preclinical 4 and early clinical studies 5 showing evidence of activity in HR positive breast cancer, everolimus was evaluated in the phase III BOLERO-2 trial in combination with exemestane, an aromatase inhibitor, in postmenopausal women with advanced HR positive breast cancer whose disease had progressed after prior endocrine therapy. 6 The everolimus combination arm was associated with a significantly improved progression free survival compared with the exemestane plus placebo arm (6.9 months compared with 2.8 months, hazard ratio 0.43; 95% confidence interval; p < 0.001). Several other expanded access studies subsequently followed, such as 4EVER, BRAWO, and STEPAUT, that evaluated everolimus plus exemestane in more diverse patient populations than did BOLERO-2, with similar results, ultimately leading to its approval in this setting. 7 Although overall survival was not shown to be significantly improved, this therapy remains a standard option for patients with this disease. 8
While generally well tolerated, 19% of patients discontinued everolimus as a result of adverse events, compared with 4% in the placebo arm in the BOLERO-2 trial. 6 Stomatitis was the most common dose limiting effect, occurring in 56% of patients, with grade 3–4 in 8%. At the time, no therapies were shown to prevent the occurrence of stomatitis, thus this side effect was usually managed by dose reduction, dose interruption or treatment discontinuation. Since then, the SWISH trial has been published showing oral dexamethasone to have efficacy in the prevention of mucositis from everolimus in the setting of a single arm trial. 9 This study presented the first commercially available and well tolerated mouth wash to be effective for the prevention and treatment of everolimus induced mucositis. The SWISH trial was published after the initiation of our trial and is now an available option for patients. However, no non-steroidal option has yet been presented as an option for patients and no regimen has been evaluated in the context of a randomized trial.
Oral mucoadhesive (MuGard), a viscous polymer-based oral mucoadhesive solution comprising glycerin, benzyl alcohol, potassium hydroxide, citric acid, phosphoric acid, emulsifier polysorbate 60, and carbomer homopolymer A, is designed to provide a protective shield over the surface of the mouth and tongue, thereby providing both relief and protection from mouth sores. This oral mucoadhesive has been studied primarily in patients undergoing radiation therapy for head and neck cancers, who are at very high risk for developing mucositis. In one study of 129 participants with cancer (one-third of whom had head and neck cancer) undergoing radiation with or without chemotherapy, the use of oral mucoadhesive was associated with an improvement in mucositis in 46%, and stabilization in oral lesions in 32%. 10 In another multicenter randomized study of 120 participants with head and neck cancer undergoing chemoradiation therapy, use of oral mucoadhesive mitigated oral mucositis symptoms as reported by patients (p = 0.034). 11 The mechanism of everolimus induced mucositis is unclear. The rapid appearance of this toxicity suggests a different mechanism from chemotherapy induced mucositis. One study using oral mucosa models showed evidence of direct everolimus induced epithelia injury followed by pro-inflammatory cytokine release and subsequent immune response resulting in the recruitment and influx of acute inflammatory cells. 12 Oral steroids may be therapeutic by helping with the second step. However, a mucoprotective product was postulated to reduce the odds of mucositis by providing an added layer of protection to the mucosa by preventing oral mucosa injury in the first place.
In the context of a randomized clinical trial, we aimed to evaluate whether primary prophylactic use of this mouth wash in patients starting everolimus for metastatic breast cancer is associated with a reduced rate of mucositis and reduction in rates of dose reduction or discontinuation of therapy.
Methods
Study design and participants
Translational Research in Oncology (TRIO)-US B-10 was a multi-site US-based phase II, open label, randomized trial to assess the use of a mucoadhesive oral solution to prevent everolimus-associated stomatitis (ClicnicalTrials.gov identifier: NCT02015559). Protocol was reviewed and approved by UCLA IRB (IRB# 13-001676) and Western Institutional Review Board (Approval # 20141123). Eligible participants were women 18 years of age or older with metastatic or locally recurrent unresectable breast cancer who were candidate to be treated with everolimus 10 mg daily combined with endocrine therapy. There was no restriction on type of endocrine therapy used. Patients were required to have estrogen receptor and/or progesterone receptor positive and HER2 receptor negative breast cancers by local assessment. Adequate baseline labs were required (Absolute Neutrophil Count >1.5, platelets >100, Hemoglobin >8, International Normalized Ratio [INR] <2, Aspartate Aminotransferase [AST] and Alanine Aminotransferase [ALT] <2.5 times the upper limit of normal [ULN] or <5 times ULN if with hepatic metastasis). Patients with severe and/or uncontrolled other medical conditions were excluded. Patients with baseline oral ulcers, prior therapy with everolimus or another mTOR inhibitor, receiving chemotherapy or radiation therapy within 4 weeks of start of therapy, and/or concurrent use of blood thinners were excluded. There were no restrictions on prior use of CDK4/6 inhibitors and no limit on number of prior lines of endocrine or chemotherapy.
Written informed consent was obtained from patients prior to screening. Patients were randomly and equally assigned to oral mucoadhesive or best supportive care (BSC). No stratification factors were used for randomization. Random permuted block design with variable block size was used to generate a randomization list and subjects were randomized according to the list by a study staff who was not involved in patient evaluation.
Procedures
Standard of care everolimus was started at 10 mg daily along with endocrine therapy of the oncologist’s choosing. Patients assigned to the mucoadhesive agent initiated this prophylactic therapy on day 1 of cycle 1 of everolimus, four times daily. Patients were instructed to gently swish 5 mL of oral mucoadhesive in the mouth for 30–60 s each time. Excess oral mucoadhesive could then be swallowed or expelled. After rinsing with oral mucoadhesive, participants were not to take anything (food, drink, medications) by mouth for at least 30 min. The control arm of the study did not receive any preventive measures and prophylactic use of steroid containing prophylactic mouth wash was specifically prohibited.
Upon occurrence of mouth sores, both arms were to receive standard of care supportive measures per the treating oncologist. Dose reduction of everolimus was per the treating oncologist’s discretion. Recommendations were as follows: for grade 1 or 2 toxicity, magic mouth wash was recommended (e.g. one part viscous lidocaine 2%, one part diphenhydramine 12.5 mg per 5 mL elixir, one part maalox). For grade 3 or 4 toxicity, a steroid containing mouth wash was recommended to be used 2–3 times a day as needed. In the oral mucoadhesive arm, oral mucoadhesive was to be continued alongside additional standard of care per above. Oral mucoadhesive was continued for the duration of everolimus therapy up to a maximum of 6 months of treatment, unless not tolerated.
Patients were evaluated at the first day of each 30-day cycle (± 3 days) and in addition called weekly during the first month of therapy. Patients were followed until time of progression, discontinuation of everolimus due to side effects or after 6 months of therapy, whichever came first. Patients kept a diary of symptoms and at each visit this was reviewed along with physical exam (including evaluation of oral mucosa according to the Oral Mucositis Assessment Scale (OMAS) scale; Table 1) and labs. The OMAS quantified degree of erythema and ulceration in the mouth. This detailed scale grades ulceration with a score from 0 to 3 at eight locations in the mouth. Score of 0 means no ulceration, score of 1 is an area of ulceration measuring less than or equal to 1 cm2, score of 2 an area of ulceration measuring 1–3 cm2 and score of 3 is an area 3 cm2 or greater. Severity of erythema in each ulcerated area is also graded by this scale from 0 to 2. Zero signifies no erythema, 1 mild erythema and 2 a severe area of erythema. The average of scores for ulceration and erythema were then assessed for each patient with an average of both erythema and ulceration being the total average OMAS score for each patient. All participating sites received training on use of this scale.
Oral mucositis assessment scale.

Area of ulceration: 0 = none, 1 = ⩽1 cm2, 2 = 1–3 cm2, 3 = ⩾3 cm2.
Severity of erythema: 0 = none, 1 = not severe, 2 = severe.
Statistical analysis
The primary endpoint was rate of grades 1–4 stomatitis in participants per Common Terminology Criteria for Adverse Events (CTCAE) grading criteria 4.03. Chi-square test was used to compare the rates between the two study arms. Secondary endpoints included rate of stomatitis as measured by the OMAS scale (Table 1), and rate of everolimus dose adjustment or discontinuation related to stomatitis in patients treated with oral mucoadhesive versus BSC.
For the primary endpoint, the assumption (based on BOLERO-2) was that grade 1–4 stomatitis would occur in 56% (P0) of participants receiving everolimus with BSC. We aimed to test whether oral mucoadhesive reduced this rate to 30% (P1) with an alpha 0.10, using Chi-square test with a one-sided significance, and 80% power, thereby requiring n = 33 in each arm, totaling a goal of 66 participants.
Results
From November 2014 to June 2017, 62 patients signed consent for the trial at 13 participating sites in the United States and were randomized. During the enrollment period, data supporting the use of prophylactic steroid mouth wash emerged, 6 resulting in a slowing of enrollment in this trial. Thus, a decision was made to end accrual early with 62 patients enrolled, just short of our goal of 66. Of 62 patients enrolled, one patient did not initiate therapy and is not evaluable. As such, 61 patients were treated on protocol (32 to mucoadhesive and 29 to BSC) and were evaluable for efficacy and safety endpoints (Figure 1). Baseline patient characteristics were similar between treatment arms and are shown in Table 2. Mean age was 65 years in the treatment arm and 62 in the control arm. The majority were White (78% in the treatment arm and 79% in the control arm). There was no statistical significance in number of prior lines of therapy between the two arms with only one patient in the investigational arm and two patients in the control arm receiving therapy in the first line setting. Similar number of patients received 2–3 and 4 or more prior lines of therapy (p = 0.78).

CONSORT diagram. Sixty-two patients were assessed for eligibility, of whom 61 patients participated in the study. Twenty-nine patients were randomized to standard of care while 32 patients were randomized to prophylactic oral mucoadhesive therapy.
Baseline patient characteristics (N = 61).
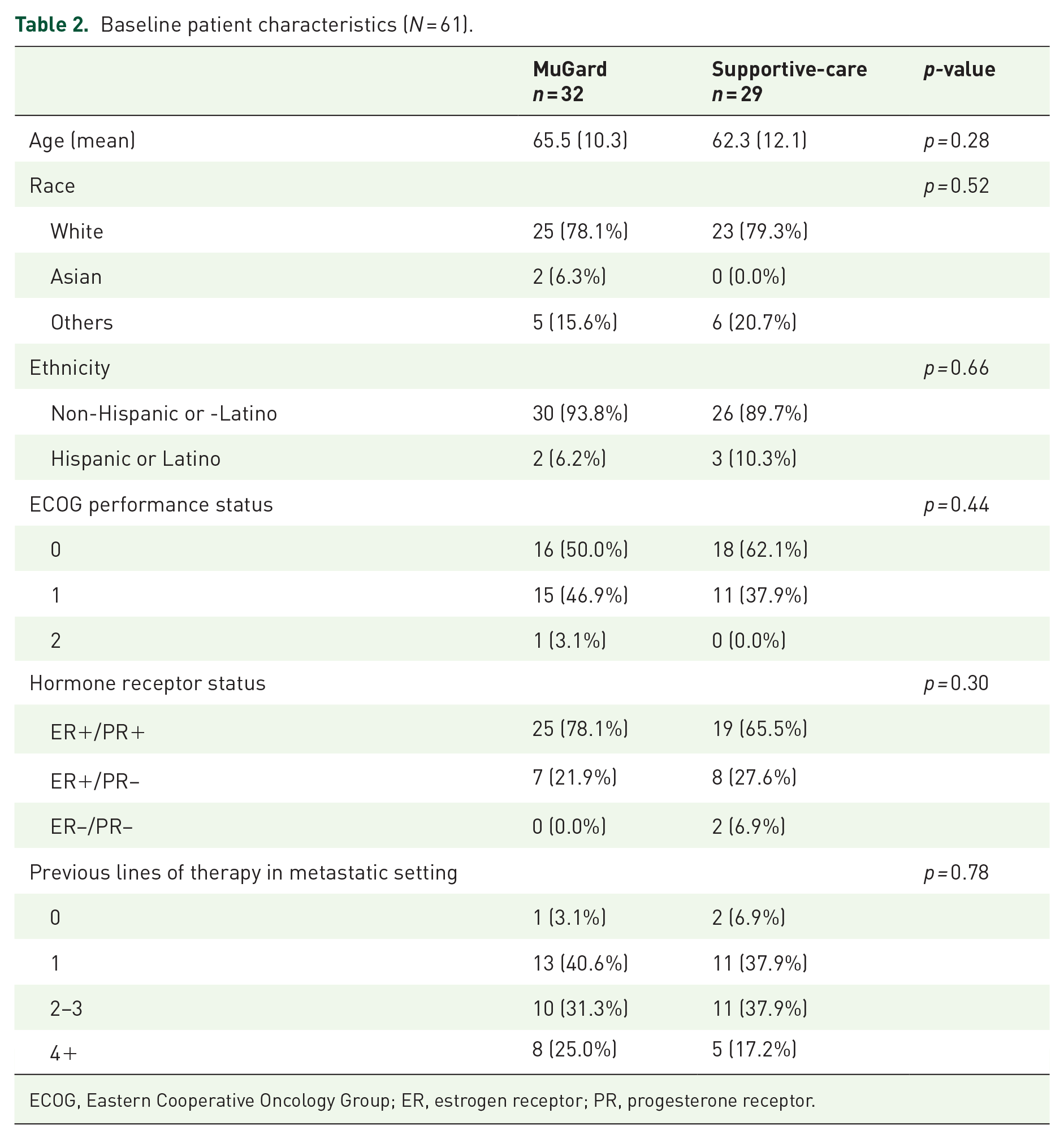
ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; PR, progesterone receptor.
Table 3 shows results of primary end point of rate of mucositis of any grade in the two study arms. Fifteen patients developed mucositis out of 32 patients in the oral mucoadhesive arm compared with 19 out of 29 patients in the control arm (46.9% versus 65.5%). However, this difference did not reach statistical significance (p = 0.14). Grade 2 or greater mucositis occurred in seven patients in the treatment arm (21.9%) compared with five patients in the control arm (17.2%). This also did not reach statistical significance (p = 0.65).
Primary endpoint: rate of grades 1–4 (Common Terminology Criteria for Adverse Events criteria) stomatitis in participants treated with everolimus a .
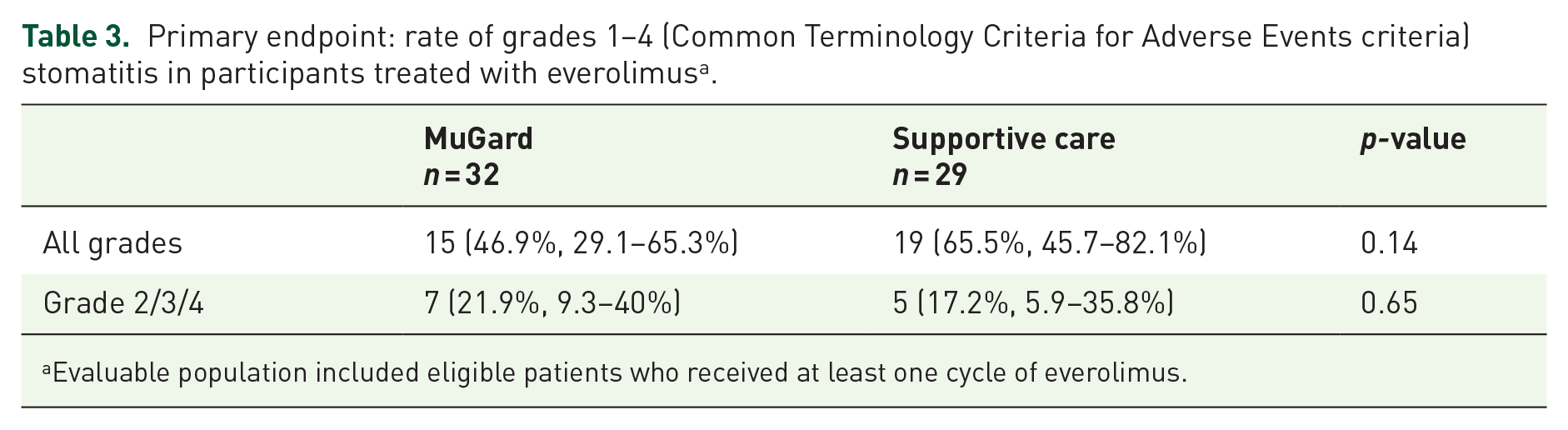
Evaluable population included eligible patients who received at least one cycle of everolimus.
Table 4 shows the results of the secondary endpoint of mucositis as measured by the treating physicians by the OMAS scale. Mucositis was less severe in the oral mucoadhesive arm with an average of 0.3 total OMAS score compared with the control arm, which had an average of 0.5 OMAS score. This end point reached statistical significance (p = 0.03).
OMAS total and subscores while on treatment a .

Evaluable population included eligible patients who received at least one cycle of everolimus.
OMAS, Oral Mucositis Assessment Scale.
More patients had everolimus held or dose reduced due to mucositis in the control arm compared with the treatment arm (31% versus 15%) (Table 5). This was not, however, statistically significant (p = 0.15).
Secondary endpoint: everolimus dose adjustment or therapy discontinuation due to stomatitis a .

Evaluable population included eligible patients who received at least one cycle of everolimus.
CI, confidence interval.
Discussion
Stomatitis is the primary dose limiting side effect of everolimus and a class effect of mTOR inhibitors. In this study, the only randomized study of a prophylactic agent for prevention of mouth sores that we are aware of, the use of oral mucoadhesive, was associated with a significant benefit in reducing the severity of mucositis as measured by a quantitative scale. While rates of all grade mucositis were not statistically significantly different between the two arms, a trend towards benefit was observed with the use of oral mucoadhesive. Rates of everolimus dose reduction and therapy discontinuation also tended to be better with oral mucoadhesive.
Since the initiation of our study, Rugo et al. published results of a non-randomized study of oral dexamethasone to prevent everolimus induced mucositis in this patient population. 9 The SWISH trial was a single arm phase II study in which 85 patients were evaluable for efficacy. By 8 weeks, the incidence of grade 2 or worse stomatitis was 2% in their patient population. This compared favorably with the rate seen in the original BOLERO 2 trial (33%). Oral dexamethasone has since become an option for patients starting everolimus therapy. This study affected accrual to our trial with enrollment stopping at 61 patients, instead of the planned 66 that was the projected number needed to treat to reach statistical significance.
The SWISH study, though with impressive results, was not randomized and therefore both degree of benefit and possible adverse events from steroid mouth are not completely clear. Furthermore, patients included in our trial were more heavily pre-treated. In the SWISH study, 62% of patients had not received any prior therapy in the metastatic setting. 9 These patients may therefore have had less mucositis due to less cumulative toxicity and decline in functional status. In clinical practice, everolimus is rarely used in the first line setting as other options such as fulvestrant, CDK4/6 inhibitors, and aromatase inhibitors are better tolerated and used first. In our study only 3% of patients in the treatment arm and 6% of patients in the BSC arm were treated in the first line setting. Therefore we believe our population more closely resembles the real world setting in which everolimus is typically used after at least one line of prior therapy. The rate of mucositis in our control arm was similar to those reported in the BOLERO-2 trial, further adding validity to the results observed in the mucoadhesive arm.
In summary, our results provide evidence supporting a non-steroidal option for the prevention of stomatitis in patients treated with everolimus. How this method would compare with a steroid mouth wash or whether addition of steroids to this oral mucoadhesive could provide additional benefit remain unclear. We believe, based on the current data, that the use of oral mucoadhesive represents at least an additional option for patients, especially for those who are not able to take oral dexamethasone. It would be interesting in future studies to combine oral dexamethasone mouth wash with oral mucoadhesive.
Footnotes
Acknowledgements
Oral mucoadhesive was provided by Access Pharmaceuticals. The study was designed by the principal investigator and conducted independently of the funding sources as an investigator initiated trial.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funding support for trial provided by Novartis with MuGard provided by Access Pharmaceuticals. Authors report no conflicts of interest. Sara Hurvitz reports funding support from the following: Contracted research from: Ambrx, Amgen, Arvinas, Bayer, Daiichi-Sankyo, Genentech/Roche, GSK, Immunomedics, Lilly, Macrogenics, Novartis, Pfizer, OBI Pharma, Pieris, PUMA, Radius, Sanofi, Seattle Genetics, Dignitana, Zymeworks, Travel reimbursed: Lilly, Novartis. Parvin Peddi reports funding support for contracted research from Novartis, Lilly and Sanofi.
